Bootcamp FL 1 - GC
1/17
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
18 Terms
Which element has the smallest first ionization energy?
A. N
B. C
C. B
D. Be
E. Li
E
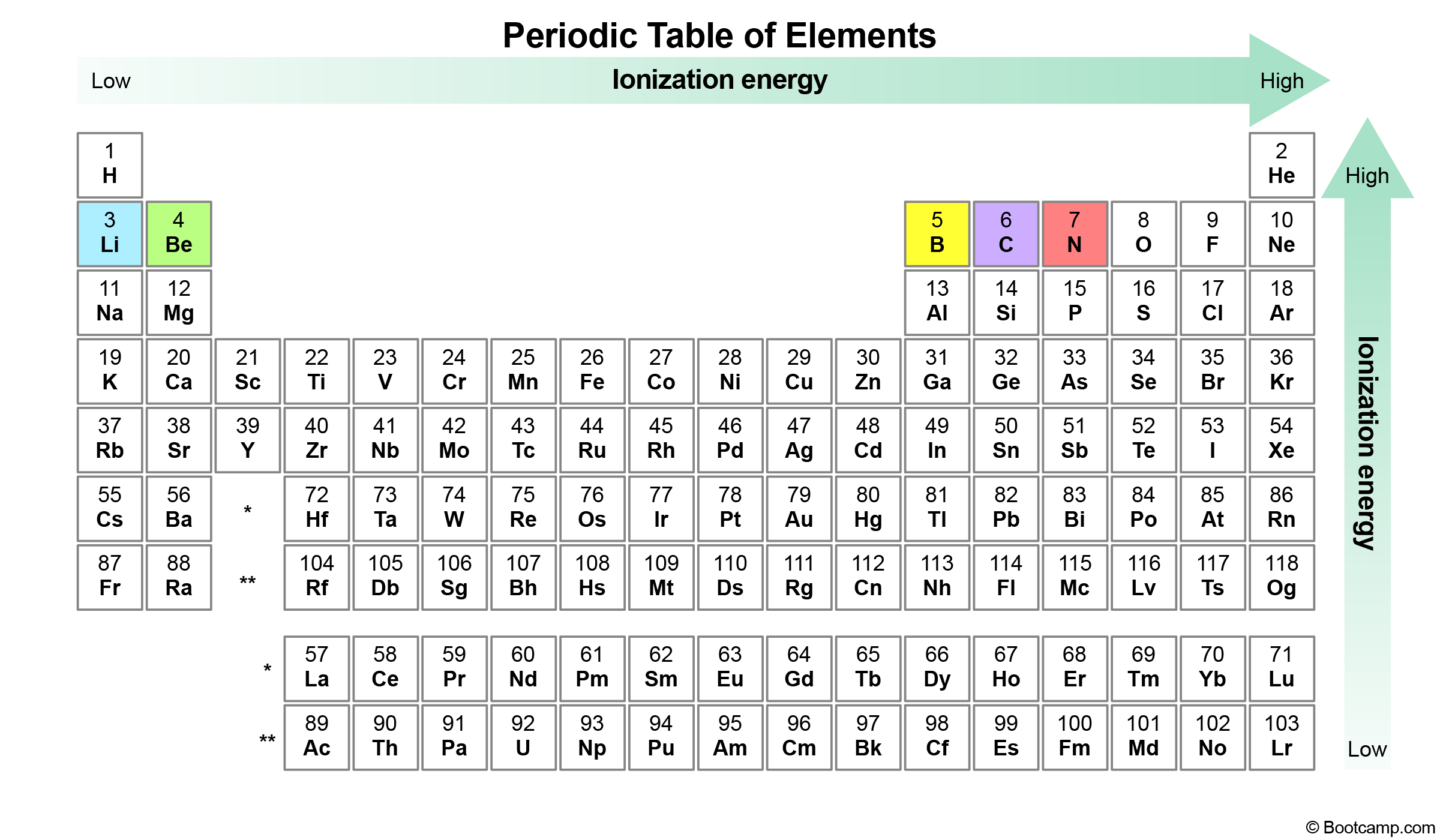
An element’s first ionization energy is the amount of energy needed to remove the first electron from a neutral atom. In general, first ionization energy increases going up and right in the periodic table.
Elements with a higher effective nuclear charge will have a higher first ionization energy as the increased attraction from the protons makes it more difficult to remove electrons from the atom (or "ionize them").
Notice that all of our answer choices are located in the same period in the periodic table. According to the periodic trend, the elements further to the left will have the smallest first ionization energy. Lithium (blue) is furthest to the left, and as a result it has the smallest first ionization energy.
Key Takeaway:
The periodic trend for first ionization energy increases as we go up and right in the periodic table.
Consider the phase diagram below for a certain substance. At which point does the gas phase start becoming indistinguishable from the liquid phase?
