5 redox equilibria
1/92
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
93 Terms
what direction are redox reactions written in
the direction of reduction
how are potentials defined
defined against the standard hydrogen electrode (SHE)
equation for E⦵cell
E⦵cell = E⦵reduction - E⦵oxidation
which half-reaction is reduction
the higher reducing potential (more positive)
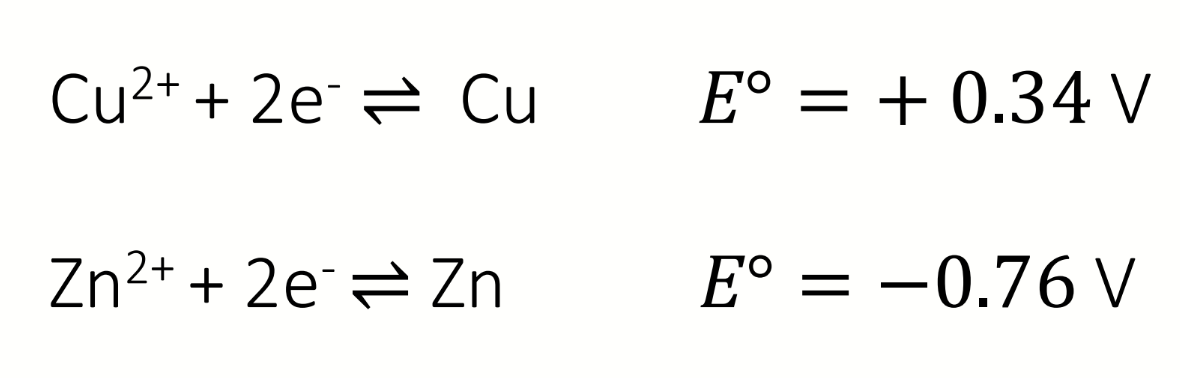
what is the overall equation?
calculate E⦵cell
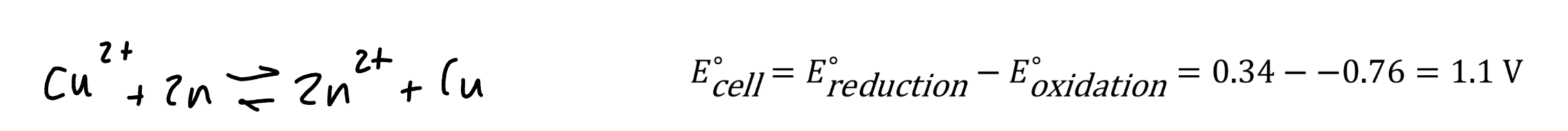
when is a reaction spontaneous in terms of E⦵cell
if E⦵cell is positive
what is the Nernst equation
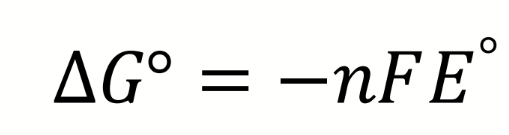
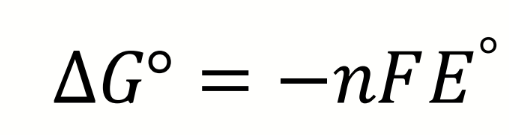
