CHEM 433 Reaction
1/12
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
13 Terms
Dissociative Substitutions
-analogous to SN1
-A two step mechanism that proceeds via a rate limiting M-X bond cleavage, leading to a
short-lived reactive intermediate, that can either back react with X or with Y to
give product—usually irreversibly
-first order
rate = d[L5 MY]/dt =
(k1 k2[L5 MX][Y])/(k-1 [X] + k2 [Y])
If k 2[Y] >> k-1[X], then rate = k1[L5 MX]
![<p>-analogous to SN1 </p><p>-<span>A two step mechanism that proceeds via a rate limiting M-X bond cleavage, leading to a</span><br><span>short-lived reactive intermediate, that can either back react with X or with Y to</span><br><span>give product—usually irreversibly</span></p><p><span>-first order</span></p><p><span>rate = d[L5 MY]/dt =</span><br>(<span>k1 k2[L5 MX][Y])/(k-1 [X] + k2 [Y])</span><br><span>If k 2[Y] >> k-1[X], then rate = k1[L5 MX]</span></p>](https://assets.knowt.com/user-attachments/c851ad3c-c0c9-41fa-ba59-6201ac911ba3.png)
Associative Substitutions
-analogous to SN2
-A two step mechanism that proceeds via a rate limiting M-Y bond forming step, leading
to a short-lived reactive intermediate with an expanded coordination number, that can
either back react by loss of Y or lose X to give product—usually irreversibly.
-second order
rate = d[L3 MY]/dt =
(k1k2 [L 3MX][Y])/(k-1 + k2)