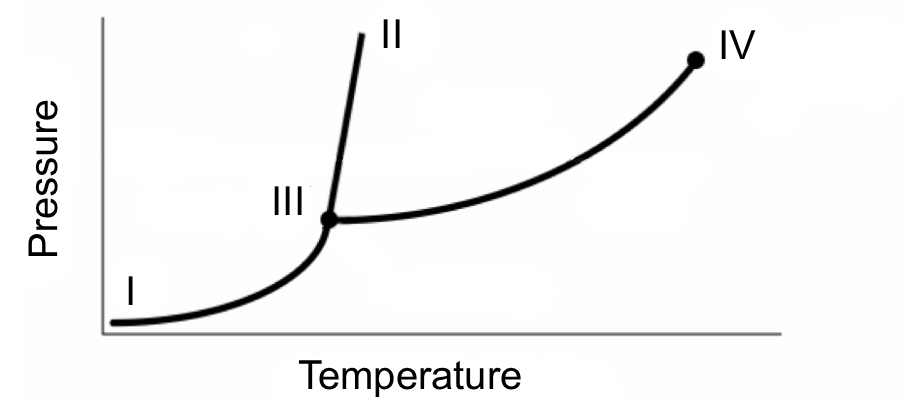
A. I
B. II
C. III
D. IV
D
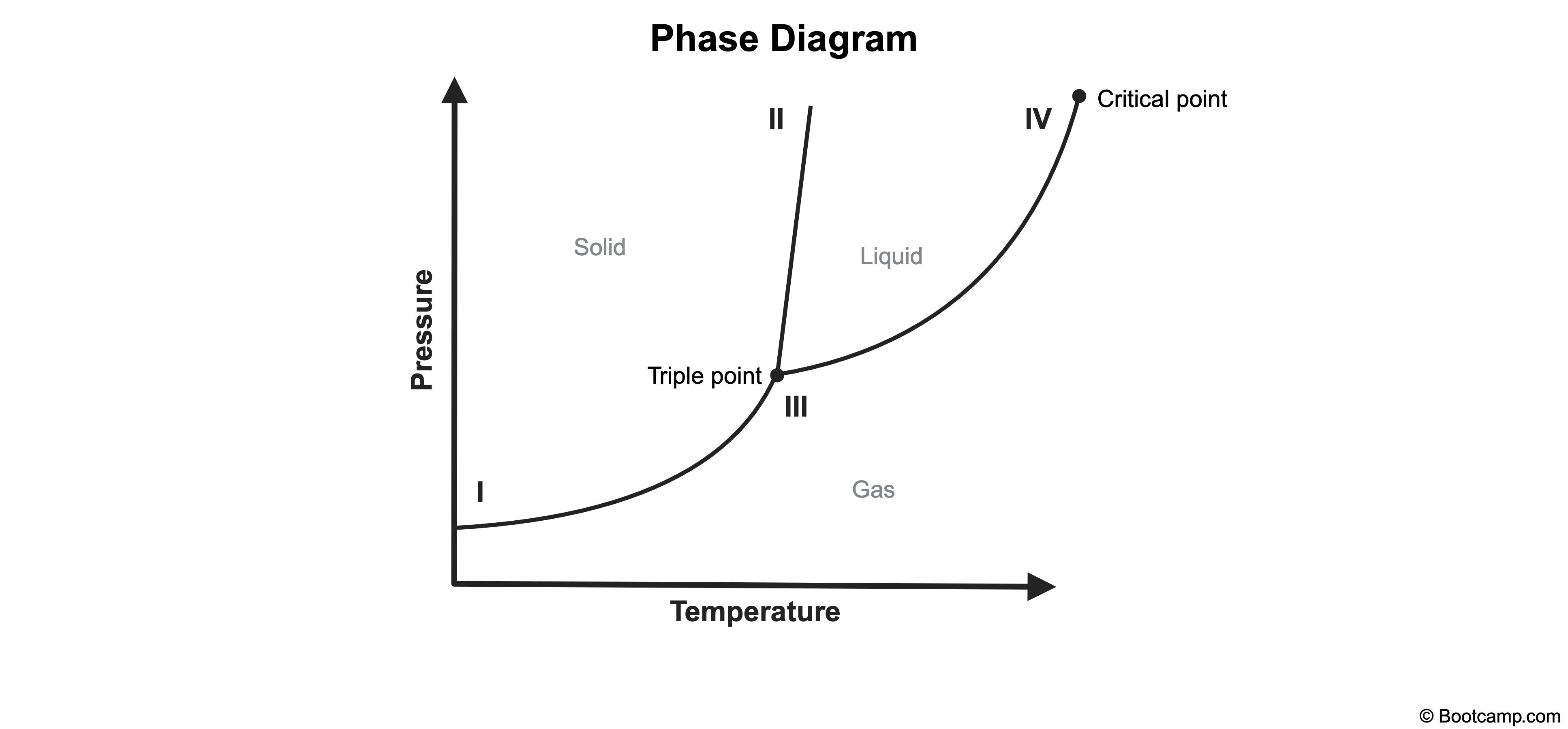
Lines of equilibrium indicate the conditions under which two phases can coexist in equilibrium or "phase transition" points. At equilibrium, the individual phases are still distinguishable from one another. However, the critical point (also called the end point) is located at very high temperatures and pressures at which the liquid and gas phases start merging together into a single phase.
Beyond the critical point, the liquid-gas equilibrium line no longer exists and liquid and gas phases become indistinguishable from one another.
A. I
Point I lies along the solid-gas equilibrium line. At point I, the solid and gas phases coexist in equilibrium and are distinguishable.
B. II
Point II lies along the solid-liquid equilibrium line. At point II, the solid and liquid phases coexist in equilibrium and are distinguishable.
C. III
Point III denotes the triple point, which designates the temperature and pressure at which the three phases (gas, liquid, solid) coexist in equilibrium and are distinguishable.
Key Takeaway:
In a phase diagram, the critical point designates the temperature and pressure beyond which the liquid and gas phases become indistinguishable.
What are the units of the rate constant (k) for the following reaction rate expression?
Rate = k[NO][CO₂]
A. s⁻¹
B. M⁻¹
C. M⁻¹s⁻¹
D. M⁻¹s⁻²
E. M⁻²s⁻¹
C
To determine the units of the rate constant (k), we must first determine the reaction order by adding the exponents in the rate law :
[NO] has 1 as its exponent.
[CO2] has 1 as its exponent.
1+1 = 2
This is a second-order reaction. The units of the rate constant (k) depend on the order of the reaction. Recall the table below to determine the rate constant units for a second-order reaction:
Reaction order | Rate constant (k) units |
First order | s-1 |
Second order | M-1s-1 |
Third order | M-2s-1 |
DAT Pro-Tip: The absolute value of adding the exponents present in the units for the rate constant (k) will be equal to the reaction order.
Key Takeaway:
First order reaction = s-1
Second order reaction = M-1s-1
Third order reaction = M-2s-1
Given the reaction 2A + B → C and the provided reaction data, what is the rate law?
Trial | [A] | [B] | Rate |
|---|---|---|---|
1 | 0.05 | 0.05 | 6.5 x 10⁻² |
2 | 0.05 | 0.1 | 6.5 x 10⁻² |
3 | 0.1 | 0.05 | 0.13 |
A. rate = k[A]¹[B]¹
B. rate = k[B]¹
C. rate = k[A]¹
D. rate = k[A]²[B]¹
E. rate = k[A]¹[B]²
C
The rate law is an expression that can be used to mathematically visualize the speed at which a reaction takes place. The general formula for the rate law is:
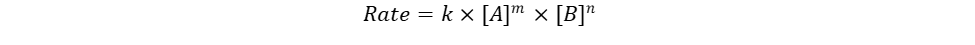
In this equation:
k is the rate constant.
