CHEM 260 + 360 UTK Reactions Quizlet
1/47
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
48 Terms
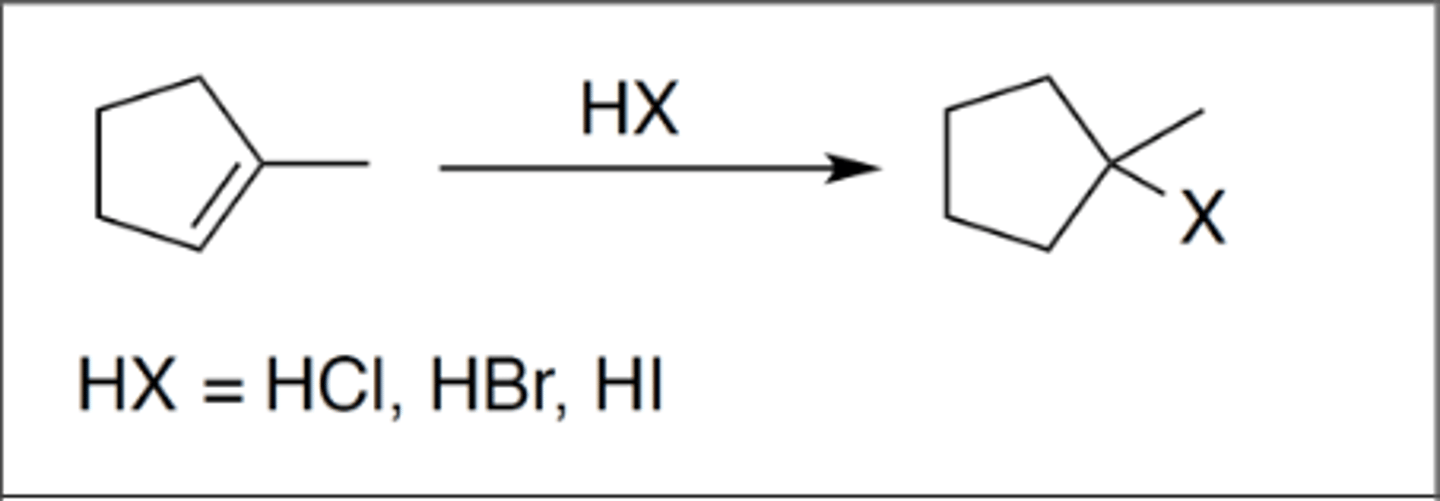
Hydrohalogenation
Carbocation: yes
Regiochemistry: mark
Stereoselectivity: none
Reagent(s): HX = HCl, HBr, HI
- X added to more stable CC+ from alkene
- Possible 1,2 shift
*KNOW MECHANISM*
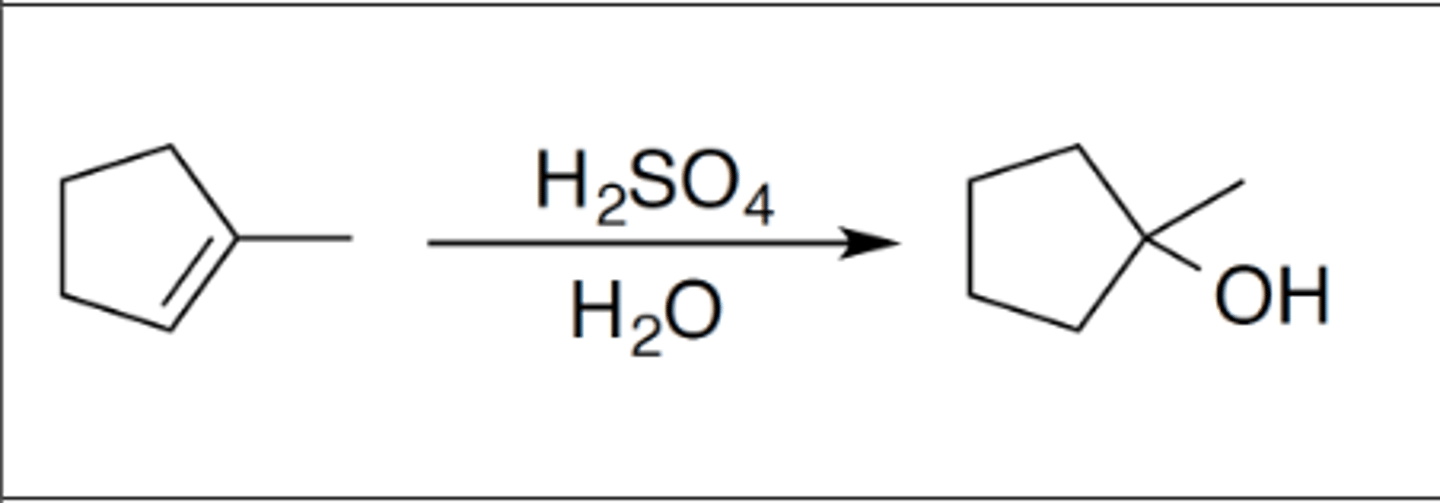
Hydration
Carbocation: yes
Regiochemistry: mark
Stereoselectivity: none
Reagent(s): strong acid (H2SO4) over H2O
- OH added to more stable CC+ of alkene
*KNOW MECHANISM*
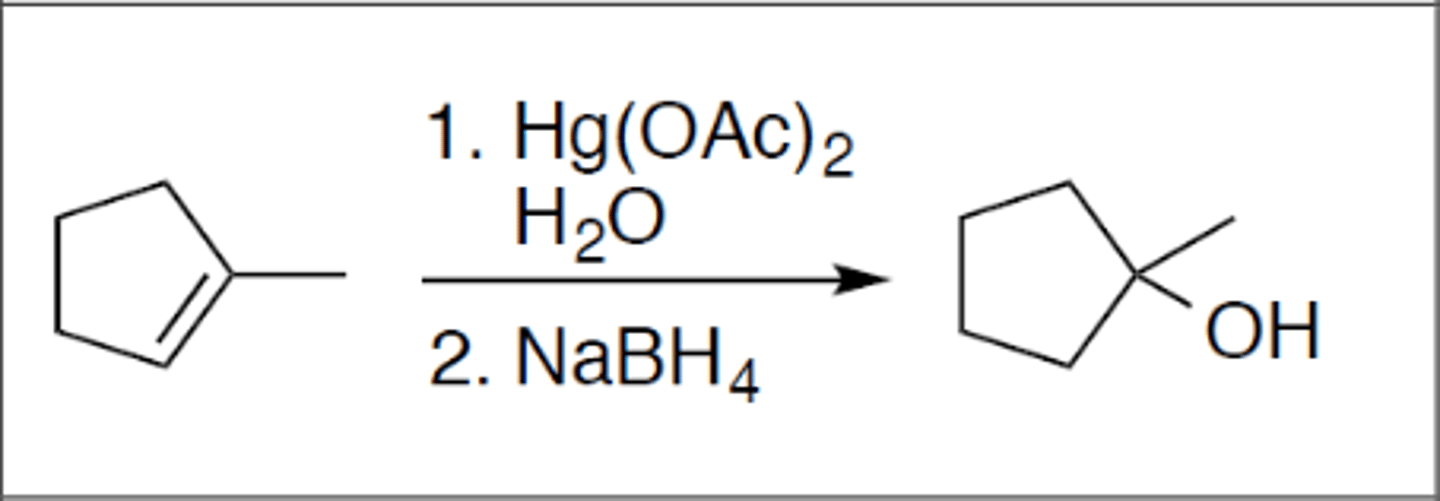
Oxymercuration
Carbocation: no
Regiochemistry: mark
Stereoselectivity: none
Reagent(s): Hg(OAc)2 and H2O over NaBH
- OH added to more stable CC+ from alkene
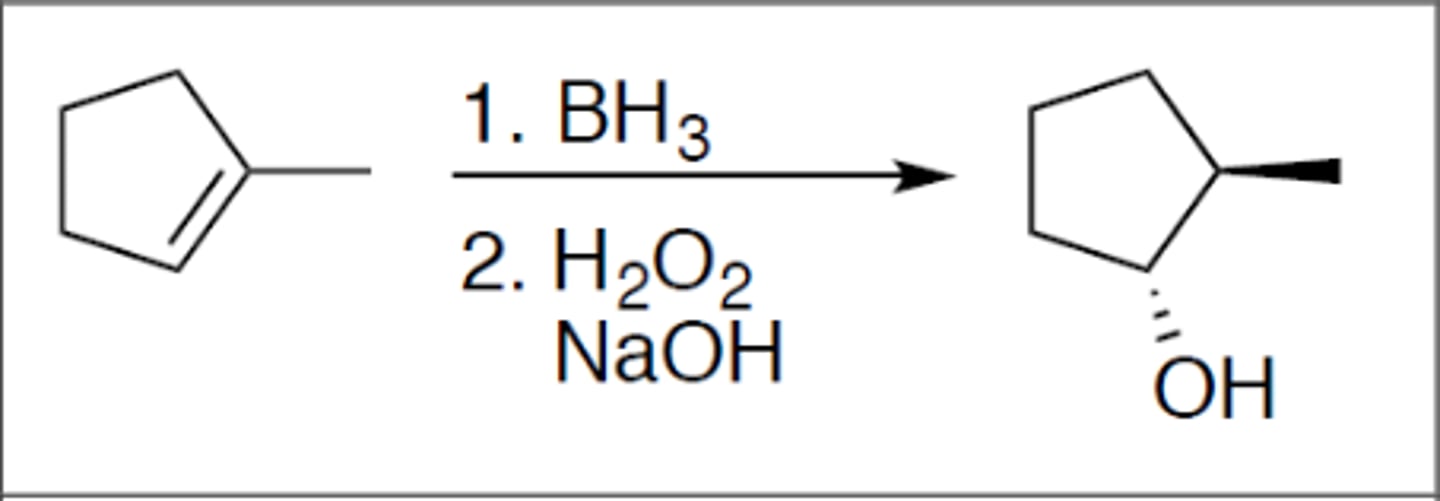
Hydroboration
Carbocation: no
Regiochemistry: anti-mark
Stereoselectivity: syn
Reagent(s): 1. BH3 2. H2O2 and NaOH
- OH added to less stable CC+ from alkene
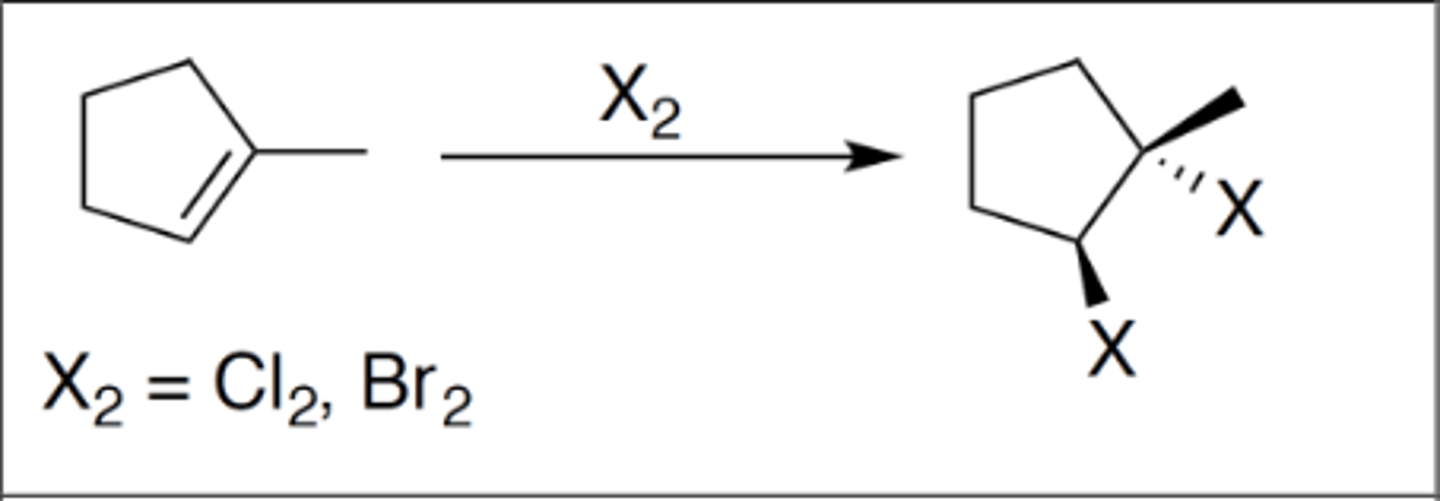
Halogenation
Carbocation: no
Regiochemistry: N/A
Stereoselectivity: anti
Reagent(s): X2 = Cl2 or Br2
- Anti addition of X to either side of alkene
KNOW MECHANISM
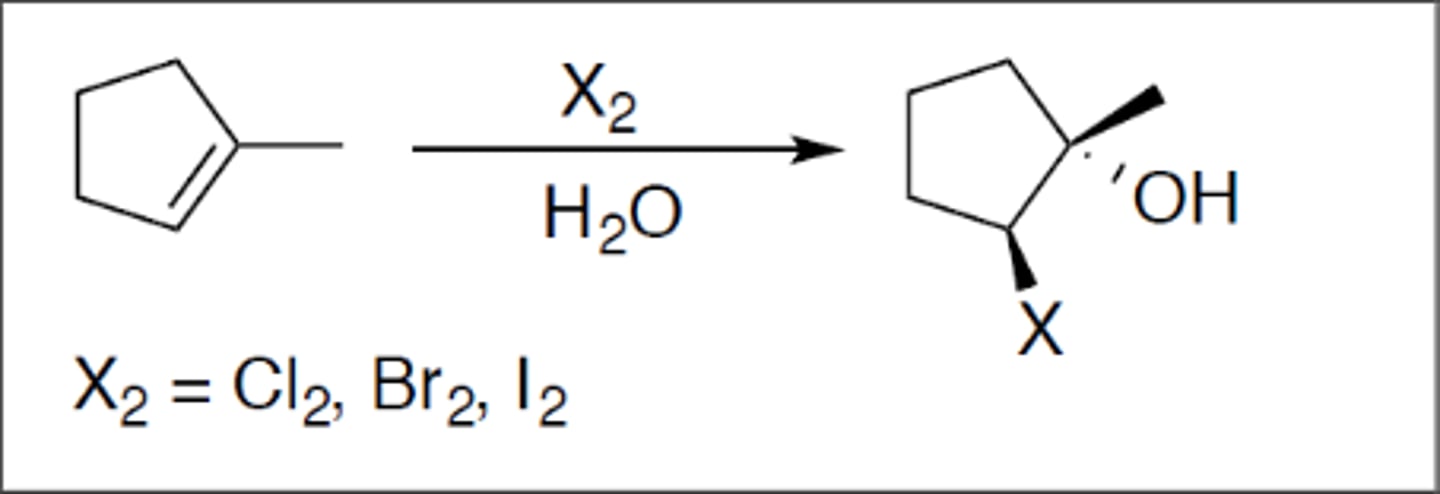
Halohydrin
Carbocation: no
Regiochemistry: mark (-OH)
Stereoselectivity: anti
Reagent(s): X2 = Cl2 or Br2 over H2O
- anti addition of OH to more stable CC+ of alkene
KNOW MECHANISM
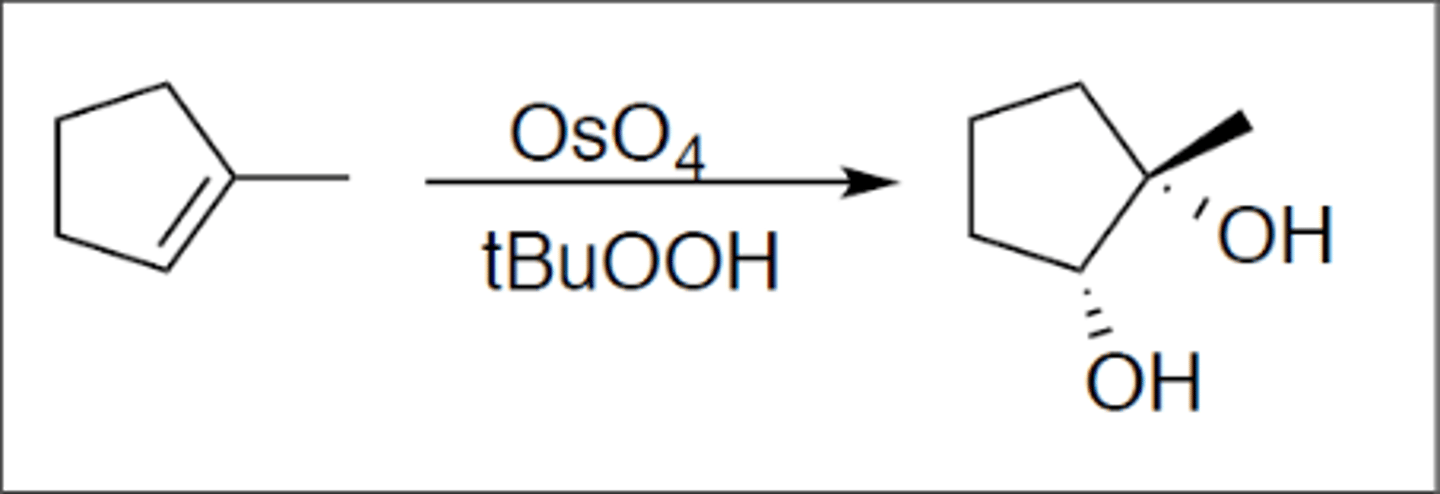
Dihydroxylation
Carbocation: no
Regiochemistry: N/A
Stereoselectivity: syn
Reagent(s): OsO4 over tBuOOH
- syn addition of two OH on either side of alkene
