Orgo 2 Lab Final
1/119
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
120 Terms
to convert a primary alcohol to an aldehyde using a recently discovered Cu-based aerobic oxidation catalyst system instead of traditional oxidizing reagents (PCC, CrO3, KMnO4)
exp 1: what is the purpose of the experiment?
often highly toxic to the environment, typically used in stoichiometric amounts (same molar amount as substrate being oxidized), they yield by-products that need to be separated from desired product, they often result in over oxidation to a carboxylic acid for example
exp 1: what are some disadvantages of traditional oxidizing reagents (PCC, CrO3, KMnO4)
their focused on the developement of catalytic systems that enable the use of atmospheric O2 as the stoichiometric oxygen source
exp 1: what are researchers now using to oxidize substrates in their experiments instead of normal oxidizing reagents?
an additive to a chemical rxn that increases the rate of a chemical rxn without being consumed
exp 1: what is a catalyst?
it must be non toxic and have byproducts that aren't harmful to the environment
exp 1: what makes an oxidizing agent "green"?
CuBr, BPY, NMI, TEMPO in combination with atmospheric oxygen
exp 1: what does the catalytic system consist of in the experiment?
atmospheric oxygen (oxygen in the air)
exp 1: what is the actual oxygen source in the experiment?
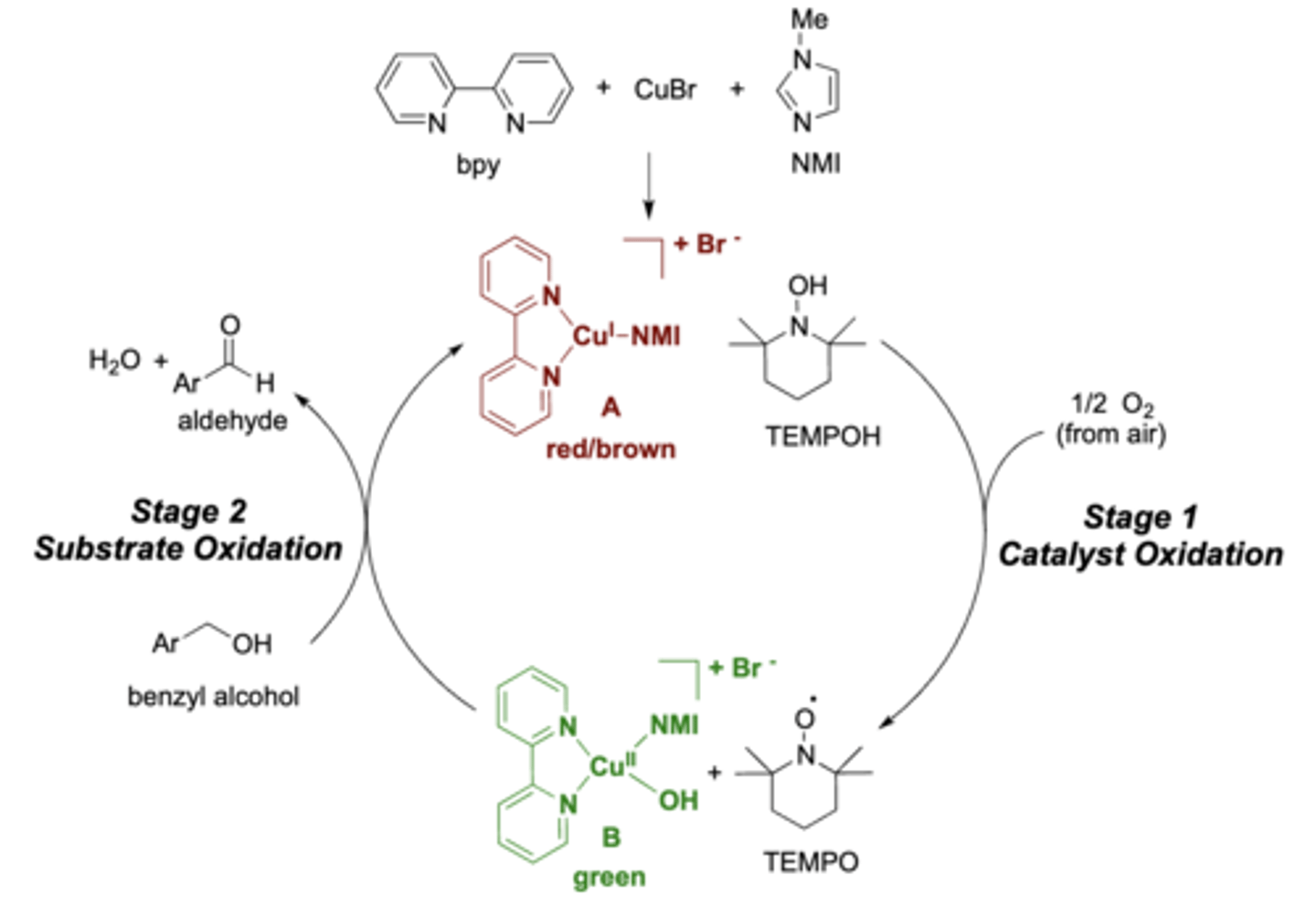
the solution will turn a green color
exp 1: how do you know the reaction is complete?
stage 1: the catalyst was oxidized by atmospheric oxygen
stage 2: the substrate (benzyl alcohol) was oxidized by the now-oxidized catalyst system
exp 1: explain stage 1 and 2 of the reaction
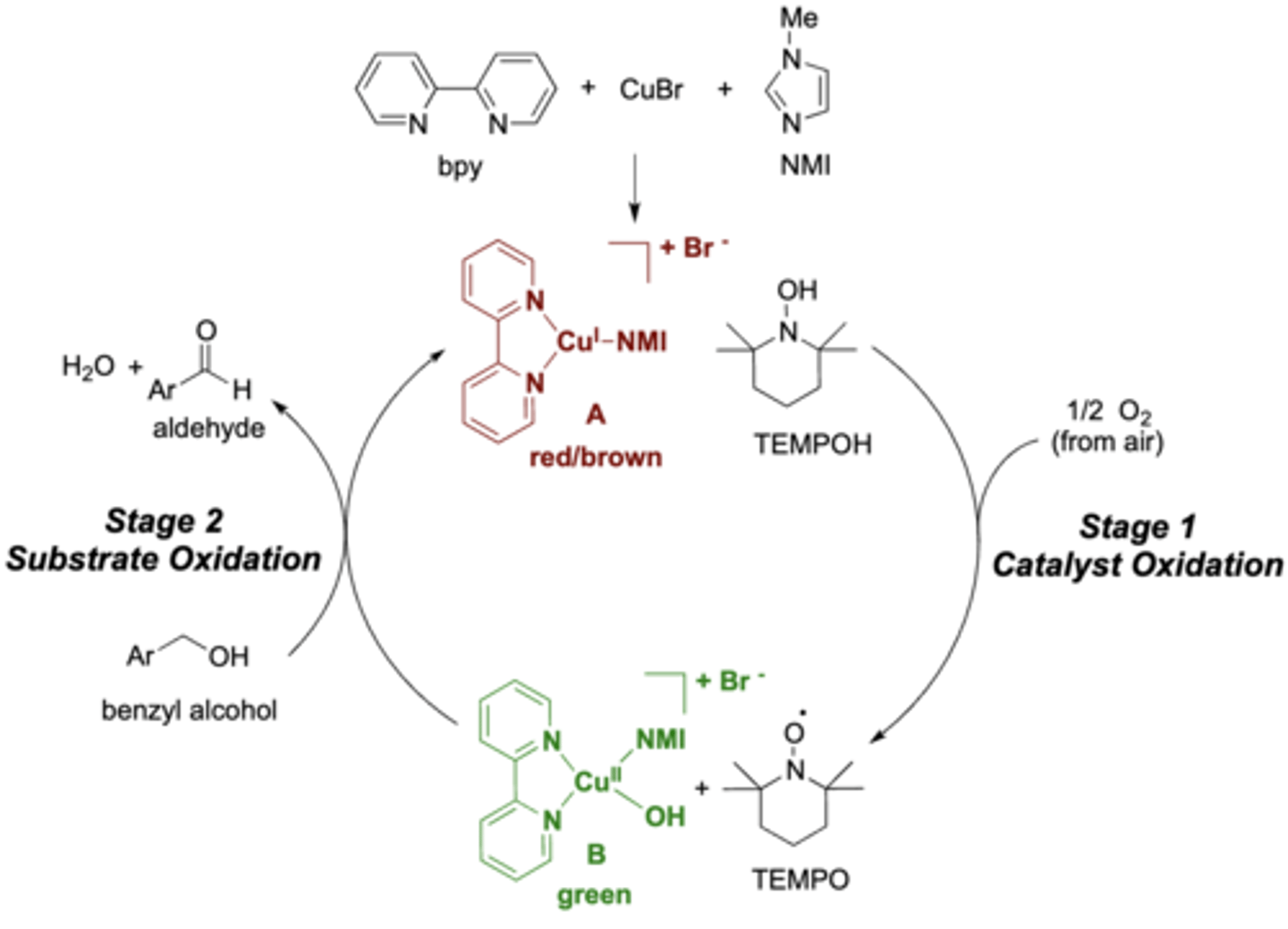
stable organic radical (on the oxygen)
exp 1: in stage 1 TEMPO formed a....?
