PP3 theory test
1/102
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
103 Terms
Cannabis ingredients
CBD & THC
CBD is responsible for?
therapeutic effect
THC is responsible for?
Adverse effects
Max THC
less than 2% otherwise will be S8
CBD only products schedule?
S4
Ratio cbd vs thc
THC:CBD
Sativex
The only THC-containing good under TGA
Special access Scheme (SAS)
A regulatory pathway that allows patients to access unapproved therapeutic goods, including certain cannabis products, under specific conditions.
How do other TGA-containing products get recieved by patients
Must be SAS approved
When do prescribers need patient-specific authorisation
The patient is <18 yrs
Patient is drug dependent/serious substance-use disorder for drugs addiction in last 5 years
History of psychosis/psychiatric issue
Dose has >40mg of THC per day for oral, or 300mg per day for inhaled/vaporised products.
If no specific patient approval required?
No needed approval to prescribe
Cannabis can be dispensed?
at any pharmacy
Requirements on prescriptions for cannabis
same as S4/8
What does the wholesaler need before supplying cannabis
Copy of TGA approval from Pharmacy, and an approval from Office of Drug Control
Top of the TGA pyramid structure?
Executive
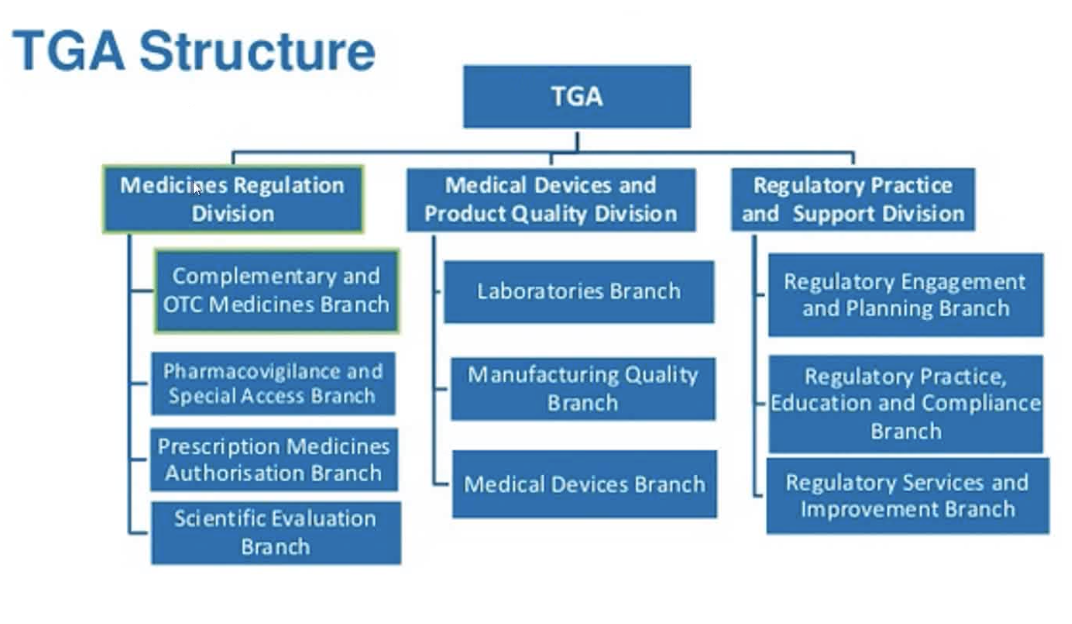
Second-line of the TGA pyramid structure?
3 divisions: Medicines Regulation division, Medical devices and product quality division, and regulatory Practice and Support Division
Role of Medication Regulation division
Monitioring medicines approved for supply in Aus.
Branches under Medicines Regulation (TGA)
OTC and complementary meds, Pharmacovigilance and SAS (Vaccines, unapproved in Australia meds from overseas etc), Prescription Medicines and Authorisation Branch, and the Scientific Evaluation (provides scientific advice about the medicines, guiding on how to regulate them).
Medical Devices and Product Quality Division (TGA)
Regulation of medical devices, quality assurance, compliance and safety monitoring, and pre-market evaluation.