[A] is the concentration of species A at a specific trial, and m is the reaction order with respect to A.
[B] is the concentration of species B at a specific trial, and n is the reaction order with respect to B.
The only unknown variables in the rate law are m and n, which represent the reaction orders. Since m represents the reaction order in terms of A, look at trials in the table where [A] changes and [B] remains constant. Trials 1 and 3 fall within these requirements; use them to calculate the reaction order m:
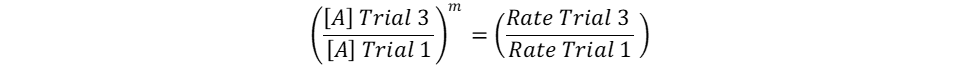
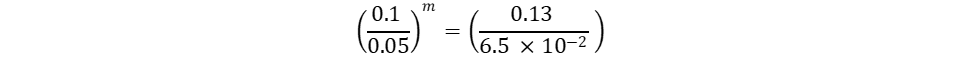
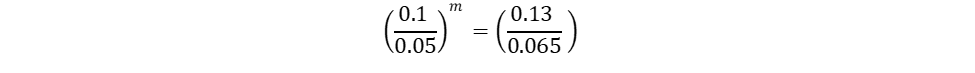
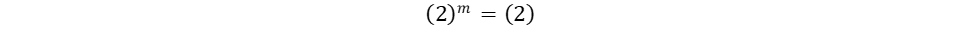
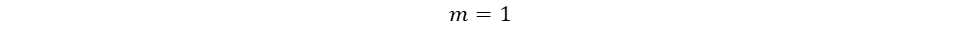
Repeat the same process for n, keeping in mind that it represents the reaction order in terms of B. Because of this, look at trials in the table where [B] changes and [A] remains constant. Trials 1 and 2 fall within these requirements; use them to calculate the reaction order n:
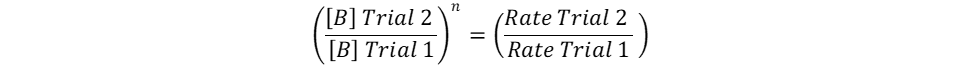
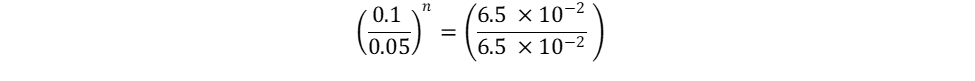
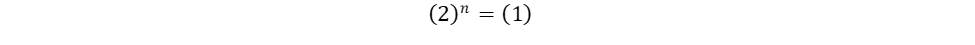
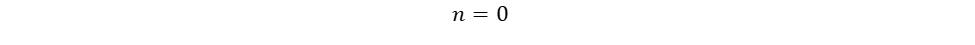
Finally, use this information to build the rate law:
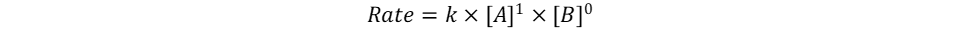
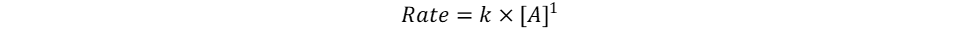
Key Takeaway:
To calculate the value of a reaction order, use the trials where the concentration of the corresponding species changes, while the concentration of any additional species remains constant.
Which of the following solutions will have a freezing point closest to that of pure water?
A. 1 m H₂SO₄
B. 1 m C₆H₁₂O₆
C. 1 m NaCl
D. 1 m AlCl₃
E. 1 m KCl
B
When a substance is dissolved in a solution, the solution's freezing point decreases. This occurs because the dissolved solutes interrupt the interactions of the solvent, making it more difficult to form a solid.
The formula for freezing point depression is:
ΔTf=iKfm
Where:
ΔTf is the change in the freezing point.
Kf is the freezing point depression constant for the solvent.
m is the molality of the solute.
i is the van’t Hoff factor.
The only two properties that are given in this problem are molality (all equal to 1 m) and the van't Hoff factor. This factor is the number of solute particles into which a compound breaks down. Because molality is the same for each solution, only the van't Hoff factor can be used to determine which compound will result in the smallest temperature change (i.e., therefore, be closest to the freezing point of pure water).
The following must be considered in the analyses of the solutions:
H2SO4: Sulfuric acid is a strong acid and dissociates completely into 2 ions: H+ and HSO4-. HSO4- can also further dissociate partially, but it is a weak acid. Therefore, 2 ≤ i ≤ 3.
C6H12O6: Glucose does not dissociate into ions in solution. Thus, i = 1.
NaCl: Sodium chloride dissociates into 2 ions: Na+ and Cl-. Thus, i = 2.
AlCl3: Aluminum chloride dissociates into 4 ions: Al3+ and 3Cl-. Thus, i = 4.
KCl: Potassium chloride dissociates into 2 ions: K+ and Cl−. Thus, i = 2.
The compound C6H12O6 has the smallest van't Hoff factor. Consequently, it results in the smallest freezing point depression and the final freezing point closest to that of pure water.
Key Takeaway:
The van't Hoff factor is directly proportional to the temperature change in the freezing point depression formula.
Which one of the following will fully dissociate in water?
A. HF
B. CH₃COOH
C. H₃PO₄
D. HNO₃
E. HSO₄⁻
D
When something completely dissociates in water it means the compound, when added to water, will completely break apart into its constituent ions. By definition, a strong acid is an acid that will completely ionize in water, while weak acids only partially ionize in water.
The following is a list of strong acids and bases that students should memorize for the DAT:
Strong Acids | Strong Bases |
HI | Group 1 metal hydroxides |
HBr | Mg(OH)2 |
HCl | Ca(OH)2 |
HClO3 | Sr(OH)2 |
HClO4 | Ba(OH)2 |
H2SO4 | |
HNO3 |
HNO3 is the only strong acid listed and will fully dissociate when added to water.
Mnemonic:
Seven strong acids: So I Brought No Clean Clothes.
So for sulfuric acid H2SO4.
I for HI.
Brought for HBr.
No for nitric acid HNO3.
Clean for HCl.
Clothes for chloric, and perchloric acid, HClO3, HClO4.
A. HF
Hydrofluoric acid is the only hydrohalic acid that is not a strong acid. It is important to know that this is due to bond length, where the relatively short (and therefore, strong) bond between the H and F atoms will not allow HF to dissociate completely in water.