what are the terms in the Nernst equation
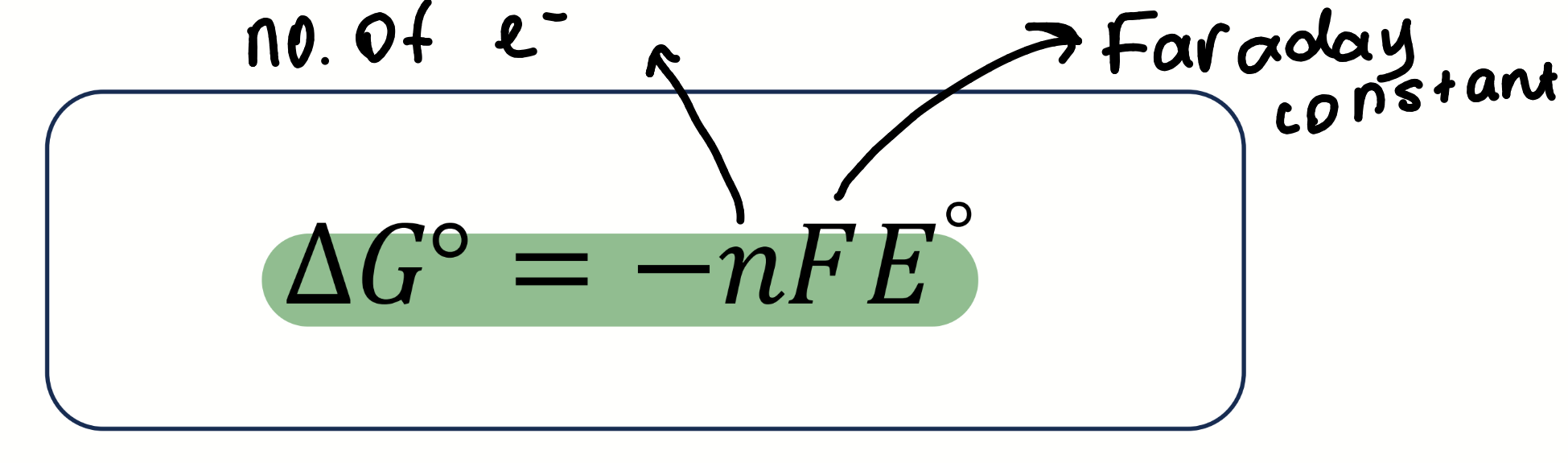

how can lnK be found
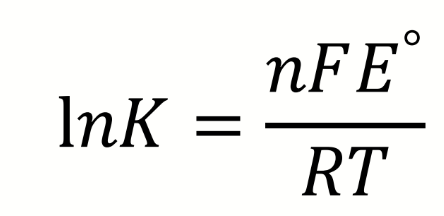
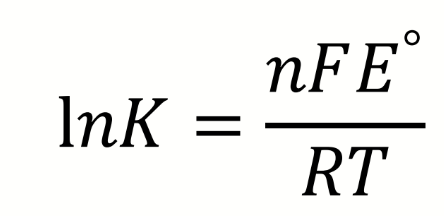
effect of E° on K
as E° increases, K increases
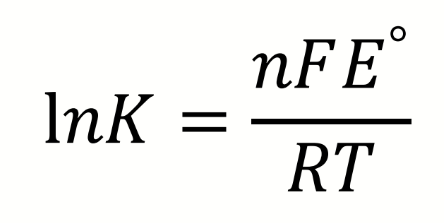
when is a reaction spontaneous in terms of E°, K and ΔG
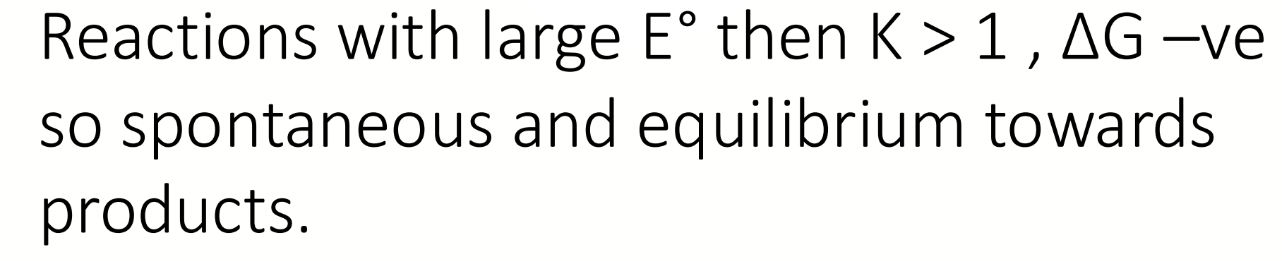
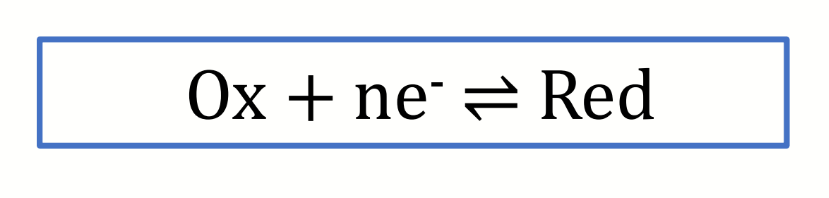
equation for Kredox
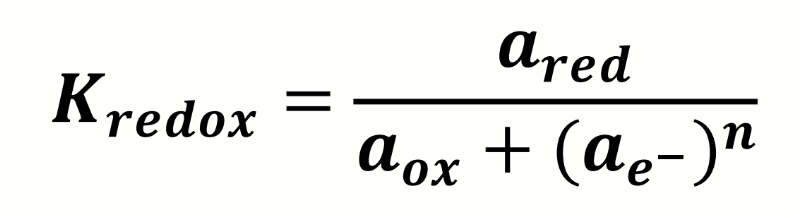
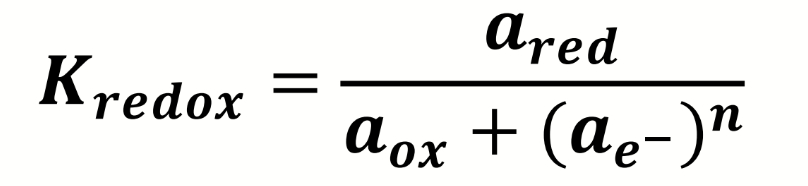
how can this be rearranged to give the activity of electrons in a redox couple
how can this be expressed
can be expressed Eh - redox potential (different to reduction potential)