![<p>-analogous to SN2<br>-<span>A two step mechanism that proceeds via a rate limiting M-Y bond forming step, leading</span><br><span>to a short-lived reactive intermediate with an expanded coordination number, that can</span><br><span>either back react by loss of Y or lose X to give product—usually irreversibly.</span></p><p><span>-second order</span></p><p><span>rate = d[L3 MY]/dt =</span><br>(<span>k1k2 [L 3MX][Y])</span>/(<span>k-1 + k2)</span></p>](https://assets.knowt.com/user-attachments/8e821061-60a3-4d31-8695-0acbafec853f.png)
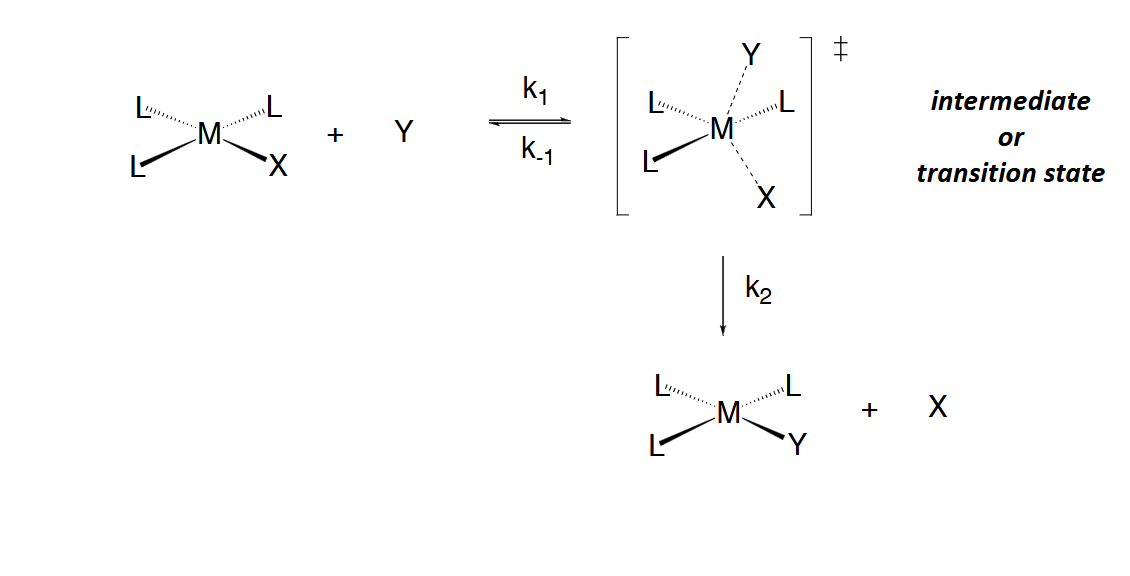
Intercahgen Mechanism
Generally, the intermediates formed in the previous mechanisms are not observable and
may even be transition states. In the interchange mechanism, a more concerted process
in which M-X bond breakage occurs simultaneously with M-Y bond formation takes place
Mechanisms are classified as Id or Ia depending on the relative degrees of M-X
bond breakage vs M-Y bond formation in the transition state:
Id à M-X bond breakage is more complete than M-Y formation
Ia à M-Y bond formation is more complete than M-X bond breakage
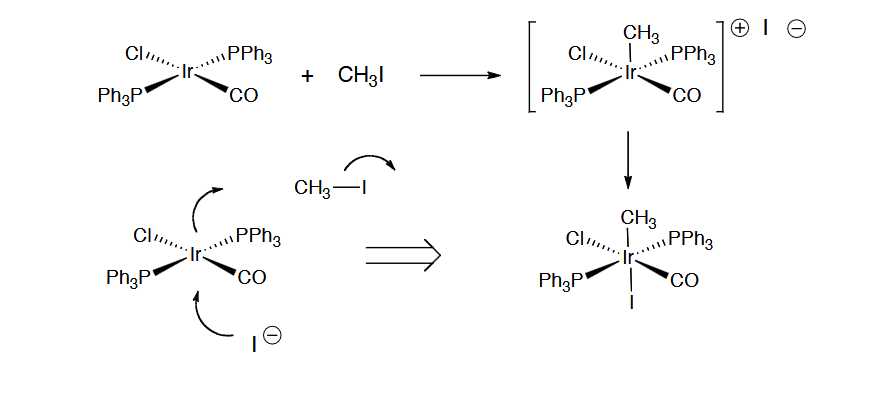
Concerted Mechanism (Non-polar) of oxidative addition
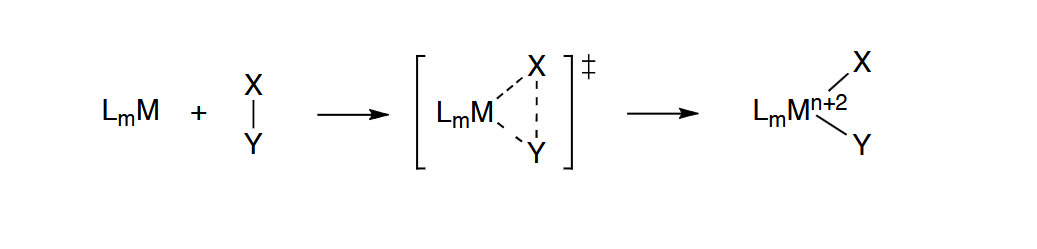
concerted Mechanism (Polar) of oxidative addition
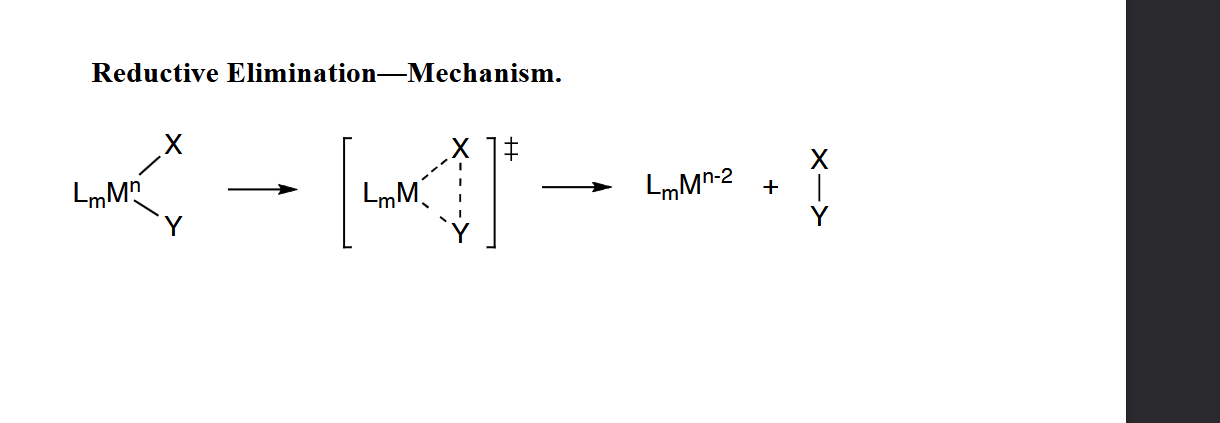
Reductive Elimination—Mechanism.
