GCSE Chemistry: C8 - Rates and equilibrium
1/84
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
85 Terms
Explain what is meant by equilibrium
- equilibrium describes where the forwards reaction is taking place at the same rate
- as the backwards reaction in a reversible reaction,
- in a closed system
Why may a solution turn cloudy?
- if sulfur is being funnelled as a solid
What does the slope of a line on a graph showing the quantity of reactants or products in a chemical reaction against the time tell use?
The slope of the line gives us an idea of the rate of reaction.
What does a steep slope on a graph showing the quantity of reactants or products in a chemical reaction against the time tell us?
- The steeper the slope of the line, the faster the rate of reaction
- when a reaction is fast because the slope of the line is steep, this tells us we are making a lot of product in a short amount of time
- this is because there is a large number of reactant molecules, so lots of these reactant molecules are reacting and forming the product over a short amount of time
What does it mean if the slope of the line then becomes less steep?
- when the slope of the line becomes less steep, this tells us that the reaction is slowing down, so the rate of the reaction is decreasing
- this is because a lot of the reactant molecules have already reacted and turned into product, so there are fewer reactant molecules available to react now
Why does the slope of the line of a graph showing the quantity of reactants or products against the time become flat at the end of the graph?
- at the end of the graph, the slope of the line is now zero, so the line is flat
- this tells us, at this point, the reaction has stopped, so no more product is being produced over the given time, because all of the reactant molecules have already reacted
How is the product from a chemical reaction measured?
Product is measured in grams (g).
How is the quantity of product measured if the product is a gas?
If the product of the chemical reaction is a gas, the quantity of the gas product is measured in cm^3.
What is the appearance of a graph showing the quantity of product produced against time?
The graph showing the quantity of product produced against time is a positive curve.
What is the appearance of a graph showing the quantity of reactant used against time?
The graph showing the quantity of reactant used against time is a negative curve.
Summarise the three main stages of a graph showing the quantity of reactant used in a chemical reaction against the time
1 - initially, the rate of reaction is fast
2 - the reaction then slows down
3 - and finally, the reaction stops
Show the two equations for rate of reaction

What is the problem with using a graph to show the rate of a chemical reaction?
- on the graph, the slope of the curve is constantly changing
- this means that the rate of reaction is also constantly changing
- this makes it very difficult to get a clear idea of the exact rate of the reaction at any specific point
What is meant by a tangent?
A tangent is a straight line which just touches the curve at the point we are interested in.
Scientists overcome the problem of the slope of the line of the graph constantly changing by drawing a tangent at any specific point on the curve.
The slope of the tangent gives us an idea of the rate of reaction.
Why may a tangent drawn onto the start of the curve be steeper?
A tangent that is drawn to nearer towards the start of the curve may be steeper, because the reaction is faster at the start of the reaction than towards the end of the reaction.
Describe the steps for an exam question on using tangents to calculate the rate of reaction
1 - Draw the tangent to the curve at the point of interest on the slope
2 - Then draw a triangle to suitable points from the tangent drawn
3 - Then work out the sides of the triangle using the scales of the graph
4 - Work out the two vertical points from the y-axis and calculate the difference (y2 - y1)
5 - Work out the two horizontal points from the x-axis and calculate the difference (x2 - x1)
6 - Then calculate the rate of the reaction using the equation:
Rate =y/x
Show the equation for rate of reaction from a tangent and triangle drawn to a point on the curve of a graph
Rate of reaction = y2 - y1 / x2 - x1
so,
Rate = y / x
Explain the idea of collision theory
Collision theory:
- chemical reactions can only take place when the reacting particles collide with each other
- the collisions must have sufficient energy
- the rate of a chemical reaction is determined by the frequency of successful collisions
In terms of collision theory, what is meant by frequency?
Frequency is the number of successful collisions per second.
Explain the stages of a chemical reaction using collision theory
- chemical reactions are rapid initially, because there are a large number of reactant molecules,
- so this means there are a large number of successful collisions per second, with sufficient energy
- then over time, reactions slow down because the number of reactant molecules starts to run out,
- so there a smaller number of collisions per second
- finally, the reaction stops because all of the reactant molecules have run out,
- so this means that the number of collisions per second between the particles is zero
Describe the effect of concentration on rate
- the higher the concentration of particles in a container, the higher the number of collisions per second
- this means, if we increase the concentration, the rate also increases,
- so the rate is proportional to the concentration
What is the effect of gas pressure on rate?
The higher the gas pressure in a container, the higher the rate.
