Mine- Chemistry 2nd Semester Final Study Guide (Morgan)
1/119
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
120 Terms
Chapter 8:
Molecular Shapes
Define: valance electrons
electron in the outermost energy level of an atom; for most atoms, it is available to be gained, lost, or shared in the formation of chemical bonds
Define: bond angle
geometric angle between two adjacent bonds in a molecule
Define: VSEPR theory
theory that explains that in small molecules, valance electrons are arranged as far apart from each other as possible; can be used to predict the shapes of molecules; known as VSEPR theory
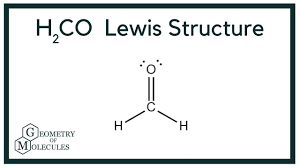
Draw the lewis structure and determine the molecular shape and polarity for the following molecules:
a. H2CO
Shape: trigonal planar
Polarity: polar
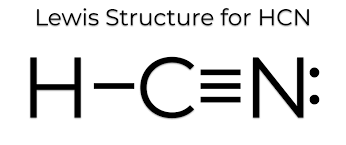
Draw the lewis structure and determine the molecular shape and polarity for the following molecules:
b. HCN
Shape: linear
Polarity: polar
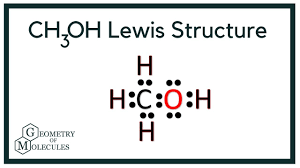
Draw the lewis structure and determine the molecular shape and polarity for the following molecules:
c. CH3OH
Shape: tetrahedral
Polarity: polar
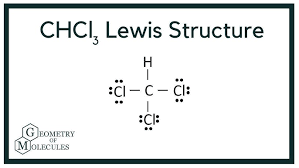
Draw the lewis structure and determine the molecular shape and polarity for the following molecules:
d. CHCL3
Shape: tetrahedral
Polarity: polar
Draw the lewis structure and determine the molecular shape and polarity for the following molecules:
e. NF3
Shape: trigonal pyramidal
Polarity: polar
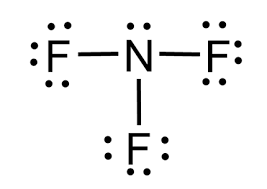
When a compound is polar, what end would be the partial negative end of the molecule?
The atom or region with the higher electronegativity. Elections shared unevenly, spending more time near electronegative atom, creating a electron density that results in a partial negative charge.
Chapter 9:
Chemical reactions- writing and balancing reactions
Define: chemical reactions
process in which one or more substances are converted into new substances with different physical and chemical properties
Define: reactant
substance that enters into a chemical reaction
Define: product
substance formed during a chemical reaction
Define: coefficient
whole number that precedes a reactant or product symbol or formula in a chemical equation and indicated the relative number of representative particles involved in a reaction
Define: synthesis reaction
two or more reactants come together to form a single product. in general, A + B → AB. Example: 2Na + Cl2 → 2NaCl
Define: decomposition reaction
chemical reaction in which a single complex compound is broken down into tow or more products; general form is AB → A + B
Define: single replacement reaction
chemical reaction in which an uncombined element replaces an element that is part of a compound; general form is A + BX → AX + B
Define: double replacement reaction
chemical reaction in which atoms or ions form two different compounds replace each other; general form is AX + BY → AY + BX
Define: combustion reaction
occurs when reactants burn, must have O2 as a reactant & a fuel that has carbon & hydrogen in it. products always CO2 + H2O. example = CH4 + 2O2 → CO2 + + 2H2O
List the seven diatomics
1- Hydrogen
2- Nitrogen
3- Oxygen
4- Florine
5- Chloride
6- Bromine
7- Iodine
what are the species called before and after chemical reactions?
The species before the reaction are reactants, and the species formed after the reaction are called products
what are coefficients and why are they used?
The large numbers placed before a chemical formula and an equation. They indicate the number of molecules or moles of the substance involved.
describe steps you would follow to balance a chemical equation
The process involves identifying elements, counting atoms, adjusting coefficient, and verifying the balance
write a balanced equation for the following reaction:
lithium metal reacts with water to produce lithium hydroxide and hydrogen gas
2Li + 2H2O → 2LiOH + H2
explain how a balanced equation verifies the law of conservation of matter
By ensuring the number of atoms for each element is identical on both the reactive and product sides
identify the five different types of chemical reactions of an example of each
1- synthesis: 2Na + Cl2 → 2NaCl
2- decomposition: 2HgO→ 2Hg + O2
3- single replacement: Pb + FeSO4 → PbSO4+ Fe
4- double replacement: AgNO3 + NaCl → AgCl + NaNO3
5- combustion: CH4 + 2O2 → CO2 + 2H2O
predict the products of the following reaction and balanced the equation:
Calcium chloride plus silver iodine yields
CaCl2 + 2AgI → CaI2 + 2AgCl
Key Details:
Products: Calcium Iodide and Silver Chloride
Reaction Type: Double Replacement (precipitation reaction)
Balancing: Two moles of silver iodide are needed to produce two moles of silver chloride
Chapter 10:
The Mole
define: mole
quantity of a substance that has a mass in grams numerically equal to its formula mass, equal to 6.02×10 to the 23rd representative particles
Define: formula (molar) mass
sum of the atomic masses of all the atoms in a compound as represented in chemical formula, measured in atomic mass units
Define: Avogadro’s number
Number of representative particles in one mole; equal to 6.02×10 to 23
Define: present composition
Mass of each element in the compound relative to the total mass of the compound, found by dividing the mass of the element by the mass of the compound and multiplying the quotient by 100%
Define: empirical formula
Chemical formula that gives the simplest whole number ratio of atoms of elements in a compound
explain the difference between formula units, molecules, and particles
Particles is a general term for any small unit of matter (atom, ions or molecules) molecules refer to covalently bonded, nonmetals and formula units represent the lowest whole number ratio of ions in an ionic compound
What is molar mass? How’s it calculated? What are their units?
Molar mass is the mass in grams of one mole of a substance. You find it by identifying the element finding the atomic mass multiplied by a subscript and some of the totals the units are grams per mole
how many moles are in 67.6 g of water
There are 3.75 moles