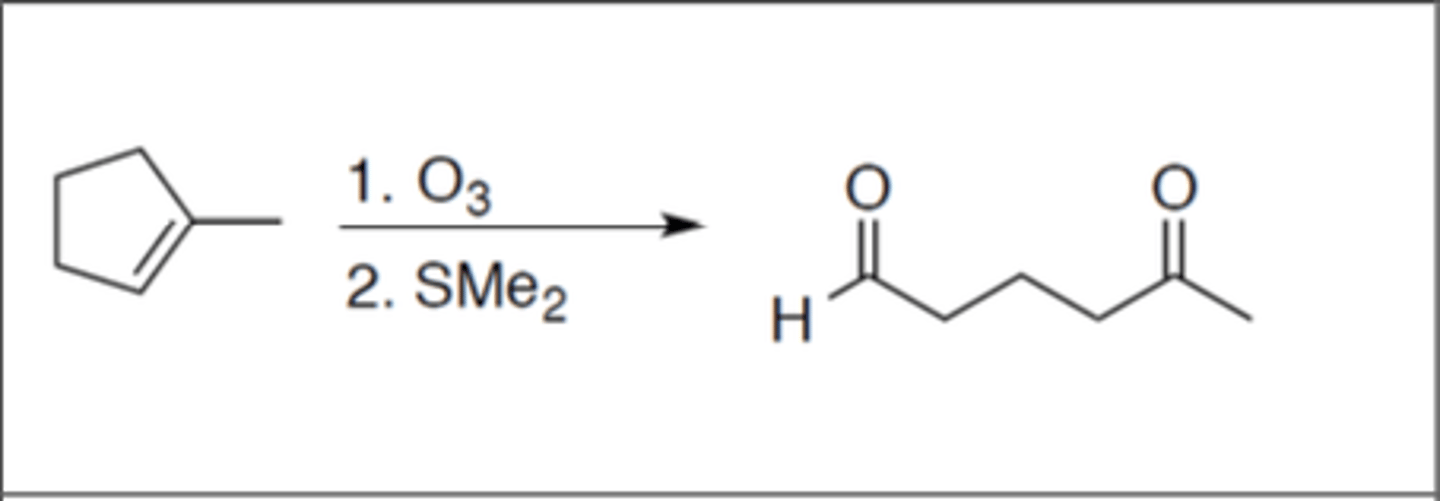
Ozonolysis
Carbocation: no
Regiochemistry: N/A
Stereoselectivity: N/A
Reagent(s): 1. O3 2. SMe2
- addition of =O on both carbons the alkene was attached to
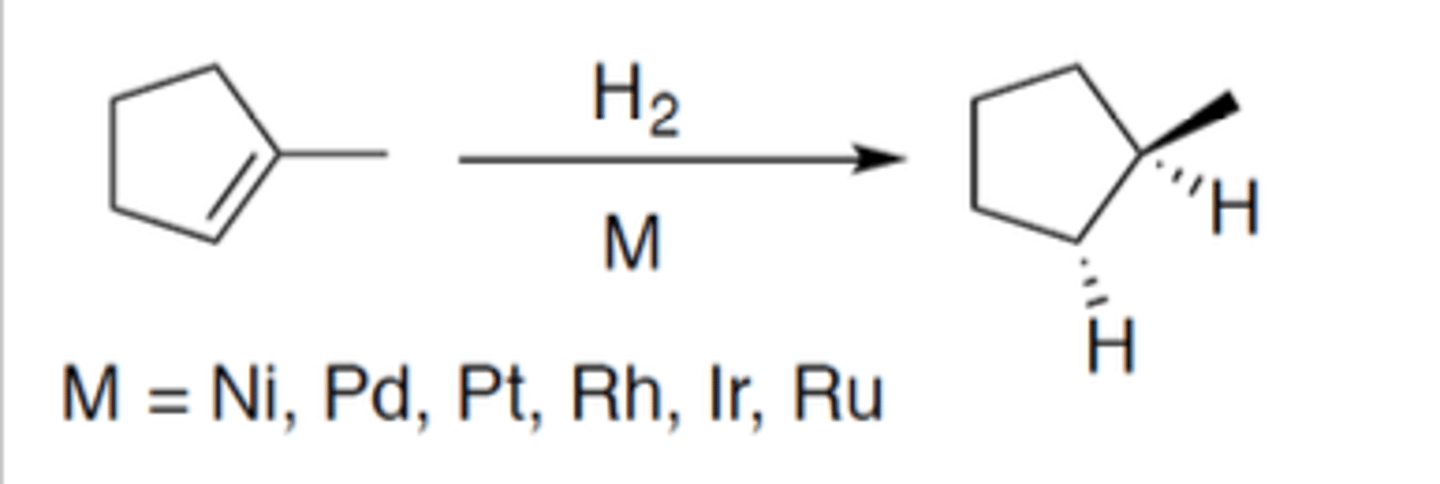
Hydrogenation
Carbocation: no
Regiochemistry: N/A
Stereoselectivity: syn
Reagent(s): H2 over M: M = Ni, Pd, Pt, Rh, Ir, Ru
- syn addition of two H on either side of alkene
SN2
Nucleophile replaces leaving group
Regiochemistry: anti with tertiary CC+
- Rate = k[substrate][nucleophile]
KNOW MECHANISM
![<p>Nucleophile replaces leaving group</p><p>Regiochemistry: anti with tertiary CC+</p><p>- Rate = k[substrate][nucleophile]</p><p>KNOW MECHANISM</p>](https://knowt-user-attachments.s3.amazonaws.com/b80f91dd-18bc-4689-ad50-236faa38a6b6.png)
SN1
Nucleophile replaces leaving group
Regiochemistry: no
- Rate = k[substrate]
KNOW MECHANISM
![<p>Nucleophile replaces leaving group</p><p>Regiochemistry: no</p><p>- Rate = k[substrate]</p><p>KNOW MECHANISM</p>](https://knowt-user-attachments.s3.amazonaws.com/1364f8e8-1b48-406b-9d71-bcafe677b7ff.jpg)
E2
Nucleophile acts as base to remove H and form alkene
- Big bases don't follow Zaitsev's rule
- Rate = k[substrate][nucleophile]
![<p>Nucleophile acts as base to remove H and form alkene</p><p>- Big bases don't follow Zaitsev's rule</p><p>- Rate = k[substrate][nucleophile]</p>](https://knowt-user-attachments.s3.amazonaws.com/62d2d5ce-aa70-4c88-9633-12b2104f8266.png)
E1
Nucleophile acts as a base to remove H and form alkene
