ch 9 organic chemistry
1/20
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
21 Terms
what makes alkynes reactive
two pi bonds → electron rich → undergo addition reaction
how are alkynes formed from dihalides?
double elimination using a strong base removes 2 HX → forms triple bond
what type of halides form alkynes via elimination
Vinylic dihalides with strong bases
what type of reactions fo alynes undergo?
electrophilic addition reaction
why are vinylic carbocations unstable
positive charge is on sp carbon (less stabilizes, only one r group)
what happens when HX is added to an alkyne
forms vinyl halide → then dihalide (markovnikov addition)
what happens when X2 (Br/Cl) is added
forms dihaloalkene → tetrahalide
product of Hg2+ - catalyzed hydration?
ketone
what is tautomerization
conversion of enol (C=C-OH) → ketone (C=O)
product for terminal alkynes
aldehyde (anti-markonikov)
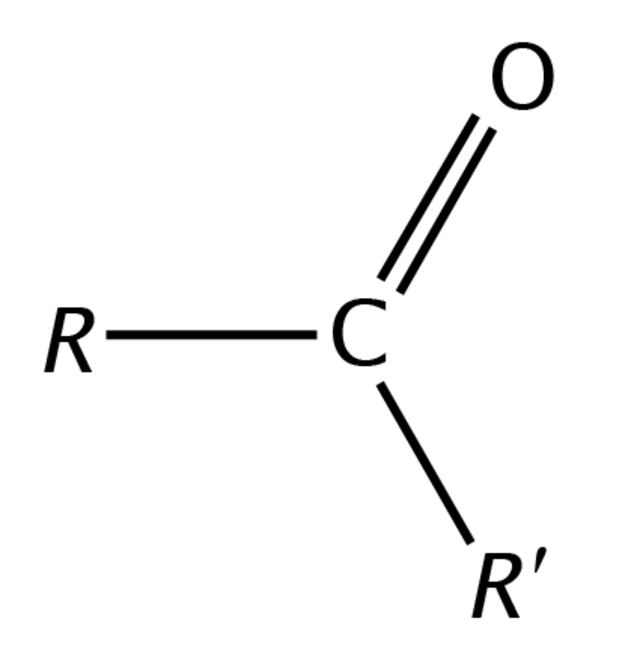
product of internal alkynes
ketone
what does H2 + Pd/C do
fully reduces → alkane
what does lindar catalyst do
partial reduction → cis-alkene
what does Li/NH3 do?
partial reduction → trans-alkene
what happens during oxidative cleavage
triple bond breaks → forms carboxylic acids or CO2
what forms when a terminal alkyne is deprotonated
acetylide anion (alkyne w a negative charge on side)
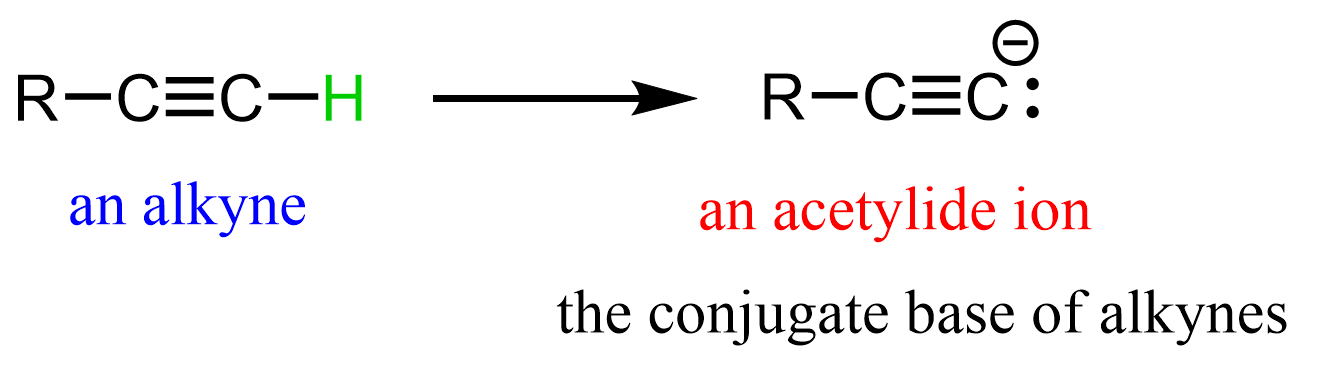
what do acetylide anions do
act as strong nucelophiles and bases
what reaction forms a longer carbocation chain
alkylation of acetylide anion (SN2)
what is the limitation of acetylide alkylation
only works with primary alkyl halides (second and third → elimination)
most stable anion
acetylide anion le
least stable anion
alkyl anion