Key Science Skills
1/83
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
84 Terms
research question structure & example
"What is the effect of [Independent Variable] on the [Dependent Variable] in [Population]?"
example: "What is the effect of daily probiotic yogurt consumption on self-reported anxiety levels in adults aged 18–30?"
Aim structure & example
"To investigate the effect of [IV] on [DV] in [Population]."
The aim of this investigation is to explore the relationship between partial sleep deprivation and low mood in young people aged 18-25.
Hypothesis structure and example
"It is hypothesised that [Population] who [IV - Group 1] will [Direction] in [DV] compared to [Population] who [IV - Group 2/Control]."
IPAD
Independent variable and dependent variables
Population
and
Direction
It was hypothesised that Australian females aged 12–16 who experienced partial sleep
deprivation would be more likely to also experience low mood than those who did not
experience partial sleep deprivation.
Independent variable
The variable that is manipulated, changed, or controlled by the researcher.
It is assumed to have a direct effect on the dependent variable.
Dependent Variable
The variable that the researcher measures.
It is observed to see how it changes in response to the IV.
Extraneous variable
any variable that is not the independent variable but may cause an unwanted effect on the dependent variable
controlled variables
variables other than the IV that a researcher holds constant in an investigation, to ensure that changes in the DV are solely due to changes in the IV
Controlled Experiment
Definition: A type of investigation used to test a causal relationship between an IV and DV in a controlled environment
Strengths:
Infer Causality: Allows researchers to conclude that the IV directly caused the change in the DV
Limitations:
conducted in a lab or highly controlled setting can mean it doesn't reflect "real life."
This may cause participants to act unnaturally
Between-Subjects Design
Different participants are randomly allocated to either the control or experimental group
Strength: No order effects (participants only do one condition).
Limitation: Requires more participants; participant differences (e.g., intelligence) can affect results
Within-Subjects Design
Each participant is in both the control and experimental groups.
Strength: Eliminates participant-related variables (everyone acts as their own control); requires fewer people.
Limitation: Prone to order effects (boredom or practice).
Mixed Design
Combines elements of both within and between-subjects designs (e.g., testing two different groups over time).
Strength: Allows researchers to see both differences between groups and changes over time.
Limitation: Can be complex and costly to implement.
Case Study
An in-depth investigation of an individual, group, or event.
Strength: Provides rich, highly detailed data; useful for rare phenomena (e.g., brain injuries).
Limitation: Results cannot be generalised to the wider population
Correlational Study
Observes the relationship between two variables without manipulating them
Strength: Can be used when experiments are unethical; identifies trends and relationships.
Limitation: Correlation does not equal causation (you don't know which variable caused the change)
Fieldwork
Gathering primary data in a "natural" setting (e.g., observing students in a playground).
Strength: High ecological validity (behaviour is natural).
Limitation: Difficult to control extraneous variables; can be time-consuming.
Literature Review
Collating and analysing secondary data from existing research.
Strength: Provides background for new research
Limitation: can be time-consuming to find relevant sources.
Modelling & Simulation
Creating physical or conceptual representations to simplify or predict reality.
Strength: Allows for testing of "what-if" scenarios safely.
Limitation: A model or simulation is only a "representation"—it can never fully capture the infinite complexity of real-world human variables.
Product, Process, or System Development
Designing something to meet a human need (e.g., a new mental health app).
Strength: Directly applies psychological theory to solve real-world problems.
Limitation: This methodology is designed to solve a problem, not necessarily to test a broad psychological theory or establish cause-and-effect.
Population vs. Sample
Population: The entire group of interest from which the sample is drawn (e.g., all VCE students).
Sample: A smaller subset or group that is actually chosen from the population to participate in the study.
What makes a sample Representative versus Biased?
Representative Sample: A sample that accurately reflects the relevant characteristics of the population (e.g., same ratio of ages/genders).
Biased Sample: A sample that does not adequately represent the population (e.g., only testing girls when the population is all students).
Convenience Sampling
Definition: Selecting participants based on their availability and readiness to participate (e.g., the first 20 people you see in the library).
Strength: Quick, easy, and inexpensive to perform.
Limitation: Highly likely to produce a biased sample; results cannot be easily generalised
Random Sampling
Definition: Every member of the population has an equal chance of being selected (e.g., pulling names out of a hat).