- Small bases usually follow Zaitsev's rule.Keep in mind situations where alkene has to form other way because LG and hydrogen must be anti/axial
- Rate = k[substrate]
![<p>Nucleophile acts as a base to remove H and form alkene</p><p>- Small bases usually follow Zaitsev's rule.Keep in mind situations where alkene has to form other way because LG and hydrogen must be anti/axial</p><p>- Rate = k[substrate]</p>](https://knowt-user-attachments.s3.amazonaws.com/c89d766c-23a5-406c-bdd3-71c36d24b3ee.jpg)
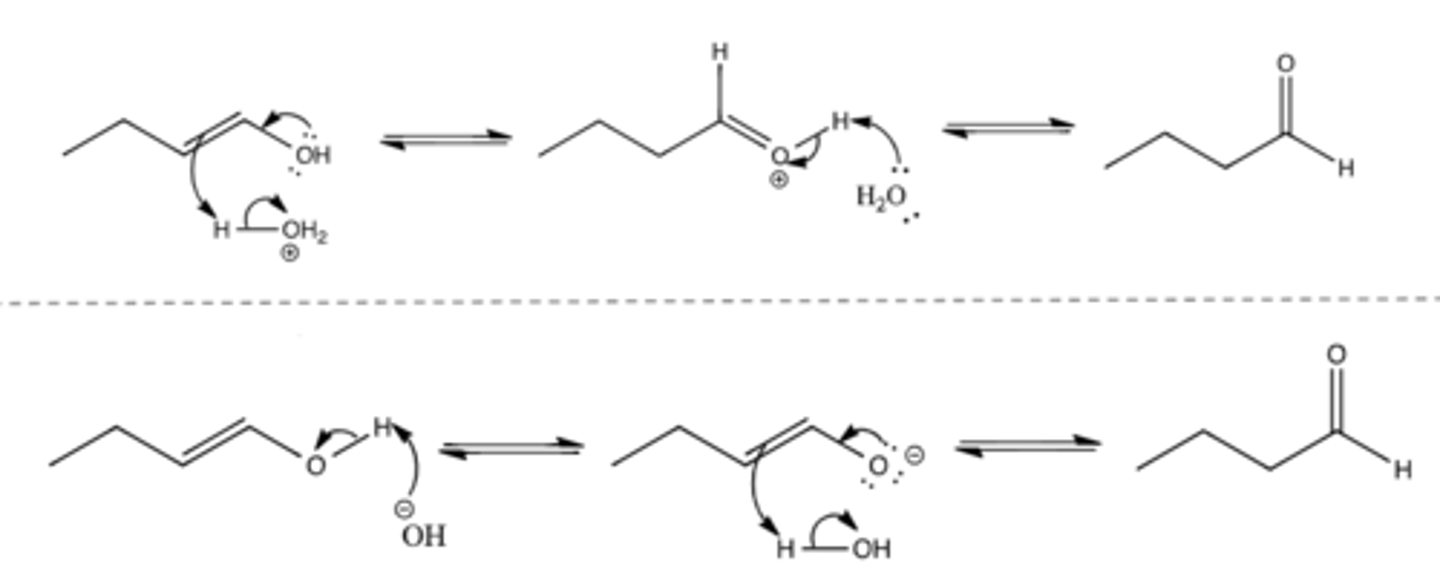
Keto-enol tautomerization (not a reaction)
Enol groups (alkene with OH attached) are unstable and tautomerize into keto/aldehyde groups
In acidic conditions:
- Arrow from O of OH to C-OH bond, arrow from alkene to H of H-OH2^+, arrow from H-OH2^+ bond to O of OH2
- Arrow from O of H2O to H of C=O-H, arrow from attached O-H bond to =O
In basic conditions:
- Arrow from O of lone OH^- to H of attached OH, arrow from attached O-H bond to O of attached OH
- Arrow from O^- to =C-O bond, arrow from alkene to H of H-OH, arrow from H-OH bond to O of H-OH
Can go forwards or reverse (draw double sided arrow)
*KNOW MECHANISM*
Deprotonation
Carbocation: no
Regiochemistry: N/A
Stereoselectivity: none
Reagent(s): Bases = C, N, H ANIONS only

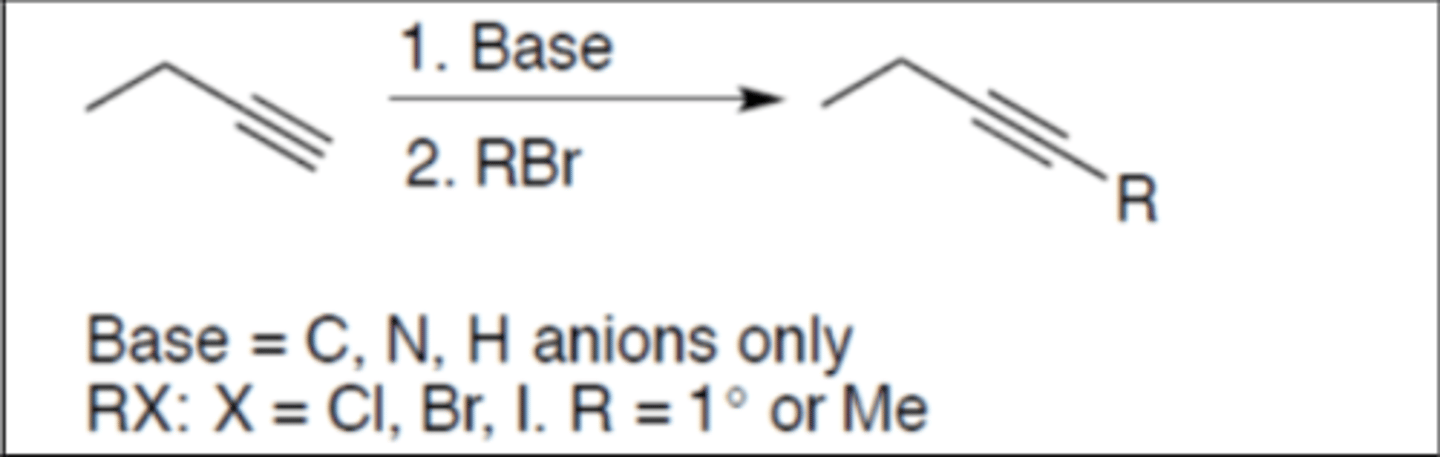
Alkylation
Carbocation: no
Regiochemistry: N/A
Stereoselectivity: none
Reagent(s): 1. Base = C, N, H ANIONS only 2. RX: R = primary or methyl and X = Br, Cl, I
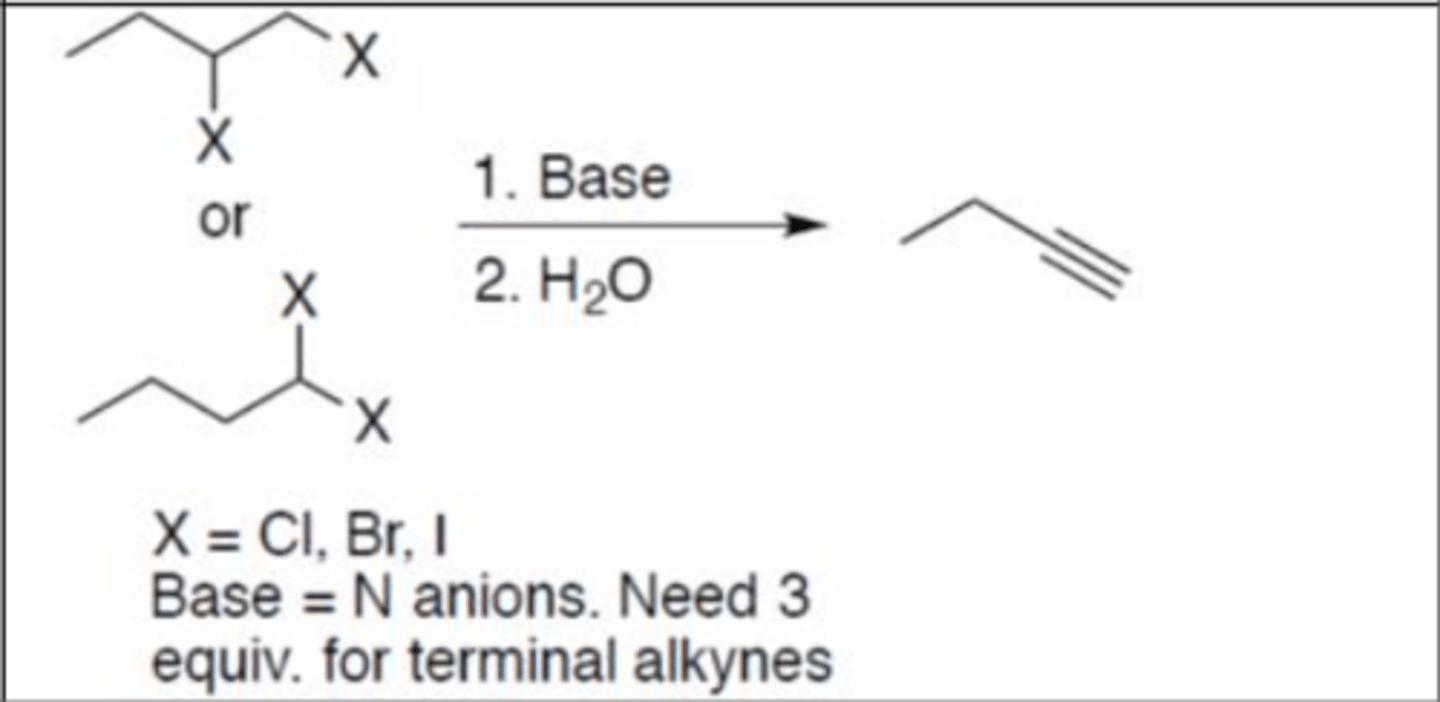
Dehydrohalogenation
Carbocation: no
Regiochemistry: N/A