B. CH3COOH
Acetic acid is another important acid to memorize for the DAT. It is a carboxylic acid and does not completely dissociate in water, and can also be called ethanoic acid.
C. H3PO4
Phosphoric acid is the acid form of the phosphate ion, PO43-, an important ion to memorize for the DAT. It is a weak acid.
E. HSO4-
Although it is tempting to think that hydrogen sulfate is a strong acid because sulfuric acid, H2SO4, is a strong acid, it is a weak acid. Sulfuric acid will dissociate into H+ and HSO4-, in water, but HSO4- will not completely dissociate into H+ and SO42-.
Key Takeaway:
HNO3 is a strong acid, meaning it completely dissociates into H+ and NO3- when placed in water.
What are the units for the rate constants of first, second, and third-order reactions?
Reaction order | Rate constant (k) units |
First order | s-1 |
Second order | M-1s-1 |
Third order | M-2s-1 |
What are the strong acids and strong bases?
Strong Acids | Strong Bases |
HI | Group 1 metal hydroxides |
HBr | Mg(OH)2 |
HCl | Ca(OH)2 |
HClO3 | Sr(OH)2 |
HClO4 | Ba(OH)2 |
H2SO4 | |
HNO3 |
Given the following standard reduction potentials:
Cu²⁺(aq) + 2e⁻ → Cu(s) E°red = +0.34 V Zn²⁺(aq) + 2e⁻ → Zn(s) E°red = -0.60 V
What is the E°cell for the following reaction?
Zn(s) + Cu²⁺(aq) → Zn²⁺(aq) + Cu(s)
A. - 0.94 V B. - 0.26 V C. + 0.26 V D. +0.94 V
D
The given E°red is the standard reduction potentials for the two half-reactions that make up our full chemical equation.
The half-reaction with the more positive E°red will be reduced (stronger oxidizing agent) in the full chemical equation.
The half-reaction with the less positive E°red will be oxidized (stronger reducing agent).
Therefore, looking at the two half-reactions we see that Cu2+ will be reduced and Zn metal will be oxidized.
To find the voltage produced by an electrochemical cell, we simply find the sum of the potentials in the circuit: E°cell = E°red + E°ox.
We can find E°ox by simply reversing the reduction half-reaction of Zn so that it reads:
Zn(s) → Zn2+(aq) + 2e-
When we reverse a half-reaction, we change the sign of its E°; thus the E°red = -0.60 V becomes E°ox = +0.60 V. Plugging this into our equation for E°cell gives the following:
E°cell = 0.34 V + 0.60 V = 0.94 V
Key Takeaway:
The half-reaction with a higher E°red will be reduced while the half-reaction with a lower E°red will be oxidized.
Which of the following is the correct hybridization of the central carbon atoms in 2-butene (C4H8)?
A. sp
B. sp2
C. sp3
D. sp3d
E. sp3d2
B
To determine the hybridization of the central carbon atoms in 2-butene (C4H8), draw the structure of 2-butene:
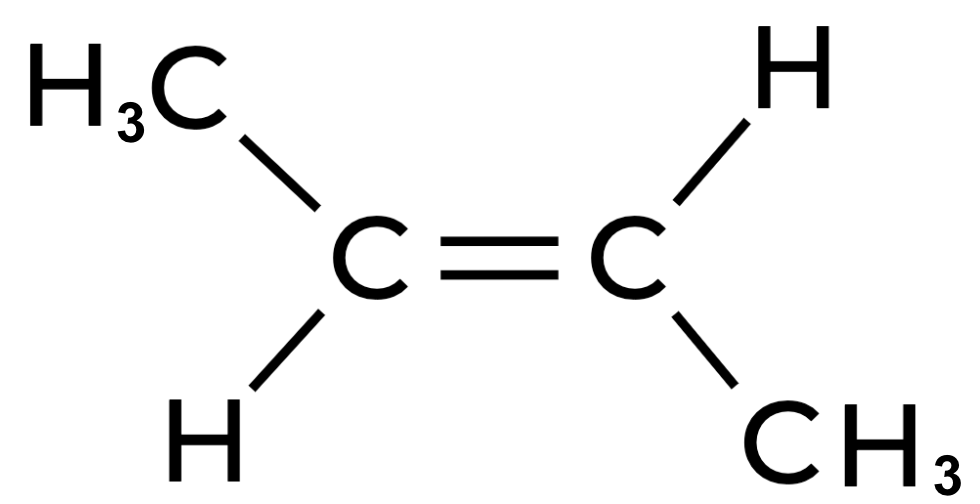
Each central carbon atom forms a single bond with a hydrogen atom, a single bond with a methyl group, and a double bond with the other central carbon atom. Thus, each central carbon atom has three electron domains, which correlates to an sp2 hybridization.
Key Takeaway:
The number of electron domains surrounding the central atom determines the central atom's hybridization.
Which of the following indicates that a reaction is at equilibrium?
