CHEM2056
1/39
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
40 Terms
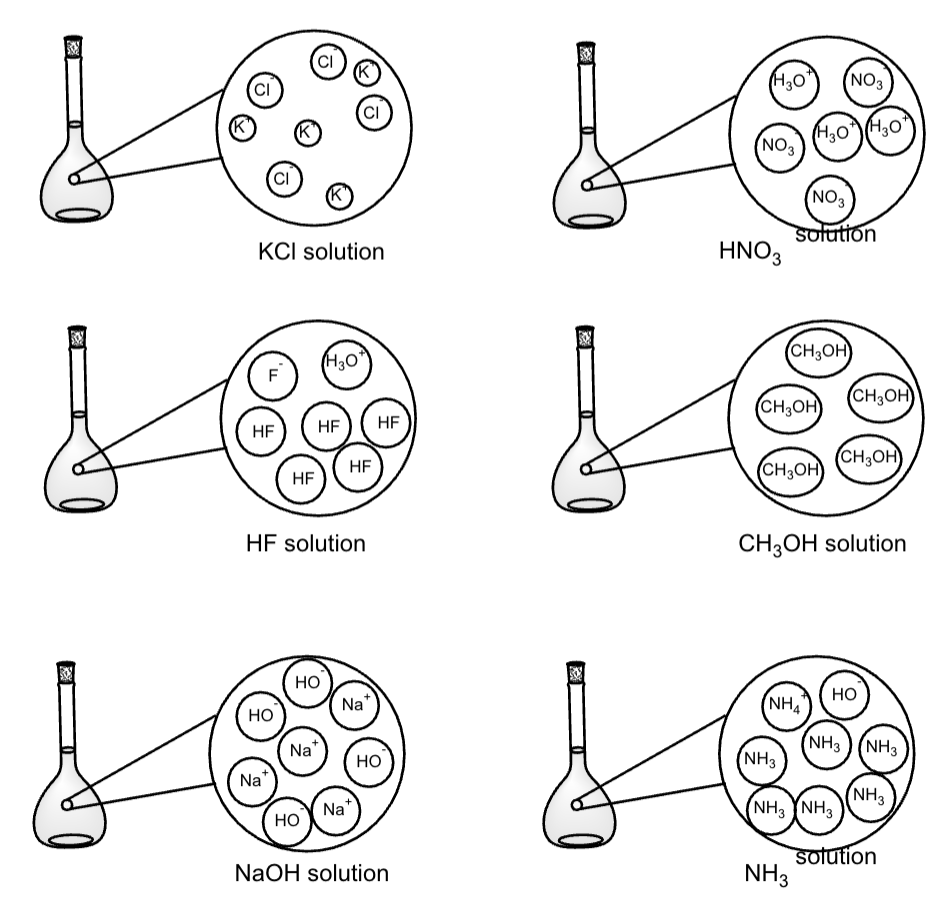
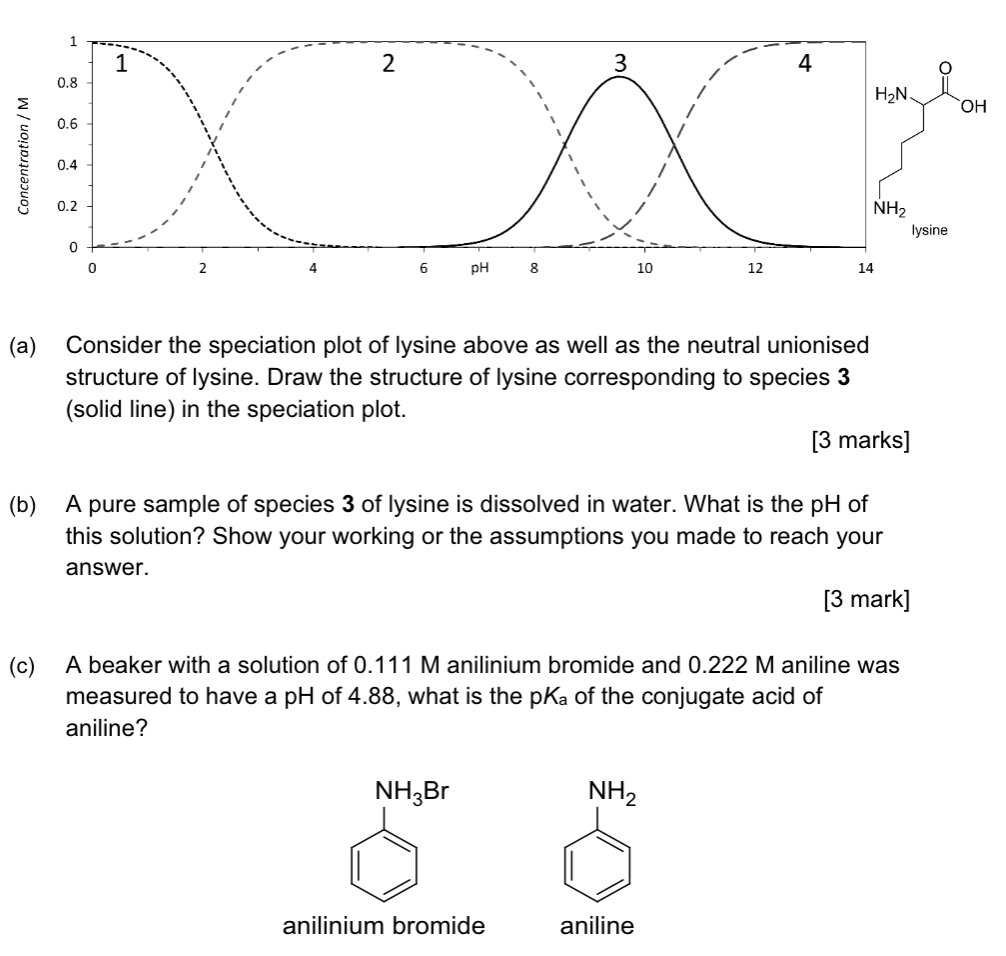
Which of the following aqueous solutions will contain the highest concentration of ions
(assuming 100% ionization and dissolution)?
(i) 1.5 M magnesium nitrate
(ii) 1.0 M aluminium chloride
(iii) 2.0 M sodium chloride
(i) 1.5 x 3 = 4.5 M ions (ii 1 x 4 = 4 M; iii 2 x 2 = 4 M)
Calculate the total concentration of nitrate ions in the final solution when the following three
separate solution volumes are combined into a single solution (assuming 100% ionization and
dissolution)
(i) 50 mL of 0.2 M potassium chloride
(ii) 150 mL of 0.1 M calcium nitrate
(iii) 300 mL of 0.05 M magnesium chloride
0.1 M Ca(NO 3) 2 = 0.2 M NO 3−, x 0.150 = 0.03 mol / (0.05+0.15+0.3) = 0.06 M
Calculate the molar concentration of 1 litre of fresh common household bleach of 4%
(40.0 g/kg) sodium hypochlorite (density = 1.08 g/mL). Your answer should be over 100mM.
1 L x 1.08 = 1.08 kg x 0.04 = 43.2 g NaOCl / 74.442 = 0.58 mol / 1 L = 0.580 M
Strontium hydroxide has a solubility of 2.25 g/100 mL water at room temperature. Calculate
the pH of a saturated solution of strontium hydroxide assuming it is a strong base. Mr = 121.63 g/mol