Stereoselectivity: none
Reagent(s): 1. Base = N anions only (3 equiv. for terminal alkynes; 2 equiv. for internal) 2. H3O+ (no 2nd step for 2 equiv.)
- Adds an alkyne to carbon(s) that the 2 Xs are attached to
- Turns alkane with X atoms into alkyne
Halogenation of alkyne (1 equivalent)
Carbocation: yes (no rearrangement)
Regiochemistry: mark
Stereoselectivity: anti
Reagent(s): 1 equivalent Cl2 or Br2
- Adds 1 X on both the alkyne carbons
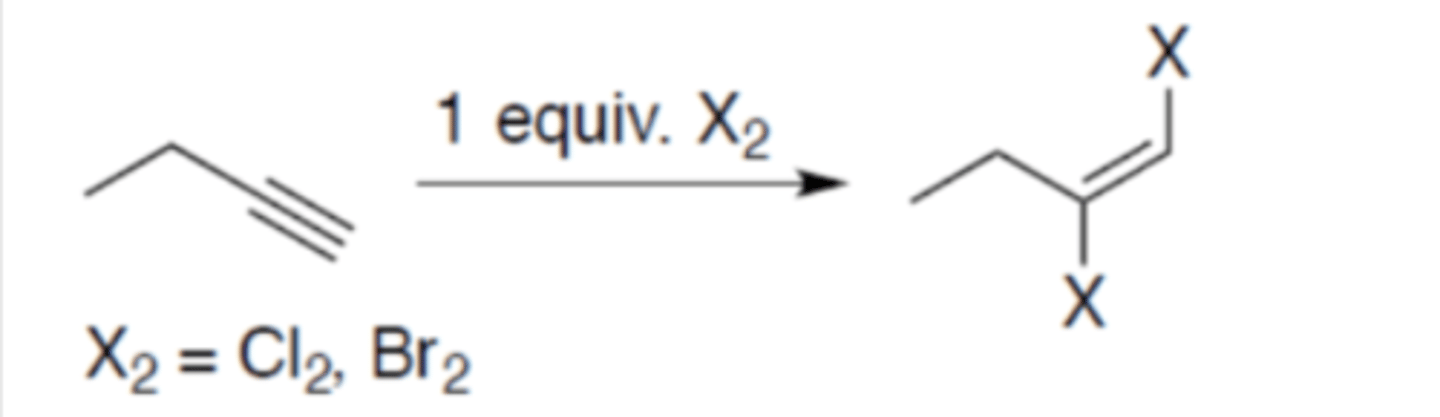
Halogenation of alkyne (2 equivalent)
Carbocation: yes (no rearrangement)
Regiochemistry: mark
Stereoselectivity: anti
Reagent(s): 2 equivalents Cl2 or Br2
- Adds 2 X on both the alkyne carbons
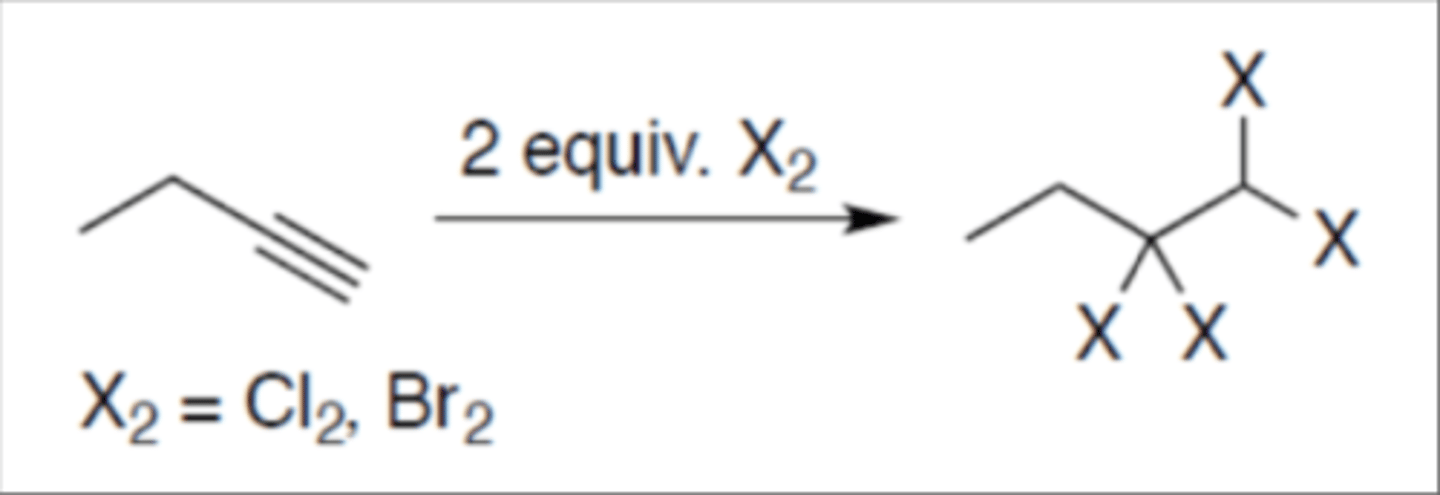
Hydrohalogenation of alkyne (1 equivalent)
Carbocation: yes (no rearrangement)
Regiochemistry: mark
Stereoselectivity: anti
Reagent(s): HX = HCl, HBr, HI
- Becomes alkene and adds X to more stable CC+ and hydrogen to other carbon
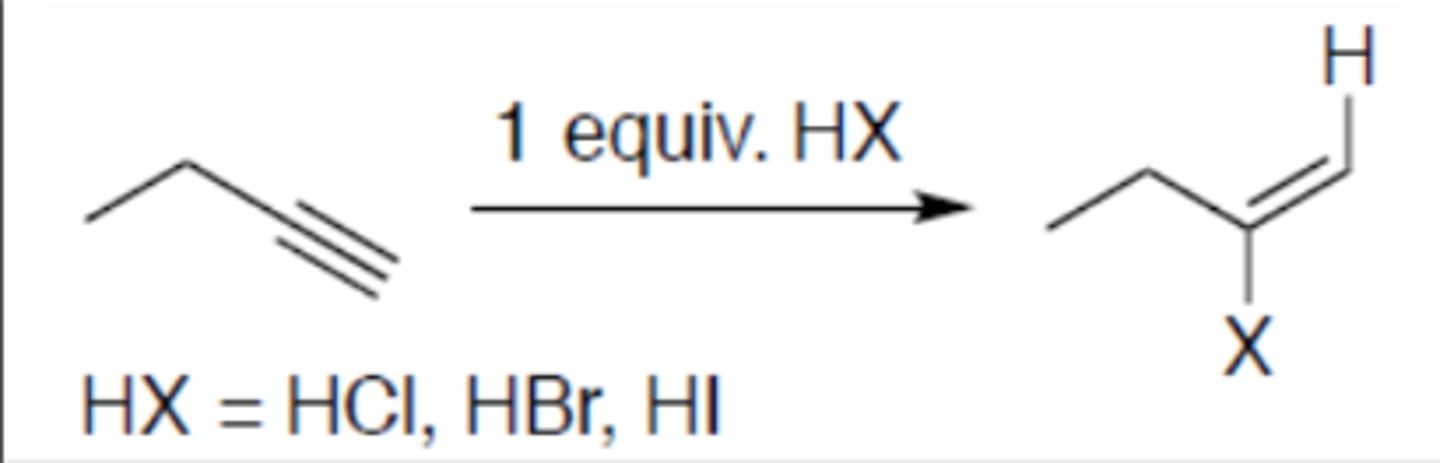
Hydrohalogenation of alkyne (2 equivalents)
Carbocation: yes (no rearrangement)
Regiochemistry: mark
Stereoselectivity: anti
Reagent(s): HX = HCl, HBr, HI
- Becomes alkane and adds 2 X to more stable CC+
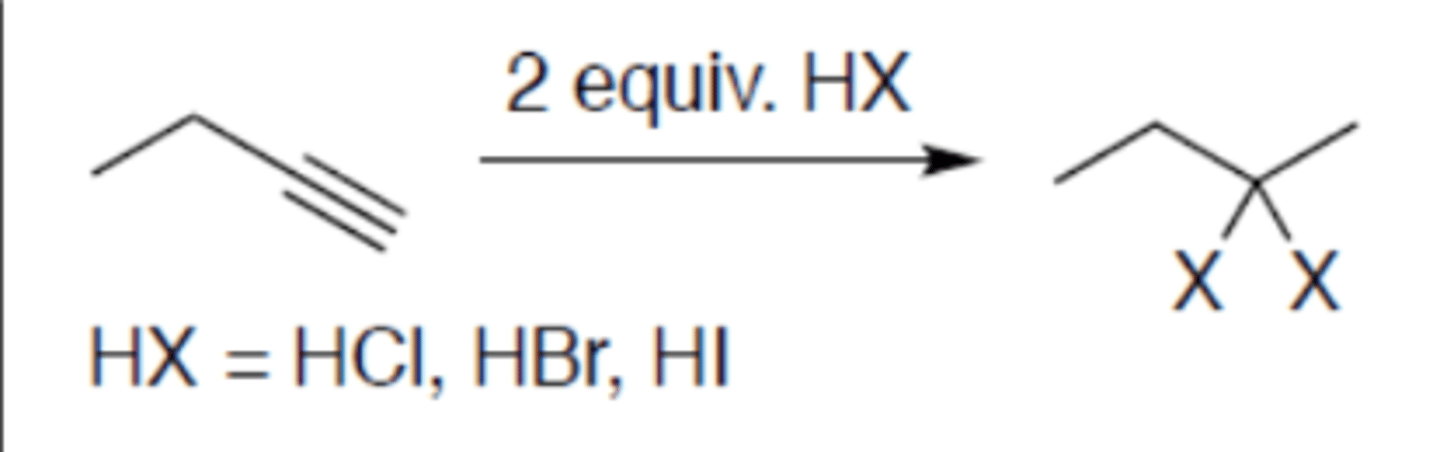
Hydroboration-oxidation (terminal alkyne)
Carbocation: no
Regiochemistry: anti-mark
Stereoselectivity: N/A
Reagent(s): 1. R2BH = (Sia)2BH or Cy2BH 2. H2O2, NaOH
- Adds a =O and hydrogen to terminal carbon (makes aldehyde)
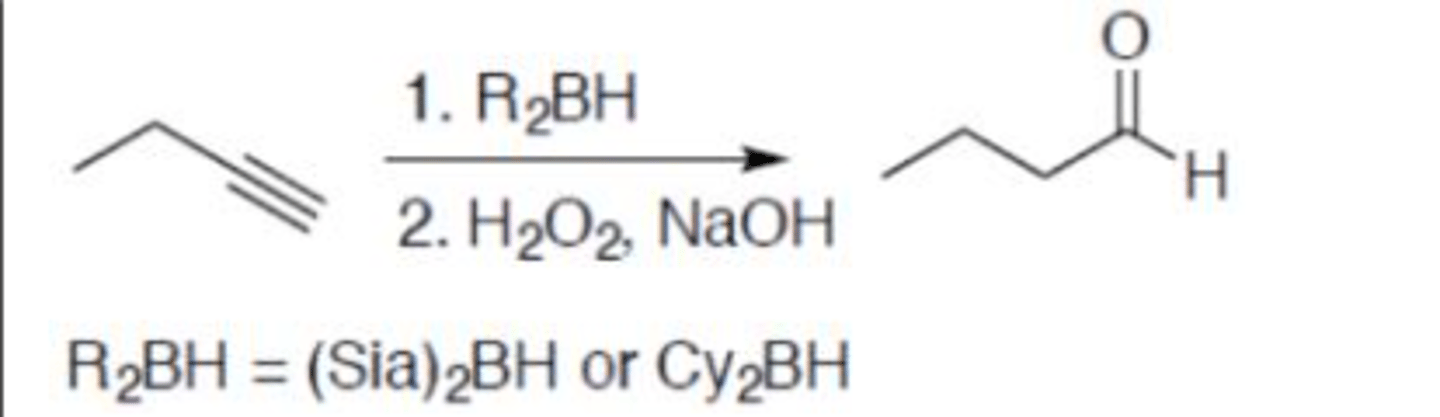
Hydroboration-oxidation (internal alkyne)
Carbocation: no