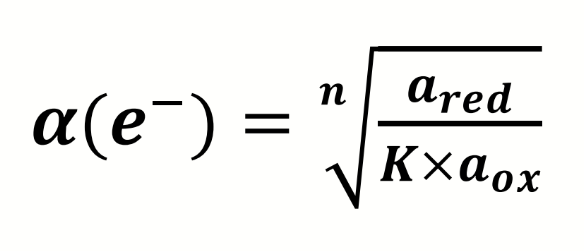
equation for redox potential

what is pE
what is it analagous to
reducing power of a solution
analagous to pH = acidity
equation for reducing power of a solution
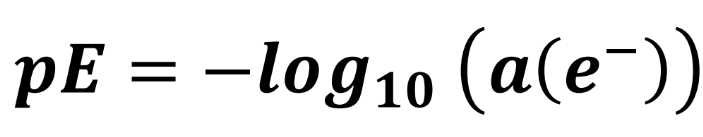
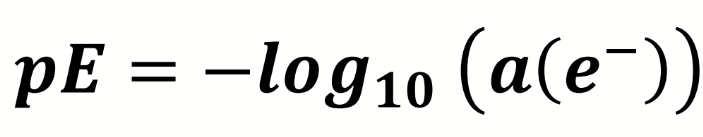
how else can this be expressed
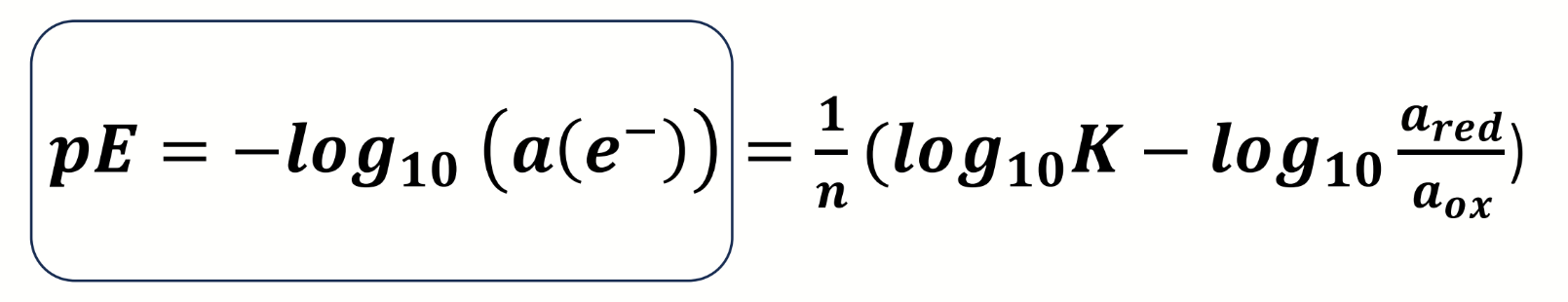
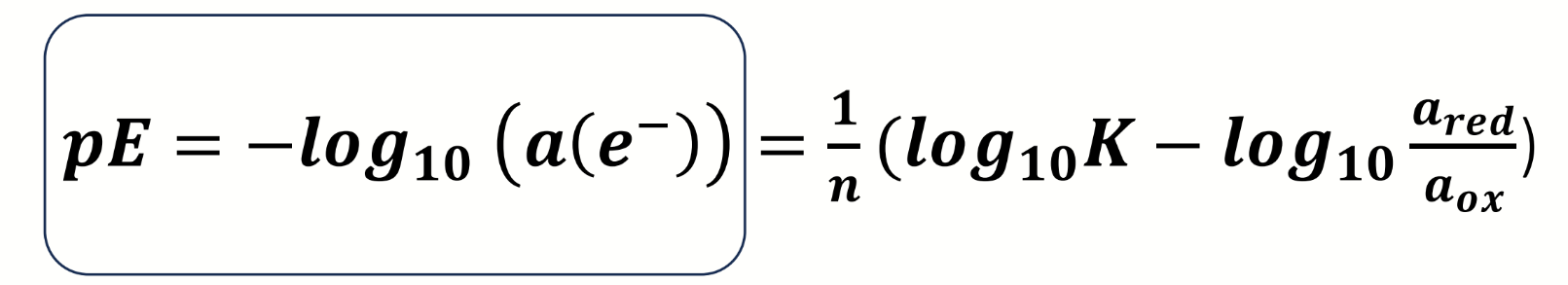
what assumption is made for this equation
αelectrons = [ ]electrons
what does the pE scale indicate
gives the ability/likelihood of a species to be oxidised or reduced in a certain environment
what are the three regions of the pE scale
oxic, suboxic and anoxic
oxic
pE
electrons?