Calculate the pH of 1 litre of fresh common household bleach of 4%
(40.0 g/kg) sodium hypochlorite (density = 1.08 g/mL). NaOCl pKb = 6.50
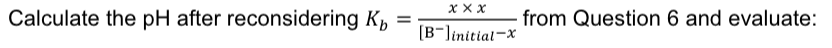


A buffer solution is made by mixing 100 mL of a 0.25 M solution of nitrous acid with 200 mL of
a 0.10 M solution of its conjugate base sodium nitrite (pKa = 3.4, 25 °C). What is the pH of this
buffer?
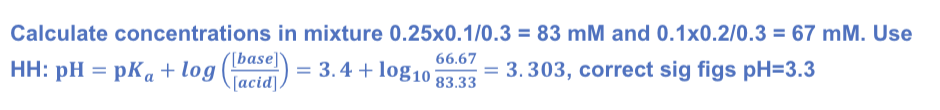
The following solutions are prepared for you:
• 2 M Sodium dihydrogen phosphate
• 2 M Disodium hydrogen phosphate
pKas for phosphoric acid are 2.14, 7.20, and 12.37.
What is the pH of each solution?

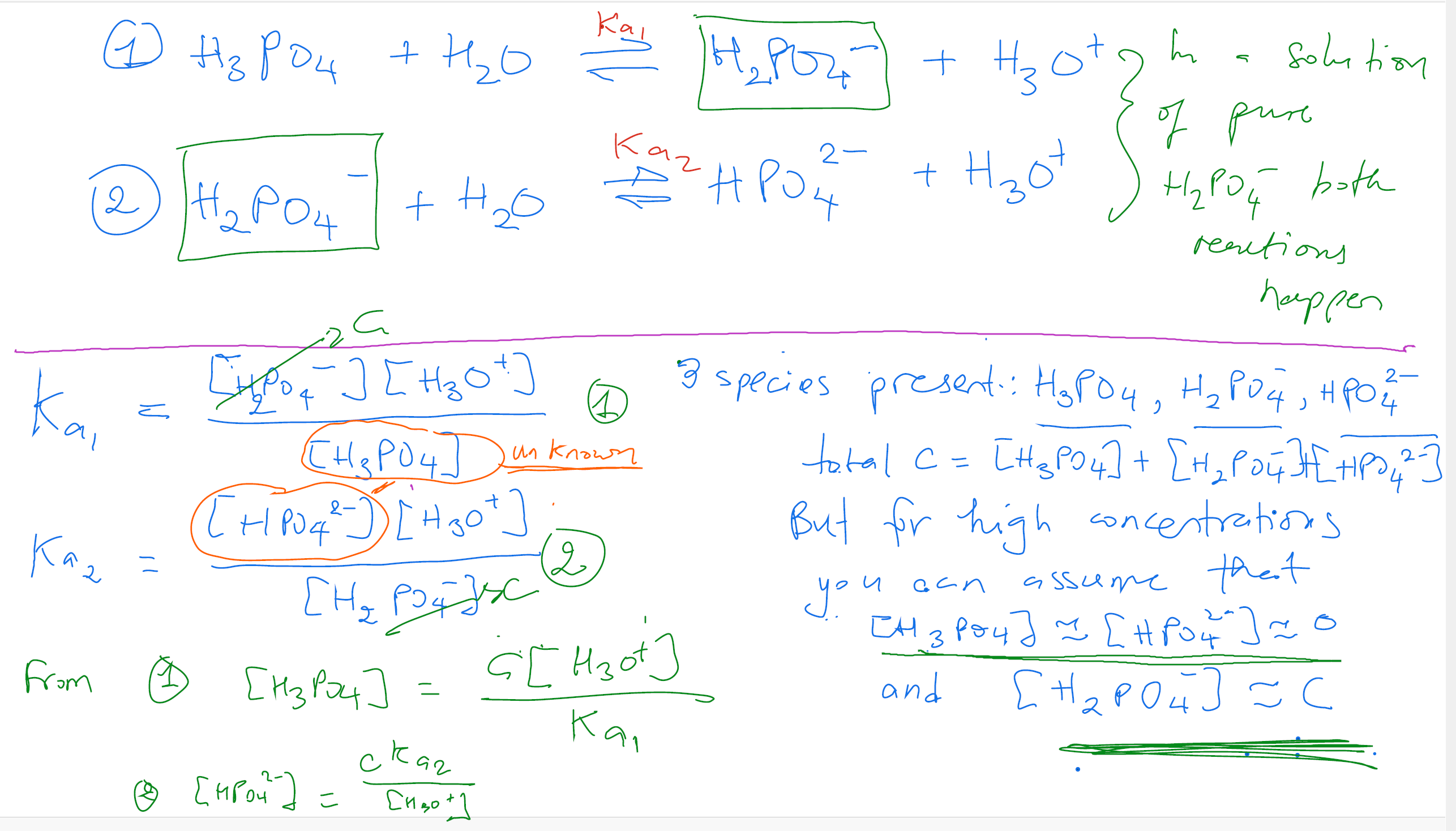
We have already made the assumption that because C is large compared to the values of the equilibrium constant, the concentrations of phosphoric acid will be equal to the concentration of hydrogen phosphate ion and negligible. This gives us:

To do this question, you need to make the assumption that the species is not dilute in solution (I was told this and could not find it in lecture slides either). Additionally, the pKa values need to be relatively spaced. Both these conditions are satisfied in this case so we can average the two pKa values surrounding the dominant species region (which will give us the pH of each 2M solution).
Since H3PO4 is triprotic, there will be 4 dominant species regions and hence 3 pKa values seperating them. At pH < 2.14, H3PO4 will be the dominant species. At 2.14 < pH < 7.20, H2PO4- will be the dominant species. And so on...
So just take the average of the two pKas surrounding each dominant species region and you should get your pH for NaH2PO4 and Na2HPO4.
You need to make 100 mL of a buffer with pH of 6.2. The following solutions are available to
you:
0.1 M Sodium dihydrogen phosphate
0.1 M Disodium hydrogen phosphate
pKas for phosphoric acid are 2.14, 7.20, and 12.37.