Regiochemistry: N/A
Stereoselectivity: syn
Stereoselectivity: N/A
Reagent(s): 1. BH3 2. H2O2, NaOH
- Adds a =O to one alkyne carbon
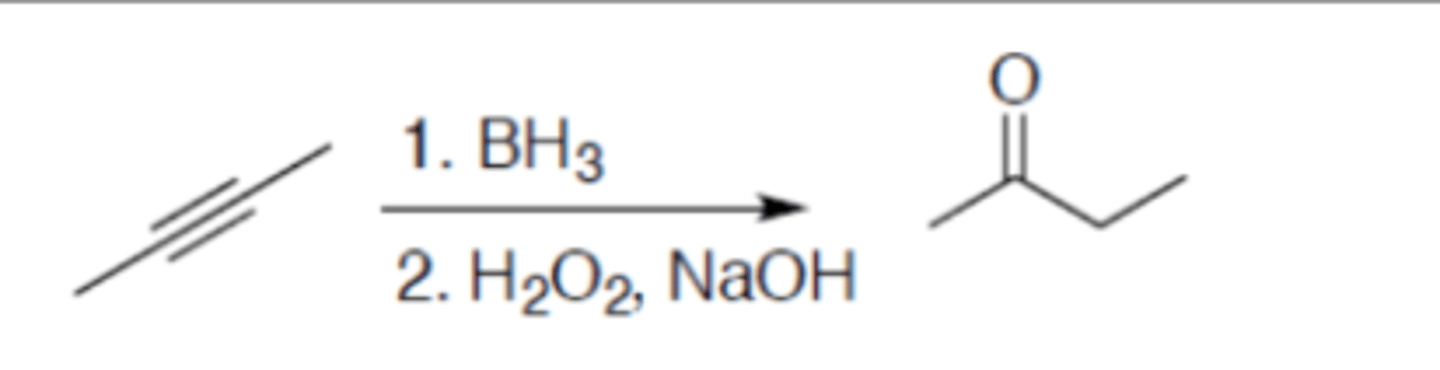
Hydration of alkyne (terminal alkyne)
Carbocation: no
Regiochemistry: mark
Stereoselectivity: N/A
Reagent(s): 1. H2SO4, H2O 2. HgSO4
- Adds =O to inner terminal carbon of alkyne
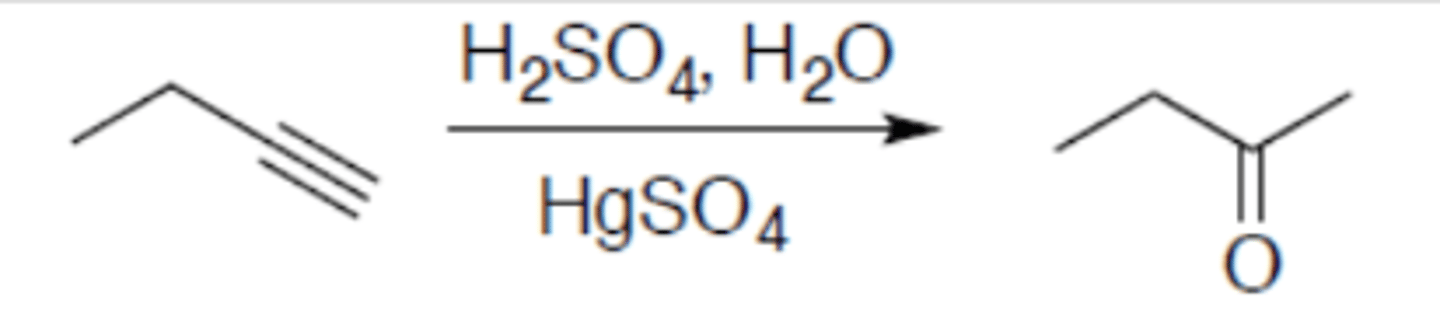
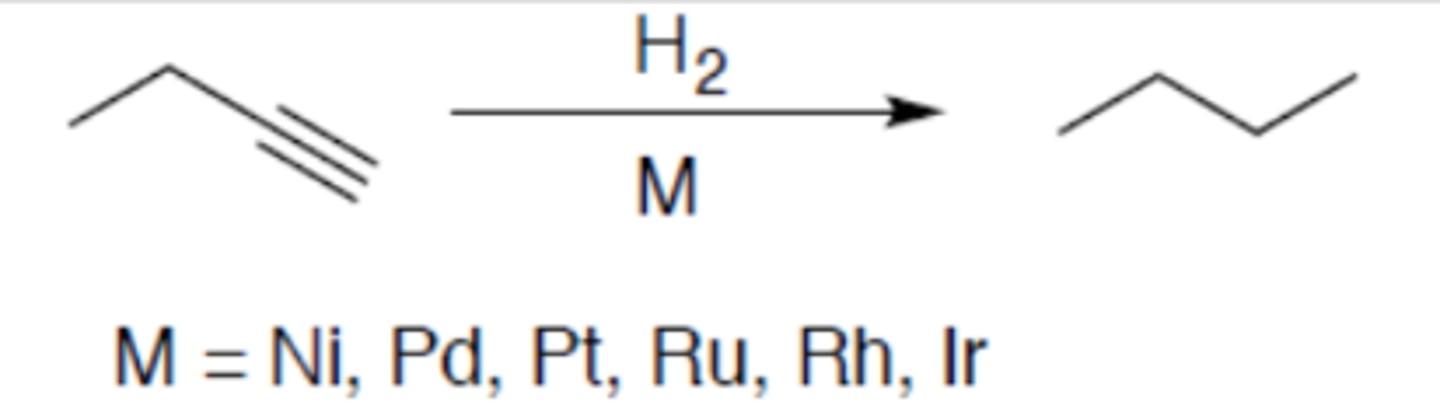
Hydrogenation of alkyne (terminal alkyne)
Carbocation: no
Regiochemistry: N/A
Stereoselectivity: syn
Reagent(s): H2 over M = Ni, Pd, Pt, Ru, Rh, Ir
- Turns terminal alkyne into alkane
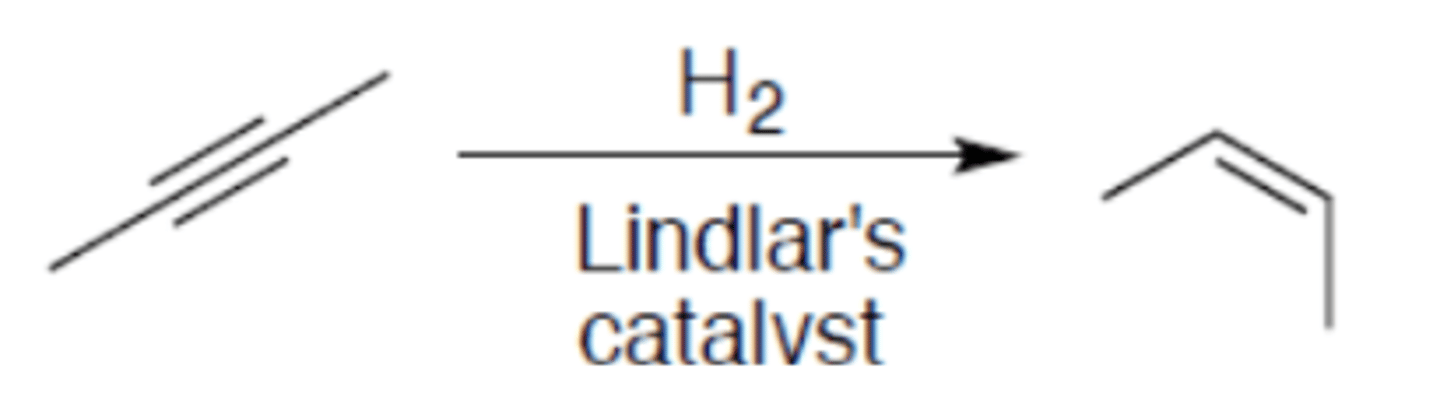
Hydrogenation of alkyne (Lindlar's Catalyst)
Carbocation: no
Regiochemistry: N/A
Stereoselectivity: syn
Reagent(s): H2 over Lindlar's catalyst
- Turns internal alkyne to (Z)-alkene
Hydrogenation of alkyne (hydroboration-protonolysis)
Carbocation: no
Regiochemistry: N/A
Stereoselectivity: syn
Reagent(s): 1. BH3 2. xs CH3CO2H
- Turns internal alkyne into (Z)-alkene
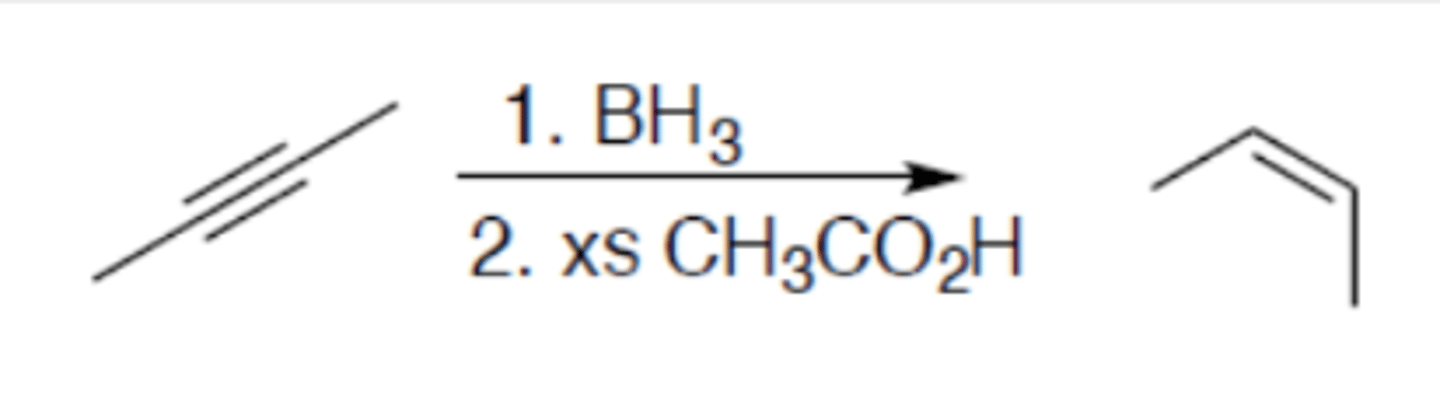
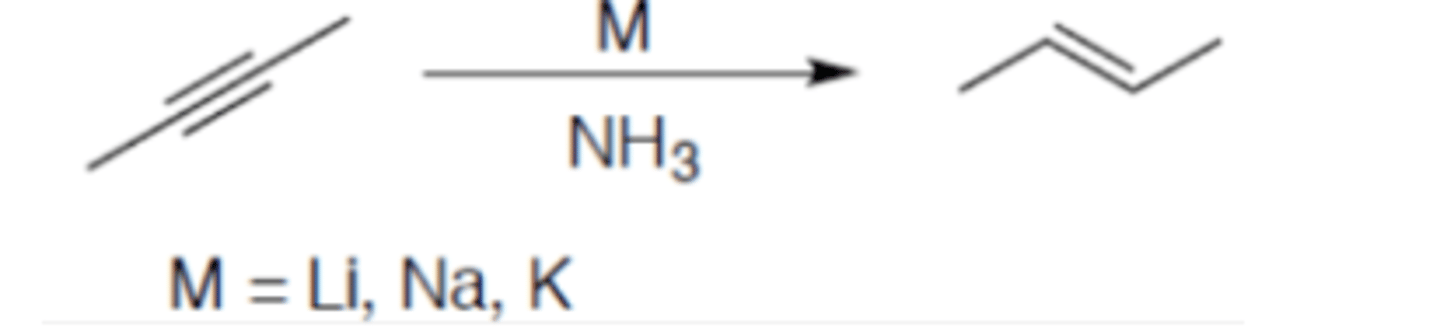
Hydrogenation of alkyne (metal reduction)
Carbocation: no
Regiochemistry: N/A
Stereoselectivity: anti
Reagent(s): M = Li, Na, K over NH3
- Turns internal alkyne into (E)-alkene
Radical halogenation
Intermediate: Radical
Regiochemistry:
- Cl2 low selectivity
- Br2 high selectivity
Stereoselectivity: none
- Reagent(s): X2 over hv (light) or heat
- Adds X to most (Br2) or least (Cl2) substituted
*KNOW MECHANISM*
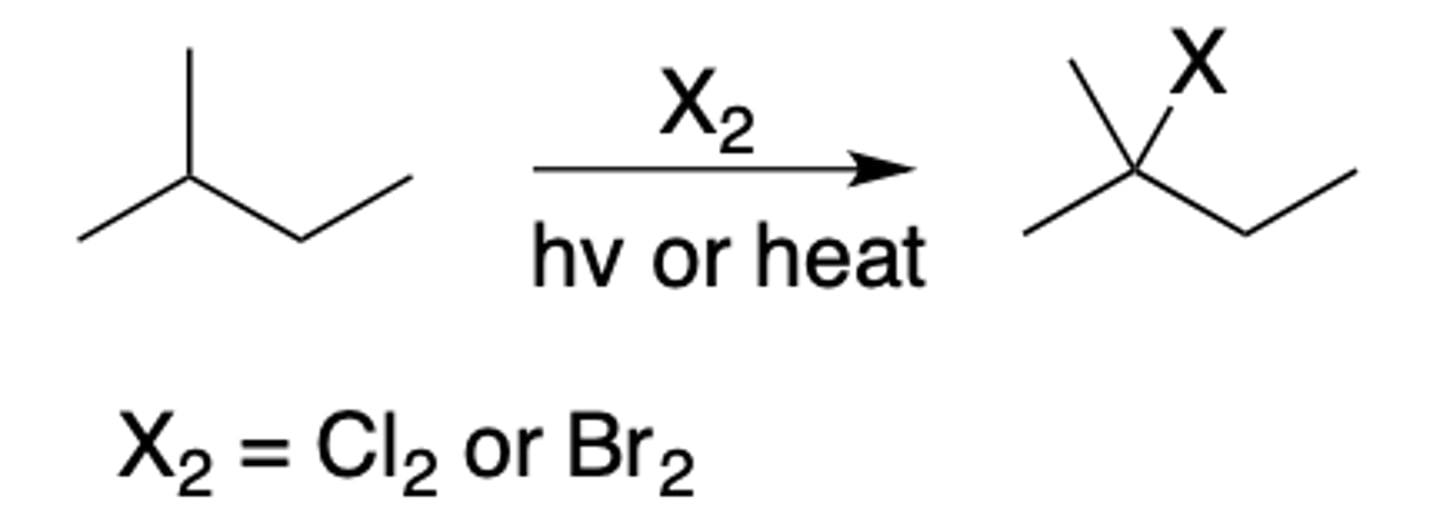