Branches under Medical Devices and Product Quality Division (TGA)
Laboratories Branch (Labs for quality assesment and procedures to make meds), manufacturing quality (ensuring manufacturers meet quality standards), and medical devices (ensuring quality of med devices).
Regulatory Practice and Support Division (TGA)
Provides guidance on regulatory practices, supports compliance, and facilitates communication with stakeholders in the therapeutic goods sector.
Branches of Regulatory Practice and Support Division (TGA)
Regulatory engagement and planning branch (media responses, communications planning, and stakeholder engagement), Regaulatory practice, education and compliance (oversight of compliance with regulations), and guidance and policy branch (developing regulatory guidance and policy frameworks, supports the rest of the TGA structure).
Statutary Advisory Comittees
Statutory advisory committees on Biologicals, on Chemical Scheduling, and on complementary medicines
Statutory advisory committees on Biologicals (ACB)
Advice + recommendation on cell and tissue products
Statutary Advisory Comittee on Chemical Scheduling (ACCS)
Advice + Recommendations on how chemicals should be made available to the public (scheduling of chemicals and not meds?).
Statutary Advisory Comittee on Complementary meds
Advice + recommendations on entering natural meds on the ARTG (refer to other flashcard for this definition). Advice and recommendations
Statutary Advisory Comittee med devices
Advice + recommendations on safety + perfomance of med devices
Statutary Advisory Comittee on meds
Advice + recommendations on entering meds into ARTG
Statutary Advisory Comittee on med schedulling
Advice + recommendations on how to make meds available to public (SUSMP came form this committee :/).
Statutary Advisory Comittee on vaccines
Advice + recommendations n safety, efficacy, quality of vaccines.
Australian Register of Therapeutic Goods (ARTG)
No product/med device that claims a therapeutic effect can be supplied in Aus without being registered on this register.
Name 2 things not regulated by TGA
Food and cosmetics (as they don’t claim therapeutic effect).
What aspects of therapeutic goods does the TGA regulate?
Manufacturing, supplying, imports and and Advertising of TGs.
Bet u didn’t know that the TGA also regulates
Anything used in the manufacture of drugs (other ingredients), and also anything to replace/modify parts of anatomy (surgery-ish prooducts/tools)
Med definition
Used on human or animal to achieve/likely to achieve, a therapeutic goal through pharmacological/metabolic/chemical/etc means.
Medical devices
Defined as being used on ONLY humans and must have a therapeutic BENEFIT (not effect). physical or mechanical effect on the body (including anything from bandages, to MRI scanners, to artificial body parts etc).
How to know if a product is to be considered a food or medicine?
Will depend on the claims it has. If it claims to have therapeautic effect —> medicine.
How to know if a product is to be considered a cosmetic or medicine?
Mainly depends on route of admin, ingredients, and claim on label/ad (If it claims to have therapeautic effect —> med. e.g., moisturiser claims to protect against UV due to sunscreen agent = med.)
Therapeutics Goods Act 1989 applies to who?
Sponsors: anyone who exports/imports meds, or who manufactures or has them imported/manufactured on their behalf. Includes authorities + hospitals and anyone involved in the provision of PBS/RPBS items.
Difference in assessing high-risk and low-risk meds
High risk is assessed for safety, quality and efficacy whereas low-risk is assessed for safety and quality only
Outer packaging TGA code
AUST L/AUST R on outer packaging indicates that it has been approved by TGA and is available for supply.
Meds and medical devices are classifies based on?
their risk level
Other things the TGA regulates
In vitro diagnostic medical devices (IVDs)
blood
blood components
plasma derivatives
tissue and cellular products
tissue and cell based derivatives
sterilants and disinfectants
Whether a medicine is listed on the ARTG or Registered on the ARTG depends on?
Ingredients
Dosage form
The claims of the therapeutic effect of the product
Listed meds on ARTG
lower risk, only have ingredients that are well-known + unschedulled, usually self-selected by consumers, not assessed for efficacy, all sponsers must hold evidence for the listed product’s claims, must have an AUST L on packaging. e.g. herbal/complementary, sunscreens etc.