A. ΔH = 0
B. ΔS = 0
C. [Reactants] = [Products]
D. Q = K
E. Forward rate = 0
D
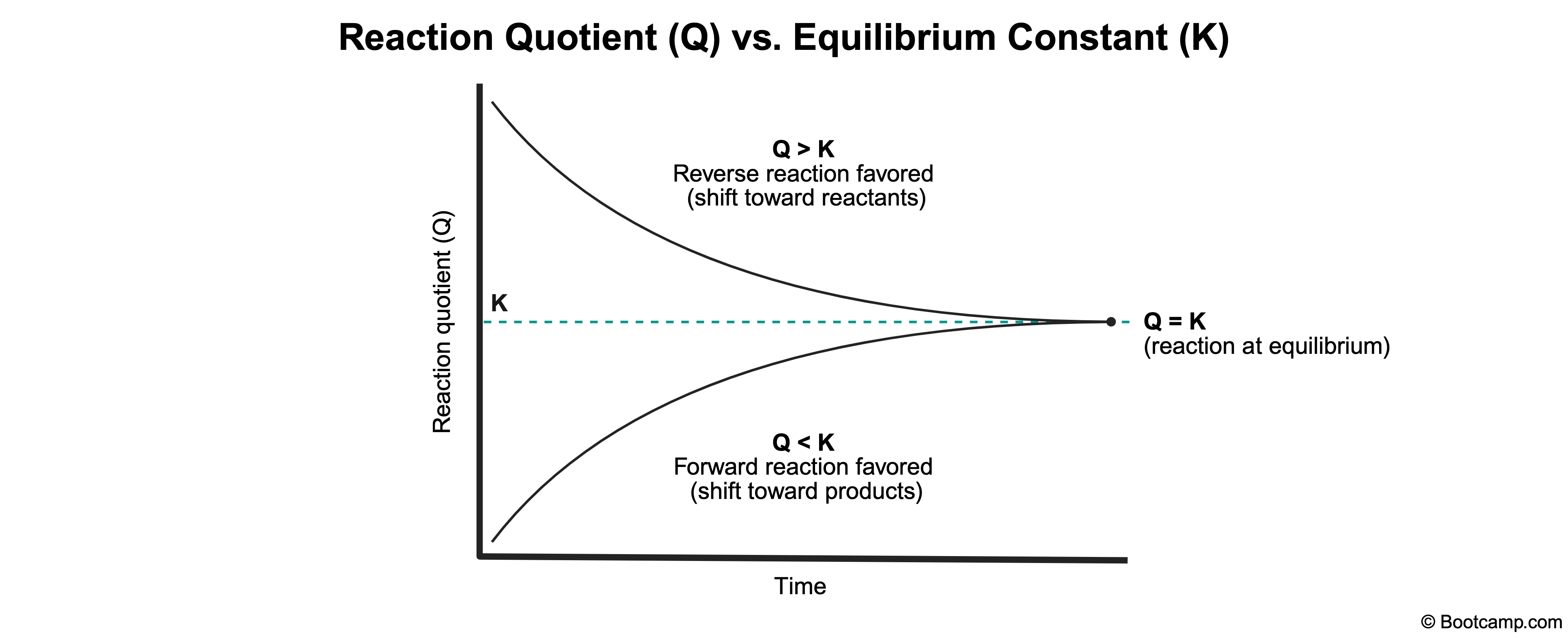
The reaction quotient (Q) and the equilibrium constant (K) both represent the ratio of multiplied product concentrations to multiplied reactant concentrations, but they differ in application. While Q can be calculated at any point during a reaction, K only applies when the system is at equilibrium.
When Q = K, the reaction is at equilibrium, meaning the rates of the forward and reverse reactions are equal.
A. ΔH = 0
The change in enthalpy (ΔH) of a reaction measures the amount of heat absorbed or released. It does not indicate whether a reaction is at equilibrium.
B. ΔS = 0
The change in entropy (ΔS) measures the amount of disorder or randomness in a system. It does not signify whether a reaction has reached equilibrium.
C. [Reactants] = [Products]
At equilibrium, the concentrations of reactants and products do not necessarily have to be equal. Instead, their concentration ratio remains constant and is defined by the equilibrium constant (K) for the reaction.
E. Forward rate = 0
A reaction is at equilibrium when the forward rate = reverse rate, not when the forward rate reaches 0.
Key Takeaway:
A reaction is at equilibrium when the reaction quotient (Q) is equal to the equilibrium constant (K).
In the process of electroplating a silver object with gold, which reaction must occur at the cathode?
A. Ag⁺ + e⁻ → Ag
B. Ag → Ag⁺ + e⁻
C. Au³⁺ + 3e⁻ → Au
D. Au → Au³⁺ + 3e⁻
C
Electroplating is an application of electrolytic cells that uses an applied electric current to plate a substance in layers of metal.
In electroplating, the anode and cathode are placed in a solution containing metal ions, and an external power source is used to drive the movement of electrons from the anode to the cathode.
This movement of electrons imparts a positive charge to the anode, and the anode gradually dissolves to donate metal ions to the solution. In contrast, the cathode is negatively charged. Positively charged metal ions in the solution are drawn to the negatively charged cathode, where they are reduced back into solid metal and deposited on the surface of the cathode.
To coat the given object in gold, gold ions must be reduced out of the solution to electroplate the cathode. The equation representing the reduction of gold is:
