Chap 24D - Amino acids
1/5
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
6 Terms
Describe amino acids + draw general structure
Contain both an acidic carboxyl group (–CO2H) and amino group (–NH2) = amphoteric
Basic building blocks of proteins and there are 20 naturally occurring a-amino acids
Both functional groups are attached to the same a-carbon atom
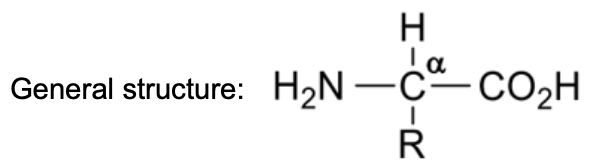
Describe mp of amino acids
Crystalline solids
High mp
Giant ionic structures containing dipolar ions held by strong electrostatic force of attraction
Describe solubility of amino acids
Soluble in water but insoluble in non-polar solvents
Ion-dipole interactions between amino acids and H2O -> energy released to overcome ionic bonds between amino acids and HB between H2O
Insufficient energy released from ion-dipole interactions between dipolar ions and non-polar solvent to overcome the ionic bonds
Describe amino acids in a neutral solution + draw the ion formed
Amino acids exist as dipolar ions with no overall charge.
The dipolar ion is formed via internal acid-base reaction between the ─NH2 and ─CO2H groups

Describe amino acids in a acidic solution + draw the ion formed
Amino group accepts a proton forming a cation
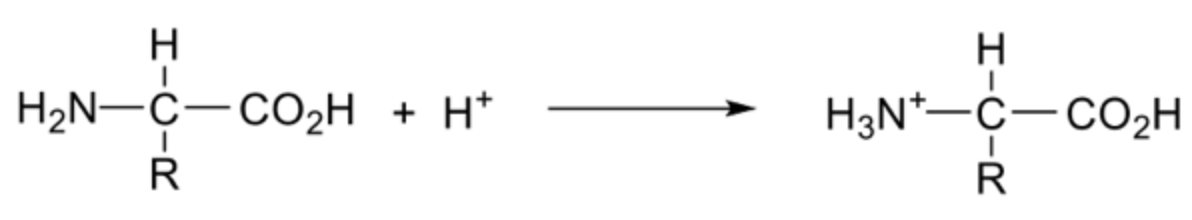
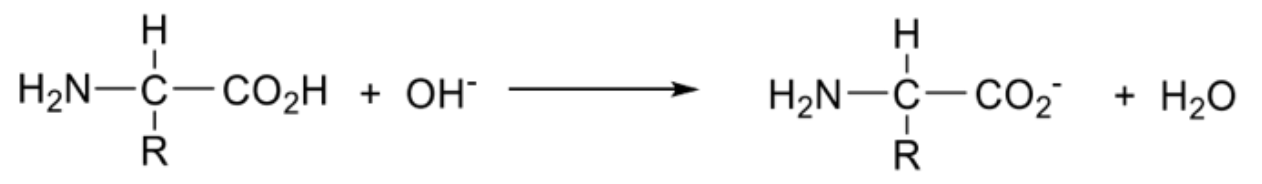
Describe amino acids in a alkaline solution + draw the ion formed
Carboxyl group donates a proton forming an anion