ALL exported meds are registered or listed?
Listed, imported ones can be either i think?
Registered meds on ARTG
Higher risk, assessed rigourously on safety, eficacy and quality. Must have an AUST R on packaging, can be categroised into high/low risk.
Categories of resitered meds on ARTG
Non-prescription (low-risk) and prescription (high-risk)
Non-prescription registered meds on ARTG
OTC & complementary, No S4, 8 or 9 ingredients. Usually contains ingredients from S2 and 3 and sometimes 5 & 6. Non-prescription meds.
Prescription registered meds on ARTG
May include ingredients of S4, 8 & S9. prescription meds.
Can complementary meds be registered?
YES! Depends on claims in the product. If claims to cure, prevent, alleviate or diagnose —> are a TG.
What meds are excluded from the TG Act 1989
Porducts (usually devices) that unintentionally come under TG definition. They are therefore excluded because it wasn’t on purpose.
What products are exempt?
Products that aren’t on the ARTG (not listed or registered) because of a specific exemption. Usually, these products pose little risk to the public. Must still comply with other TGA standards and regulations, including advertising and labelling.
Products can be exempt from what? (under the TGA)
ARTG or from ARTG AND Manufacturing requirements. These are neither listed or registered under the ARTG.
Can exemptions be revoked?
YES!
What is an unapproved drug?
A drug that is not classified as listed or registered or exempted on the ARTG.
Access to unapproved Drugs
Authorised prescriber, SAS, personal importation scheme, or participating clinical trial.
Must an approved prescriber (to supply unapproved drugs) report to TGA when supplying?
No, but must submit a record of all treated individuals every 6 months.
Category A of patients under the SAS scheme
Patient is seriously ill —> prescriber gives out unapproved drug—> must complete Category Approval to Supply form and submit within 28 days to the TGA nd supplier. TGA does not reply though. Only for medical practitioners. Suppliers cannot supply without form.
Category B of patients under the SAS scheme
Patients that don’t fit category A or C (no history of using the Unapproved Drug). The prescriber fills out the Category B form (or document with same info), preferably with handwriting. Usually prescribers are dental or medical. TGA must reply for approval before supply.
Category C of patients under the SAS scheme
Patients with a history of using the unapproved drug. The prescriber must complete the Category C form and submit it to the TGA. TGA does not reply, and supply can occur after submission. Can be meds, med devices or biologicals. There is a list available for the products and their indications, as well as the prescribers that can apply. The drug being on that list acts as the approval for the supplier to supply.
Personalised Importation scheme
Brought a med with you from overseas, or imported it within Australia from overseas. Must be for you or immediate family members. Order as much as you want of the drug within a 12-month period at max dose recommended by manufacturer, but cannot exceed 15 month supply. Can’t be an illegal drug, or have human/animal remains. If it’s a S4 or S8, must have a prescription from authorised prescriber. Want more supply than 15 month? Doctor must apply from permission under SAS.
Manufacturing (TGA definition)
Producing goods or being involved in the process of producing goods
Good manufacturing Practice guidelines
Describe the principles and practices that are necessary to follow in order to provide assurance that each batch of a therapeutic good is safe, reliable, and of consistent high quality. Manufacturers must hold a licence and also their factory must meet GMP guidleines via audits
GMP guidelines not met?
License can be revoked, TGA officer can walk in anytime. Refusal to give samples for testing = fine.
compounded medicines must meet GMP guidelines?
No, they are exempt from requiring manufacturing licences. But encouraged to meet guidelines anway. But compounded goods for sale on large scale = pharmacist must apply to TGA for license.
Advertising restriction
OTC = allowed most of the time.
Prescription and some pharmacist-only meds = prohibited to make ads abt them
Advertising prescription-only
Not allowed to consumers
Allowed to healthcare professionals under self-regulatory scheme operated by Medicines Australia
Also must meet the Competition and consumers Act
Schedule 3
Some may be advertised to consumers, not all.
Non-prescription ads
Ok mostly. Regulated by both self-regulatory and co-regulatory arrangements.
Ads that require approval
TV, Cinema, Radio, outdoors (billboards, etc.), and print media (magazines, newsapapers).
