Water Treament
1/34
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
35 Terms
How is Silica removed?
Along with the precipitated Magnesium Hydroxide, Mg(OH)2
The silica is removed from the water through electrostatic adsorption onto the surface of the Mg(OH)2 precipitate
Mg(OH)2 forms as MagOx is added as a dry chem at the top of the WLS
How can more Mg(OH)2 be added?
By adding MgO (Magnesium Oxide - aka MagOx) that then forms Mg(OH)2 upon contact with water
What is hard water?
Water that contains a lot of dissolved ions → particularly Mg+2 and Ca+2
Why does water become hard?
Water is a strong solvent
It picks up minerals as it moves through rock
Produced water has been in contact with the formation for millions of years - amplifying its hardness compared to typical groundwater
Why is water hardness a problem in SAGD operations?
The SPF is essentially a steam factory
hard water causes efficiency and mechanical problems for steam generators
What are the 3 main functions/processes in a SAGD CPF
Treat the produced emulsion
ie. separate the oil, water, and gas
Treat the produced water
Reduce hardness
Generate steam for injection
Water treatment is a balancing act between __________ and _________
corrosion and scale control
Why is hardness a problem in SAGD SPFs?
Because hardness causes severe scale in in steam generators
The magnesium and calcium ions precipitate as scale in boilers and exchangers
This scale reduces the efficiency and shortens the life of the equipment used in steam generation (ie. boilers - BFW)
Hardness also damages the heat transfer equipment, which requires downtime for acid cleaning
Heat exchangers
Produced water coolers
Name 3 types of common SAGD scale minerals
CaCO3 → carbonate
CaSO4→ anhydrite
Calcium-magnesium-silicates
What are the 3 main methods of reducing water hardness at Christina Lake?
WLS → Warm Lime Softening
Bulk removal (80-90% of the load)
Solids settle as sludge and are physically removed
WAC → Weak Acid Cation exchange
removes hardness associated with alkalinity (bicarbonate hardness)
SAC → Strong Acid Cation exchange
removes permanent hardness
removes remaining Ca2+ and Mg2+
final polishing before boilers
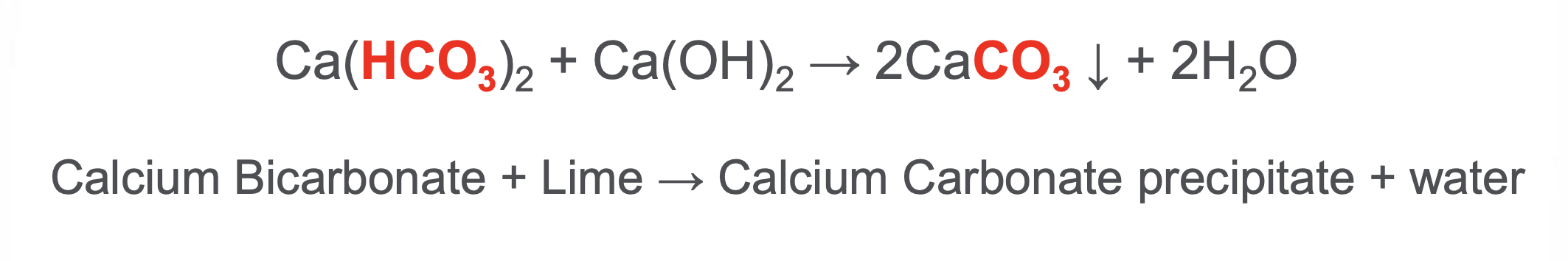
What is the difference between temporary and permanent hardness?
Temporary → carbonate hardness → caused by magnesium and calcium bicarbonates dissolved in water
Ca(HCO3)2
Mg(HCO3)2
temporary because it can be removed by lime addition (WLS)
WLS + WAC
Permanent → non-carbonate hardness → caused by sulfates, nitrates or chlorides of calcium and magnesium that cannot be removed by lime addition (WLS)
CaSO4
MgSO4
CaCl2
MgCl2
SAC
What is the device used to measure turbidity?
Nephelometer
measures tubidity in units of NTU → Nephelometric Turbidity Units

What is alkalinity?
A measure of the water’s ability to neutralize acids
depends mainly on the amount of bicarbonate present
Too high → scale deposits can form
Too low → corrosion
Scale formation rate ______ with increasing pH
increases
What do the following acronymns stand for?