top layer: organic (pentane solvent is less dense than water)
bottom: aqueous
exp 1: which is the top and bottom layer? why?
We used IR to check if the obvious alcohol peak disappeared, should have become an aldehyde instead (should see peak around 1700 cm^-1)
exp 1: how do we know that the rxn worked?
over oxidizing would have gone past the aldehyde to the carboxylic acid, carboxylic acids have a distinct IR peak and an extremely shifted hydrogen (10-12 PPM) in NMR that would indicate if this happened
exp 1: what would have been the result of over-oxidizing the product? How could we tell with IR/NMR?
nitrogen gas
exp 1: what did we use to evaporate the solvent?
add benzyl alcohol, acetone, CuBr to rxn flask, rapidly stir then add bpy, TEMPO, and NMI in order, rapidly stir until significant color change, transfer mixture to seperatory funnel and add water and pentane to form organic and aqueous layers, extract organic layer twice with added pentane (remember pentane is less dense than water so organic layer will be at top), dry organic product with MgSO4, use nitrogen gas to evaporate solvent, analyze product with IR
exp 1: summarize the procedure
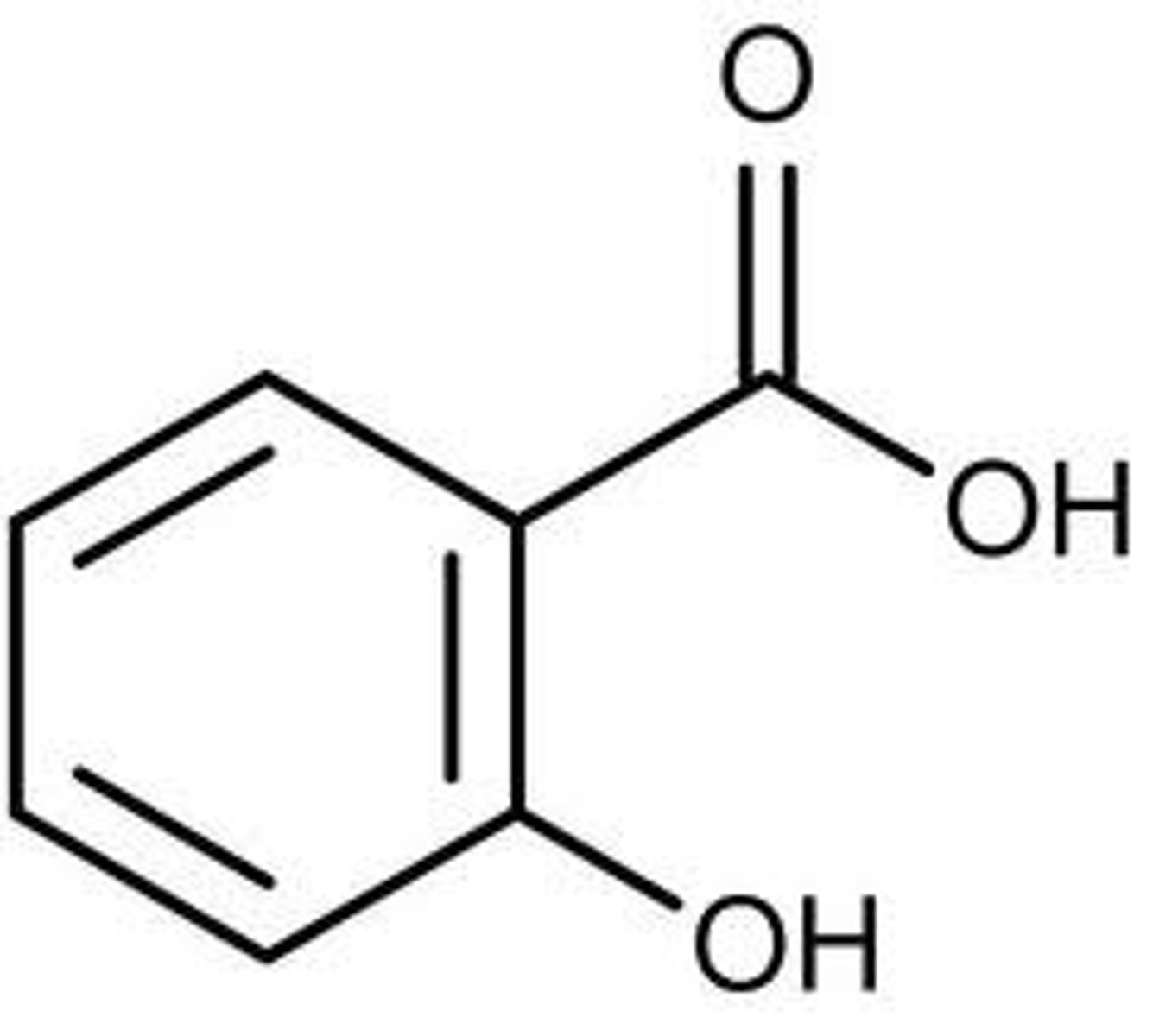
its a basic intermediate in the synthesis of mesalazine, a compound used to treat ulcerative colitis, crohn's disease, irritable bowel disorder)
exp 2: why should 5-nitrosalicyclic acid be synthesized?