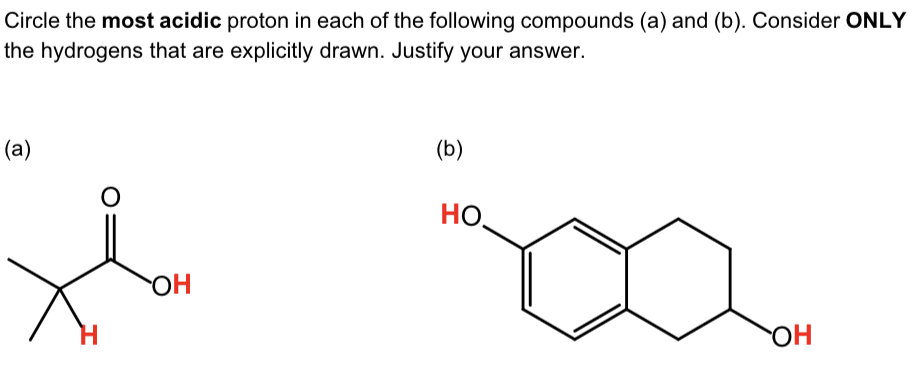
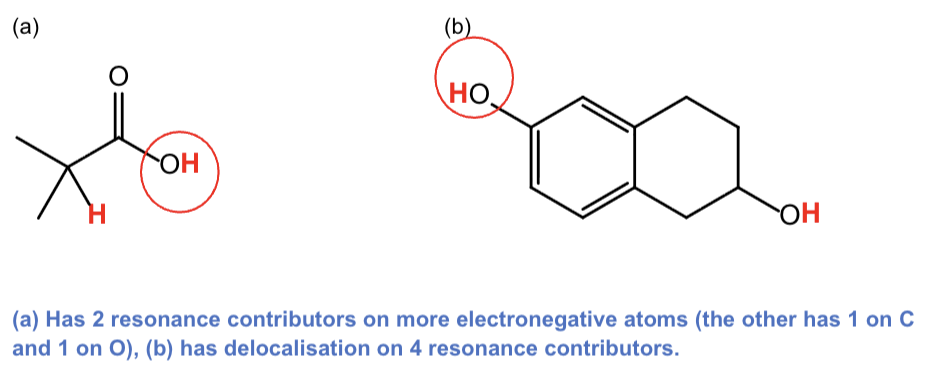
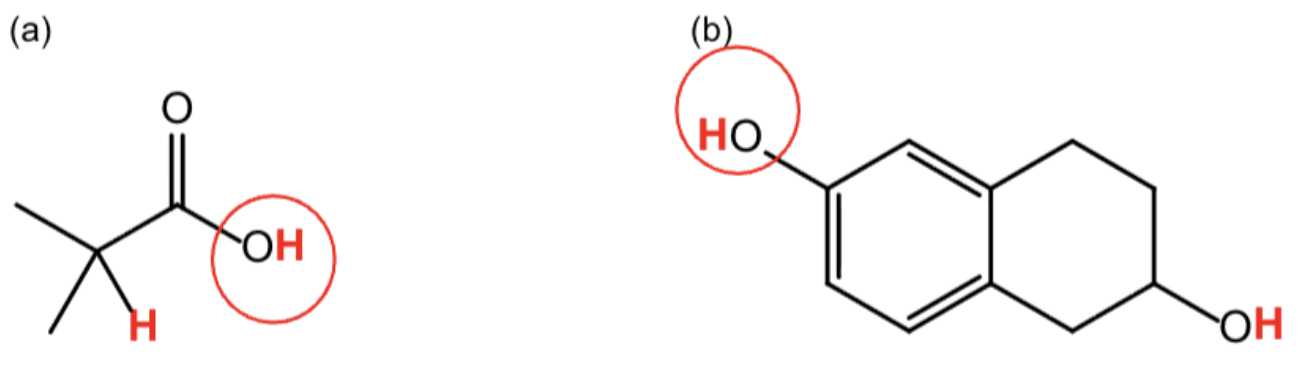
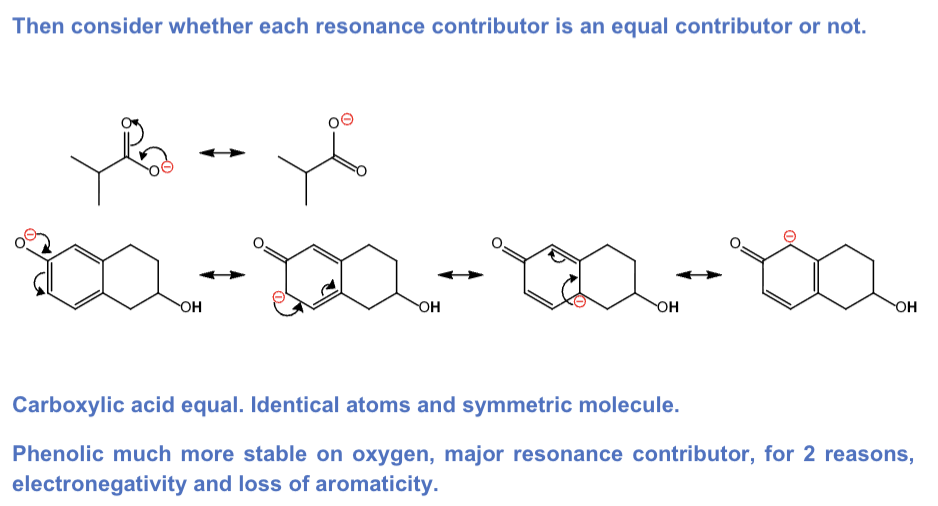
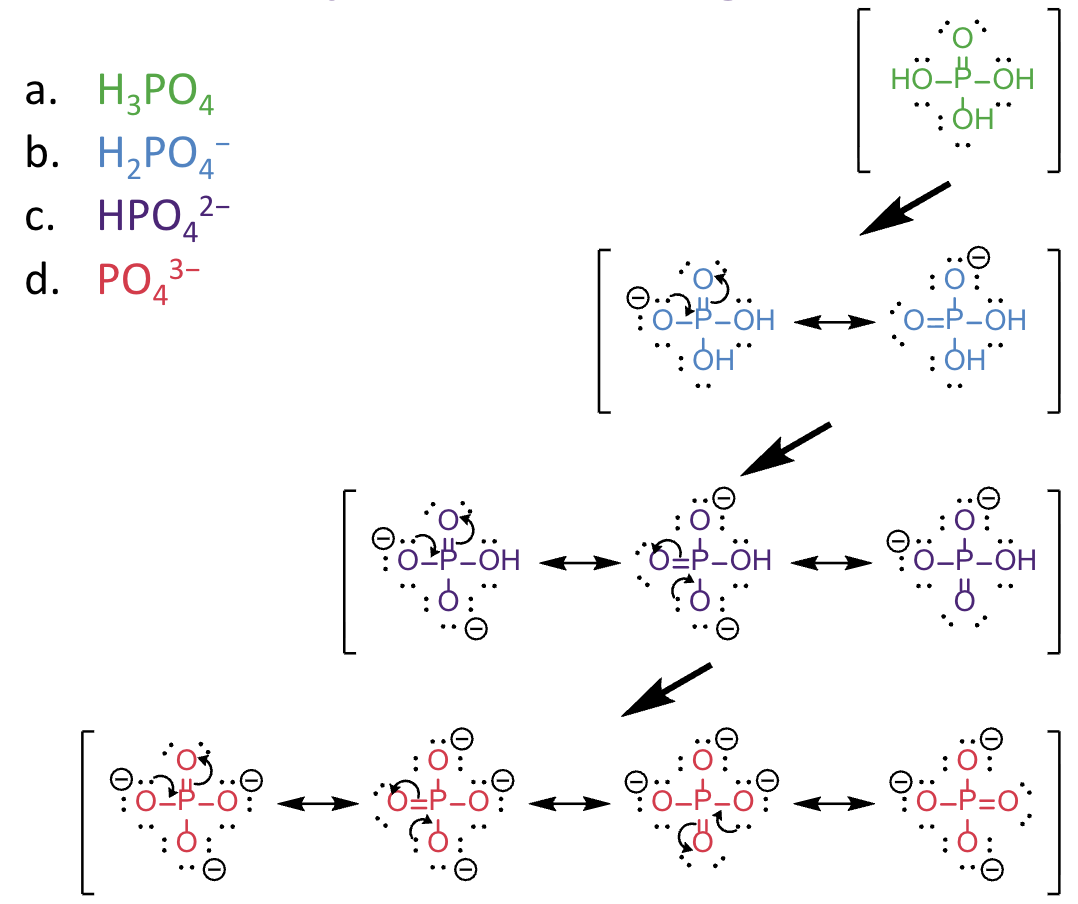
For a) b), draw all resonance contributors for
the conjugate base of the acidic proton you have circled.
Calculate the pH of 1.0 g NaF(s) dissolved in 1 L water. pKa = 3.17