what happens
high pE, low electron availability, species oxidised
what happens in the suboxic region
species likely to be half oxidised, half reduced
anoxic
pE?
electrons?
what happens
low pE, high electron availability, species reduced
equation for redox potential
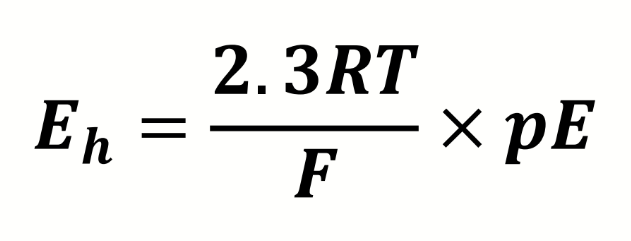
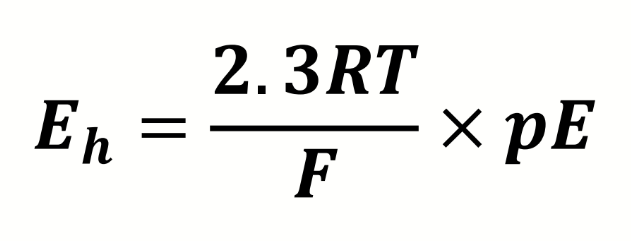
what do the terms mean
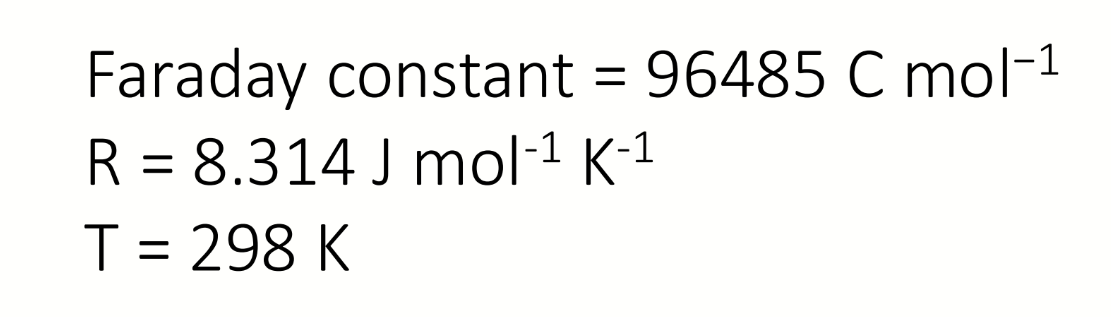
what is the conversion factor from electron activity to redox potential (in Volts)
298K
pE is dimensionless
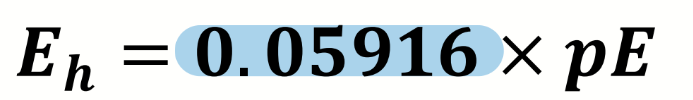
what happens in an electrolytic cell
an external voltage is applied to drive a nonspontaneous reaction
how can Ksp/Ka & Kb/Kf be found using redox
why is this effective
practical example?
couple to redox reactions and measure using half-cell potentials
coupled equilibria change the value of E°
e.g. potentiometric titrations
what can be added to balance redox reactions
H+, OH-, H2O
steps to balance a chemical reaction in acidic solution?
write out half reactions
balance all atoms except H and O
balance O with H2O
balance H with H+
balance charge with e-
multiply by lowest common multiple so same number of electrons on both sides
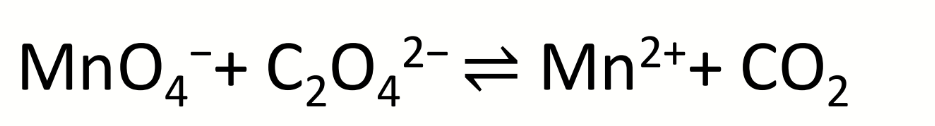
balance this equation in acidic solution
what happens if pH is increased?
reaction depends on [H+] so if the pH is increased the reaction proceeds to the right
![<p>reaction depends on [H<sup>+</sup>] so if the pH is increased the reaction proceeds to the right</p>](https://assets.knowt.com/user-attachments/d04e1a5d-8d88-40ee-a640-1c85562a2d5c.png)
what can be used to indicate water quality
dissolved oxygen concentration
what is a Winkler titration
[O2(aq)] is measured using iodine-thiosulfate titration
what happens in the first step of a Winkler titration
what is mixed
pH of solution?