CPF
WLS
WAC
SAC
BFW
CPF → Central processing facility
WLS → Warm lime softening
WAC → Weak acid cation exchange
SAC → Strong acid cation exchange
BFW → Boiler feed water
WRU → Water recovery unit
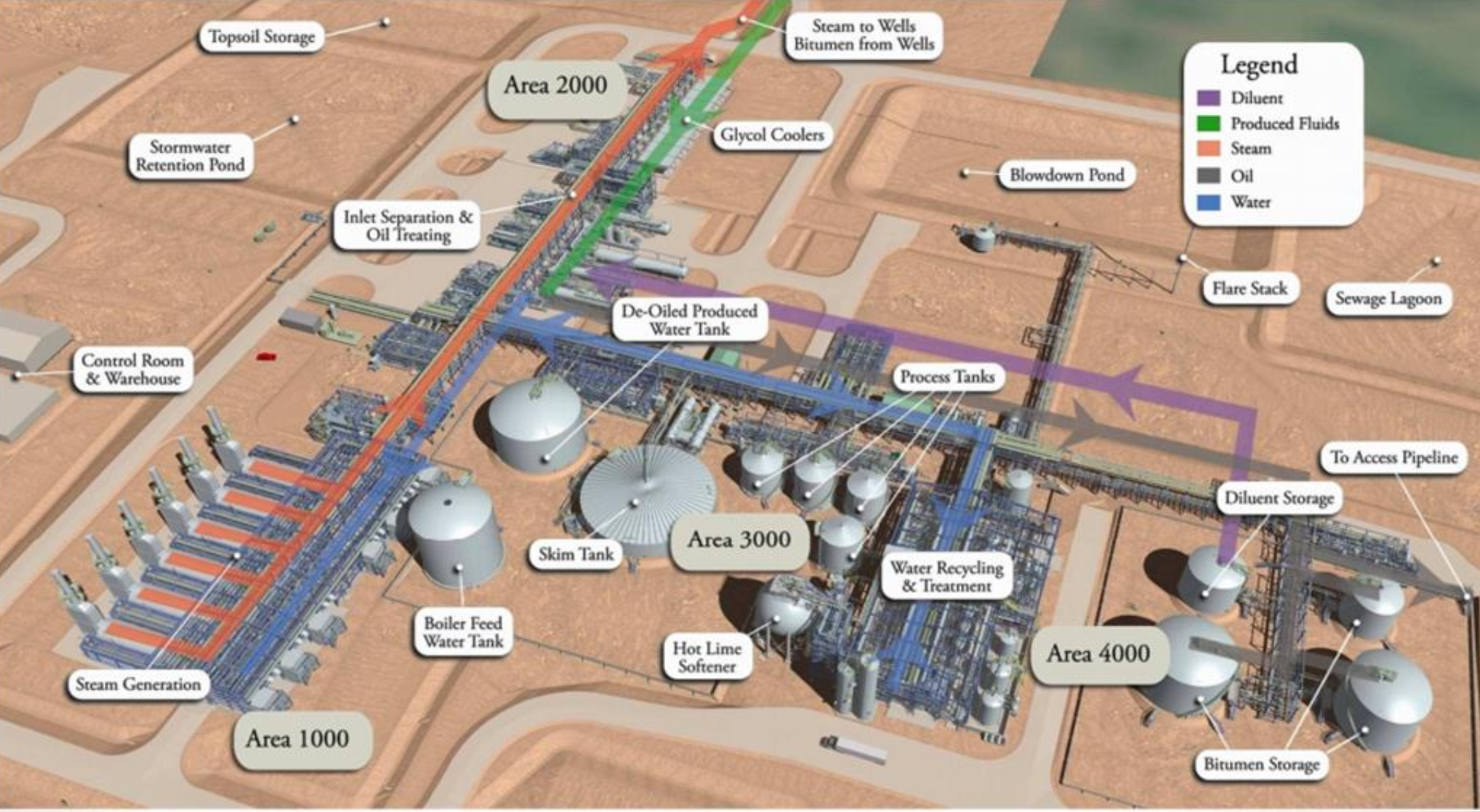
What is the skim tank used for?
Removes free and weakly emulsified oil from the produced water before it continues on for treatment
Situated after the FWKO and Treaters
Before WLS
What are the main processes in HLS?
Deaeration to remove CO2 and Oxygen
Induce precipitation reactions
Silica reduction
Clarification
What are the 3 main chemicals used by SLB at the CPF?