find the empirical formula for a compound which contains 6.5 g potassium, 5.9 g chloride, and 8.0 g oxygen
The empirical formula of the compound is KClO3

A compound has a mass of 0.2370 g. 0.09480 g is carbon, 0.1264 g is oxygen and 0.0158 g is hydrogen. What is the percent composition of a compound?
The compound is composed of 40.00\% carbon, 53.33% oxygen, and 6.67% hydrogem

what is a mole and how is it used in chemistry?
The SI unit for the amount of a substance, 6.0 2214076×10 23 (atoms, molecules, or ions). It functions as a bridge between microscopic particles and macroscopic measurements allowing chemist to weigh out specific quantities of atoms or molecules using grams.
what is Avogadro’s number and what is it used for in chemistry?
A fundamental constant known as 6.02214076×10 to 23rd atoms, molecules or ions per mole. It is used to convert moles to particles, converting particles to moles, stoichometry, and molar mass calculations
what is the difference between an empirical formula and a molecular formula?
Empirical formula show the simplest whole number ratio of atoms in a compound, well molecular formula show the actual number of atoms in a molecule
how many molecules are in 1.5 moles of CH4
The total number of molecules is 9.033 times 10^{23}

Chapter 11:
Stoichiometry- using the mole in reactions
Define: stoichiometry
Study of quantitative relationships that can be derived from chemical formulas and equations
Define: limiting reactant
Reactant that is completely used up in a chemical reaction and that therefore determines the maximum amount of product that can be formed
Define: excess reactant
The substance in a chemical reaction that remains unreacted after the limiting reactant is completely consumed
Define: actual yield
Would be what you actually produce in lab
Define: theoretical (expected) yield
what the math says you should get
Define: percent yield
Actual yield divided by expected yield times 100%
according to the equation, how many moles of FECL3 would be produced from six moles of CL2?
The number of moles of FeCl3 produced is 4 moles.

according to the equation, what volume of CO2 would be produced when 128.0 g of C6H6 reacts at STP
The volume of CO2 produced is 220.2 L

identify the limiting reactant when 10.0 g of water reacts with 4.5 g of NA to produce NAOH and H2
The limiting reactant is Sodium (Na) because it provides fewer moles than are required to react with the total amount of water available.

5.00 g of copper is placed in a solution of silver (I) nitrate containing excess AgNO3. The silver metal produce has a mass of 15.2 g what is a percent yield for this reaction?
Rounding to three significant figures based on the provided data, the final result is 89.6%

describe the difference between the terms expected yield, actual yield percent yield
expected yield is the calculated maximum product, actual yield is what is measured in the lab and percent yield is the ratio between between them, indicating efficiency
what is meant by stoichiometric proportions (mole to mole ratio)?
The exact, just right ratio of reactants required to completely consume each other, producing a specific amount of products based on a balanced chemical equation
explain why all the available reactants might not be converted to products in a chemical reaction
most reactants reach a dynamic equilibrium where formed and reverse reactions happen at the same time leaving a mixture of both
Chapter 12:
Thermochemistry- heat in chemical reactions
Define: thermochemistry
Study of changes in heat energy that accompany, chemical reactions and physical changes
Define: exothermic reaction
process that releases energy
Define: endothermic reaction
process that absorbs heat
Define: calorimetry
measurement of the amount of heat released or absorbed during a chemical reaction
Define: heat capacity
amount of heat energy needed to raise a temp of a given sample a matter by 1°C
Define: specific heat
amount of heat energy required to raise the temperature of 1 g of a substance by 1°C
understand the difference between Endo/exothermic reactions
Endothermic reactions absorb heat from their surroundings, causing the temp to drop. An exothermic reaction reactions releases heat into their surroundings, causing the temperature to raise.
how much heat will be released when 6.44 g of silver reacts with excess estrogen according to the following reaction
The amount of heat released is 79.5 kJ or 79.4 J depending on rounding.