Describe a graph to show the effect of concentration on rate
- on a graph, a higher concentration of reactant will have been involved in the reaction if the reaction is a fast reaction,
- and we can tell if the reaction is fast if the line is steep(er)
- a higher concentration reactant will also give more product at the end of the reaction, as the reaction would have started with more reactant molecules at the start if a high concentration reactant was used
What is a hypothesis?
A hypothesis is a proposal that could explain a fact or an observation.
What must a scientific hypothesis be?
In science, a hypothesis must be testable.
Name the experiment that can be used in order to explore the effect of concentration on the rate of a reaction.
The disappearing cross reaction can be used for when exploring the effect of concentration on the rate of a reaction.
In the disappearing cross reaction, what product is produced when sodium thiosulfate solution is reacted with hydrochloric acid?
If we react sodium thiosulfate solution with hydrochloric acid in the disappearing cross reaction, one of the products is sulfur. The sulfur produced is a solid.
Why may the solution in the disappearing cross reaction turn cloudy?
- In the disappearing cross reaction, the solution may turn cloudy
- this is because sulfur is being funnelled as a solid
What is meant by turbidity?
Turbidity describes the cloudiness of a solution due to sulfur being funnelled as a solid.
What can the cloudiness (turbidity) caused in the solution due to sulfur being funnelled as a solid during the disappearing cross reaction tell us?
The turbidity of the solution can be used to see how long the reaction takes to finish.
Required Practical 5: Rates of Reaction
Describe the disappearing cross reaction for exploring the effect of concentration on the rate of a reaction
1) Use a measuring cylinder to put 10 cm^3 of sodium thiosulfate solution into a conical flask
2) Place the conical flask onto a printed black cross
3) Then add 10 cm^3 of hydrochloric acid into the conical flask
4) Swirl the solution and start a stopwatch
5) Look down through the top of the flask, as after a certain time, the solution will turn cloudy, so we stop the stopwatch when we can longer see the cross due to the cloudiness
6) Carry out experiment again using lower concentrations of sodium thiosulfate solution
7) Then repeat the whole experiment and calculate mean values for each concentration of sodium thiosulfate solution, but remove any anomalous results when calculating the mean
What is meant by reproducibility?
Reproducibility:
- a measurement is reproducible if it can be repeated by another investigator using a different technique or different equipment, and the same result is still gained
Suggest a problem with the disappearing cross reaction to explore the effect of concentration on the rate of reaction
- a problem with the dispappearing cross experiment is that different people have different eyesights
- this means that, as some people can see the cross for longer than others, they may not get the same results
- however, because all of the students use the same size printed cross, this problem may not be too great
State the word equation for the reaction involved in the experiment measuring the volume of a gas produced by a reaction in order to explore the effect of concentration on the rate of a reaction
magnesium+hydrochloric acid->magnesium chloride+ hydrogen
Required Practical 5: Rates of Reaction
Describe the experiment measuring the volume of a gas produced by a reaction in order to explore the effect of concentration on the rate of a reaction
1) Use a measuring cylinder to place 50 cm^3 of hydrochloric acid into a conical flask
2) Attach the conical flask to a bung and a delivery tube
3) Now place the delivery tube into a container filled with water
4) Place an upturned measuring cylinder, also filled with water, over the delivery tube
5) Add a 3 cm strip of magnesium to the hydrochloric acid and start a stopwatch
6) The reaction produces hydrogen gas, which is trapped in the measuring cylinder
7) Every 10 seconds, measure the volume of hydrogen gas in the measuring cylinder, and continue doing this until no more hydrogen is given off
8) Repeat the experiment using different concentrations of hydrochloric acid
What do both the disappearing cross experiment and the experiment measuring the volume of a gas produced tell use about the effect of concentration on the rate of a reaction?
- Both the disappearing cross experiment and the experiment measuring the volume of a gas produced show that the greater the concentration of a chemical in a reaction, the faster the reaction takes place
Why can it be said that the finding that the greater the concentration of a chemical in a reaction, the faster the reaction takes place, is reproducible?
Because the result is shown by two experiments, we can say that the finding that, the greater the concentration of a chemical in a reaction, the faster the reaction takes place, is reproducible.
Describe how the particles in solution are able to react with the particles in a solid
- particles in solution can only react with particles on the surface of a solid
- however, a lot of particles in a solid are not on the surface, so these particles that are not on the surface can not react with the particles in solution
How can the surface area be increased in a solid?