Au3+ + 3e- → Au
Key Takeaway:
In electrolytic cells, the anode is positively charged, and the cathode is negatively charged.
Electroplating relies on the reduction of metal ions to coat a substance in metal.
How does the viscosity of a liquid change with an increase in temperature?
A. Decreases as intermolecular attractions are disrupted
B. Decreases as intramolecular bonds are broken
C. Increases as molecules gain kinetic energy
D. Increases as the liquid solidifies
E. Increases due to more frequent molecular collisions
A
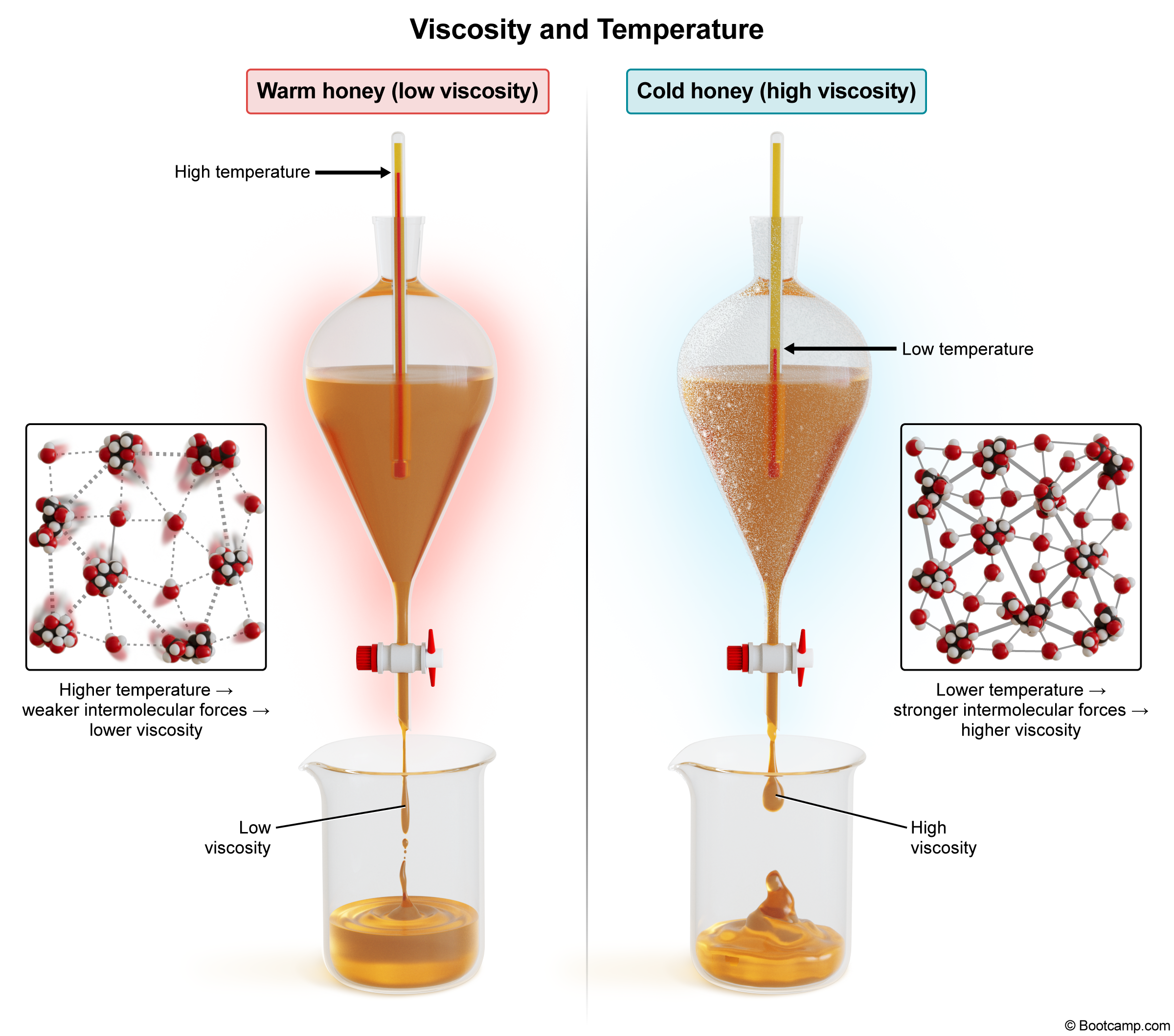
Viscosity refers to a liquid's resistance to flow. It is influenced by the strength of the intermolecular forces between the liquid's molecules. As temperature increases, the molecules gain kinetic energy and move more rapidly. This increased motion weakens the intermolecular forces, allowing the liquid to flow more easily.
Thus, viscosity decreases as temperature increases because intermolecular attractions are disrupted.
B. Decreases as intramolecular bonds are broken
Intramolecular bonds, which are bonds within molecules, typically remain intact when a liquid is heated. A decrease in viscosity results from weakened intermolecular forces, not from the breaking of intramolecular bonds.
C. Increases as molecules gain kinetic energy
Higher kinetic energy allows molecules to overcome intermolecular forces, decreasing viscosity.
D. Increases as the liquid solidifies
As a liquid cools and solidifies, viscosity increases. However, since the temperature is increasing, the liquid moves away from solidification, causing viscosity to decrease.
E. Increases due to more frequent molecular collisions
Although an increase in temperature results in more frequent molecular collisions, it does not increase viscosity. Instead, higher temperatures decrease viscosity by reducing intermolecular attractions.
Key Takeaway:
Viscosity decreases as temperature increases because intermolecular attractions are disrupted.
A buffer is prepared from H₂PO₄⁻ / HPO₄²⁻ with a pKa of 7.2. If the pH is adjusted to 10, which species is present in a higher concentration?
A. H₃PO₄
B. H₂PO₄⁻
C. HPO₄²⁻
D. PO₄³⁻
C
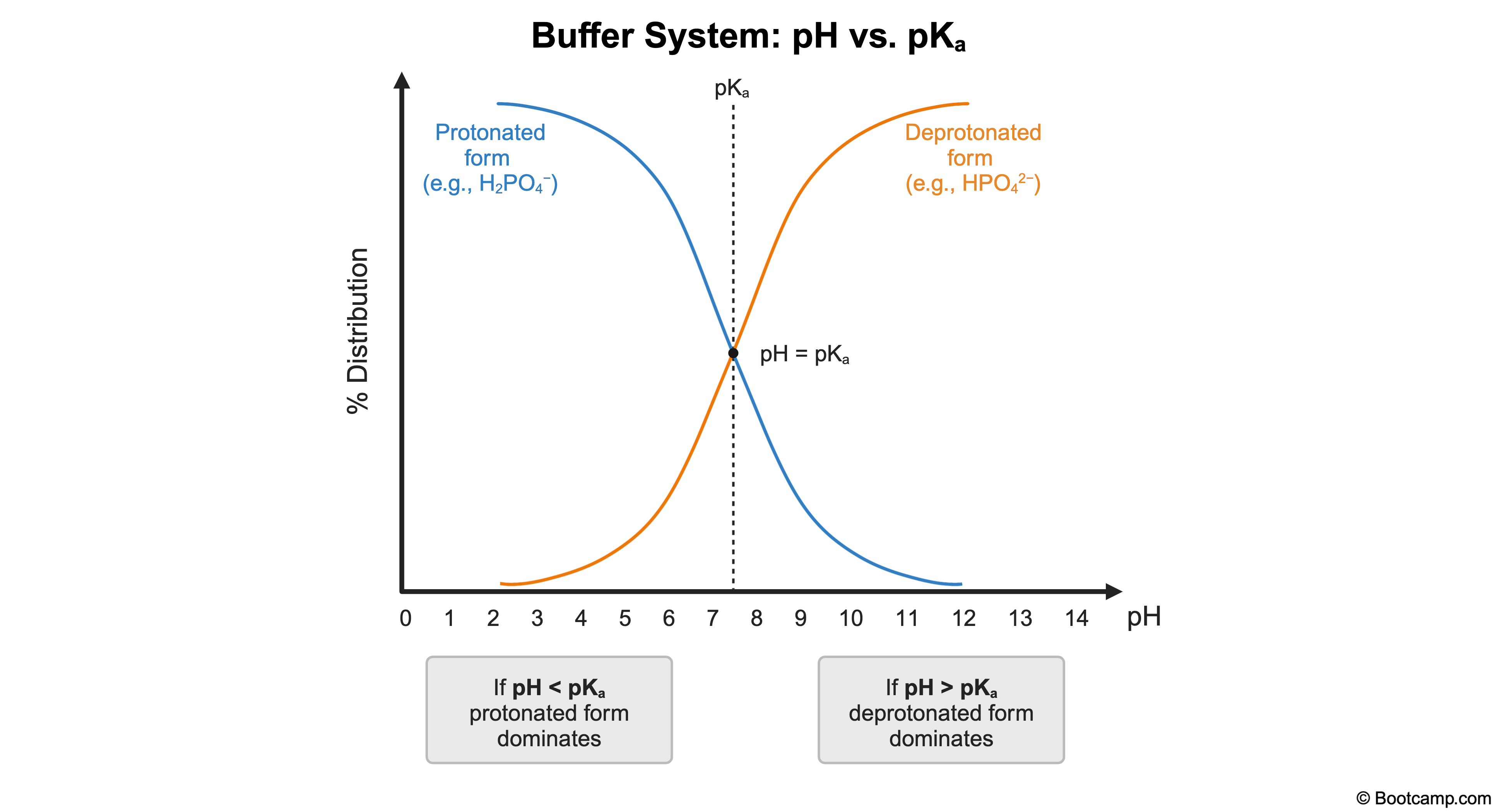
The pKa of a buffer system represents the pH at which the protonated and deprotonated species exist in equal concentrations.