Strength: Reduces bias; the sample is more likely to be representative than convenience sampling.
Limitation: Can be time-consuming to obtain a full list of the population; may still accidentally miss small sub-groups by chance.
Stratified Sampling
Definition: Dividing the population into distinct sub-groups (strata) based on shared characteristics, then selecting a sample from each stratum in the same proportion as they exist in the population.
Strength: The most representative method; ensures all sub-groups are accurately represented.
Limitation: Very time-consuming and expensive to identify and calculate sub-groups.
Differentiate between Sampling and Allocation.
Sampling: The process of choosing people from the population to be in the study. (Population →Sample).
Allocation: The process of assigning the chosen participants into groups within the experiment (e.g., Experimental group vs. Control group)
Random Allocation
Assigning participants to groups so that each person has an equal chance of being in the experimental or control group
Experimental Group
Exposed to the Independent Variable (the "treatment" or "intervention").
Control Group
Not exposed to the IV; provides a baseline to compare the experimental results against
Identification Example: In a study on caffeine and memory:
Experimental: Group drinking 2 cups of coffee (IV).
Control: Group drinking plain water (No IV).
Extraneous Variable
Any variable other than the IV that may cause an unwanted effect on the DV.
They should be controlled or monitored.
Confounding Variable
A variable other than the IV that has directly and systematically affected the DV.
Crucial Rule: Confounding variables can only be identified at the end of an experiment after the results have been analysed.
Participant Variables
Personal characteristics of the participants (e.g., age, intelligence, prior experience, mood).
Example: In a memory test, one participant naturally has a better memory than another.
Situational Variables
Factors in the environment that can affect the results (e.g., background noise, temperature, time of day).
Example: One group takes a test in a quiet room while the other deals with a loud construction site outside.
Systematic Errors
Errors that differ from the true value by a consistent/predictable amount (e.g., a scale always being 100g light).
Affects: Accuracy (how close the result is to the "true" value).
Random Errors
Unsystematic errors that occur due to chance and vary unpredictably (e.g., a participant being momentarily distracted).
Affects: Precision (how consistent the measurements are with each other).
How can extraneous variables be reduced, prevented, or accounted for?
Random Allocation: Spreads participant variables evenly across groups.
Single-Blind Procedure: Participants don't know which group they are in (prevents Placebo Effect).
Double-Blind Procedure: Neither participants nor researchers know who is in which group
Uncertainty
The lack of exact knowledge of the "true" value of the quantity being measured. All data has some uncertainty due to errors.
Outliers
Data points that differ significantly from other observations in the same sample.
Impact: Outliers can distort the mean (average) and increase uncertainty.
Repeatability
the closeness of results when the same researcher repeats the experiment under the same conditions.
Reproducibility
the closeness of results when different researchers conduct the experiment using different equipment or settings to test the same hypothesis.
Internal Validity
Does the study actually measure what it claims to measure? (i.e., was the change in the DV only caused by the IV?).
External Validity
Can the results be generalised to the wider population or other settings?
Robust
Data is considered robust when it remains valid and reliable despite small changes in the conditions or errors.
Improvements to reduce uncertainty in data and avoid errors
Increase Sample Size
Standardise Procedures
Refine Measurement Tools
Informed consent
Participants should be informed of the nature, purpose and risks involved with a study prior to giving their consent to participate
If under 18, consent must be given by a legal guardian
Consent is usually provided in writing.
Beneficence
The consideration of the benefits or gains from research in relation to the risks (MAXIMISE benefits and MINIMISE risks/harm)
Non-maleficence (also known as the no-harm principle)
the idea that any potential harm should be avoided and minimised as much as possible
Integrity
the commitment to search for knowledge, and then honestly report information and findings.
Justice
The distribution of fair access to the benefits of research to everyone,
ensuring that there is no burden on one group,
and ensuring that any opposing claims are considered.
Respect
the belief that everyone has value in regards to their welfare and beliefs, and has a right to make their own decisions.
Withdrawal rights
Participants have the right to withdraw from a study at any time, and/or may also elect to have their results withdrawn from a study.
Voluntary participation
Participants must not be coerced or forced into participating, and must do so freely. There must not be adverse consequences for choosing not to participate.