Allylic halogenation
Intermediate: Radical
Regiochemistry: N/A
Stereoselectivity: selectivity for most substituted double bond in product
Reagent(s): NBS or NCS over hv
- NBS: X = Br
- NCS: X = Cl
- Adds X to most substituted allylic carbon
*KNOW MECHANISM*
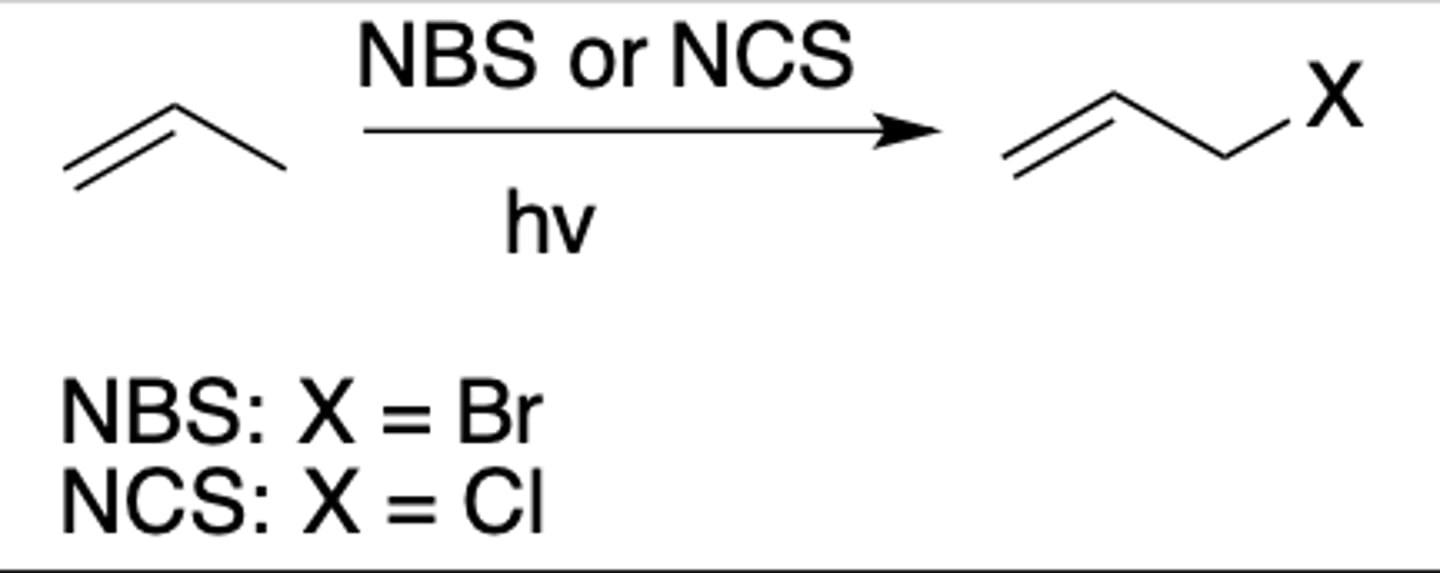
Radical HBr addition (with peroxides)
Intermediate: Radical
Regiochemistry: anti-mark
Stereoselectivity: none
Reagent(s): HBr over peroxides = tBuOOtBu
- Radical goes to more substituted carbon of double bond, Br joins other carbon
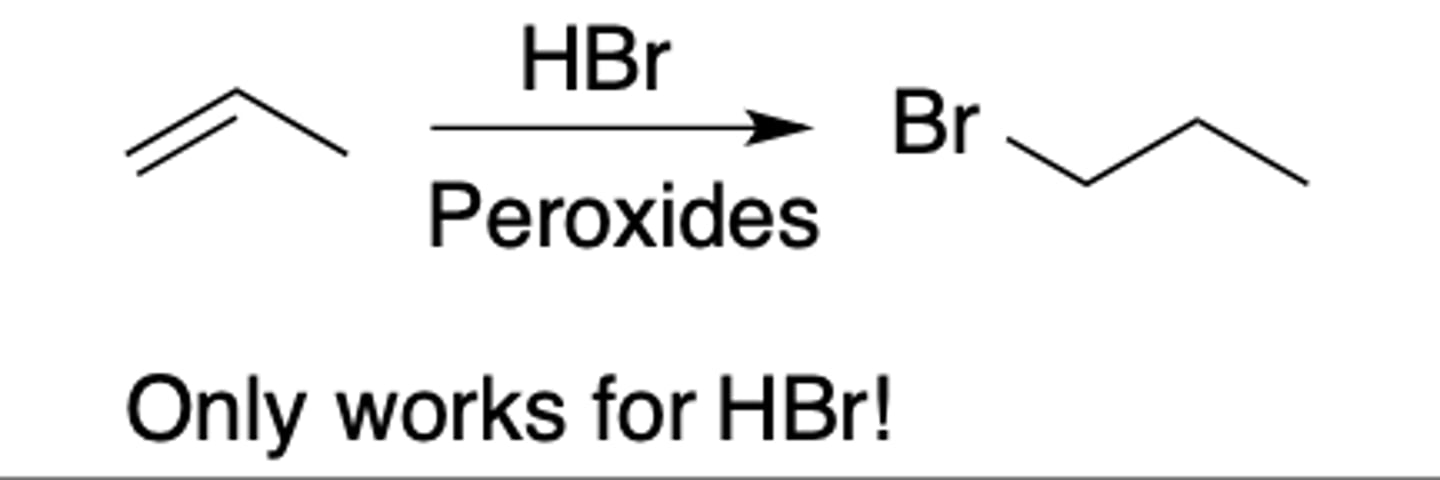
Active metals reaction
Intermediate: none
Regiochemistry: N/A
Stereoselectivity: N/A
Reagent(s): M = Li, Na, K
- Turns OH into O^-
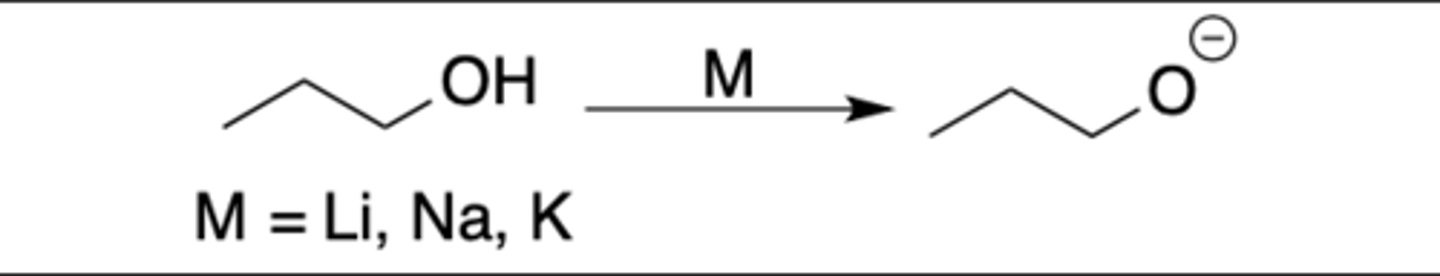
Hydride
Intermediate: none
Regiochemistry: N/A
Stereoselectivity: N/A
Reagent(s): NaH
- Turns OH into O^-
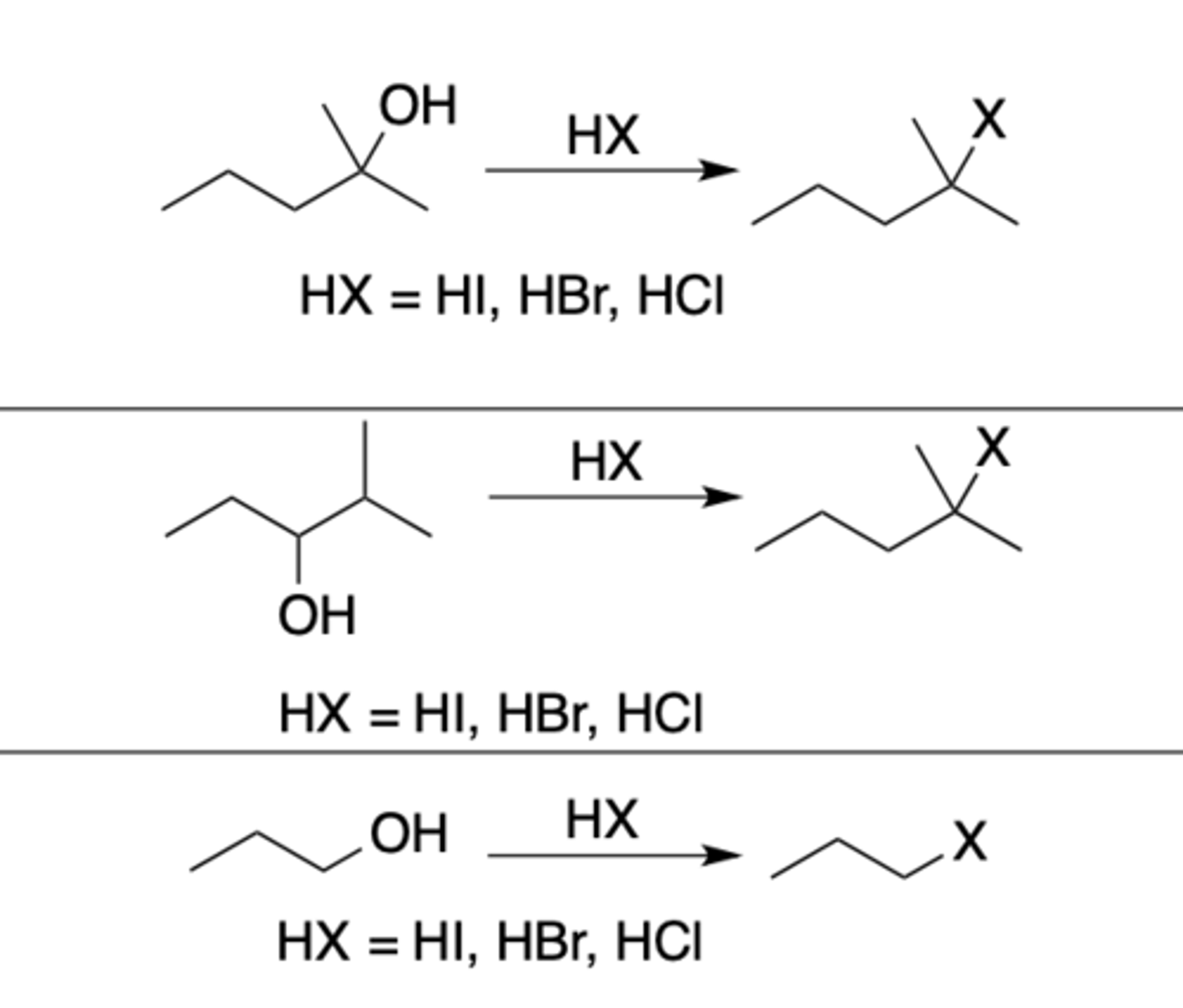
Hydrohalogen acid
Intermediate: none
Regiochemistry: REARRANGEMENT only possible if following CC+ can form:
- tertiary
- secondary benzylic/allylic
- tertiary benzylic/allylic
(reaction can still happen with primary, secondary, and tertiary)
Stereoselectivity: none
Reagent(s): HX: X = I, Br, Cl
- Turns OH into X
*With primary branched alcohols, simultaneous 1,2 shift can happen (methyl can move from most substituted to replace LG, leaving CC+ where the methyl left* - CANNOT occur with primary unbranched alcohols
*KNOW MECHANISM*
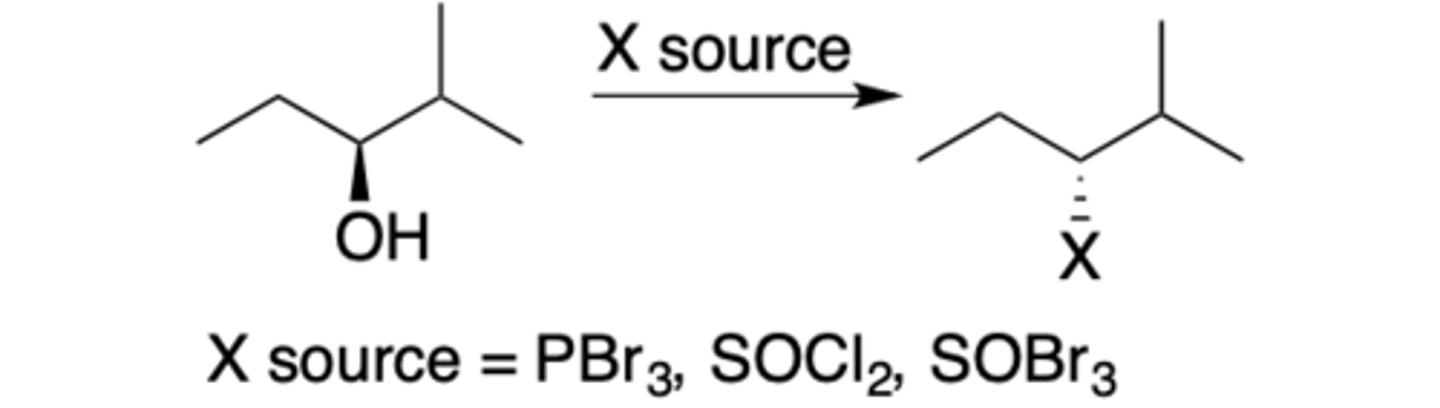
PBr3, SOCl2, SOBr3 reaction
Intermediate: no CC+
Regiochemistry: no rearrangment