For an H2PO4- / HPO42- buffer with pKa = 7.2:
When pH < pKa, H2PO4- (protonated form) predominates.
When pH > pKa, HPO42- (deprotonated form) predominates.
Since the pH = 10 (pH > pKa), the predominant species is HPO42-.
Key Takeaway:
When pH < pKa, the protonated form predominates.
When pH > pKa, the deprotonated form predominates.
A mixture of NO₂ and CO was allowed to react in a closed container, establishing equilibrium according to the following exothermic reaction (ΔH < 0):
NO₂(g) + CO(g) ⇌ NO(g) + CO₂(g)
Which of the following changes would increase the concentration of CO₂?
A. Adding NO to the container
B. Adding NO₂ to the container
C. Decreasing the pressure
D. Increasing the temperature
E. Removing CO from the container
B
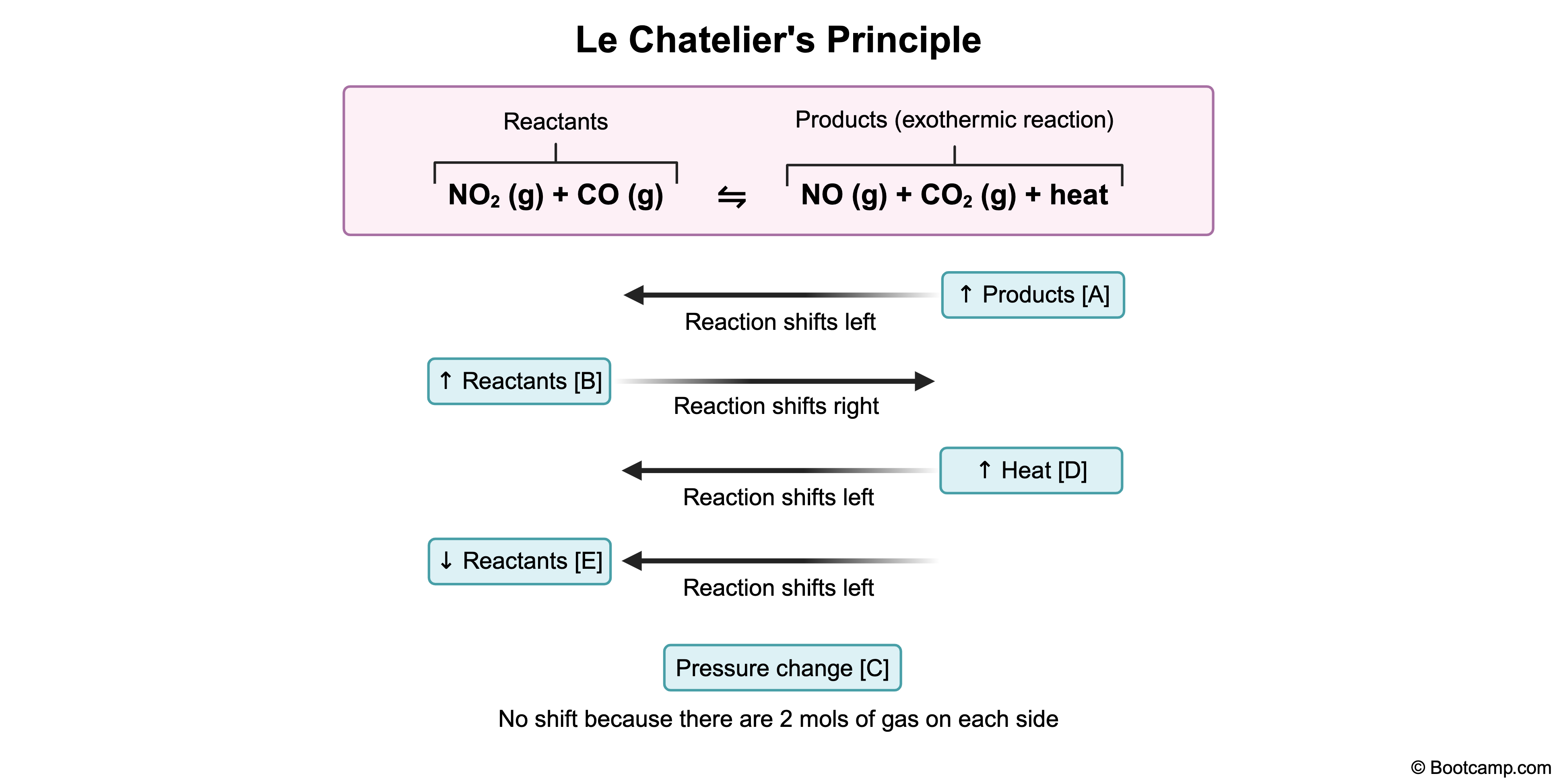
NO2(g) + CO(g) ⇌ NO(g) + CO2(g)
According to Le Chatelier's principle, a system at equilibrium will adjust to counteract any change imposed upon it. To increase the concentration of a product such as CO2, the equilibrium must shift to the right, favoring product formation.