- If a solid is split into two parts, this means that there is now a greater surface area,
- and this is because there are now more particles on the surface to react
Describe the effect of surface area on the rate of a reaction
- when we increase the surface area of a solid reactant, there are more successful collisions with sufficient energy per second,
- as there are more reacting particles on the surface now,
- so this means the rate also increases
Explain why smaller sized blocks of a solid reactant increase the rate of reaction
- smaller sized blocks of a solid reactant have a greater surface area to volume ratio than larger blocks
- this means smaller sized blocks have more particles on the surface to react compared to larger blocks, so there are more collisions per second,
- and so this increases the rate of reaction
Describe an investigation to test the effect of surface area on the rate of reaction by measuring the volume of carbon dioxide produced
- marble chips contain the chemical calcium carbonate, which reacts with hydrochloric acid to produce carbon dioxide gas
- the volume of carbon dioxide gas can be measured in order to determine the rate of reaction
- we can then change the surface area of the marble chips used in the investigation, and test the reaction again
- as the bubbles of carbon dioxide gas in this test can be quite rapid, measuring the volume of carbon dioxide gas can be difficult using a measuring cylinder, so a gas syringe can be used instead, in order to get more accurate results
Describe an alternative test on the effect of surface area on the rate of reaction that involves measuring the mass of carbon dioxide gas lost in order to work out the rate of reaction
- firstly, place the reaction on a balance
- as carbon dioxide is produced, the mass decreases, so we can use this to calculate the rate of reaction
- cotton wool is used in this test to allow carbon dioxide gas to escape, and the cotton wool also prevents acid from splashing out of the flask
- this is important, as if any acid did splash out of the flask, it would cause the mass to fall more than it should, and that would in turn produce an anomalous result
What is activation energy?
Activation energy is the minimum amount of energy that the particles must have in order to react and collide successfully.
In diagrams of particles, what does the size of an arrow represent?
The size of an arrow in diagrams of particles represents the energy of each particle.
Describe what would likely happen if two particles with a small amount of energy collided
- If two particles with a small amount of energy collide, because they have low energy,
- they can not overcome the activation energy barrier
- this means the two particles do not collide successfully, so no reaction occurs
Describe what would likely happen if two particles with a large amount of energy collide
- If two particles with a large amount of energy collide,
- they have enough energy to overcome the activation energy barrier and collide successfully
- this means a reaction takes place
Explain why increasing the temperature increases the rate of reaction
- Increasing the temperature increases the rate of reaction,
- because increasing the temperature increases the energy of the particles
- as the particles now have a higher amount of energy, the particles now move more quickly as they have more kinetic energy
- this increases the frequency of collisions between reacting particles, as well as the energy of each collision
- this means more particles can overcome the activation energy barrier and collide successfully
What is the relationship between temperature and the rate of reaction?
Because the rate of reaction increases with the temperature, we can say that the rate of reaction is proportional to the temperature.
What is meant by a catalyst?
A catalyst is a substance that increases the rate of a chemical reaction, but is not used up during the reaction.
Describe the reasons why catalysts are important
- catalysts allow us to carry out reactions quickly without needing to increase the temperature,
- so this saves money
- and because catalysts are not used up during the reaction, we can reuse them again and again, so this is sustainable
What does the rate of reaction depend on?
The rate of reaction depends on the number of particles that have enough energy to cross the activation energy barrier and collide successfully.
How do catalysts increase the rate of reaction?
- Catalysts increase the rate of reaction by lowering the activation energy and providing a different pathway for the reaction
- this means, as the particles require less energy to cross the activation energy barrier, more particles can overcome the activation energy barrier and react and collide successfully per second
Why are catalysts not included in the chemical equation for a reaction?
Catalysts are not included in the chemical equation for a reaction, because they are not used up in the reaction, so catalysts are not a reactant. However, the catalyst may be included/need to be included above the arrow symbol in a chemical equation for a reaction.
Are the same catalysts always used in chemical reactions?
Different reactions need different catalysts.
What are the catalysts in living organisms?
Enzymes act as catalysts in living organisms.
Show the forwards arrow that tells us that a reaction only goes forward

Show the arrow used to show a reversible reaction, which is a reaction where the products can react to reform the original reactants.