Medical devices
A similar regulation to non-prescription; however, no prior approval needed.
AD regulations
Must be realistic of effectiveness, not unwarranted, and have no incorrect or unbalanced statements.
Mustnt lead consumers to self-diagnosing or inappropriate treatment
No taking advantage of lack of knowledge of consumers.
Musnt encourage inappropriate/excessive use.
Must not claim it has no side effects
No ads towards minors
No personal incentives in the ad
Cannot say it is recommended by an authoritative body (like TGA/governemnt etc).
Who do the advertising regulations not apply to?
These regualtions do not apply when advertising to most healthcare professionals
Don’t apply to non-human use meds.
What must an AD contain
The drug name, the indication, the dose form, and trade name.
Also USE ONLY AS DIRECTED SEE DOCTOR IF SYMPTOMS PERSIST
ASK YOUR PHARMACIST
ALWAYS READ THE LABEL
No ads for:
unlisted, unregistered, and non-exempted (unapproved drugs).
Recalling is responsibility of?
Sponspor that caused the fault to recall.
Recalling classifications
Class 1 - Life-threatening or serious risk to health
Class 2 - can cause defect or mistreatment, not as risky as Class 1
Class 3 – no risk to health, but recalled for other reasons.
stages of recall
notify the coordinator of crisi management
collect info to conduct assessment
assess the recall
recall
notify federal minister of Consumer Affairs
conclude recall and report
follow-up actions
Bioequivalent
same mechnaism of action + absorption.
Generics
Bioequivalent to the brand product, it may or may not be more expensive. Made because the brand product is not protected by patent. Allows for brand substitution.
Generic vs Brand
Same absorption and MOA, same strength and quantity, but different excipients. Can be switched to/from generic by a pharmacist or prescriber.
Substitutes in the Schedules of Pharmaceutical Benefits,
Flagged with an "a". These brands can be interchanged without any clinical effect. Anything flagged like this can be substituted by the pharmacist.
Biosimiliar
Highly similiar biological drug to already existing biological drug. Not identicle.
Interchangeability
Can only be changed by the prescriber. Drugs that are interchangeable (often of the same class).
Marked “b” on schedule of pharmaceutical benefits
We don’t know if they’re bioequivalent to the brands marked "a".
What if there is no brand on prescription?
Pick up anything flagged “a,” that is biosimiliar or equivalent. No need to notify prescriber.
The patient wants a brand when “brand substitution is not permitted” on the prescription.
No option for patient. Discuss with prescriber.
When is substitution not allowed?
When brands are not bioequivalent
Medicines with narrow therapeutic index (e.g. epeliptic meds). Seek prescriber advice first
Risk of confusion anxiety/harm
Negative impact on adherence
Allergy/intolerance/concern to excipients
Different device used to admin med
Why are biosimilars not identical?
Bcause they are made from biological cells —> making an identical is impossible, as they are very complex
Drug-Related Problem (DRP)
A medication-related issue that can potentially harm a patient or prevent optimal pharmacotherapy. This includes issues like adverse drug reactions, drug interactions, or inappropriate medication use.
Clincial Interventions
It involves identifying current/potential DRPs and changing patient drug therapy.
SOAP
Subjective info, objective info, assess the info collected (S & O) and plan
Clinical problem solving framework
Collect info, Identify problem (DRP), and then Resolve DRPs.
Problem-solving
Nature of problem, its consequence, and plan to manage it
Identifying DRPs
Identify the drug
Identify indications for each med
Dose/s prescribed
Drug-drug Interaction
Drug-disease interactions
8 categroies of DRPs
Drug selection
Over/underuse
Compliance
Undertreated
Monitoring
Education
Not classifiable
Adverse effects/toxicity
My health record
Record of medical history, medications, Procedures the patient underwent available to Health professionals 24/7. All Australians have one/an individual healthcare identifier.
Safe to assume MHRa is thorough?
Nope. The patient has control over what you see. They may have deleted some information or requested restrictions. Never safe to assume that.
Can a healthcare professional access MHR without the patient being present?
Yes!