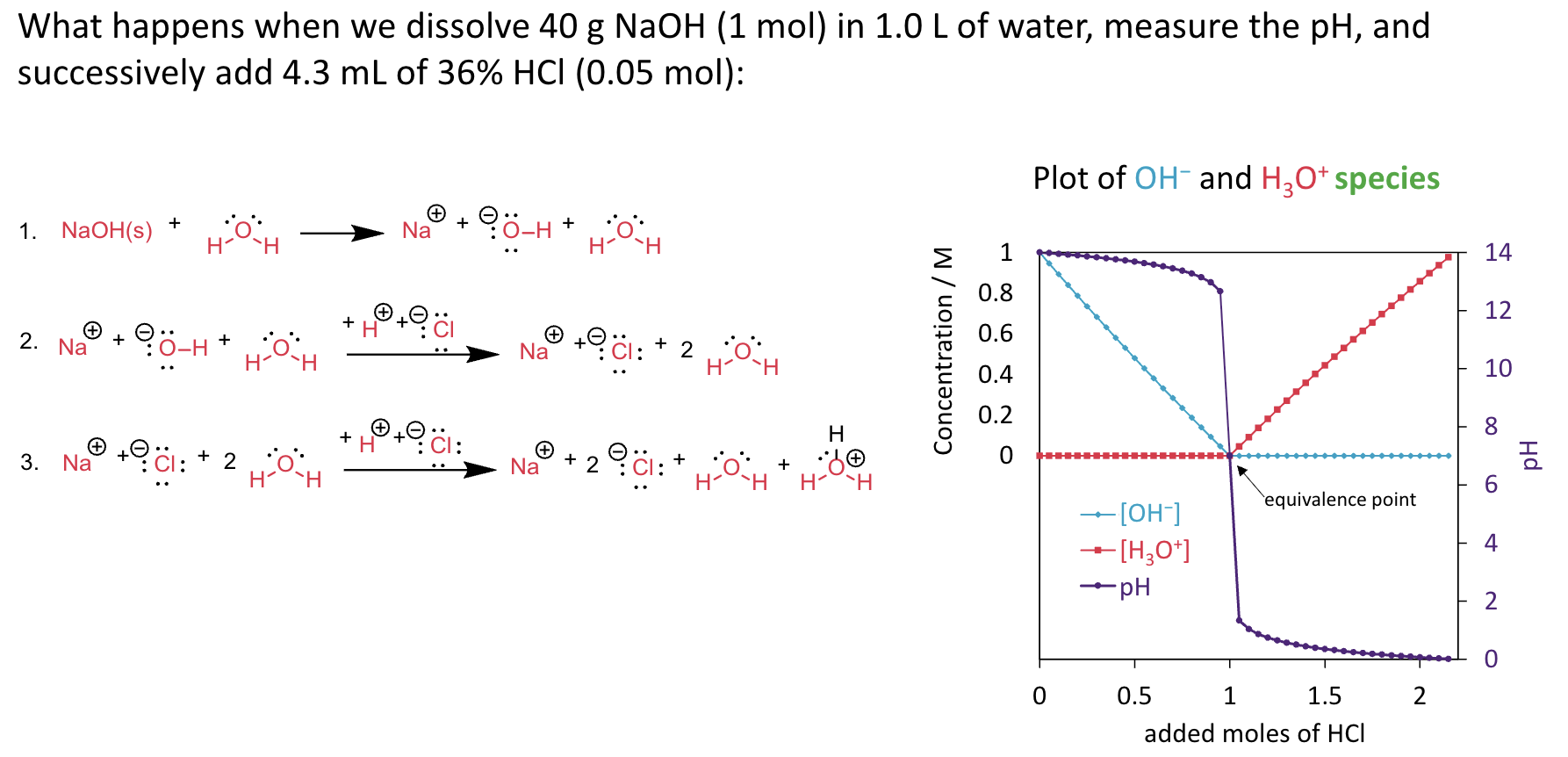
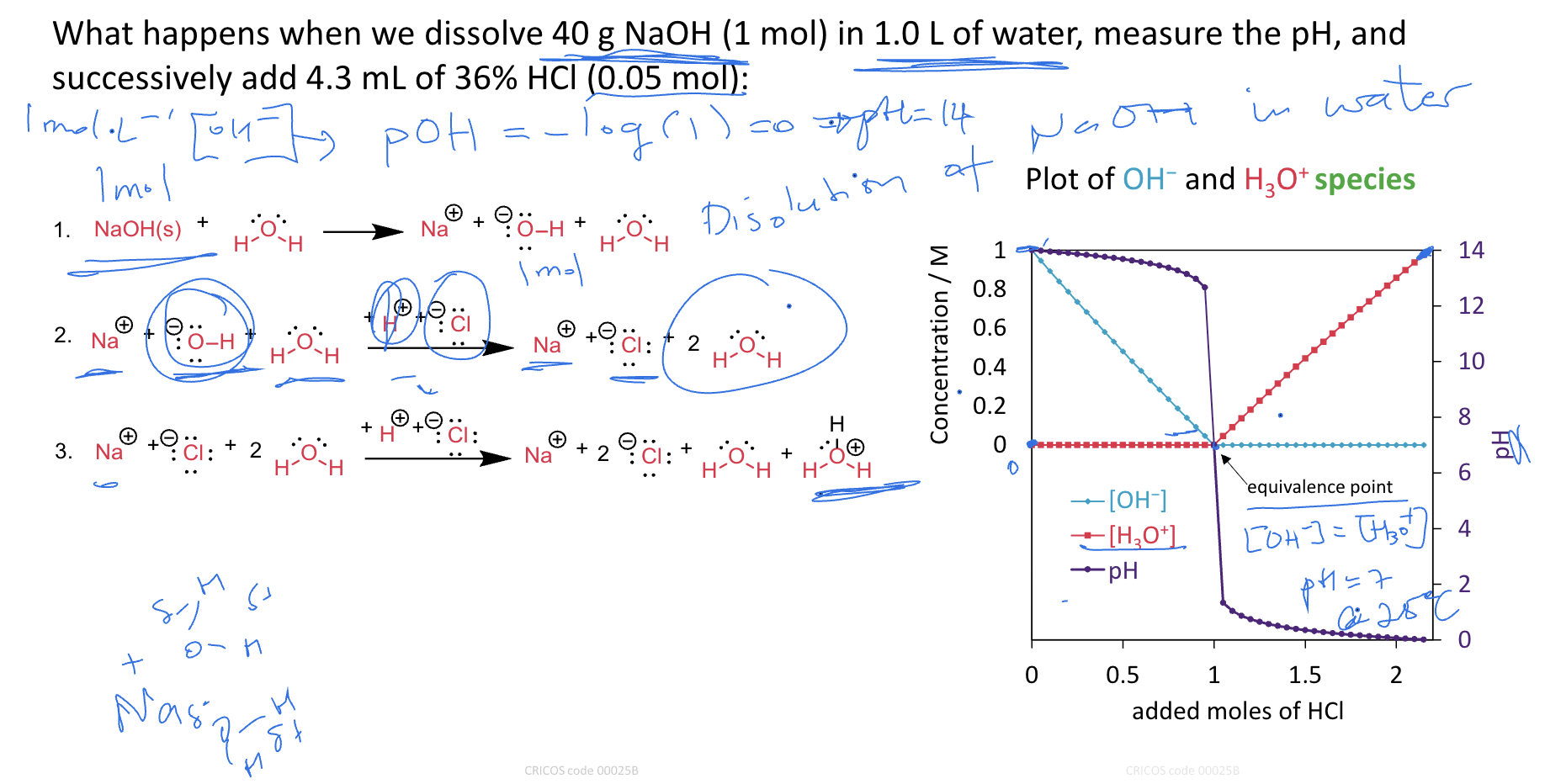
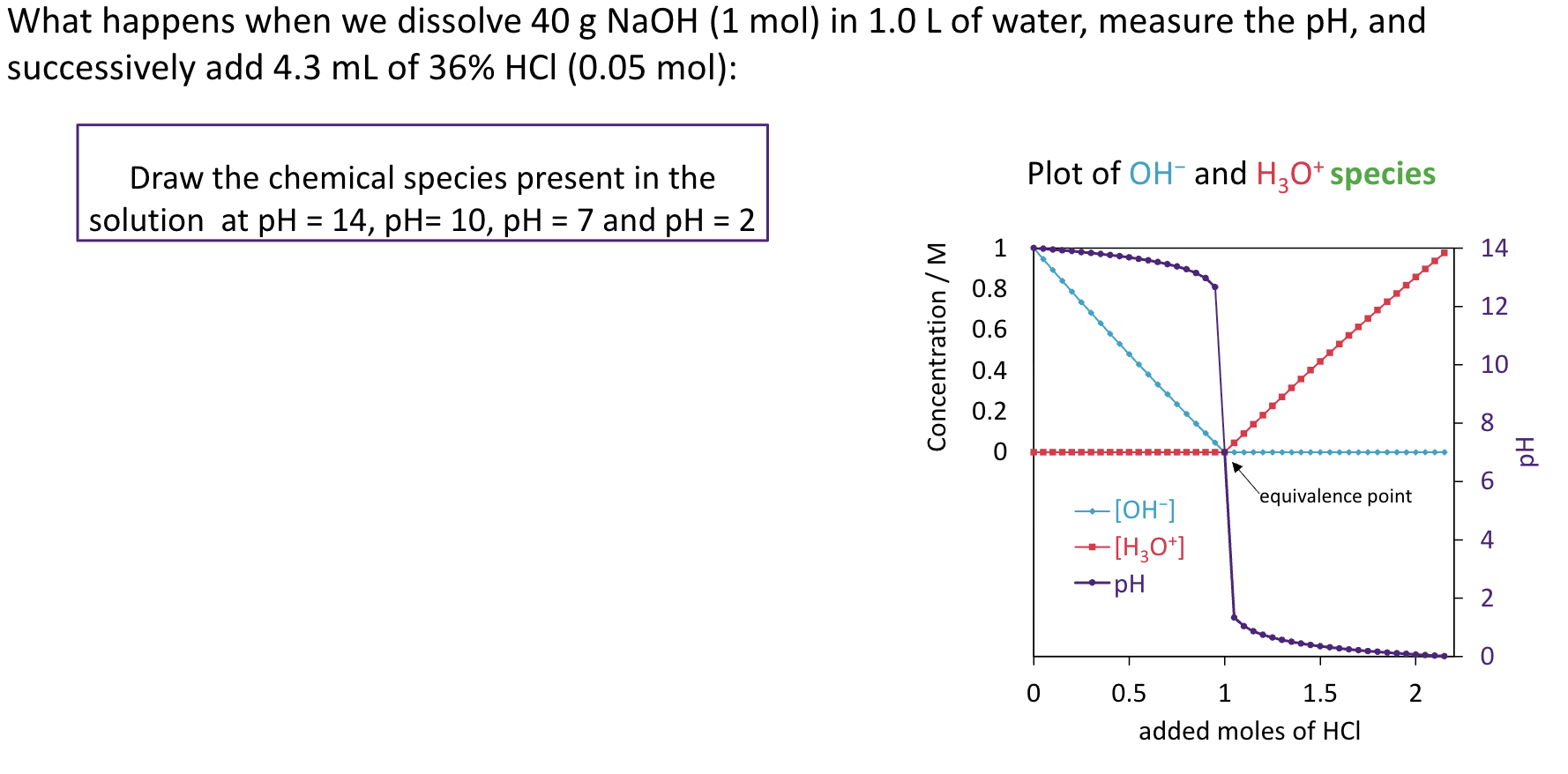
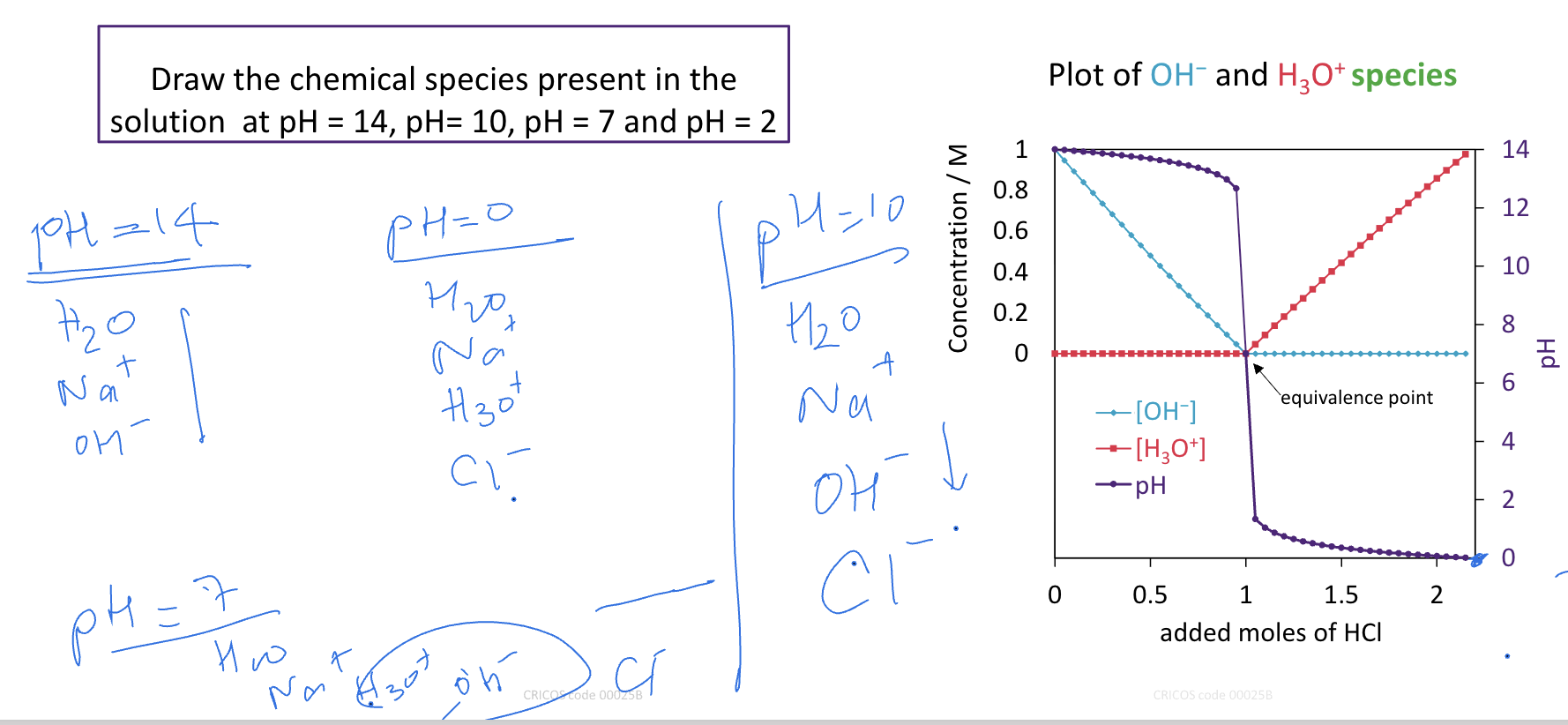