what reacts
what is formed
the water sample is mixed with manganese (II) in a basic solution
manganese (II) ions react with hydroxide ions to form a white precipitate
equilibrium equation for the first step of Winkler titration (mixing of manganese with hydroxide)
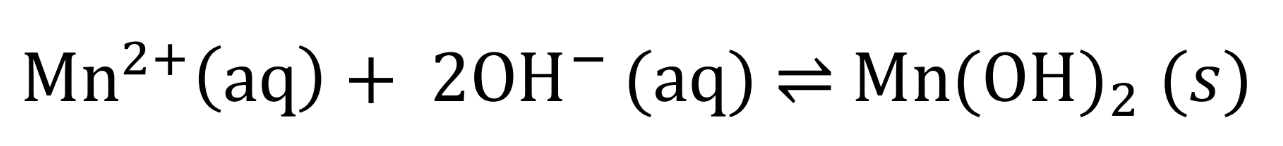
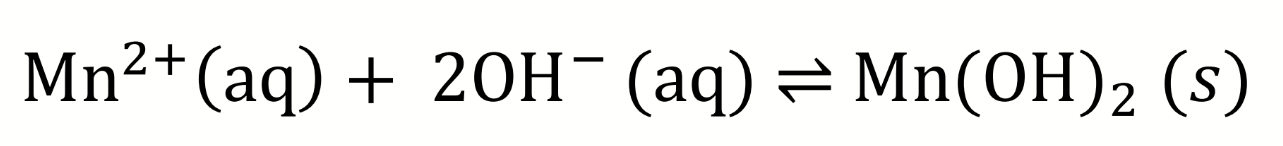
Ksp for Mn(OH)2?
is this low or high? what does this tell you?
Ksp = 5 × 10-2
low, not very soluble (forms white ppt)
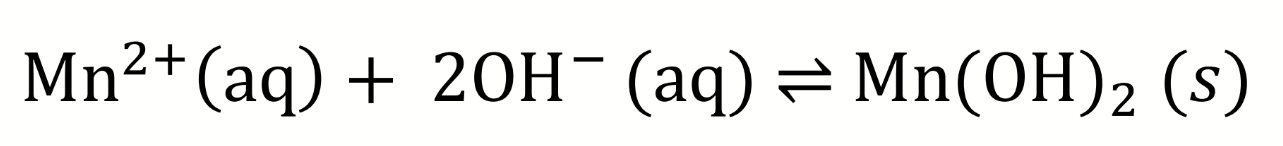
what effect means solubility depends on pH? what is the effect on this reaction for higher pH
due to the common ion effect
at higher pH, the concentration of [OH-] increases, pushing equilibrium to precipitation
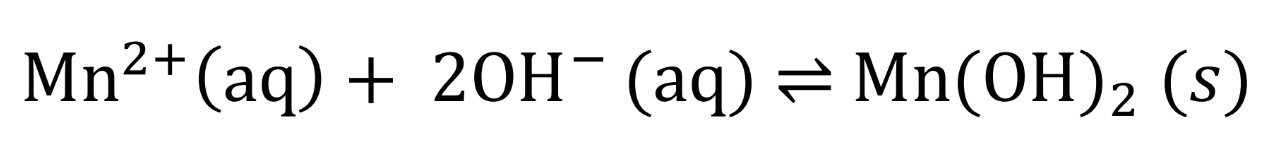
what happens if oxygen is present
show chemical eqn and charges of Mn
a redox reaction takes place
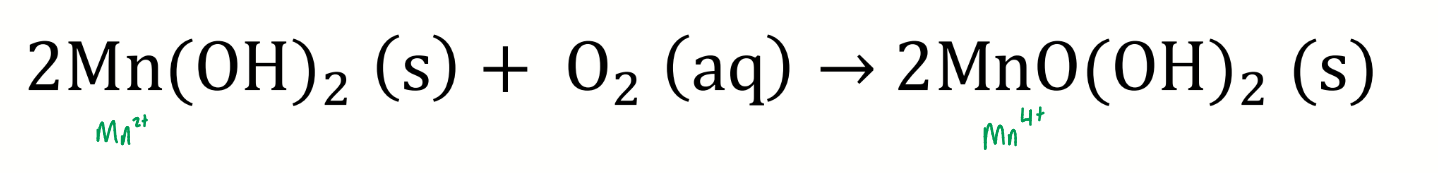
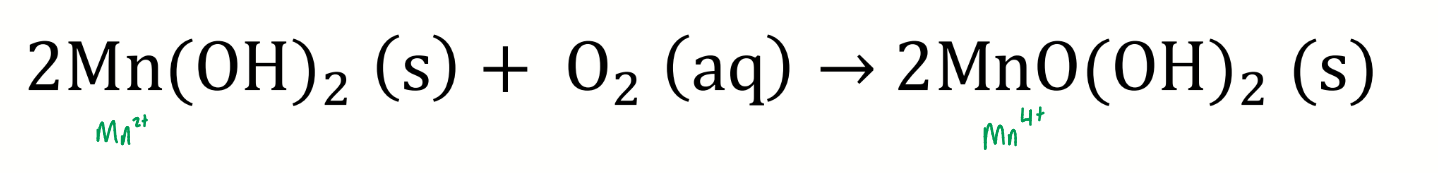
what are the oxidising equivalents?