Confidentiality
Participants names or identifying details must not be revealed.
*Deception
When the true nature or purpose of a study is not revealed to participants, it must be because doing so would undermine the results
*Note - If deception is used, debriefing MUST occur to explain the true nature/purpose of the study, and why the deception was necessary.
Debriefing
The participants should be told of the results of a study after its completion
They may also be offered counselling (depending on the nature of the study) and be allowed to have their results removed.
Primary data
Data that is sourced first-hand.
Secondary data
Data that is sourced through someone else’s research.
Primary data strengths
specific + detailed
Inform future research
current
Primary data weaknesses
greater cost involved
greater sample size restrictions
Secondary data strengths
Cheap, easy to obtain
Large amounts available
Collected over long periods of time
Secondary data weaknesses
Might not be up to date
May not be specific to research
Quantitative data
Data that are numerical and categorical.
Quantitative data strengths
Easy to compare values between participants or groups
Quantitative data weaknesses
Some psychological data can be difficult to quantify.
Qualitative data
Data that are descriptive.
Qualitative data strengths
Can be a rich source of data on people’s thoughts, feelings and observations on behaviour
Qualitative data weaknesses
can be difficult to compare.
Subjective data
Data that relies on assumptions or personal experience.
Subjective data strengths
offering nuanced, qualitative understanding
Subjective data weaknesses
high variability, cognitive biases
Objective data
Data that can be directly observed or measured.
Objective data strengths
enhances the consistency and validity of assessments and research
Objective data weaknesses
can lack depth and nuance
failure to capture the subjective human experience
Mean
The ‘average’ score
Add all scores together and divide by the number of pieces of data.
Mean usefulness
Large amounts of data, even distribution of data around the centre
Mean disadvantages
when data values are widely distributed, the data set is likely to be influenced by outliers.
Median
The ‘middle’ score: Place all scores in order and find the centre
Median usefulness
When there are outliers
Median disadvantages
May not be representative in datasets with large gaps or uneven distributions.
Mode
The most ‘frequent’ score:
Tally the number of times each score appears.
Mode usefulness
Identifying patterns, categorical data
Mode disadvantages
may not exist, may be multiple
standard deviation
Measures the spread of scores around the mean.
The higher the standard deviation, the greater the range of values within the sample. (larger spread away from mean)
A low standard deviation means that scores are clustered around the mean (not a lot of variation away from mean)
how to label data tables
IV goes in the first/left hand column
DV goes in the second/right hand column
Why?
because the IV is what you’re changing or categorising, and the DV is what you’re measuring as a result
Bar charts
a good way to show comparisons between groups.
They require:
Title
Labelled axes
Accuracy
IV= x axis
DV= y axis
Line graph
a good way to show trends over time.
They require:
Title
Labelled axes
Accuracy
IV= x axis
DV= y axis
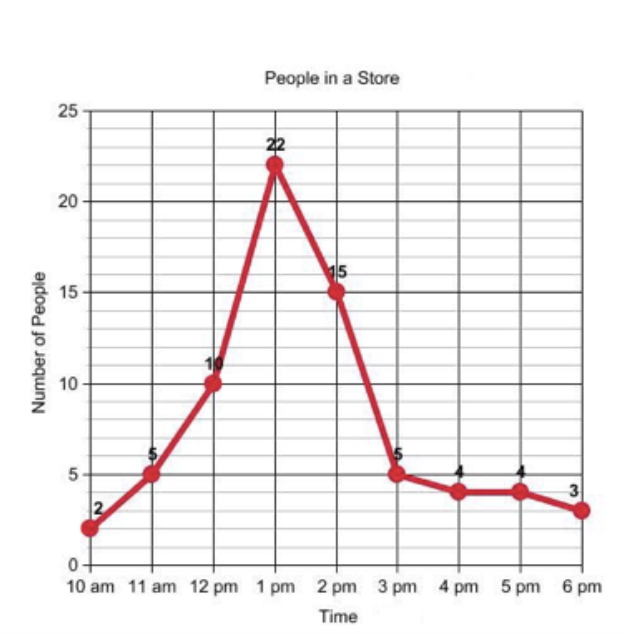
Percentage change