Gen Chem: Final Exam
1/77
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
78 Terms
First Law of Thermodynamics
the internal energy of a system changes through heat exchange with the surroundings or work on or by the system
enthalpy
the heat flow for a process at constant pressure
state function
its value only depends only on the state of a system, and not the path taken to reach that state
exothermic
delta H is less than 0; thermal energy is transferred from the system to its surroundings
endothermic
delta H is greater than 0; thermal energy is transferred to the system from its surroundings
Hess’s Law
if a process can be written as the sum of several stepwise processes, the enthalpy change of the total process equals the sum of the enthalpy changes if the various steps
standard state
pure solid and liquid, pressure 1 atm (g), concentration 1 M (aq), and T = 298K
spontaneous process
process that takes place without a continuous input of energy from an external source
nonspontaneous
process that requires continual input of energy from an external source
entropy
measure of the disorder of the system
microstate
possible configuration or arrangement of matter and energy within a system
Second Law of Thermodynamics
all spontaneous processes involve an increase in the entropy of the universe
Third Law of Thermodynamics
entropy of a perfect crystal at absolute zero (0K) is zero
standard entropy
entropy for one mole of a substance at 1 atm pressure
Gibbs free energy change
thermodynamic property defined in terms of system enthalpy and entropy
electrochemistry
the study of the interconversion of electrical and chemical energy
electricity
presence and flow of electrical charge
active electrodes
the anode and cathode materials participate in the reactions
inert electrode
the half-reaction does not contain an active electrode
standard hydrogen electrode (SHE)
the reference for all half-cell potential values with an assigned value of 0 V for the oxidation reaction H2 (g) —> 2H+ + 2e-
standard reduction potentials (SRP)
determined by the determination of the potential of a cell made up of a reduction reaction and the SHE
Chloralkali Process
aqueous sodium chloride can be electrolyzed to produce NaOH (aq) and Cl2 (g)
Hall-Héroult process
a molten mixture of aluminum ore (bauxite, AL2O3) and cryolite (Na3AlF6) are electrolyzed at (>900oC) to produce very pure aluminum metal
batteries
single or series of galvanic cells designed for use as a source of electrical power
primary cell
non-rechargeable battery, suitable for single use only
secondary cell
batter designed to allow recharging
fuel cell
galvanic cells that require a continuous feed of redox reactants
corrosion
degradation of metal via a natural electrochemical process; always oxidation
dispersion forces
fleeting, temporary dipoles resulting in weak electrostatic forces between molecules when very close together
instantaneous dipoles
result from constant motion of electrons in molecules and atoms
induced dipoles
result when an instantaneous dipole in one molecule or atom distorts the electrons in a neighboring atom or molecule
polarizability
measure of how easy or difficult it is for another electrostatic charge to distort a molecule’s charge distribution
hydrogen bonding
an unusually strong type of dipole-dipole attraction
what is the ranking of intermolecular forces?
hydrogen bonding > dipole-dipole > dispersion
cohesive forces
attraction between identical molecules of a substance
adhesive forces
attraction between different types of molecules
viscosity
the measure of a liquid’s resistance to flow
surface tension
the energy required to increase the area, or length of a liquids surface by a certain amount
capillary action
liquid flow within a porous material due to attraction of the liquid to the surface of the material and to other liquid molecules
vaporization
endo or exo?
liquid → vapor
endo
condensation
endo or exo?
vapor → liquid
exo
melting
endo or exo?
solid → liquid
endo
freezing
endo or exo?
liquid → solid
exo
sublimation
endo or exo?
solid → gas
endo
deposition
endo or exo?
gas → solid
exo
dynamic equilibrium
reciprocal process occur at the same rates
vapor pressure
the pressure exerted by a vapor in equilibrium with a liquid in a closed container at a given temperature
normal boiling point
the temperature at which the liquid boils when the pressure above the liquid is 1 atm
enthalpy of vaporization
endo or exo?
the energy required to vaporize one mole of a liquid substance
endo
enthalpy of fusion
endo or exo?
the energy required to melt one mole of a solid substance at a temperature
endo
enthalpy of sublimation
endo or exo?
the energy required to convert one mole of a solid substance directly to the gaseous state
endothermic
where are the phases located on a phase diagram?
left
right
bottom
solid
liquid
gas
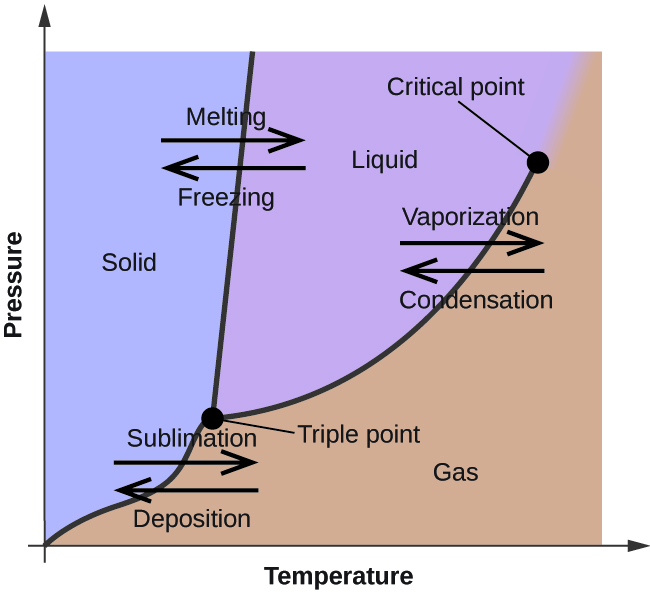
triple point
the temperature and pressure at which solid, liquid, and vapor of a substance are all in equilibrium
critical point
the temperature and pressure above which a gas cannot be condensed into a liquid
supercritical fluid
the high density, low viscosity phase intermediate between liquid and gas
crystalline solid
solids in which atoms, ions, or molecules are arranged in a definite repeating pattern
amorphous solids
glassy or noncrystalline solids that lack an ordered internal structure
ionic solid
solid composed of cations and anions held together by strong electrostatic attractions
metallic solid
solid composed of metal atoms