what does Hesse’s law state?
The total empathy change for a chemical reaction is the same regardless of whether the reaction occurs in one step or several steps
when a 13.7 g sample of solid Pb(NO3)2 dissolves in 85.0 g of water in a calimoter the temperature drops from 23.4°C to 19.7°C calculate the change of empathy for the solution process
Answer: +37 kJ/mol

what is the difference between heat and temperature?
Heat is a transfer of thermal energy between systems due to temp difference measured in joules, and the temp is the measure of the average kinetic energy of particles and a substance measured in kelvin or celsius
what is meant by standard conditions of a reaction?
A specific set of environmental parameters used as a universal reference point to ensure consistency when measuring or comparing thermodynamic data like entropy entropy and Gibbs free energy
how does a chemical reaction produce heat?
When the energy released forms new bonds, and the product products is greater than the energy required to break the bonds in reactants
what is the difference between heat capacity and specific heat?
Heat capacity is a total energy required to raise a temp of an entire object by 1°C flow specific heat is energy required to raise 1 g of a substance by 1°C he capacity depend depends on the mass and specific heat is independent of the mass
how does the food you eat provide for the energy needs of your body? What happens if you eat more food than your body needs for its daily energy requirements?
Your body generates energy by digesting food into basic nutrients like glucose which cells then combine with oxygen to create ATP to power all the bodily functions, the body stores is excess energy, first as glycogen in the muscle and liven then permanently as fat in adipose tissue leading to weight gain
explain how heat/cold packs are useful in terms of energy
manipulating chemical energy to create thermal energy, heat, or observed thermal energy, cold through chemical reactions. heat packs, release energy to surroundings (exothermic) and cold packs absorb heat from surrounds (endothermic)
Chapter 13:
Gases- KMT
Define: elastic
ability of a substance or object readily spring back to his original size or shape after being squeezed, hit, or subjected to similar disturbances
Define: manometer
Instrument used to measure the pressure of a gas in a close container
Define: standard temperature and pressure
Standard temperature and pressure, designated as 0°C or 273 K and one atmosphere
Define: absolute zero
theoretically lowest temperature that can be reached, -273.15°C or 0K
Define: ideal gas
theoretical gas described perfectly by the kinetic molecular theory of gases
what is the significance of an absolute temperature in terms of KMT?
it is directly proportional to the average kinetic energy of the gas particles. Absolute temperature provides a direct measure of energy and motion within a system.
what are two false assumptions of KMT?
gas particles have negligible volume and there are no intermolecular forces between them
explain how a gas exerts pressure on a container in terms of KMT
gas pressure is caused by constant, random, and rapid movement of gas particles colliding with an exerting force on the inner walls of their container
why do your ears pop when you ride in a plane?
The rapid change in altitude during takeoff and landing, creates a pressure difference between your middle ear and the airplanes cabin
what is Boyles, Charles, Gay-Lussacs, Avogadro, combined, Daltons, and the ideal gas
they explain how pressure, volume, temperature and amount of gas affect each other. Boils relates pressure and volume, Charles and Gay-Lusscas involve temp, change, avocado, volume, and moles, Dalton’s adds gas pressure together and the ideal gas law combines all the major gas relationships into one equation
A sample of nitrogen at 20°C was compressed from 300 mL to 0.360 mL and it’s new pressure was found to be 400.0 PA what’s it original pressure in kPa?
Answer: 0.00048 kPa

5.00 L of a gas is collected at 30°C and then allowed to expand to 20.0 L what must the temperature be?
1212.6 K or 939.45 degrees Celsius

if I have 17 L of a gas at a temperature of 67°C and a pressure of 88.89 ATM, what will the pressure of the gas if I raise the temperature to 94 degrees Celsius increases the volume to 12 L
Answer: 135.92 ATM

if I haven’t unknown quantity of oxygen gas at a pressure of 0.5 ATM, a value of 25 L, and a temperature of 300 K, how many grams of oxygen gas do I have?
Answer: 16.25 g

Chapter 14:
Liquids and Solids- KMT applies to condensed states
Define: intermolecular force
relatively weak force of attraction that exists between neighboring molecules
Define: hydrogen bond
strong Temecula force between the hydrogen atom of one molecule and a highly electronegative atom of another molecule, such as fluorine, oxygen or nitrogen
Define: induced dipole
dipole created by the presence of a neighboring dipole
Define: dipole-dipole force
intermolecular force of attraction between neighboring permanent dipoles that is created when the dipole lineup so that the positive and negative ends are close to each other
Define: viscosity
resistance to motion that exists between the molecules of a liquid when they move past each other
Define: amorphous solid
solid in which the arrangement of the representative particles, lack a regular, repeating pattern
Define: sublimation
conversion of a solid directly into a gas, without first becoming a liquid
What types of intermolecular forces are in the following compounds:
a. CO2
London dispersion forces
What types of intermolecular forces are in the following compounds:
b. CH3OH
hydrogen bonding, dipole-dipole, and london dispersion forces
What types of intermolecular forces are in the following compounds:
c. C6H12
london dispersion forces