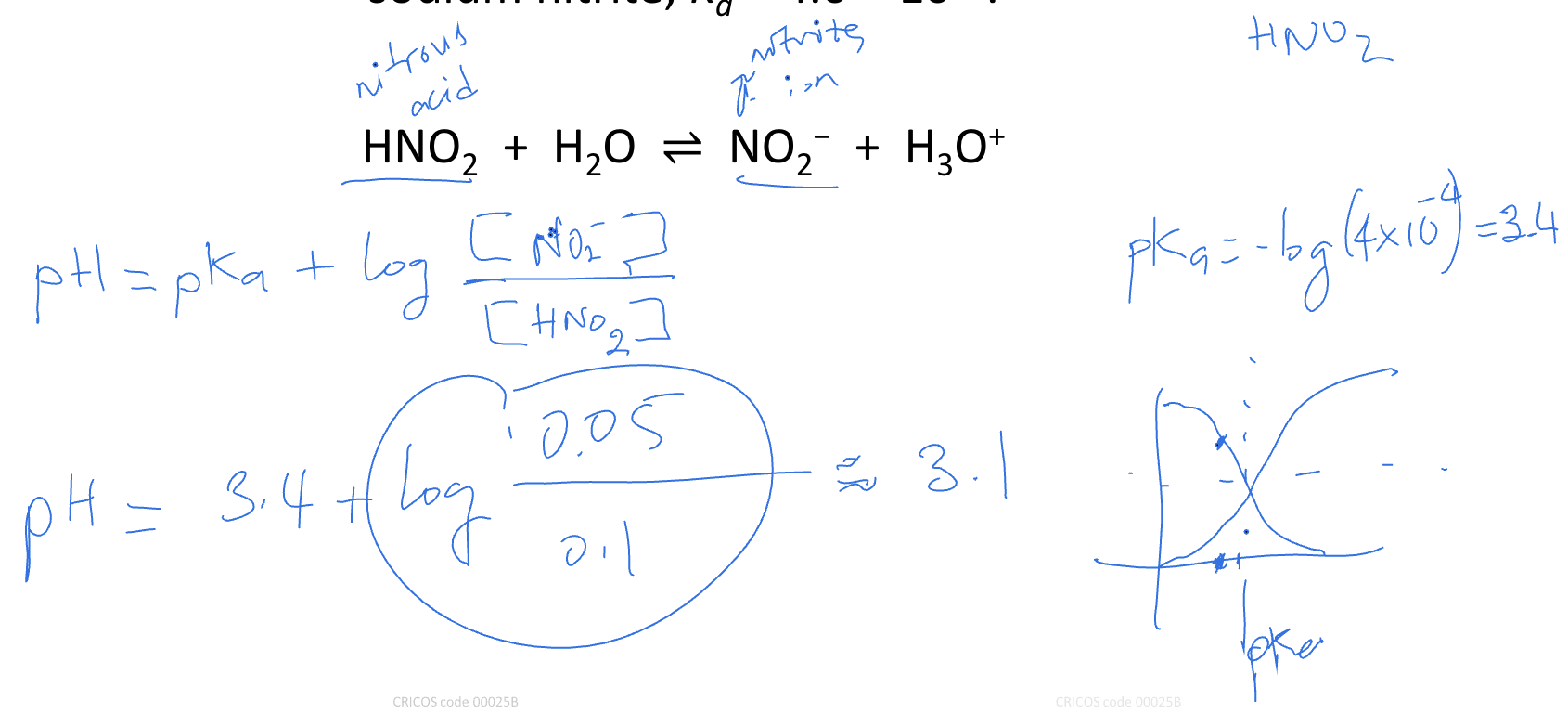
Calculate pH of a solution containing 0.100 M nitrous acid and 0.050 M of
sodium nitrite, Ka = 4.0 × 10−4.
HNO2 + H2O ⇌ NO2− + H3O+
How many moles of sodium acetate do we need to add to a 1 L solution with 0.050 moles of acetic acid to
make a buffer with a pH of 5.0? pKa = 4.75.