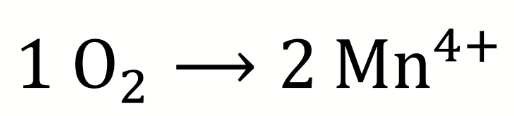
how can Gibbs free energy of the redox reaction be found
using standard redox potentials
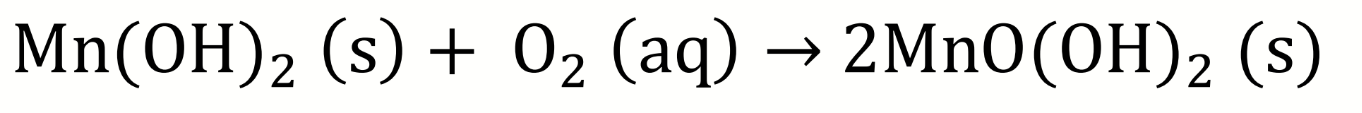
write the half equations for O and Mn
which is reduced and which is oxidised?
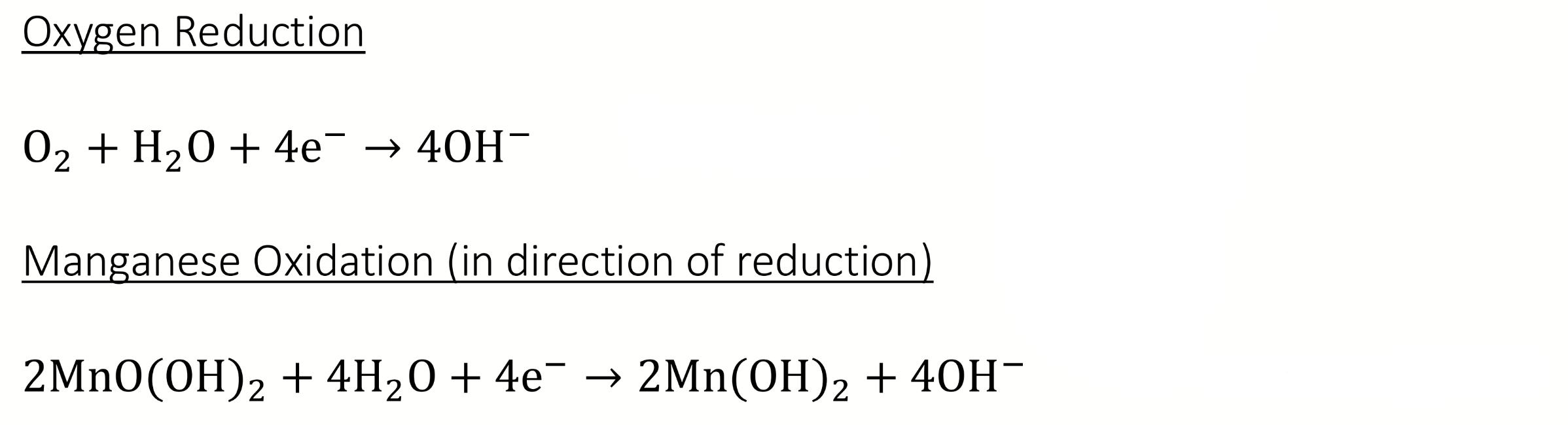

calculate E°cell?
E°cell = 0.4 - 0.2 = 0.2V

calculate lnK
where does the equilibrium lie and what does this mean for oxygen

what is step 2 of the Winkler titration
(after Mn mixed with hydroxide then Mn(OH)2 has reacted with O to form MnO(OH)2)
the solid MnO(OH)2 is added to a solution containing sulfuric acid and potassium iodide
chemical equation for solid MnO(OH)2 being added to sulfuric acid and potassium iodide?