Emulsion Breaker (EMBR 10070A)
Coagulent (CLAR 18114A)
Flocculent (CLAR 01601A)
What is the purpose of EMBR 10070A and where is it used at the plant?
It is used to break emulsions in the emulsion line feeding the plant
Added into the line at loop stations to the north of the plant
What is the purpose of CLAR 01601A and where is it used at the plant?
Flocculent
Used to bind the destabilized particles into large, dense flocs that settle
Added at the spray box on the WLS
What is the purpose of CLAR 18114A and where is it used at the plant?
Coagulent
Used to destabilize the suspended particles by neutralizing the surface charge on them
Once particles are neutralized they can collide and stick together (ie. coagulate)
What is sludge recirculation used for in the WLS?
Provides seeds to increase precipitation
These recirculated solid particles provide a surface for the hardness ions to attach to
This improves floc size and settling rates
What is the process for checking Treater taps?
Open sample line
Open by-pass/slop line
Open the tap you want to check
Go to hood and open valve to check tap
Find CW and label with clip or magnet
Describe the chemicals used in WLS
Into the top → lime, magox, soda ash (dry chems)
Inlet produced water stream → Coagulent (CLAR 18114A)
Fed into spray box → Flocculant (CLAR 01601A)
What is the primary role of lime in WLS?
Alkalizing agent that forces precipitation of temporary hardness
What is the primary purpose of magox (MgO) in WLS?
Reduction of silica
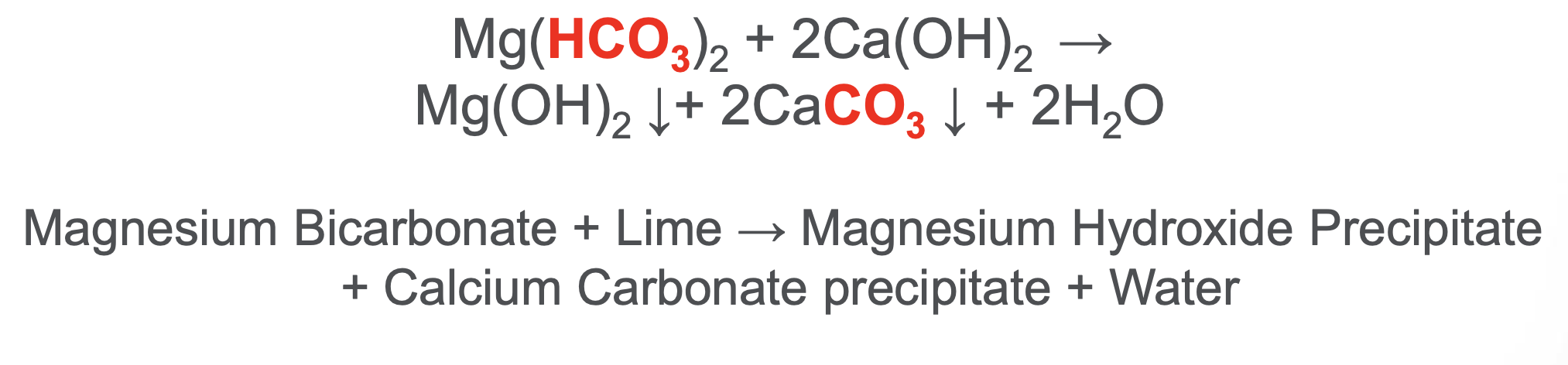
Where is the OIW concentration evaluated?
Describe the process
Off the water dump line of the FWKOs and treaters
Take 2 50ml samples of water off the dump line
Add 2 pumps of acid to one and shake
Add 50ml Varsol to each
Shake both and compare colour of Varsol
Darker = more residual hydrocarbons = higher ppm
Describe the process of testing sales oil and FWKO oil
Purpose → shows treater performance by testing BS&W before and after the treater
Collect a samples from the FWKO outlet and the sales line
Bring to oil lab
In centrifuge tubes add Varsol to 100 and then the oil to 200 for both samples
Place in the centrifuge on opposite ends (to counter-balance)
Spin for 10mins and read the BS&W
What can happen if too much floc or coag is added in WLS?
You will overcharge the particles and they will repel eachother
What is the name of the cones used when testing the taps on the WLS?
Imhoff Cones

Describe the process of testing the taps on the WLS units
Fill your cones to 100 and start the stop watch
After 10mins take the sludge % reading from each cone
What is meant by carryover?
An unwanted substance being carried past where it should have been removed.
ie. oil carryover from a treater