Orgo unit 1
1/54
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
55 Terms
Alkane (Substituent: alkyl)
a hydrocarbon with no multiple bonds
nonpolar
tetrahedral (sp3 hybridized)
free radical reactions
Alkene (Substituent: alkenyl)
a hydrocarbon with at least 1 C-C double bond
nonpolar
trigonal planar (sp2 hybridized)
addition reactions, oxidative cleavage
stability increases with # carbons attached
Alkyne (Substituent: alkynyl)
a hydrocarbon with at least 1 C-C triple bond
nonpolar
linear (sp hybridized)
addition reactions, oxidative cleavage, acid-base reactions
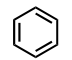
Benzene Ring (Substituent: phenyl)
a 6-membered ring containing 3 alternating double bonds
substitution reactions (less reactive than normal alkenes bc aromaticity)
Alcohol (Substituent: hydroxy)
OH group attached to C (unless attached to C=O)
polar (OH does hydrogen bonding)
acid-base reactions, substitution reactions, oxidation reactions
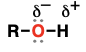
ether (Substituent: alkoxy)
an oxygen flanked by two carbons
borderline between polar and nonpolar (dipole-dipole)
acid-base reactions
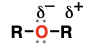
alkyl halide (Substituent: haloalkyl)
an alkyl group attached to a halogen
considered nonpolar but more polar than alkanes
substitution and elimination reactions
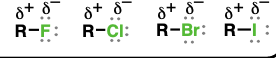
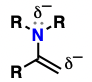
amine (Substituent: amino)
a nitrogen attached to a simple carbon or hydrogen atoms
polar (N-H does hydrogen bonding)
acid-base, substitution reactions
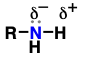
aldehyde (Substituent: oxo)
a carbonyl (C=O) attached to hydrogen and another carbon
C=O bond is somewhat polar
addition reactions
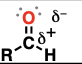
ketone (Substituent: oxo)
a carbonyl (C=O) flanked by two carbons
C=O bond is somewhat polar
addition, acid-base reactions
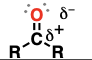
carboxylic acid (suffix: -oic acid)
a carbonyl (C=O) adjacent to a hydroxyl (OH) and an R group
acid-base, acyl substitution reactions
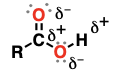
ester
a carbonyl group adjacent to an alkoxy (OR) and an R group
acyl substitution reactions and addition reactions
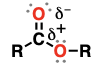
thiol (Substituent: mercapto)
sulfur-containing analogs of alcohols
acid-base reactions, substitution reactions
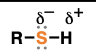
sulfide (suffix: sulfide)
a sulfur flanked by two carbon atoms
reduction reactions
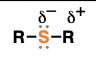
epoxide (suffix: oxide)
cyclic ethers consisting of a 3-membered ring containing 1 oxygen and two carbons
ring opening when treated with nucleophiles
nitrile (suffix: nitrile)
a molecule containing the CN group
reduction reactions, hydrolysis reactions
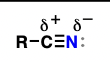
imine (suffix: imine)
nitrogen-containing analogues of ketones and aldehydes
reduction reactions, hydrolysis
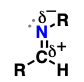
acyl halide (suffix: -oyl halide)
Carboxylic acid derivatives where OH has been replaced by a halogen
acyl substitution reactions
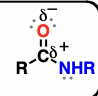
amide (suffix: amide)
nitrogen-containing analogues of esters
reduction reactions, hydrolysis reactions
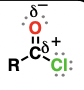
anhydride (suffix: anhydride)
oxygen atoms flanked by two acyl groups
acyl substitution reactions, reduction reactions
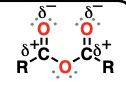
Nitro
a molecule containing the NO2 group
reduction reactions, acid-base reactions
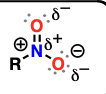
Enol
alkenes attached to a hydroxyl substituent
good nucleophiles, will perform addition reactions to aldehydes and ketones
also will tautomerize to aldehydes and ketones
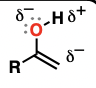
Enolate
the conjugate bases of enols
excellent nucleophiles
addition reactions to aldehydes and ketones
substitution reactions with alkyl halides
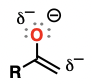
Enamine
alkenes attached to an amino substituent
excellent nucleophiles
addition reactions to aldehydes and ketones
substitution reactions with alkyl halides
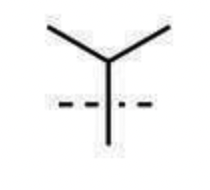
tert-butyl

isopropyl
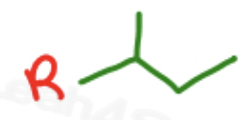
isobutyl

sec-butyl
Formula for Degrees of Unsaturation
A formula used to determine the number of rings and multiple bonds in a molecule. It is calculated as DU=1+n−2m+2o−2q, where (n) is the number of carbons, (m) is the number of hydrogens, (o) is the number of nitrogens, and (q) is the number of halogens.