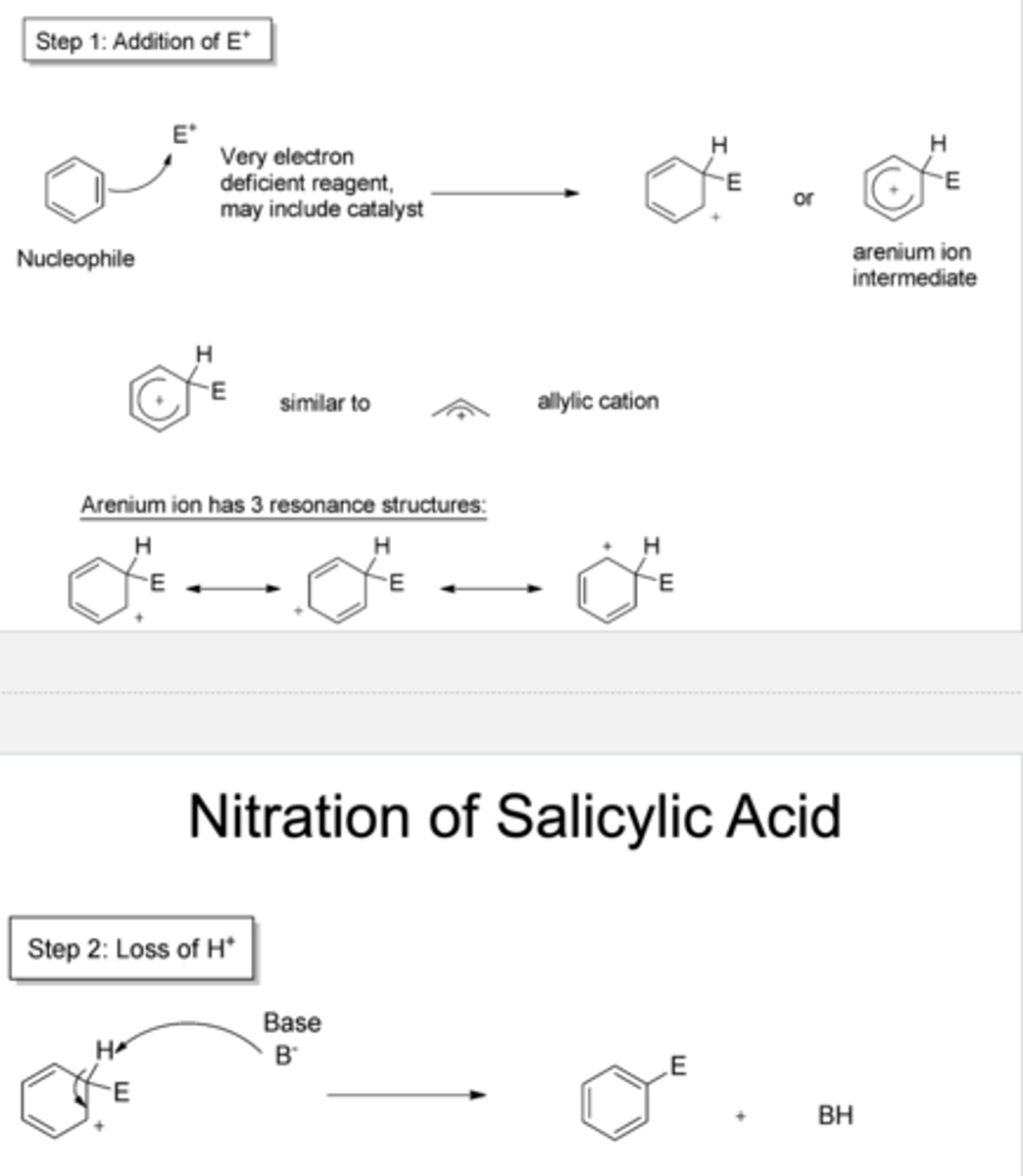
electrophilic aromatic substitution
exp 2: what type of rxn is the nitration of salicyclic acid?
they're electron rich and they have resonance
exp 2: why are aromatic compounds more stable that non-aromatic?
step 1 is slow since aromaticity is lost
step 2 is fast since aromaticity is gane
exp 2: why is step 1 of the nitration rxn slow & step 2 fast?
depending on the substituents, it can make the reactant more reactive and result in specific regiochemistry (ortho, para, meta)
exp 2: what happens if there are already substituents on the aromatic ring prior to EAS, as in the salicylic acid starting material?
more
exp 2: is salicyclic acid more reactive than benzene?
position meta to COOH, and para to OH. COOH is a meta director (deactivator) and OH is a ortho para director (activator)
exp 2: if a reagent was to be added to salicyclic acid, where would it get added to and why?
nucleophile: salicyclic acid
electrophile: +NO2 (nitronium ion)
exp 2: whats the nucleophile and electrophile in the rxn?
resonance stabilized EAS intermediate
exp 2: whats an arenium ion/sigma complex?
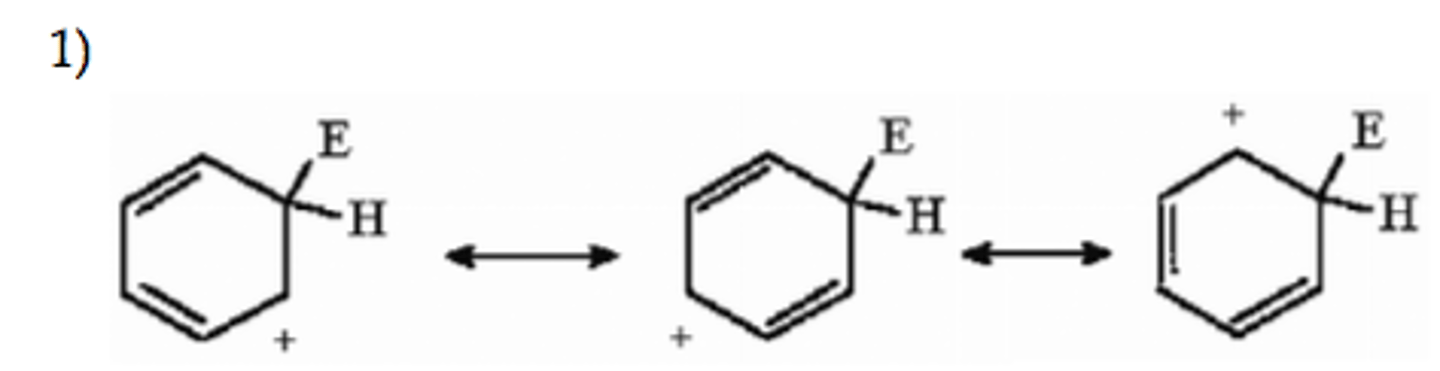
the first step is limiting/rate determining because it breaks aromaticity
exp 2: what is the rate determining step in the reaction?
add salicyclic acid, nitric acid, and sulfuric acid (catalyst) to rxn mixture, stir mixture at low temp (ice water bath), isolate product by adding more ice cold water, collect precipitate by vacuum filtration, add more cold water and ethanol to fully dissolve product and recrystallize with vacuum filtration, analyze dry product with IR, HNMR, and melting point analysis
exp 2: summarize procedure
steric hindrance from OH discourages nitration. nitration will occur at the 5th carbon as long as low temperature is maintained
exp 2: why is the 5th carbon position (kinetic product) favored in the reaction?
nitration would occur at the 3rd carbon (thermodynamic product) because the heat would overcome activation energy
exp 2: what would happen if the reaction was run in a hot water bath instead of cold?
in theory nitration can occur at either the 5th or 3rd carbon
exp 2: why might we not get 100% yield of our target product in the reaction?
salicyclic acid. because it's the main molecule you want to modify, it's the one being nitrated to form your product, so you want all to be converted to the target product
exp 2: whats the limiting reagent in the reaction? why?
sulfuric acid. because it helps start the reaction without being consumed overall
exp 2: whats the catalyst in the reaction? why?
sulfuric acid acts as a catalyst, not a reactant that gets consumed in the overall balanced equation. Meanwhile, salicylic acid is a reactant — it's actually undergoing a chemical change (getting nitrated), so it needs to be added in a stoichiometric (measured, reactant-proportional) amount.
