TRIPLE ONLY: Polymers part 2
1/12
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
13 Terms
What is condensation polymerisation?
Polymerisation of monomers in which each time two monomers combine, a small molecule such as water or hydrogen chloride is removed
Condensation polymerisation can make____
A polyester
Polyester is NOT made from addition polymerisation
Difference between addition polymerisation and condensation polymerisation?
Condensation polymerisation forms the polymer molecule and one water molecule (or hydrogen chloride molecule) per linkage
Condensation requires 2 types of monomers
What two monomers are required for a condensation type of polymerisation?
A diol and a dicarboxylic acid
What is a diol?
What is the structural formula of ethane-1,2-diol?
A diol is a compound containing two hydroxyl (—OH) groups
Structural:
OHCH2CH2OH
or CH2OHCH2OH
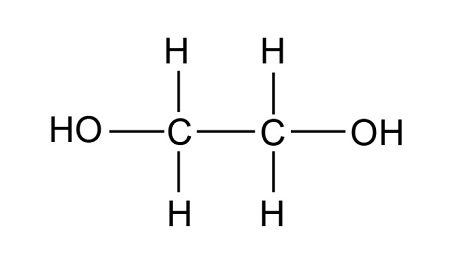
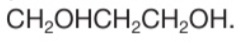
What is the name of this compound?
Propane-1,3-diol
3 carbon so ‘prop’
diol because 2 OH
1,3 because OH are on carbons 1 and 3
KEY POINT

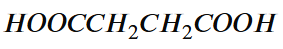
What is a dicarboxylic acid?
What is this dicarboxylic acid called? Draw the displayed formula as well
A carboxylic molecule with one —COOH at each end
Hexane-1,6-dioic acid
Hexanoic acid for carbxylic acid
Ending = ‘dioic acid’
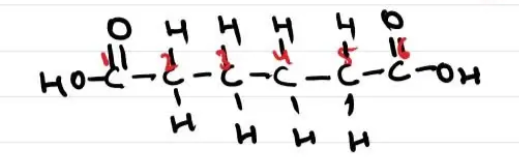
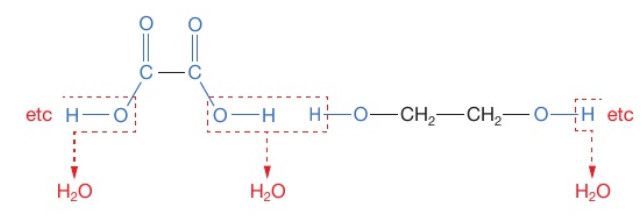
Draw the equation (displayed formula) when you react ethanedioic acid and ethanediol with the polymer as the repeating unit
Remove the OH and H as they will form water
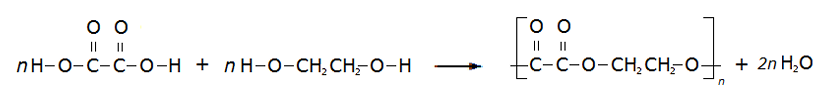
Lo de buscar monomers from a polymer (en el movil
As with addition polymers there are environmental issues with the disposal of condensation polymers.
Why are condensation polymers more reactive?
Because of their ester linkage
What are biodegradable polyesters?
A polyester that is biodegradable
Biodegradable is being able to be broken down into smaller components
How are biodegradable polymers made?
Made from lactic acid
Lactic acid can be obtained from corn starch and when it undergoes polymerisation it forms a biodegradable polyester