a. H 3PO4
b. H 2PO4−
c. HPO42−
d. PO43−
Which species has the greatest resonance stabilised charge?
Consider a balloon that has been inflated with a He/air mixture to a volume of 3.0 L at a temperature
of 25°C and atmospheric pressure (101.325 kPa). The balloon can stretch to a maximum capacity of
3500 cm3 .
The balloon is released and rises 330 m through the atmosphere. There, the external pressure is 0.89
atm and the external temperature is 9.0°C.
Will the balloon burst?

Calculate the change in Gibbs free energy of a sample of 2.00 mol of argon when
the pressure is changed from 1.00 x 10 5 N m−2 to 2.00 x 10 5 N m−2 at a constant
temperature of 298 K

The normal boiling point of ethanol is 78.4°C, and at this temperature
∆H vap = 38.7 kJ mol−1 . To what value must the pressure be reduced to
make it boil at 25.0°C in a vacuum distillation?

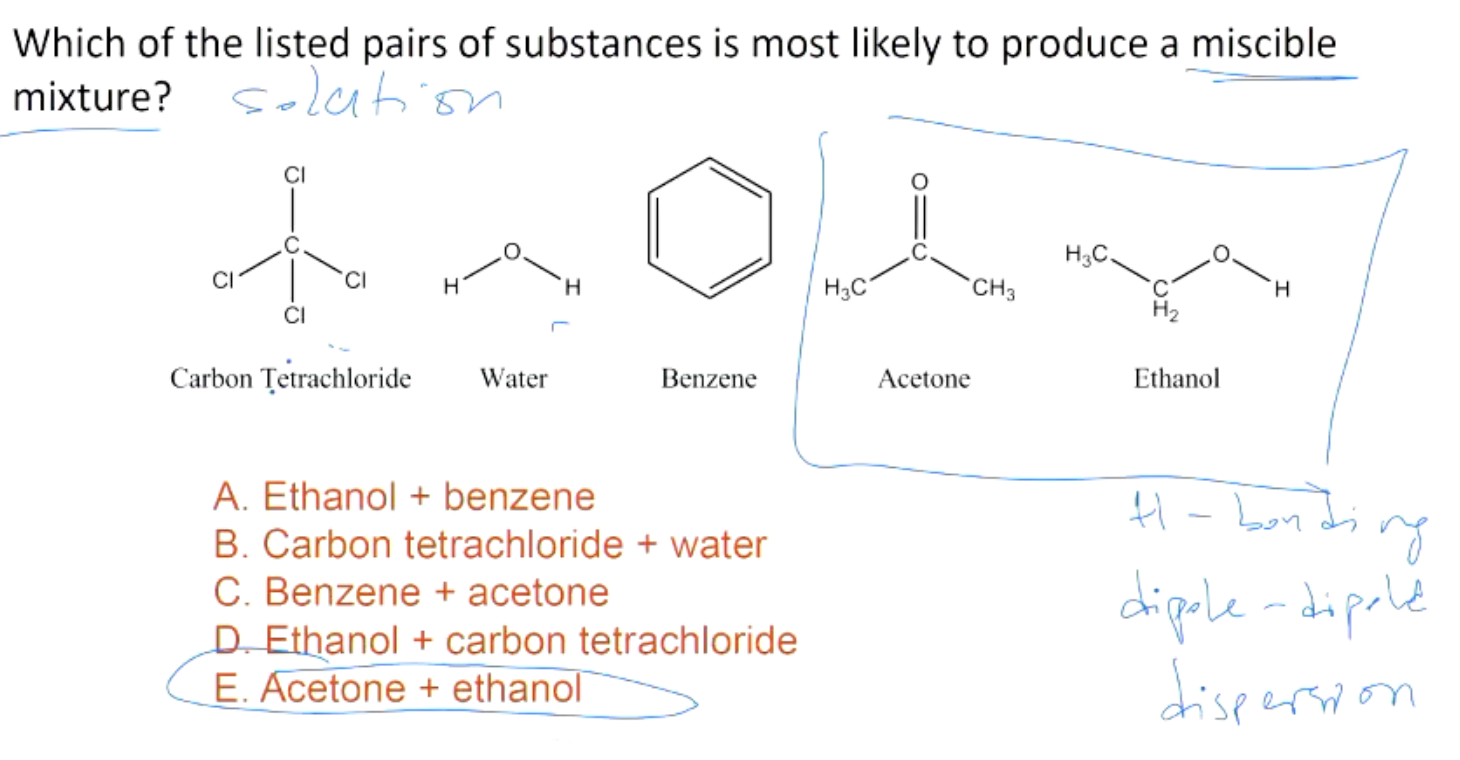
The saturated vapour pressures at 298 K for benzene and toluene are
12.85 kPa and 3.85 kPa respectively.
(i) What is the total vapour pressure over a solution containing 2.00 mol of
benzene and 3.00 mol of toluene?
(ii) What is the percentage composition of a solution of benzene and toluene
with a total vapour pressure of 9.50 kPa?


A solution is prepared from of 0.18 mol ethanol (C2H5OH) and 0.32 mol
methylcyclohexane (C7H14) and placed in a sealed container at 55°C. At
equilibrium, the partial vapour pressures above the solution of ethanol and
methylcyclohexane are 26.2 kPa and 24.2 kPa respectively.
The vapour pressures of the pure substances at 55°C are:
Ethanol = 37.3 kPa
Methylcyclohexane = 22.4 kPa
Calculate the mole fraction, vapour pressure and activity coefficient
above the solution for each component (assuming
ideal behaviour).

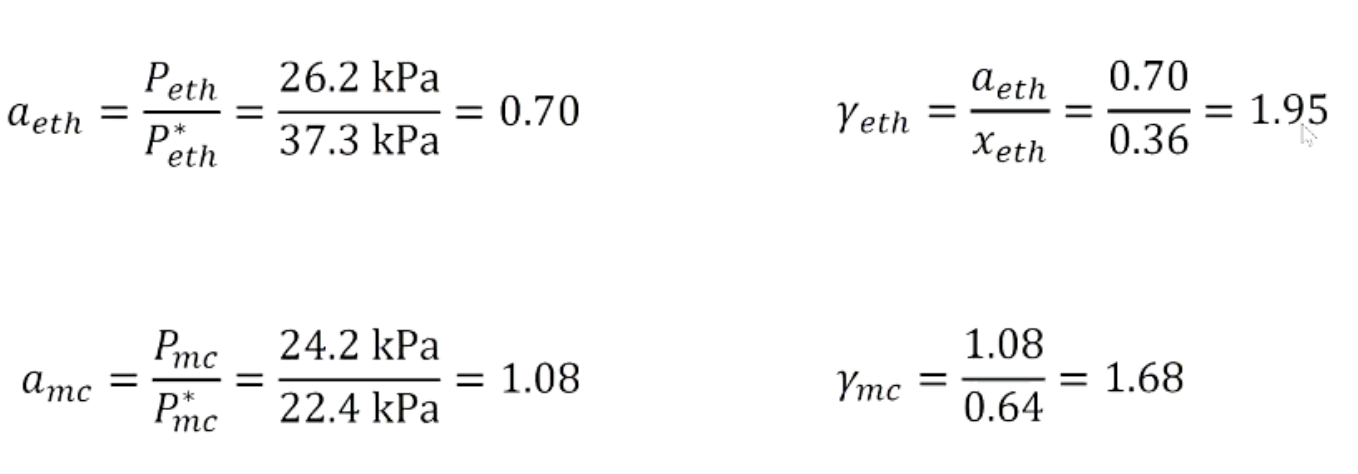
What is the value of the freezing point constant for water?
For water, Tfus = 273 K, M1 = 18.02 x 10⎼3 kg mol−1
∆Hfus = 6.00 kJ mol−1