Polar Effect
An atom not connected to H draws e- density due to its electronegativity which weakens H and other atom bond —> dissociation —> stronger acid.
Charge Effect
Atom connected to H has + formal charge —> atom less stsable —> easier to remove H —> neutral or stable covalent bond
Element effect
atom connected to H is lower on periodic table —> larger atom —> weaker and longer bond —> easier for H to dissociate. can also argue atom connected to H is more electronegative so greater e- density and weaker bond —> easier for H to dissociate
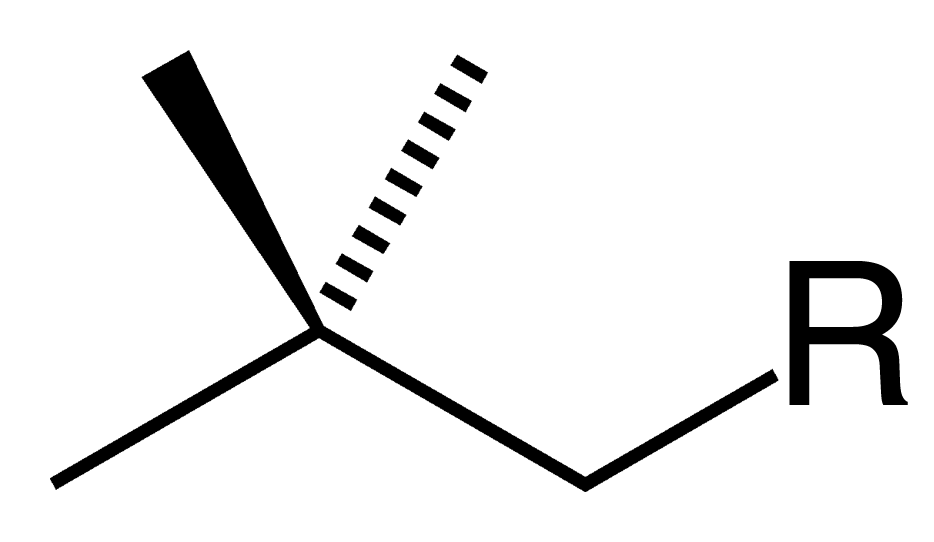
Neopentyl
Anti conformation - newman projection
lowest staggered confirmation
Gauche conformation