has to be cis to each other
Insertion reactions (1,1)
d[P]/dt = k 1k 2 [L'][SM]
/(k -1 + k 2 [L'])
![<p><span>d[P]/dt = k 1k 2 [L'][SM]</span><br>/(<span>k -1 + k 2 [L'])</span></p>](https://assets.knowt.com/user-attachments/522c080b-7860-4412-83b7-c3e94ca1a4ba.png)
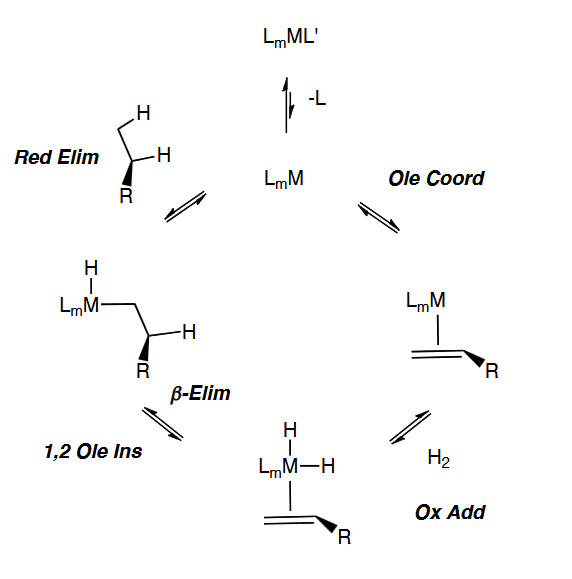
Olefin Insertion and Beta-Hydride Elimination
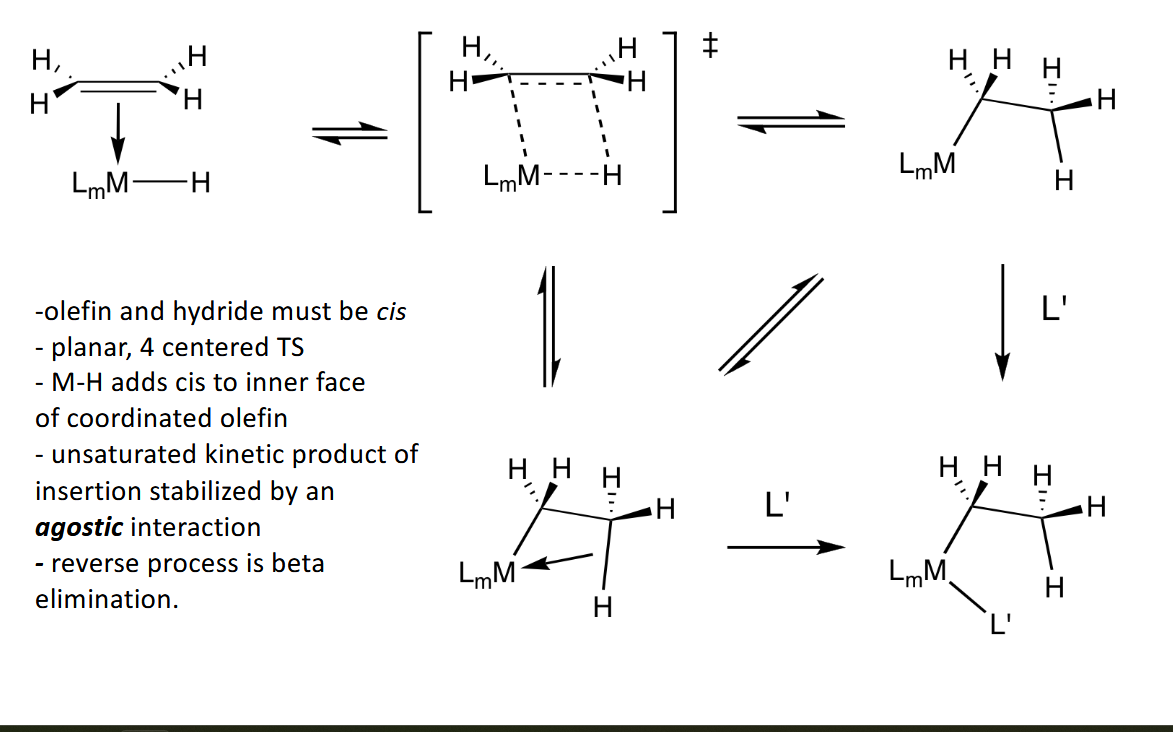
Olefin Hydrogenation Hydride Path
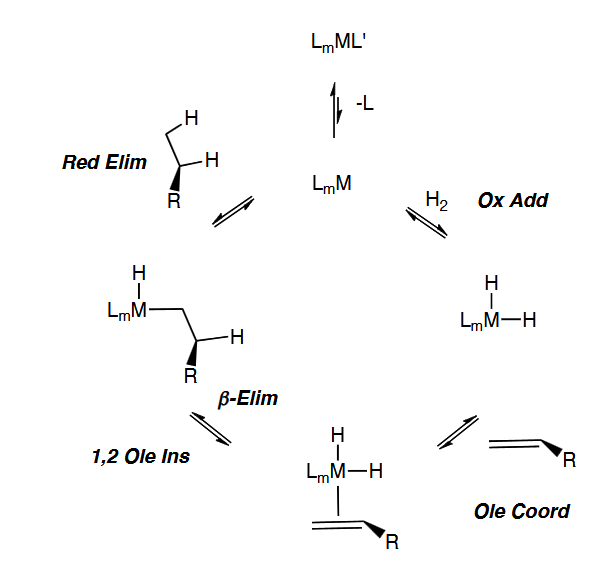