Calculate the freezing point of an aqueous solution containing 39.5 g ethylene glycol
(HOCH 2CH 2OH, MW = 62.1 g mol−1) dissolved in 750 mL water (assume density of
water = 1.00 g mL−1).
𝐾𝑓 = 1.86 K kg mol−1
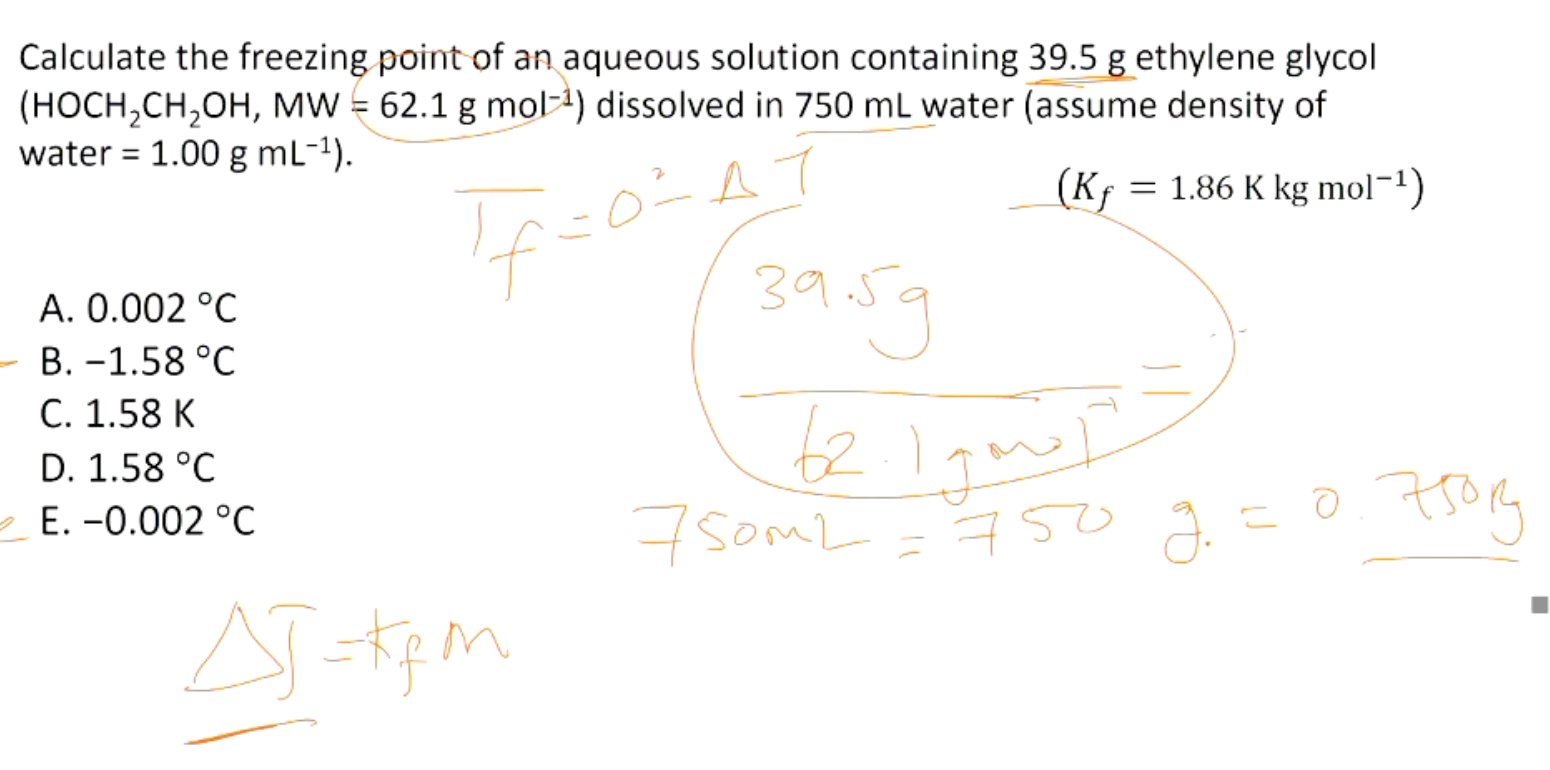
The osmotic pressure of a solution of poly(vinyl chloride),
PVC, in cyclohexanone at 298 K, expressed as height of solution, was
measured to be 8.13 cm. The concentration was 9.00 g L−1 and the
solution density 0.980 g cm−3. Calculate the molar mass of the polymer
Butane (C4H10) is a hydrocarbon that has various applications as fuel, for example in
barbeque cooking. The combustion of butane according to the equation below has been
found to have ∆H° = –5314 kJ and ∆S° = +311 J K –1 at 25.0 °C.
2 C4H10 (g) + 13 O 2 (g) → 8 CO 2 (g) + 10 H2O (g)
(a) Calculate the value of ∆G° for the combustion of one mole of butane at room
temperature.
(b) Calculate the value of ∆G° for the combustion of one mole of butane at 75.0 °C,
assuming that the standard enthalpy and entropy of combustion of butane are
temperature independent.
(c) Over what temperature range is the combustion of butane a spontaneous process?
(d) A chef uses some butane to cook a BBQ meal. Assume that (i) the cooking takes place
at 75.0 °C and atmospheric pressure, and (ii) the butane is completely burned (i.e. it is
converted completely into CO 2 and H2O, without forming any side products). What is
the total Gibbs energy change involved in burning 1 gram of butane?

Consider a piston containing 10.0 g of octane at 130 °C. At this temperature, octane is a
gas. If the external pressure is decreased from an initial value of 0.99 atm to a final value of
60 kPa at constant temperature, by how much will the Gibbs energy of the gas change?
What assumptions are required?
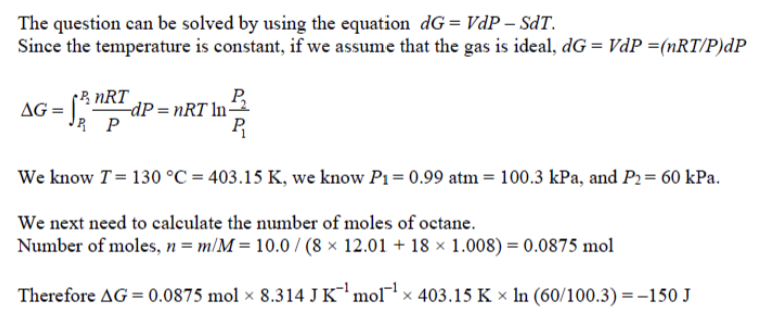
At 90ºC, the vapour pressure of methylbenzene is 533 mbar, and that of 1,2-
dimethylbenzene is 199 mbar. Assuming that they form an ideal solution, what is the
composition of a liquid mixture when the vapour pressure of the mixture is 480 mbar?

(a) An ideal binary solution has composition, described in terms of mole fraction, of
(x 1, x2). If we describe the composition of the vapour in equilibrium with this
solution by (y1, y2), show that:
Hint: The ratio of the mole fractions in the vapour is equal to the ratio of the partial
pressures of each component (Dalton’s Law)
(b) Calculate the composition of the vapour over the solution described in Question
3. At 90ºC, the vapour pressure of methylbenzene is 533 mbar, and that of 1,2- dimethylbenzene is 199 mbar. Assuming that they form an ideal solution, what is the composition of a liquid mixture when the vapour pressure of the mixture is 480 mbar?
(c) Comment on the difference between the compositions of the liquid and the
vapour.

The vapour pressure of 2-propanol (CH3CHOHCH3) is 50.00 kPa at 338.8°C,
but it fell to 49.62 kPa when 8.69 g of an involatile organic compound was
dissolved in 250 g of 2-propanol. Calculate the molecular mass of the
compound.
Hint: Use Raoult’s Law to calculate the mole fractions of the components and therefore
the number of moles of the compound, assuming ideal behaviour.


Strontium hydroxide, Sr(OH)2 , has a solubility of 9 g/100 mL water at room temperature. Calculate the pH of a saturated solution of strontium hydroxide.
14.17
If 100 mL of the saturated strontium hydroxide solution was mixed with 200 mL of 2 M HCl, what would be the pH of this solution? Strontium hydroxide, Sr(OH)2 , has a solubility of 9 g/100 mL water
0.08