Adding NO2 increases the concentration of a reactant. In response, the system shifts the equilibrium to the right to use up the excess NO2, converting more reactants into products. As a result, the CO2 concentration increases.
A. Adding NO to the container
Adding NO to the container increases the concentration of a product. In response, the system shifts the equilibrium to the left, decreasing the concentration of CO2.
C. Decreasing the pressure
Both sides of the reaction have two moles of gas. Therefore, changing the pressure does not affect the equilibrium position of this reaction.
D. Increasing the temperature
This is an exothermic reaction, meaning heat functions as a product. Thus, when temperature increases, the equilibrium shifts to the left, decreasing the concentration of CO2.
E. Removing CO from the container
Removing CO from the container decreases the concentration of a reactant. In response, the system shifts the equilibrium to the left to replenish the CO, decreasing the concentration of CO2.
Key Takeaway:
According to Le Chatelier's principle, increasing the concentration of a reactant shifts the equilibrium to the right, favoring product formation.
Which of the following reactions will be spontaneous at all temperatures?
A. 2H₂(g) + O₂(g) → 2H₂O(g), ΔH = –483.6 kJ
B. 2NCl₃(g) → N₂(g) + 3Cl₂(g), ΔH = –460.1 kJ
C. 2Na(s) + Cl₂(g) → 2NaCl(s), ΔH = –821.8 kJ
D. N₂(g) + O₂(g) → 2NO(g), ΔH = +180.5 kJ
E. C(s) + 2S(s) → CS₂(g), ΔH = +92.0 kJ
B
ΔH | ΔS | ΔG | Impact on the reaction |
– | + | – | Spontaneous at all temperatures |
+ | – | + | Nonspontaneous at all temperatures |
– | – | Depends | Spontaneous at low temperatures |
+ | + | Depends | Spontaneous at high temperatures |
A spontaneous process is one where ΔG is negative, according to the equation:
ΔG=ΔH−TΔS
For a process to be spontaneous at all temperatures, the following must be true for enthalpy (ΔH) and entropy (ΔS):
ΔH < 0 (exothermic)
ΔS > 0 (increasing in disorder)
ΔH is given in the answer choices, so to find ΔS, calculate the change in the number of moles of gas. Because gases have greater entropy than solids and liquids, a decrease in the number of gas moles leads to a negative ΔS, while an increase in the number of gas moles leads to a positive ΔS.
Choice [B] has a negative ΔH coupled with a positive ΔS due to more moles of gas on the products side. This makes it spontaneous at all temperatures.
A. 2H2(g) + O2(g) → 2H2O(g), ΔH = –483.6 kJ
The reactants have 3 moles of gas, whereas the products have 2 moles of gas; therefore, ΔS is negative and the reaction will be spontaneous only at low temperatures.
C. 2Na(s) + Cl2(g) → 2NaCl(s), ΔH = –821.8 kJ
The reactants have 1 mole of gas, whereas the products have 0 moles of gas; therefore, ΔS is negative and the reaction will be spontaneous only at low temperatures.
D. N2(g) + O2(g) → 2NO(g), ΔH = +180.5 kJ
E. C(s) + 2S(s) → CS2(g), ΔH = +92.0 kJ
When ΔH is positive, the reaction will not be spontaneous at all temperatures.
Key Takeaway:
A reaction is spontaneous at all temperatures only when ΔH is negative and ΔS is positive.
ΔH | ΔS | ΔG | Impact on the reaction |
– | + | – | Spontaneous at all temperatures |
+ | – | + | Nonspontaneous at all temperatures |
– | – | Depends | Spontaneous at low temperatures |
+ | + | Depends | Spontaneous at high temperatures |
Memorize spontaneous VS nonspontaneous @ high VS low temperatures
Which of the following represents a conjugate acid-base pair?
A. H₂O and H⁻
B. HCO₃⁻ and CO₃²⁻
C. H₃PO₄ and PO₄³⁻
D. H₂O and O²⁻
E. H₃O⁺ and HCO₃⁻
B
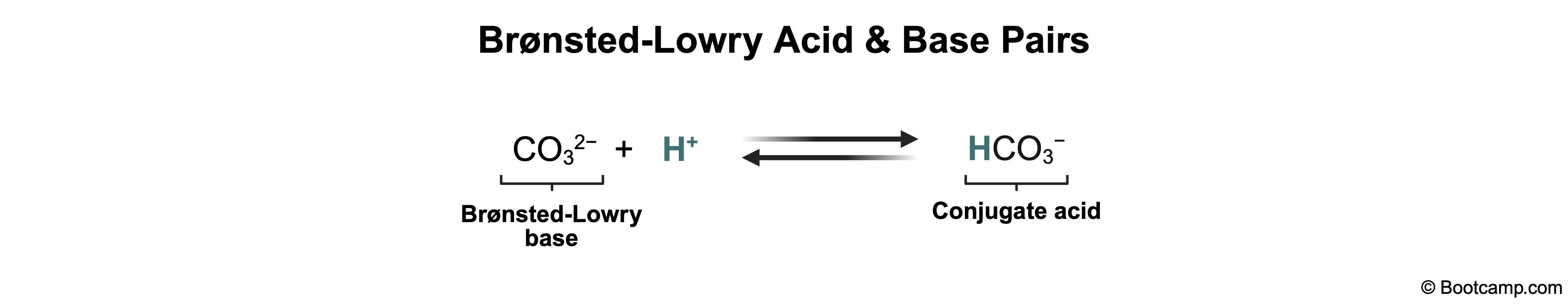
In an acid-base reaction, a Brønsted-Lowry base is any species that accepts a proton (H+). When a base gains a proton, it forms its conjugate acid. Thus, the base and its conjugate acid differ by exactly one proton.
CO32- acts as a base, forming HCO3- when it accepts one proton. Therefore, HCO3- and CO32- are a conjugate acid/base pair.
A. H2O and H-
E. H3O+ and HCO3-
These pairs differ by more than just one proton. They also differ in the number or types of other atoms, meaning they are not conjugate acid/base pairs.
C. H3PO4 and PO43-
D. H2O and O2-
These pairs differ by multiple protons. Conjugate acid/base pairs differ by only one proton, so these answer choices do not represent conjugate acid/base pairs.
Key Takeaway:
When a base accepts a single proton, it forms its conjugate acid.