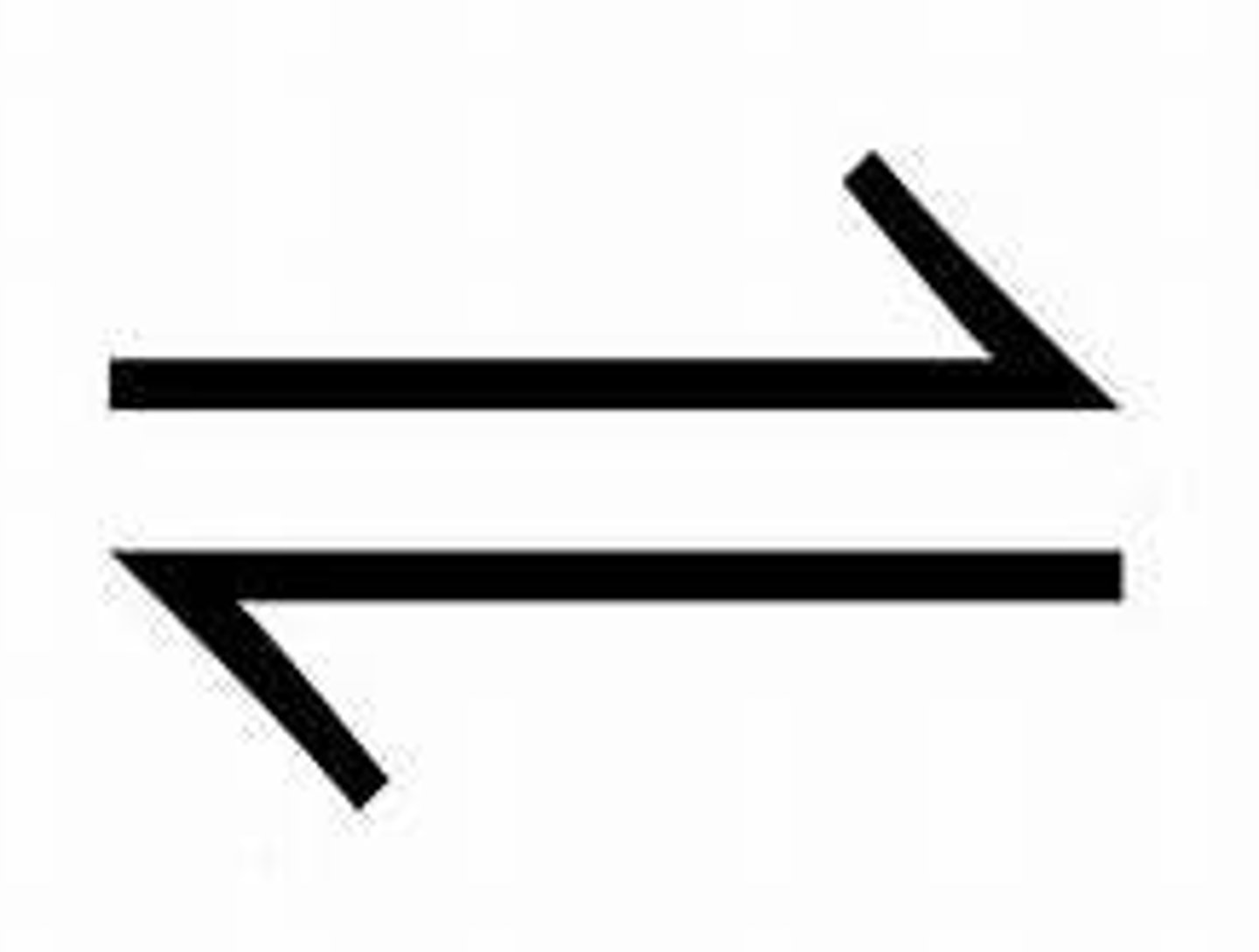
How can the direction of a reversible reaction be changed?
The direction of a reversible reaction can be changed by changing the conditions.
What is the backwards reaction in a chemical reaction if energy is released and the reaction gets hot?
If energy is released and the reaction gets hot in the backwards reaction, this means the backwards reaction is exothermic in this case.
What is the forwards reaction in a chemical reaction if the reactants are heated?
If the reactants in the reversible reaction are heated, this means that the forwards reaction is endothermic in this case, as energy is being taken in/gained by the reacting molecules.
If a reversible reaction is exothermic in one direction...
If a reversible reaction is exothermic in one direction, it is endothermic in the opposite direction.
The same amount of energy is transferred in each case.
What colour is hydrated copper sulfate?
Hydrated copper sulfate is a blue substance
What forms when hydrated copper sulfate is heated?
When hydrated copper sulfate is heated, it reacts to form anhydrous copper sulfate. Water is also produced in the reaction.
What colour is anhydrous copper sulfate?
Anhydrous copper sulfate is white.
How is the reaction for heating hydrated copper sulfate to form anhydrous copper sulfate reversed?
If the water is added to the anhydrous copper sulfate produced when hydrated copper sulfate was heated, the reaction reverses.
What is used to stop any reactants or products from escaping a chemical reaction?
A sealed container is used to stop any reactants or products from escaping a chemical reaction?