Since sulfuric acid helps the reaction but isn't used up permanently, you don't have to match it stoichiometrically. Instead, you just add enough to:
fully protonate HNO₃
soak up the water
maintain the right acidic environment.
In fact, it's usually added in excess to make sure the reaction runs efficiently.
exp 2: why doesnt sulfuric acid need to be added in stoichiometric amounts like salicylic acid?
NO2 peak at 1350 cm^-1 (unreliable)
exp 2: what should we see on the IR to confirm we have our product?
only 3 aromatic hydrogens, and they would be more downfield
exp 2: what should we see at on the HNMR to confirm we have our product?
to aid in dissolving of all components in reaction mixture
exp 2: why is ethanol added after dissolving the crude product in water?
we put them in conical vials and transported them that way
exp 2: how did we get the nitric and sulfuric acid to our hood from the reagent hood?
it is less sterically hindered (not next to the alcohol substituent) and therefore formed more quickly
exp 2: why is the 5-nitro product the kinetic product?
to synthesize (E,E)-1,4-diphenyl-1,3-butadiene so it can beused in the Diels-Alder reaction toward synthesizing p-terphenyl
exp 3: what is the purpose of this experiment?
first combine benzyl alcohol and triphenylphosphine (Sn2 rxn) and p-cymene, heat the rxn mixture under reflux and stir, then cool in ice water bath, collect product by vacuum filtration, then dry benzyltriphenylphosphonium chloride (product of first step), then in a conical vial combine trans-cinnamadehyde, DCM and our first product benzyltriphenylphosphonium chloride, stir and then add sodium hydroxide while stirring, then transfer mixture to centrifuge tube and separate using DCM and water, extract product into the DCM layer (organic layer) and dry with anhydrous sodium sulfate, evaporate DCM to leave solid product, add ethanol to solid product and triturate (grind down solid), this will remove triphenylphosphine oxide (by product) and other impurities. collect solid by vacuum filtration (wash filter with ethanol) and then recrystallize then dry the product again and analyze product by measuring mass & melting pt
exp 3: summarize procedure
combined an ylide and an aldehyde (trans-cinnamaldehyde) to form an alkene
exp 3: this experiment combined a ___________ and ____________ to form a __________
to remove the triphenylphosphine oxide byproduct of a wittig reaction
exp 3: why was the product "triturated"?
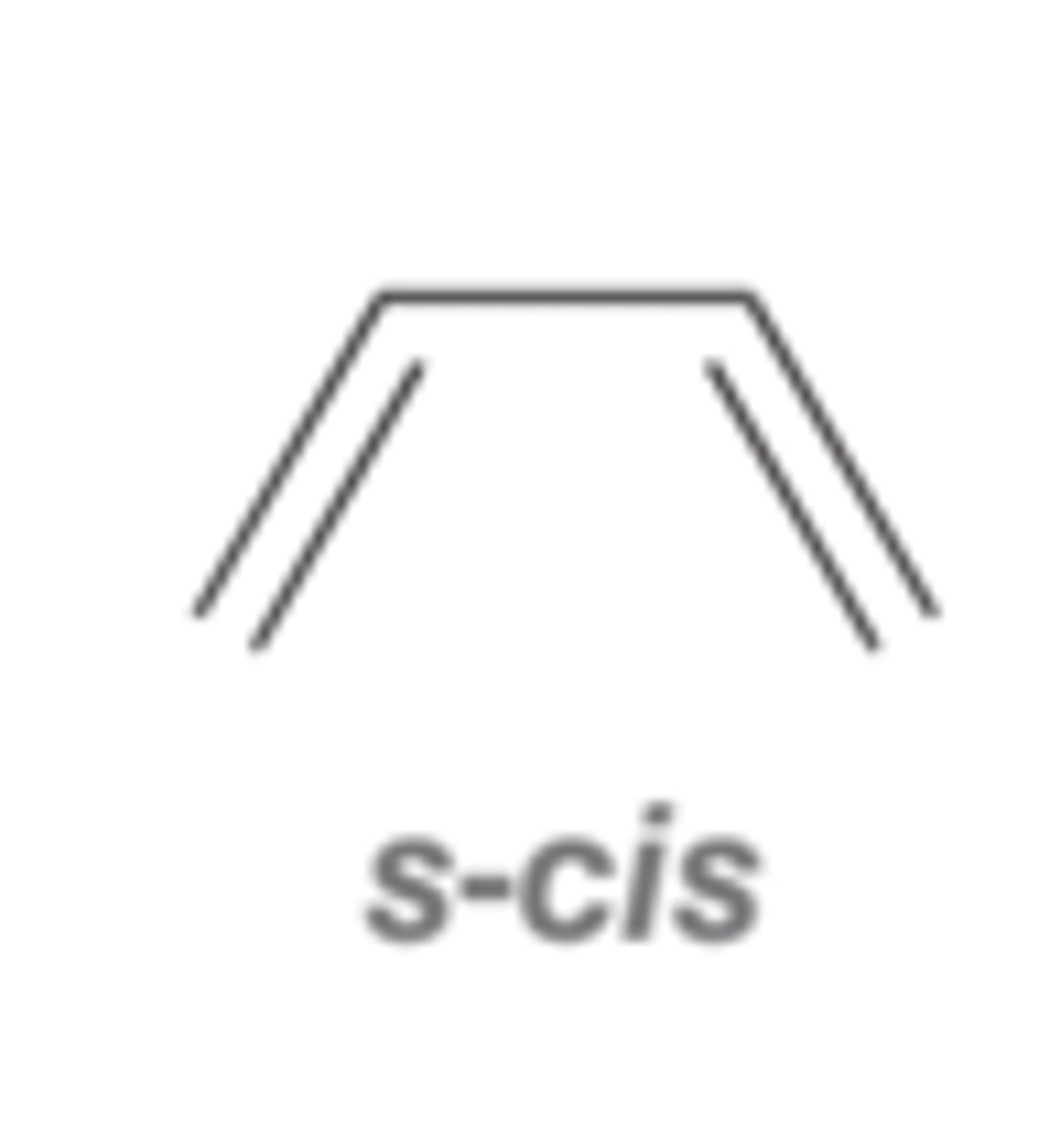
we were roleplaying like we wanted to use this product in a diels-alder reaction
the double trans combo would make it easiest (least hindrance) for the double bonds to adopt the "s-cis" conformation you need for diels-alder reactions
exp 3: in this experiment we attached two benzenes together with a alkyl chain between them that had 2 double bonds in it. We wanted these double bonds to be trans - why?