Stereoselectivity: Inversion (SN2)
Reagent(s): PBr3/SOCl2/SOBr3
- Turns OH into X source and flips stereochemistry
- X source = PBr3, SOCl2, SOBr2 (image has typo)
*KNOW MECHANISM FOR PBr3*
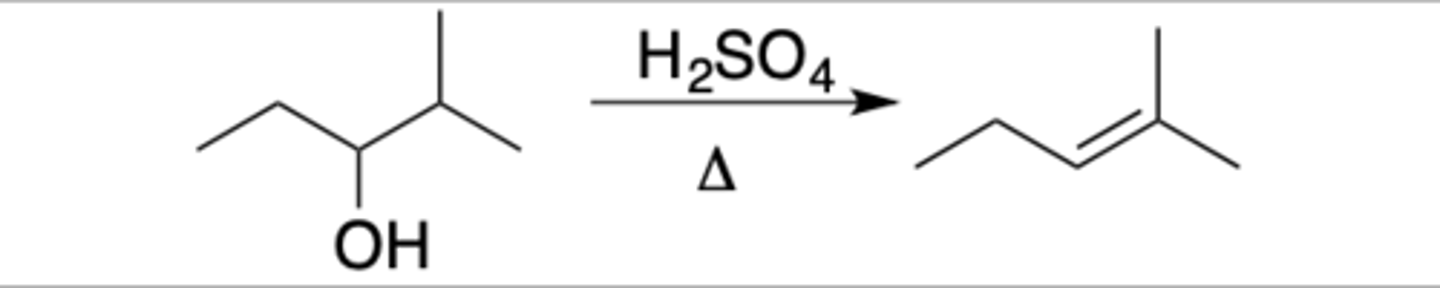
Dehydration
Intermediate: CC+
Regiochemistry: unlike HX reaction, rearrangement will happen for all alcohols
Stereoselectivity: none
Reagent(s): H2SO4 over delta (heat)
- OH protonated, leaves, alkene is formed to CC+
*KNOW MECHANISM*
Nucleophilic Opening (Epoxide Opening)
SN2
Reagent(s): NON-POOR Nu^- (NH3/C^- anion) OVER H2O/EtOH, etc.
OR
1. LiAlH4 2. H2O (H^- is Nu^-)
NEED: EPOXIDE -> Nu-R-OH
Nu^- ATTACKS LEAST SUBSTITUTED
Acid Catalyzed Opening (Epoxide Opening)
SN2
Reagent(s): POOR Nu^- OVER STRONG ACID
OR
STRONG ACID (HX)
NEED: EPOXIDE -> Nu-R-OH
Nu^- ATTACKS MOST SUBSTITUTED
POOR Nu^- WILL PROTONATE/DEPROTONATE
USE ALREADY PROTONATED POOR Nu^- (CH3OH2+)
Grignard (Basic)
Reagent(s): Mg OVER Et2O (aprotic solvent/no H-bonds)
NEED: R-X -> R-MgX
MUST PROTECT AN EXISTING OH on R-X WITH SILYL ETHER
R-MgBr/R-Li Reactions (Epoxide Opening)
VERY STRONG BASES
-MUST PROTECT EXISTING OH WITH SILYL ETHER
Reagent(s): MUST HAVE R-MgBr OR R-Li AND PROTON SOURCE (H3O+)
Nu^- ATTACKS LEAST SUBSTITUTED (UNLESS CATALYZED BY ACID)
SAME AS Nu^- OPENING
Carbonyl under Acidic/Basic Conditions
Acidic: R-C=O -R -> R-C-OH+ -R (causes resonance)
Then Basic attacks C, causes Resonance and balance
Basic (Strong Nu^-): R-C=O -R -> R-C-O^- -Nu - R (causes resonance)
Then O^- attacks acid, causes balance