other 2 staggered conformations - not anti
eclipsed - newman projection
steric hinderance, without hyperconjugation
staggered - newman projection
less steric hinderance and hyperconjugation can occur. this hyperconjugation leads to lower energy and thus makes for a more stable conformation
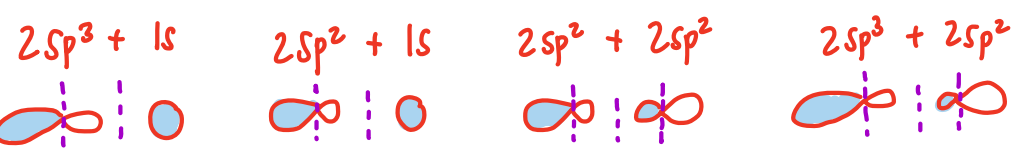
2sp3 hybridization electron energy diagram
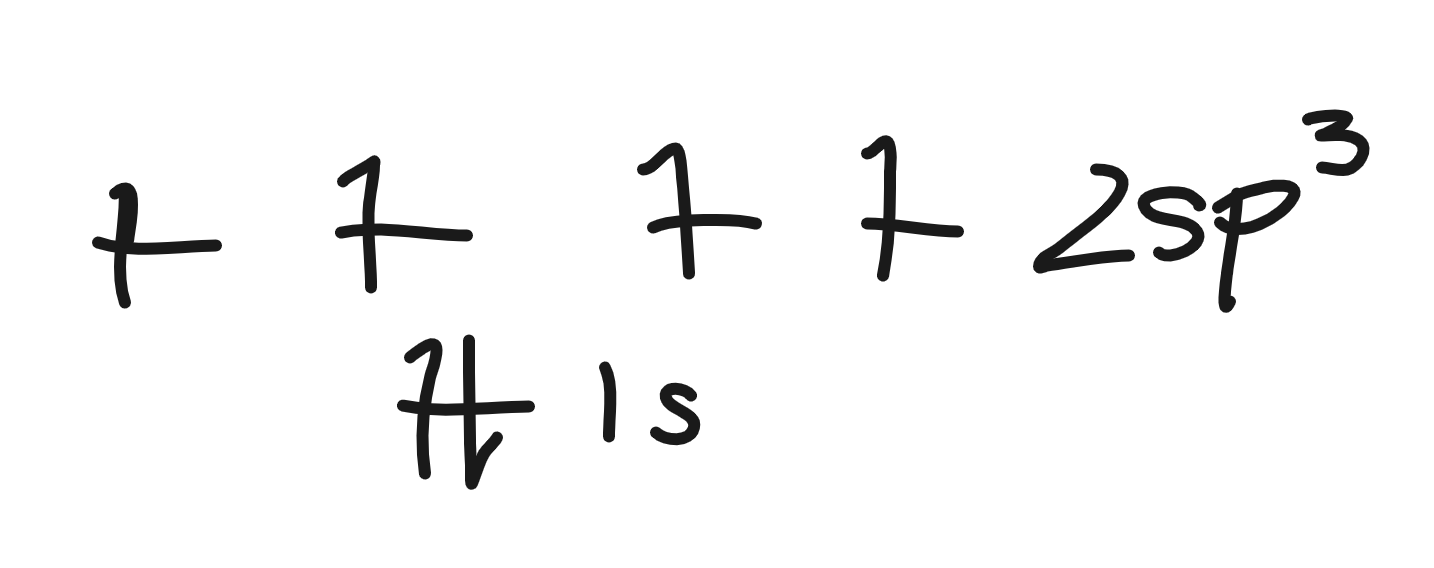
4 sigma bonds
how many bonds can sp3 hybridization make
2sp2 hybridization electron energy diagram
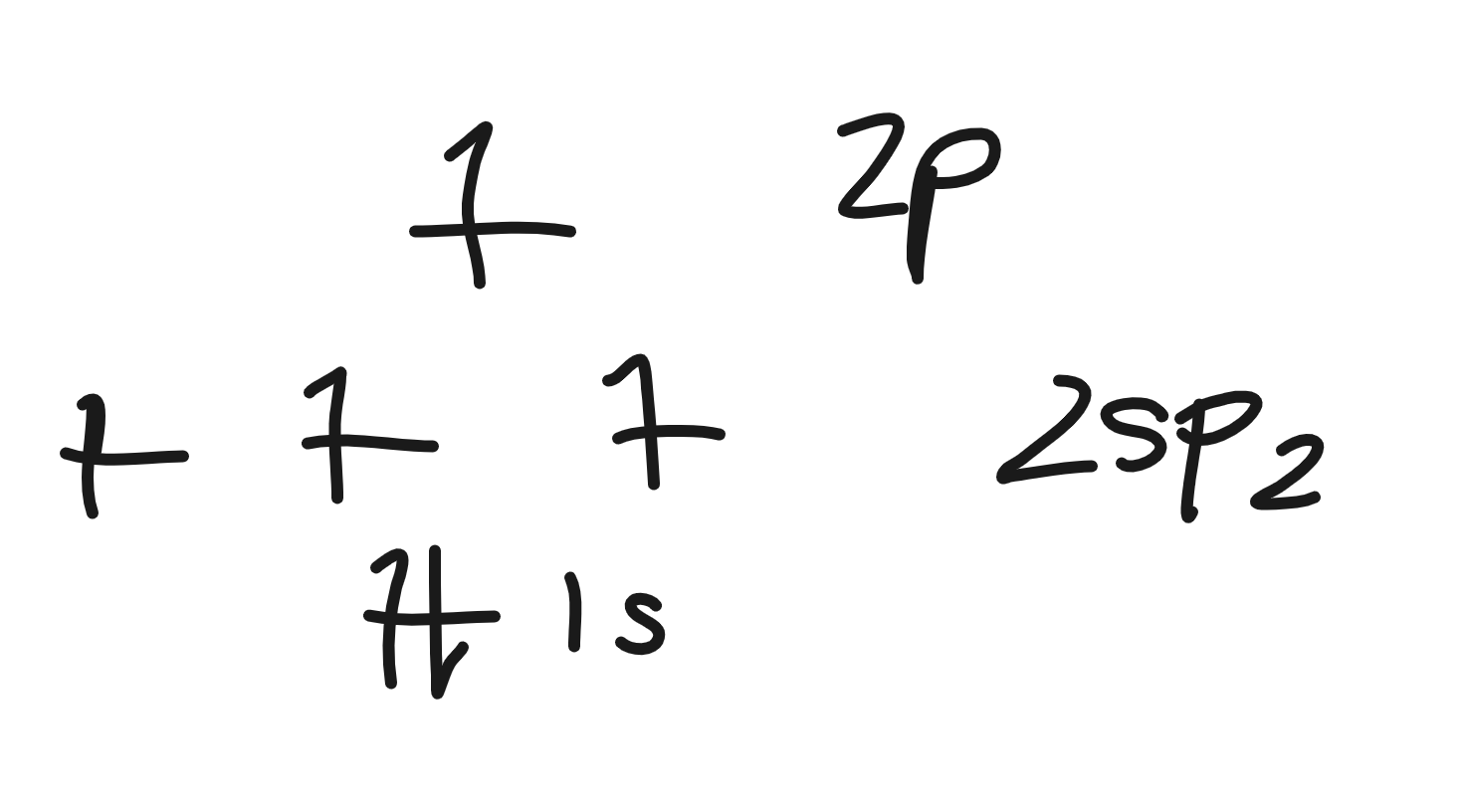
3 sigma bonds, 1 pi bond
how many bonds can sp2 hybridization make
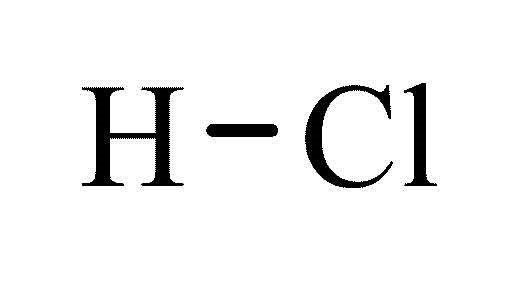
HCl pKa
-7 pKa
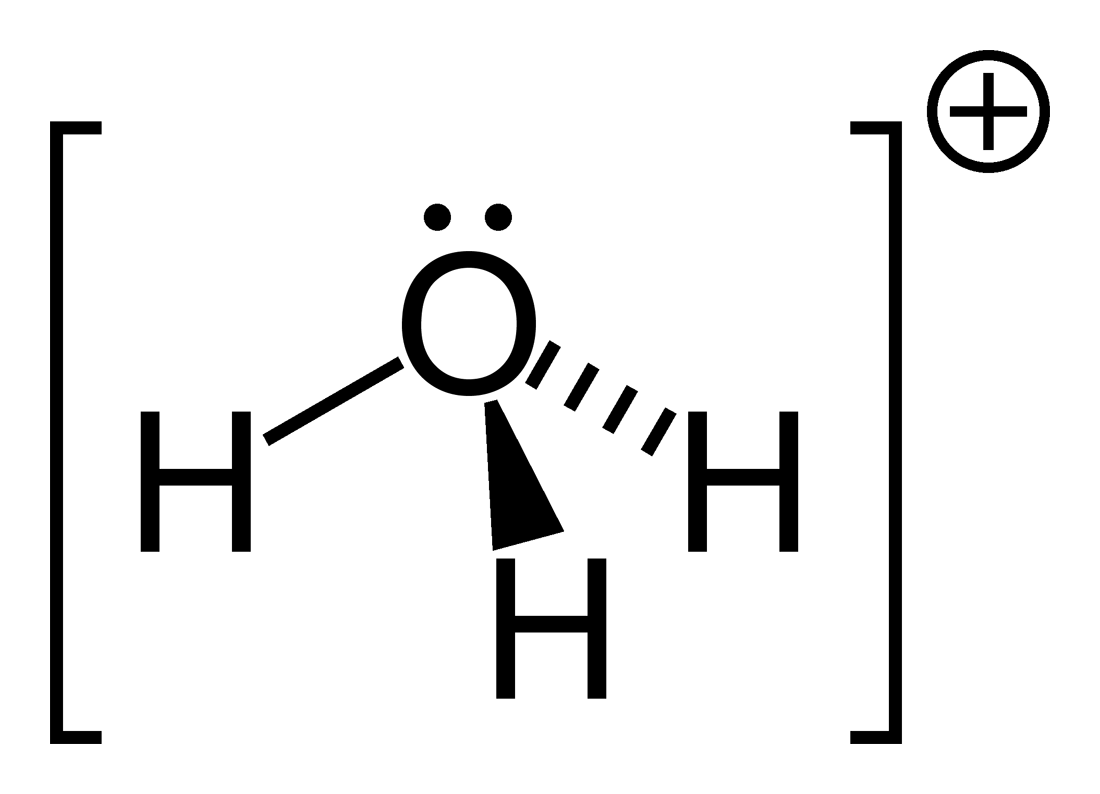