What is meant by a reversible reaction?
A reversible reaction is a chemical reaction that can be reversed so that the products react to re-form the original reactants.
What is meant by a closed system?
A closed system is where no reactants or products can enter or leave the reaction
State Le Chatelier's Principle
If a system is at equilibrium and a change is made to the conditions, then the system responds to counteract the change.
Scientists call this Le Chatelier's Principle.
Show the general structure for an exam question on changing equilibrium concentrations.
If the concentration of ______ is _____, equilibrium will want to _____ the concentration. The equilibrium will do this by shifting to the _____ and favouring the _____ reaction. This means that the yield of _____ will _____.
If the forwards reaction in a reversible reaction is exothermic, ....
If the forwards reaction in a reversible reaction is exothermic, energy is released and the reaction gets hot, so the temperature of the system increases.
If the reverse reaction in a reversible reaction is endothermic, ....
If the reverse reaction in a reversible reaction is endothermic, energy is taken in, and so the temperature of the system decreases.
State the general structure for an exam question on changing equilibrium temperature
If the temperature is _____ ,equilibrium will want to _____ the temperature. The equilibrium will do this by shifting to the _____ and favouring the _______ reaction, as this is the _______ direction. This means that the yield of ______ will _______ .
Which types of reactions does pressure affect?
Pressure affects reactions involving gases.
What does the pressure of a gas depend on?
The pressure of a gas depends on the number of molecules.
Describe the effect of increasing the pressure on a reversible reaction at equilibrium.
If we increase the pressure on a reversible reaction at equilibrium, the position of the equilibrium shifts to the side with the smallest number of moles.
Describe the effect of decreasing the pressure on a reversible reaction at equilibrium
If we decrease the pressure on a reversible reaction at equilibrium, the position of the equilibrium shifts to the side with the largest number of moles.
What is the effect on equilibrium if the number of molecules is the same on both sides of a reversible reaction?
If the number of molecules is the same on both sides of a reversible reaction, changing the pressure has no effect on the position of equilibrium.
Show the general structure for an exam question on changing equilibrium pressures.
If the pressure is ______ , equilibrium will want to _____ the pressure. It will do this by shifting to the _____ and favouring the _____ reaction, as this is the side with the ______ number of molecules of gas. This means that the yield of _____ will _______.
Describe what is meant by the compromise between rate and equilibrium.
The compromise between rate and equilibrium describes where, often, in industrial processes, a compromise has to be made between using the fastest rate of reaction, and using the maximum yield of the product produced from the reaction.
Describe what the general structure for a 6 mark question on the compromise between rate and equilibrium is
Recommended to use headings and bullet points for an exam question answer to a 6 mark question on the compromise between rate and equilibrium.
1 - suggest how a compromise between fast rate of reaction and high equilibrium yield has been made for the temperature, using the temperature for the given reaction
2 - suggest how a compromise between fast rate of reaction and high equilibrium yield has been made for the pressure, using the pressure for the given reaction
3 - if the given reaction also involves a catalyst, suggest why a catalyst has been included in order to improve the compromise for fast rate of reaction
Show the general structure for a paragraph on the compromise for rate and equilibrium for the temperature
Temperature:
- high equilibrium yield may need a low temperature, but this would slow down the rate of the reaction
- alternatively, a high temperature would mean faster rate of reaction, but this would possibly lower the equilibrium yield
- therefore, a compromise between high equilibrium yield and fast rate of reaction is made, by using a temperature of (actual reaction temperature given in the exam question) *C, to ensure as much product is produced as quickly as possible.
Show the general structure for a paragraph on the compromise for rate and equilibrium for the pressure
Pressure:
- a high pressure means there is a faster rate of reaction, but this also means there is a lower equilibrium yield of the product
- also, using a high pressure can be dangerous for workers
- as well as this, strong, pressurised containers are required for using high pressure, but these are expensive to buy and maintain
- alternatively, a low pressure means there is a higher equilibrium yield, but there is a slower rate of reaction
- therefore, a compromise between high equilibrium yield and fast rate of reaction is made, by using a pressure of (actual pressure used ) atm, to ensure as much product is produced as quickly as possible
Show the general structure for a paragraph on the compromise for rate and equilibrium if the exam question also states that a catalyst is used in the reaction
A catalyst:
- a catalyst can speed up the reaction without being used up, by lowering the activation energy, so that more particles can collide and react successfully in order to overcome the activation energy barrier
- this means that adding a (name of catalyst) catalyst increases the rate of reaction, so that the product of the reaction is produced as quickly and economically as possible