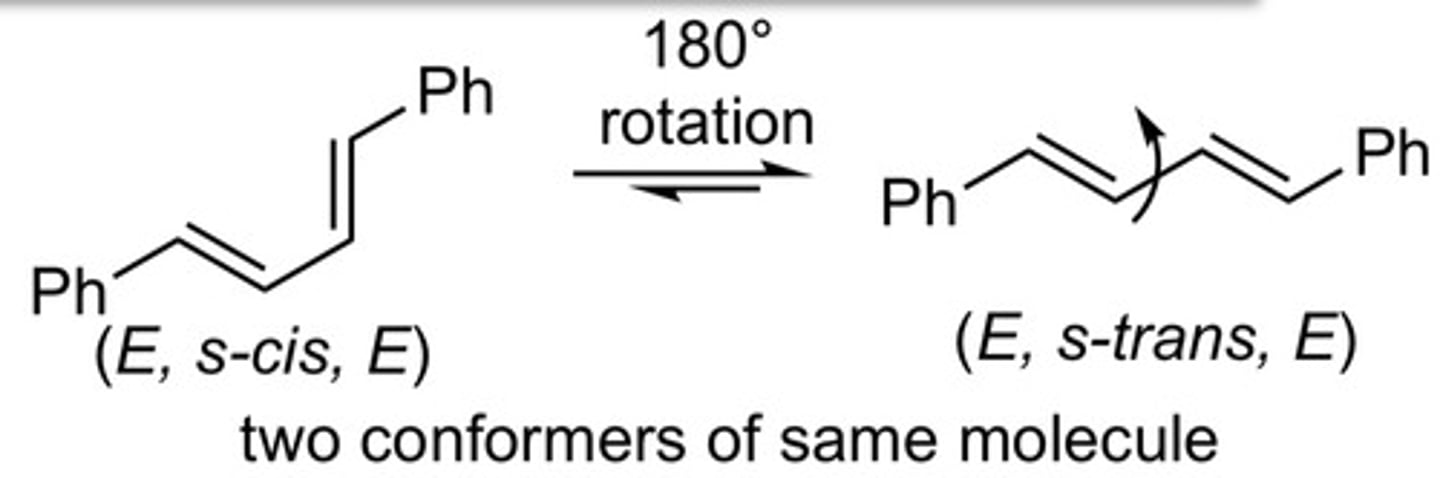
E,E
exp 3: the reaction was going to give the E,Z or E,E product. which one was preferred?
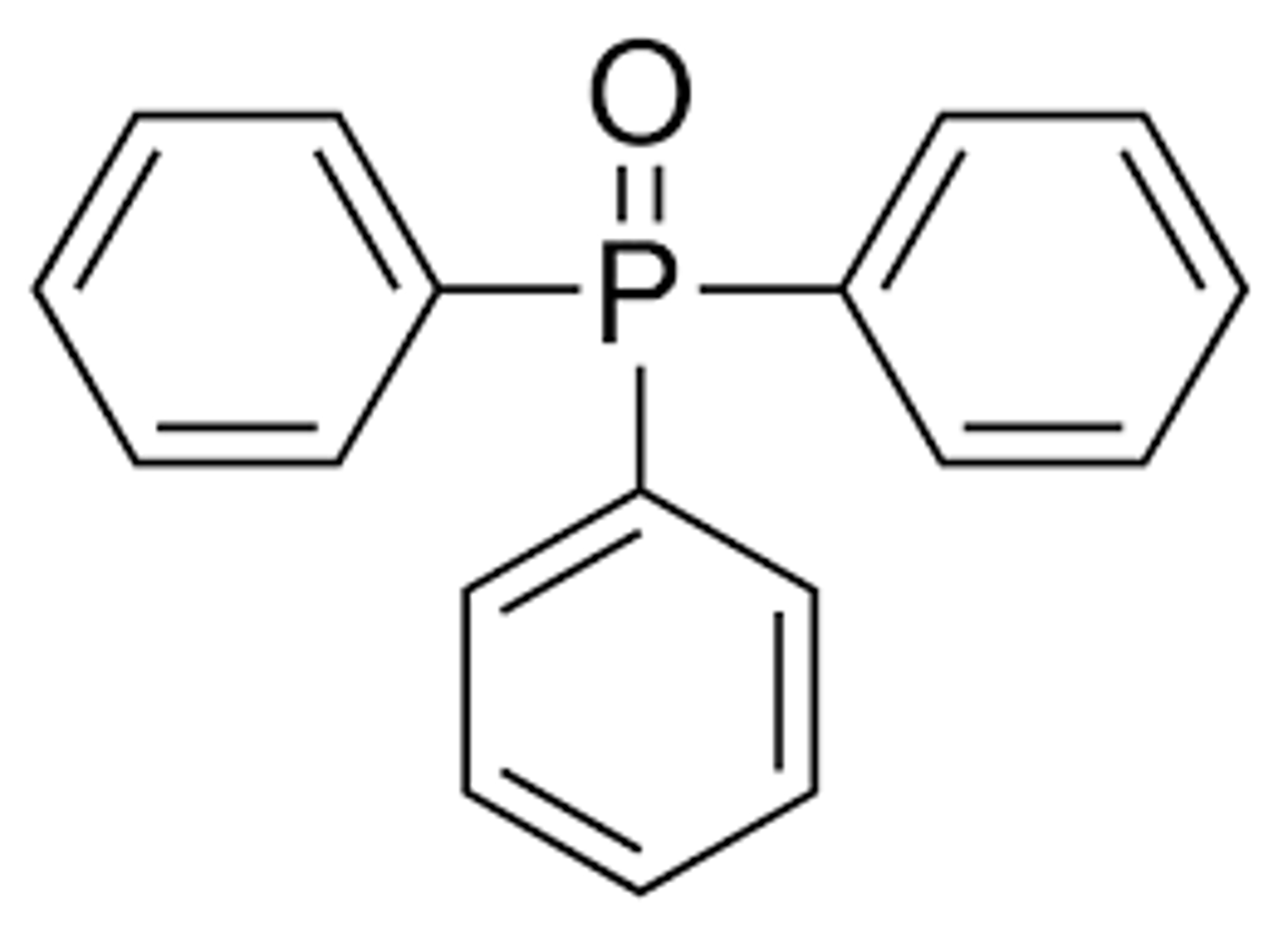
phosphorous really likes to be double bonded to oxygen, so the phosphorous exchanges its alkyl chain for the carbonyl
exp 3: What is the driving force of the wittig reaction?
kinetic control and form the Z-isomer due to sterics
exp 3: wittig reactions are normally under __________ control and form the ___-isomer of the alkene due to ____________
e-isomer so we ran it at room temp (as opposed to on-ice for kinetic product). If there an EWG adjacent to the carbanion of the ylide that can do resonance with that lone pair it will help make the E-isomer as well
exp 3: we wanted the ___-isomer so we ran the reaction at what temp? What other factor, discussed more in lecture, also prefers this isomer?
the larger 14-15 Hz J-value = E/trans
the smaller 8-10 Hz J-value = Z/cis
exp 3: we used the "J value" of the alkene hydrogens in the HNMR to figure out if we got the E or Z isomer, how does this work?
because there are two solvents, DCM (organic layer) and NaOH (aqueous layer). DCM layer contains our reactant (trans-cinnamaldehyde), the phosphonium salt (benzyltriphenylphosphonium chloride), the ylide (benzyltriphenylphosphorus ylide), and the
exp 3: why is the experiment "bisphasic"?