hydronium pKa
-1.5 pKa
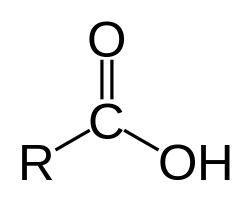
carboxylic acid pKa
~5 pKa
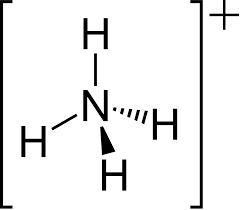
ammonium pKa
~9 pKa
H2O pKa
15.7
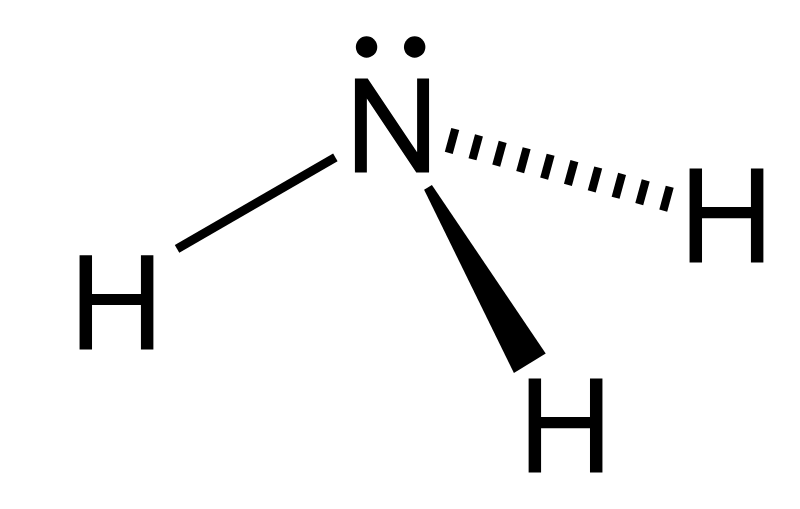
ammonia pKa
35 pKa
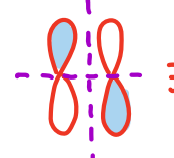
molecular orbital diagram for pi bond
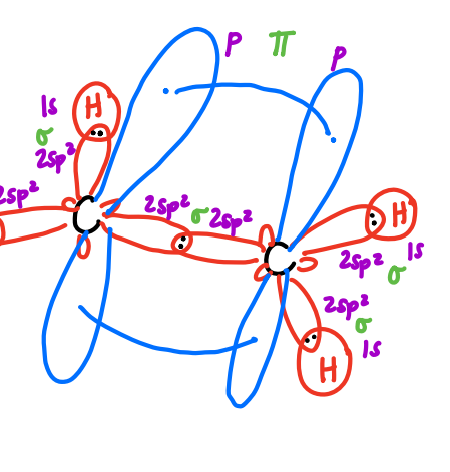
sigma anti-bonding molecular orbital
pi anti-bonding molecular orbital
stereoisomer
molecules that have the same molecular formula and sequence of bonded atoms (constitution), but differ in the three-dimensional orientations of their atoms in space
constitutional isomer
compounds with the same molecular formula but different bonding arrangements. While they share the exact same number and types of atoms, the atoms are connected in a different order, leading to distinct physical and chemical properties
when does an acid dissociate (explain using pKa values)
if solution pKa is less than acid pKa, the acid will not dissociate. if the solution has a higher pKa than the acid, then the acid will dissociate.
Markovnikovs rule
a more substituted carbocation will form as an intermediate. this is more stable because there are more instances of sigma bonds aligned with empty p orbitals and thus more instances where hyperconjugation is possible
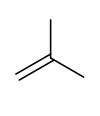
molecular orbital diagram justification for markovnikovs rule