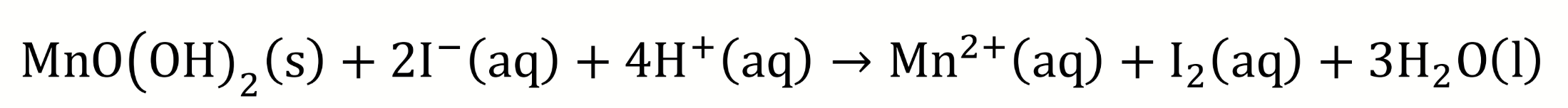
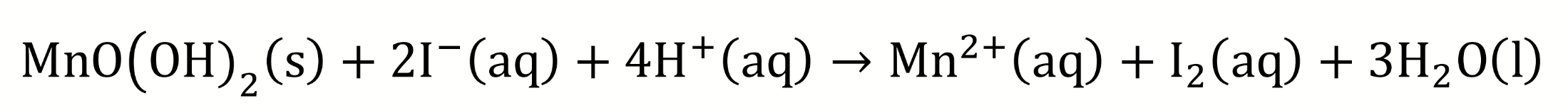
what does acidification cause?
MnO(OH)2 dissolves as OH- reacts with H+ which forms H2O and pushes the equilibrium to the right
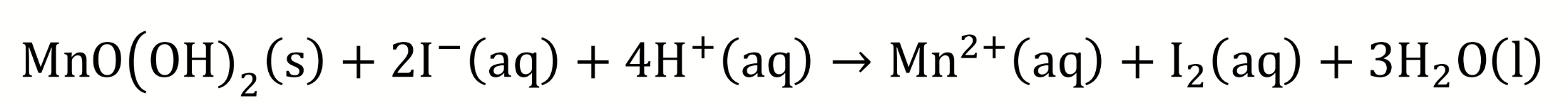
is manganese oxidised or reduced? why? what are the oxidation states?
what other species is involved and oxidised/reduced (which?)?
manganese (IV) is reduced to manganese (II) due to the coupled reaction with iodide which is oxidised to iodine