NaOH
exp 3: what is the base used in our biphasic Wittig reaction?
Trituration and recrystallization
exp 3: how do we purify our compound?
to synthesize DEET (N,N-diethyl-m-toluamide) using the schotten baumann reaction which uses a cheaper and more environmentally friendly solvent like water instead of diethyl ether
exp 4: what is the purpose of the experiment
an insect repellant (as opposed to an insecticide)and supposedly acts by blocking the neural stimulatory response to lactic acid and/or warm moist air currents.
Used to repel mosquitoes and ticks, these insects are vectors for several diseases, including: MalariaViral encephalitis (e.g., West Nile Virus)
exp 4: what is DEET?
carboxylic acids will just get deprotonated by an amine (unless there's an activation or high heat), so they need to be converted to a more reactive molecule like an acyl chloride that is highly reactive
exp 4: we first convert a carboxylic acid to an acyl chloride, so then the amide can be created. why?
synthesizes amide from acyl chloride or an anhydride using an amine and a base
exp 4: what is the schotten baumann reaction?
In the Schotten–Baumann reaction, an acid chloride reacts with an amine under mild, biphasic conditions, producing an amide and HCl (neutralized by base). It is highly reactive and does not require strong heating.
In contrast, a general nucleophilic carbonyl reaction involves a carboxylic acid or ester reacting with an amine, typically needing heat or activation. It produces water as a byproduct, which must be removed to drive amide formation, and usually requires harsher conditions.
its also easier for purification
exp 4: why is the schotten baumann reaction used in this experiment instead of converting straight from a COOH?
NaOH is cheaper and more readily available than amines, especially if the amine is complex or expensive.
NaOH efficiently neutralizes the HCl produced without wasting valuable amine.
NaOH prevents protonation of the amine — if HCl builds up, it could protonate the amine to form R-NH3+, making it a poor nucleophile and slowing down or stopping the reaction.
Using less amine keeps the reaction mixture simpler and avoids difficult purification steps to remove large amounts of leftover amine.
exp 4: Why might using NaOH be advantageous compared to the use of excess amine?
first m-toluic acid and SOCl2 are reacted under reflux and are stirred, then theyre cooled. then NaOH and diethylamine hydrochloride are combines in an ice water bath and are stirred. the amine salt will convert to diethylamine. then mix in sodium lauryl sulfate. then add the solution to the m-toluic acid solution and heat the reaction with vigorous stirring. then prepare for separation by first cooling it to room temp and adding it to separatory funnel and add diethyl ether to separate. organic layer will be at the top with diethyl ether. extract organic and wash with HCl to neutralize any base. wash with NaCL to rmove residual HCl. dry organic with anhydrous sodium sulfate and evaporate ether. purify by column chromatography by adding impure DEET to column with hexanes. elute compound and obtain TLC, IR. submit pure samples for HNMR and GC/MS analysis
exp 4: summarize procedure
GC tells us how many molecules are in the product (each peak is a molecule and the width of each peak is the amount of that molecule), which in turn determines if the product is pure or not, and the retention time, which tells us the polarity and BP (short retention time = non polar/low BP and vice versa)
MS gives us the mass and fragments of the compound so we can determine the exact compound
exp 4: why do we use GC/MS in the experiment?
column chromatopgraphy has a polar stationary phase (alumina) and a non-polar mobile phase (hexanes) so the polar impurities will stick to the stationary phase and elute out much slower, while non polar impurities will dissolve in the hexanes and elute fast
exp 4: why do we use column chromatography to purify our compound?
TLC to assess polarity and find Rf value to compare to literature and see if we synthesized the correct compound
IR to find functional groups and see if correct compound was made
exp 4: why do we use TLC and IR to analyze the compound?
distance traveled by the compound/distance traveled by the solvent front
exp 4: whats the Rf value formula
we add it to the diethyl amine solution, which is then added to the m-touluoyl chloride in order to:
if you just dumped water or aqueous amine straight onto the acyl chloride, it could hydrolyze (turn into a carboxylic acid) instead of reacting cleanly with the amine.
By emulsifying the diethylamine first with sodium lauryl sulfate, you:
Make a very fine suspension of the amine in a little bit of water.
Create small droplets that can better mix with the organic layer (where the acyl chloride is).
Control the reaction — the amine reacts preferentially with the acyl chloride, not water.
Then, by slowly adding the emulsified amine to the acyl chloride, you:
Minimize hydrolysis (you don't want the acyl chloride to see too much water all at once!).
Ensure efficient amide formation (DEET).
Avoid too much heat or side reactions.
exp 4: why do we add diethylamine to SAS insead of acyl chloride first?
heating will increase rotation, making the ethyl hydrogens indistinguishable (cannot tell the difference)
if we cool the sample, this should decrease rotational rate, and the ethyl hydrogens for each of the 2 ethyl groups will be indistinguishable
exp 4: amides have restricted rotation around the C-N bond. why is this a problem in our experiment and how did we deal with it?
N,N-diethyl-m-toluamide (most polar)
exp 4: Which of the following is expected to elute last during column chromatography:
Acid chloride
N,N-diethyl-m-toluamide
diethyl amine
m-toluic acid
the prevent the reformation of carboylic acids from acyl chlorides from NaOH (hydrophilic, think aqueous)
exp 4: why are hydrophobic acyl chlorides preferred?
DEET is a liquid, recrystallization is for solids
exp 4: why do we use column chromatography to purify instead of recrystallization?
Deposit four spots a TLC plate using a sample of your synthetic DEET. The spots arecrude DEET, pure m-toluic acid, overlapping ("mixed") spots of toluic acid andDEET, and purified DEET. Run the separation about halfway up the TLC plate in a100-mL beaker containing 2-4 mm of 1:1 (v/v) ethyl acetate/hexanes.
exp 4: explain the TLC procedure in the experiment
false
exp 4: True or false, We added diethylamine directly from the bottle to our reaction?
thionyl chloride (SOCl2) decomposes violently when in contact with water
exp 4: Why was it very important to have completely dry glassware for this lab?
sodium hydroxide is cheap and easy to remove, amines are expensive and annoying to remove
exp 4: why did we use NaOH for the base insetad of the 2 equivalent amine like usual?
"sodium lauryl sulfate" there were distinct organic and aqueous layers in this reaction and they interact poorly normally (at the border where they touch) an emulsifier kind of break up the organic layer and get more surface area for the reaction to go
exp 4: What was the emulsifier and what purpose does it serve?
small amines are nasty chemicals but ammonium salts are easy to handle powders
the ammonium salt got deprotonated with some NaOH into normal diethylamine and this is called "freebasing" the amine.
we did this in a separate conical vial and then used the syringe to inject it into our acyl chloride reaction vessel
exp 4: Why did we introduce our diethylamine as a diethylammonium chloride salt rather than just as diethylamine? What is this process called and where did we do it?
Column chromatography.
The stationary phase was the polar "alumina" - the silvery junk in between the sand and cotton in that pipette we made.
The "mobile phase" were the hexanes we kept putting into it
exp 4: We purified our DEET by __________________________. It had a "mobile phase" that was...? Stationary phase that was......?
