Halogens (Group 7)
1/8
Earn XP
Description and Tags
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
9 Terms
What colour and state is chlorine?
yellow - green gas
What colour and state is bromine?
red - brown liquid
What colour and state is iodine?
purple solid
What is the trend going down group 7 in terms of state?
There is a trend in state from gas to liquid to solid down the group
This is because the melting and boiling points increase as you go down the group
From this, you can predict that any halogens above chlorine will be gases (their boiling points will be even lower), and any below iodine will be solids (their melting points will be even greater)
What is displacement?
Displacement is the process where an atom in a compound is replaced by a more reactive compound.
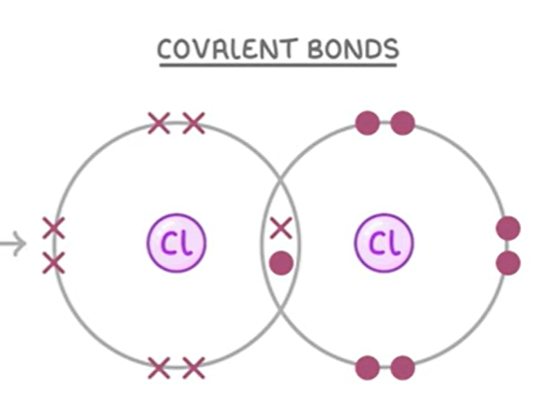
Halogens come in groups of_____. What is this called?
Halogens come in pairs
This is called diatomic molecules
It is where two of the same group 7 element form a covalent bond and share an electron to have a full outer shell.
What is the trend going down group 7 in terms of reactivity?
As you go down group 7, the reactivity decreases
This is because the less energy shells the halogen has, the more reactive it is because the outer energy level is closer to the nucleus and so the force of attraction between the nucleus and another element’s outer shell is greater.
Halide
Where a halogen gains an electron and becomes a 1- charged ion
What is the chemical equation for displacement in group 7?
E.g potassium bromide + chlorine —> potassium chloride + bromine
2KBr + Cl2 —> 2KCl +Br2