Rates of Ligand Substitution
1/5
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
6 Terms
What are inert complexes
Complexes that are thermodynamically unstable but survive for long periods (at least a minute) are called inert
What are labile complexes
Complexes that undergo more rapid equilibration are called labile
Describe how lower activation energy increases rate of reaction
stabilisation of the transition state (TS)
destabilisation of the ground state
How does oxidation state affect rate
Oxidation state of the metal will affect rate.
Rates are slower for ions with higher charge
Due to the fact that
metal ligand bonds are harder to break
there is less space for attack by the incoming ligand
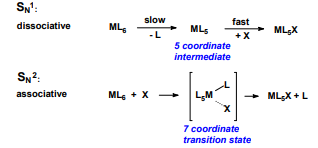
Give the dissociative and associative reactions for metal ligands
Detail how distribution of 3d electrons effects rate
d0 → d3 : rates drop
Metal ion nuclear charge increases. SN1 becomes harder. Since the electrons go into the orbitals pointing in between the ligands, Zeff felt by the ligands increases and the M-L bonds get stronger.
SN2 gets harder as well. The electrons in dxy, dxz, dyz protect the metal from an attack by an incoming ligand.
d3 → d5 : rates rise
SN1 easier, The 4th and 5th electron go into the orbitals pointing at the ligands. This makes the M-L bonds weaker.
SN2 easier, gets easier as well since the electrons don’t protect from attack.
d5 → d8 : rates drop
The electrons go into the orbitals lying in between the ligands
d8 → d10 : rates rise
The electrons go into the orbitals pointing at the ligands