As the liquids pass through the column, they stick to the stationary, polar alumina to different degrees and separate into bands. Ideally you know which band the thing you will want to be in and collect that "fraction"
We tossed the first 1 mL to go thru the column, collected the next 4 mL, and then tossed the remainder. So I guess the deet was somewhere in the middle.
exp 4: How did the column chromatography purify our deet?
big change would be loss of the big obvious carboxylic acid peak while keeping the 1700 carbonyl peak
DEET would not actually have any N-H bonds to make an amine or similar peak
exp 4: for IR of DEET, what big change would we expect from the reactant? would DEET have an amine peak?
Hydrogens that look equivalent on the ethyls attached to the nitrogen may have been split into separate peaks, or messily combined into broad peaks. They do this because resonance of the nitrogens lone pair towards the carbonyl gives that bond some "double bond character" and as a result it actually rotates quite slowly
this slow rotation makes the pair of CH2s and CH3s of the two ethyls not-quite-equivalent even though they look like they should neatly combine in NMR
exp 4: What strange NMR behavior did DEET exhibit? Why does it occur?
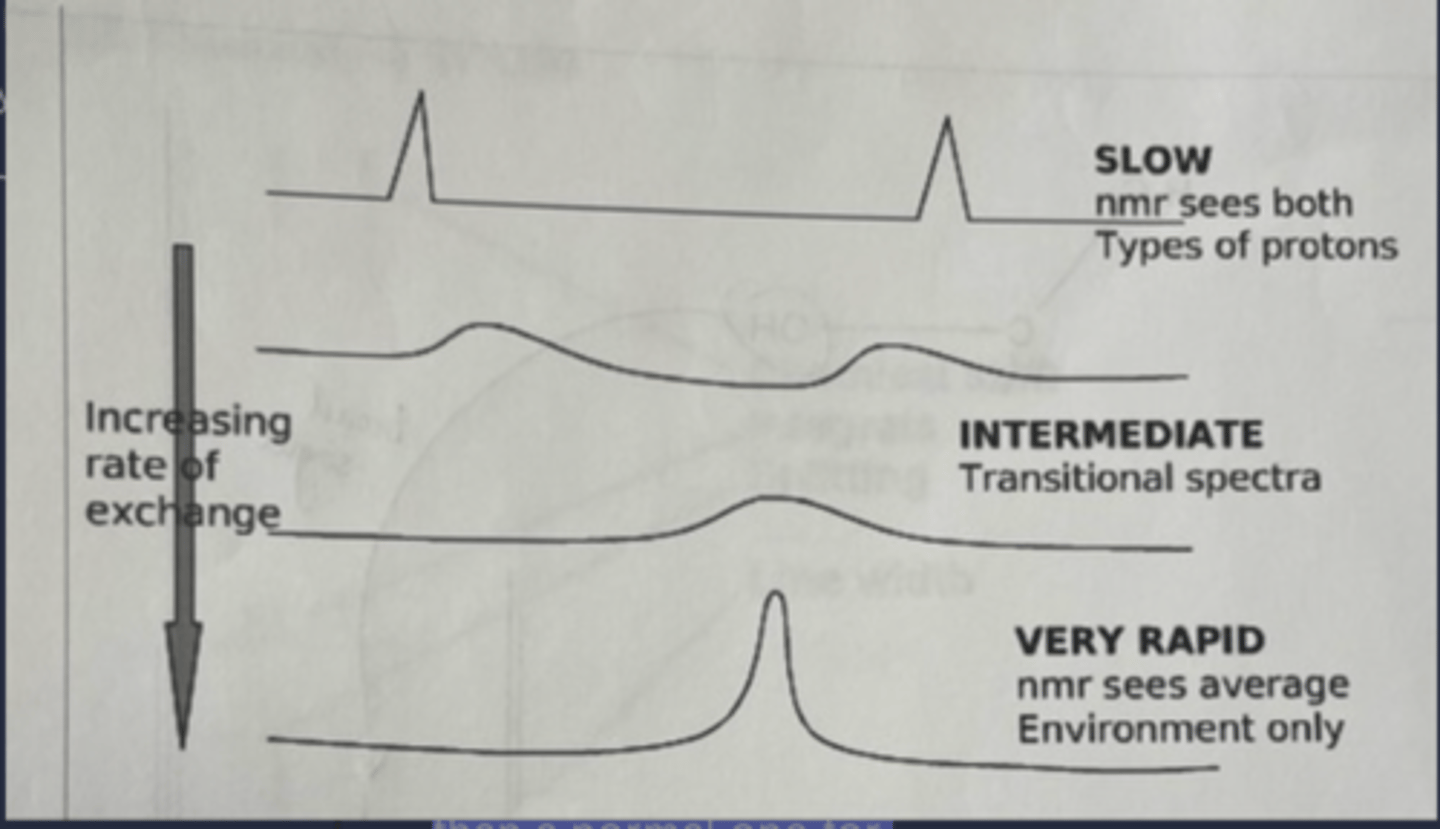
At low temps that nitrogen-carbonyl carbon bond rotates really slow and the hydrogen groups on the ethyls that look like they should be equivalent split into entirely different, separate peaks (top of image)
At higher temps the rotation is faster and they kind of smash together into a single peak but it is still broader than a normal one for equivalent hydrogens (bottom of image)
exp 4: The behavior discussed in the prior card is called "exchanging" - how is it altered by different temperatures?