Carbonyl Reactions With Carbon Nu^-
CREATES C-C BONDS
- Grignards
- Organolithium
- Alkyne anion
- Hydrocyanation (equilibrium arrows)
Reagent(s): ANY ABOVE + MUST HAVE PROTON SOURCE (H3O+)
NEED: C=O -> OH + new C-C
Carbon Nu^- attacks C of C=O, kicks up double bond to lone pair, oxygen protonated
Hydride Reduction
Reagent(s): 1. LiAlH4 2. H3O+ OR 1. NaBH4 2. ROH/H2O
H^- Nu attacks C=O
LiAlH4 = more reactive, less selective
NaBH4 = less reactive, more selective
C=O -> C-OH
H2/Rh
Reagent(s): H2/Rh
Reduces alkene, LEAVES C=O ALONE
Making Carboxylic Acids
Oxidation of Primary -OH with Jones' Reagent
Grignards + CO2
Haloform
Hydrolysis of Carboxylic Derivatives
Reduction of Carboxylic Acids
Hydride Reduction: 1. LiAlH4 2. H3O+
- C.A. -> -OH
Diazomethane: H2C=N^+=N^-
- C.A. -> Ester + N2
Beta-Keto Acid: Heat
- Beta-Keto Acid -> Ketone + CO2
- Gets rid of a single C.A.
Nucleophilic Addition to Carboxylic Acids (Recap)
Carbon Nu: (Grignard for example = MgBrR)
- C.A. -> Carboxylate + R (steals H)
- Acid/Base because C.A. is not as E^+ as ketones/aldehydes and C^- is EXTREMELY STRONG
Hydrogen Reduction: 1. LiAlH4 2. H3O^+
- C.A. -> Alcohol
Nitrogen Nu: H2NR
- C.A. -> Carboxylate + H3N^+R
- Acid/Basebecause C.A. is not as E^+ as ketones/aldehydes and N^- Nu is VERY STRONG
Oxygen Nu: ROH, Acid
- C.A. -> O=C-OR + H2O
Carboxylic Acid Derivative + Grignard
Reagent(s): 1. 2 equiv. MgXR 2. H3O^+
Ester
- O=C-OR -> HO-C-R
- Ketone intermediate attacked by grignard again then H3O^+ to make alcohol
Acid Halide
- R-X -> R-HO-R
- R added to C-OH
MAKES AN ALCOHOL