Olefin Hydrogenation Olefin Path
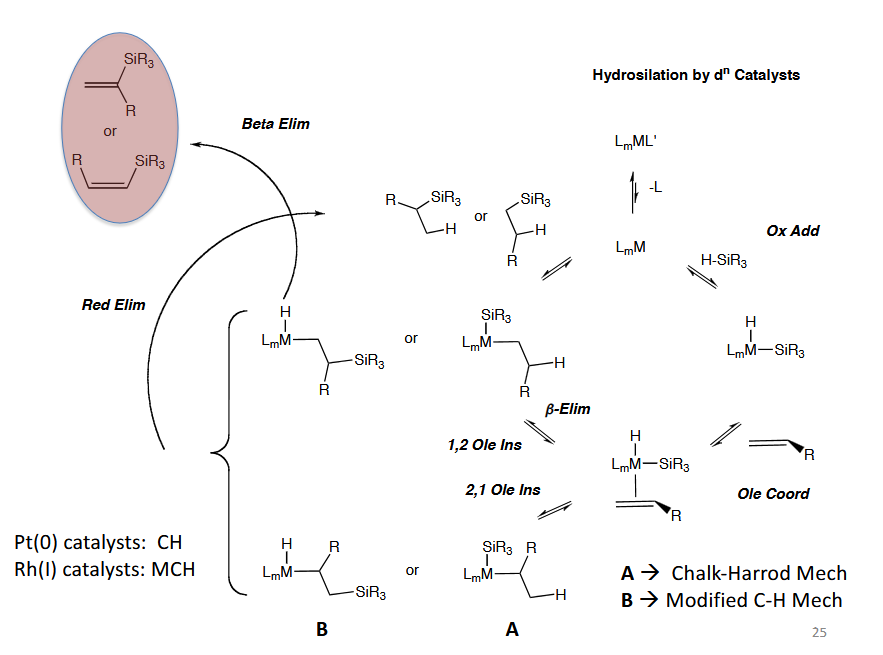
Hydrosilation by dn Catalysts
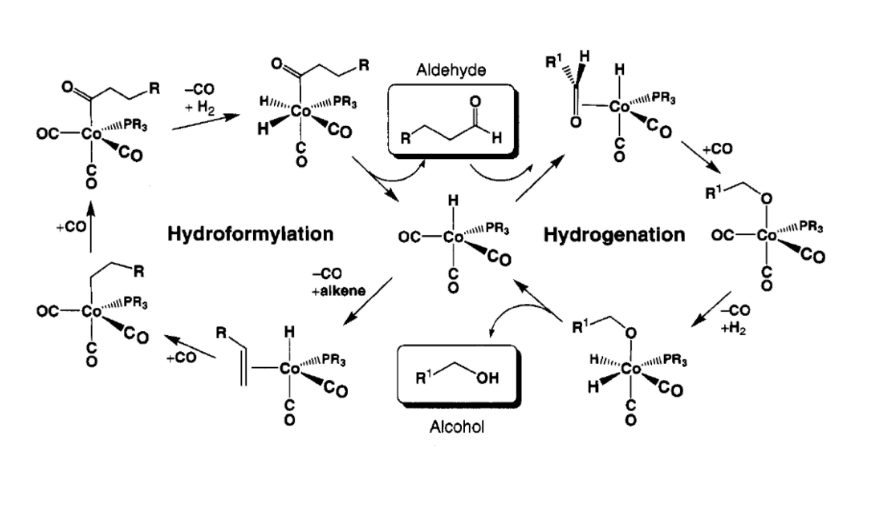
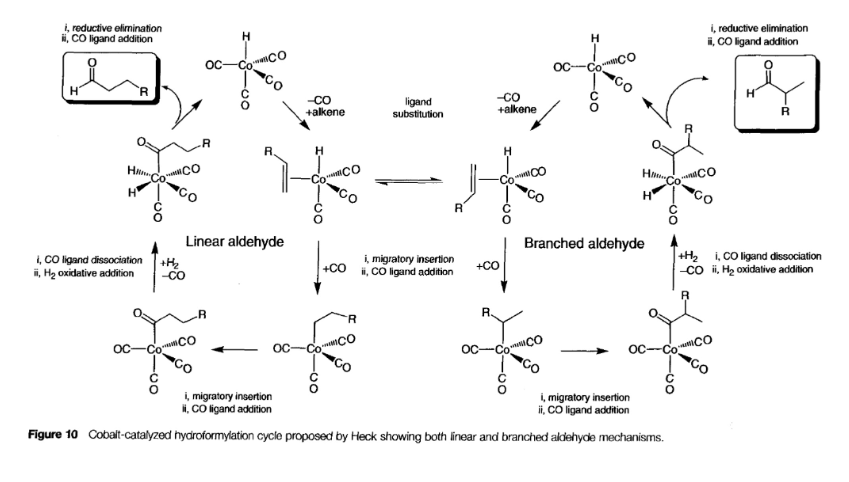
Hydroformylation
hydroformylation vs hydrogenation