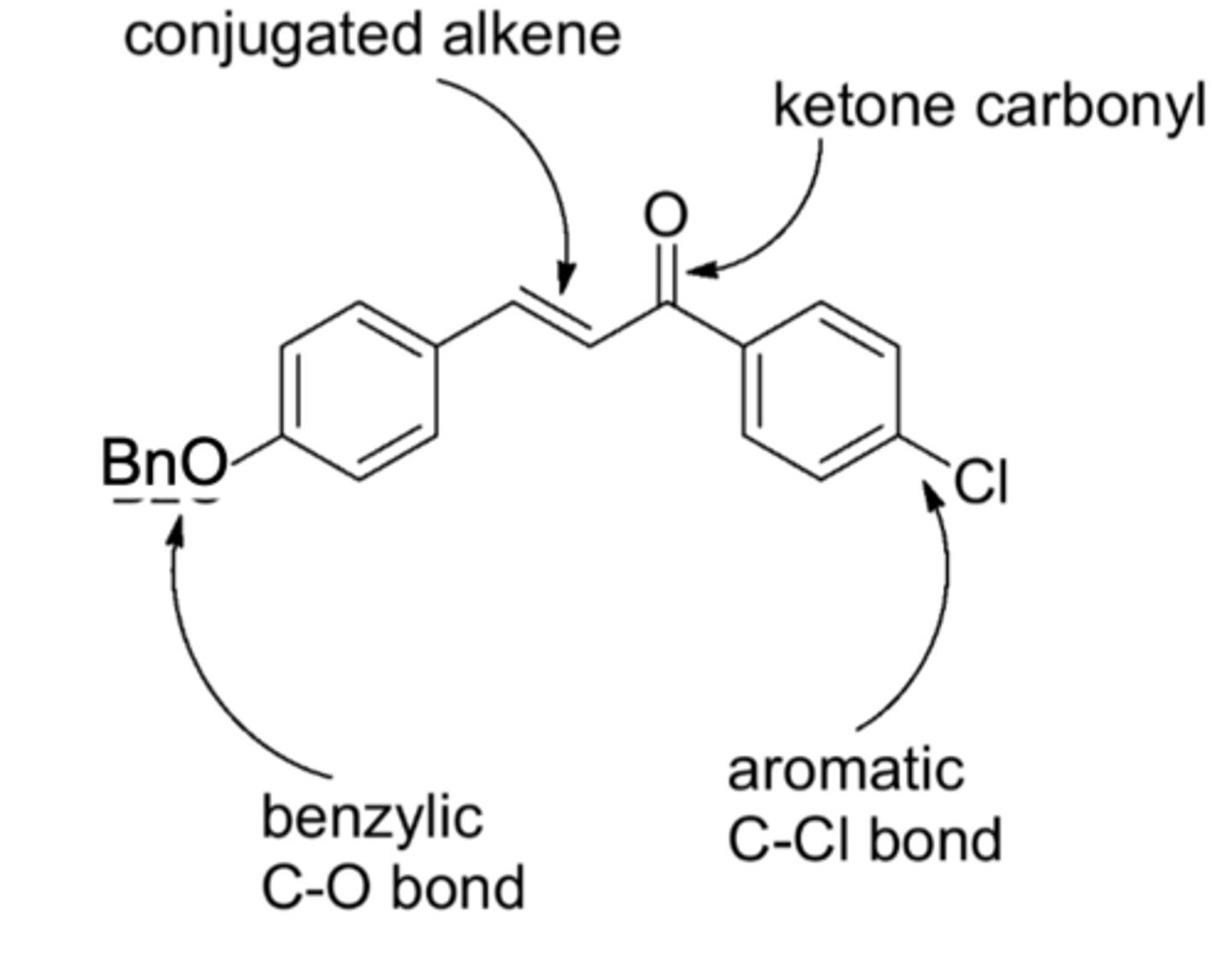
class of molecules with significant activity in antitumor, antifungal, antibacterial, and anti inflammatory screens
basically they are possible drug precursors
exp 5: what is chalcone?
We will vary reaction conditions and monitor reaction products from the chalcones related to the structure in the image. By varying substituents and conditions we learn more about the reaction and its mechanism
exp 5: what is the goal in the experiment?
Heat a water bath to and maintain it at 50-75°C. In an Erlenmeyer flask, combine 1.5 mmol each of 4-chloroacetophenone and 4-methylbenzaldehyde with 5 mL EtOH. Spot each starting material on a TLC plate using pre-diluted solutions. After mixing, spot the reaction mixture on the TLC plate. Add 1 mL NaOH, then heat and stir the reaction for 30 min. Spot the product on TLC; run the plate using 9:1 hexanes:EtOAc. If starting materials remain, heat for 10 more min. Once complete, cool in an ice bath for 5-10 min, then vacuum filter and rinse with 5 mL cold water. For recrystallization, dissolve ~100 mg product in hot 5-10 mL isopropanol in the warm bath; add acetone dropwise if needed. Vacuum filter and let air-dry.
exp 5: explain day 1 procedure
Heat a water bath to 70 °C. Finish recrystallizing chalcone using ~5 mL isopropanol and up to 5 drops acetone if needed. Dry product in oven. Prepare NMR and Mel-Temp samples. Pack Pasteur pipet with cotton and Celite for Pd/C filtration. In a round-bottom flask, combine ~50 mg chalcone, 10 mg Pd/C, 69 mg ammonium formate, and stir bar. Add 5 mL alcohol (or additive mix, which was HCl for me), place flask in hot bath with condenser, and reflux for 15 minutes. Filter mixture through Celite, rinse with ethanol. Wash filtrate with brine and diethyl ether, extract organic layer. Dry with sodium sulfate, filter, and save liquid. Prepare GCMS sample with hexanes. Submit all samples and clean up Pd/C waste properly.
exp 5: explain day 2 procedure
analyzed GC/MS and everyone submitted their data on an excel sheet. we were able to find how much of our product was reduced, and to what it was reduced to thanks to the GC/MS
exp 5: explain day 3 procedure
It was an aldol condensation like from chapter 21, producing a product very like the image (right benzene had a chlorine, left had a methyl). Lab material also calls it a "Claisen-Schmidt condensation"
The right side was a benzene with a ketone and chloride that enolate attacked the left side, a benzene with a methyl and aldehyde. The resulting alcohol was replaced in an elim reaction ("condensation") by heating.
exp 5: We had to make our chalcone before we tried to reduce it. What kind of reaction did we do? What were our two reactants?
The catalyst can be recovered (doesn't dissolve in the solvent)
You can modify the reaction by changing solvents, or adding additive
seasier purification
exp 5: What are the advantages of a heterogeneous catalyst over a homogenous one?
The carbon-carbon double bond, the chlorine, and the carbonyl.
Alkene usually easiest to reduce (256 kJ/mol) C-Cl bond next easiest (358 kJ/mol) C=O bond hardest (441 kJ/mol)
exp 5: What 3 groups were possible reduction targets in our chalcone? How difficult is each to reduce under typical conditions - think in terms of bond energy.
Neighboring functional groups, other molecules present, etc. can alter the bond energies from the usual state. For example, A C=C alkene might be harder to reduce when it is conjugated with other pi systems (e.g. benzene is extremely hard to reduce)
So it wasn't immediately obvious which of our functional groups would reduce
exp 5: Why is predicting what would be reduced first not as simple as looking at typical bond energies?
In the presence of a catalyst the ammonium formate breaks down into H2 gas, CO2 gas, and ammonia. So it indirectly provided the H2 gas. H2 gas is flammable, requires pressurization, and is generally just a pain to use
exp 5: We reduced our chalcone with AMMONIUM FORMATE and Pd/C instead of the H2 + Pd/C we see often in lecture. How does this work? Why didn't we just use H2 gas?
Reduction of the pi bond vs reduction of the carbonyl was hard. Each requires the addition of two hydrogens to reduce them, so the amount of "weight" that changes is the same. You would have to analyze the fragmentations to tell the two apart.
THe chlorine was easy because the product would be much heavier if it was not reduced, and much lighter if it was.
exp 5: What two possible reductions were hard to tell apart on the mass spec output? Which was easy? How would you figure it out?
It altered the behavior of the reduction reaction, causing different things to be reduced/not reduced than without the additive. Choice of solvent can also alter what gets reduced.
exp 5: What was the role of the additive in the chalcone lab (I used HCl)? What other molecule could potentially affect reductions?
to synthesize compounds that can inhibit the main protease enzyme so covid 19 can be treated
exp 6: what is the purpose of the experiment?
ammonia
exp 6: what reagent cleaves the resin?
TFA/DCM, NMP
exp 6: what reagent do we use to wash the vessels?
DIEA
exp 6: what reagent do we use to neutralize (boc removal) the resins?
there is no color change
exp 6: what happens if cleavage is unsuccesful?
static electricity
exp 6: why may the analytical balance flucturate?
they ahve a vinyl group in conjugation with a carbonyl
exp 6: why are michael acceptors reactive?
triethylamine
exp 6: what can be used if DIEA is not available?
neutrally buoyant
exp 6: what does isopycnic
UV
exp 6: whats used to visualize crude product on the TLC plates?
drip procedures
exp 6: what type of procedures are carried out for deprotection and neutralization steps?