write the reduction half reactions for the acidic step 2 of the Winkler titration
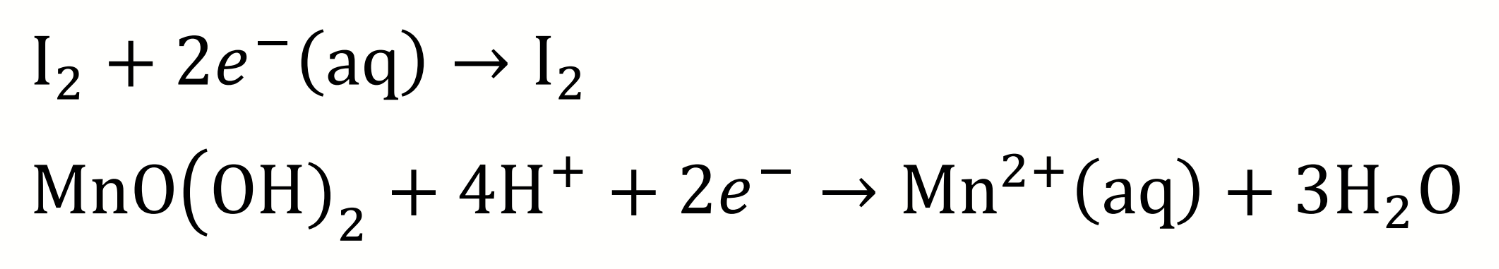
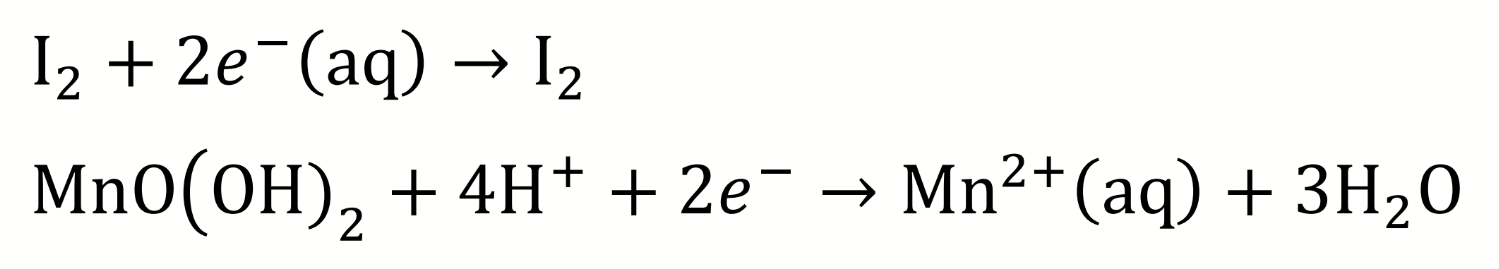
what are the oxidising equivalents for the acidic step 2 of the Winkler titration
include step 1’s oxidising equivalent
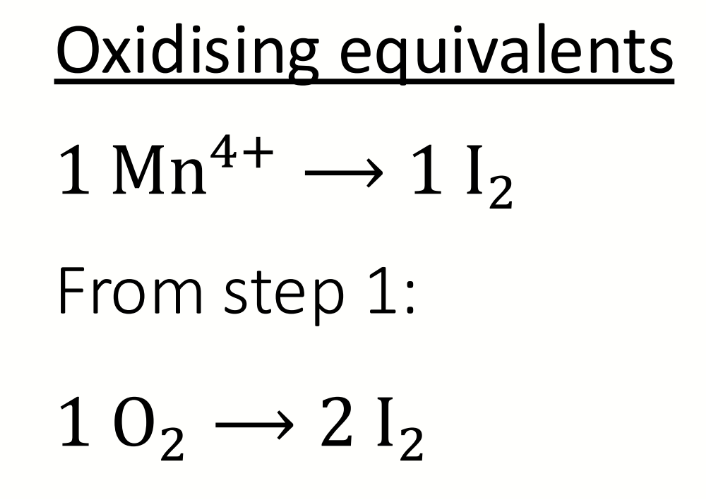
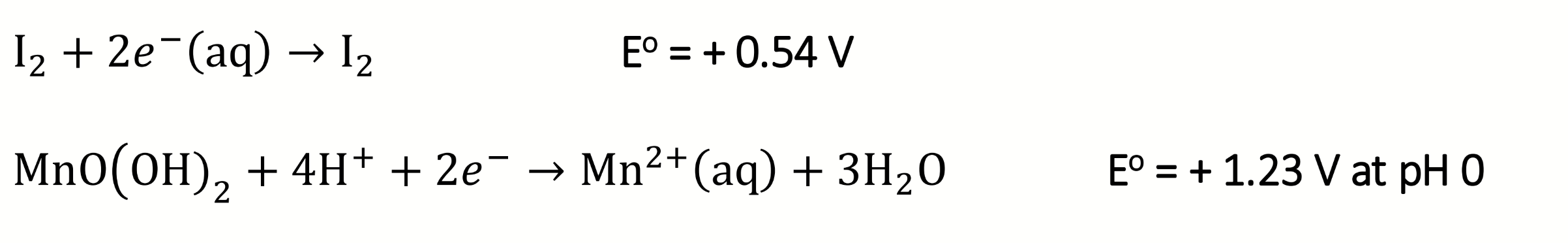
calculate E°cell
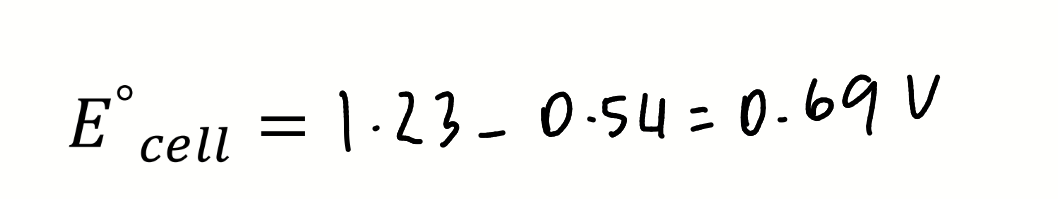
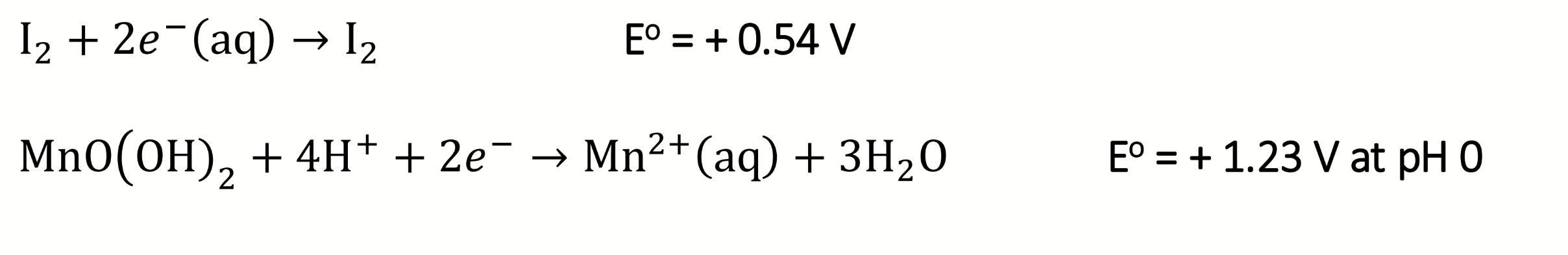
calculate the E°cell values at pH 0 and pH 10
E° for Mn(IV)/Mn(II) at pH 0 is +0.2V
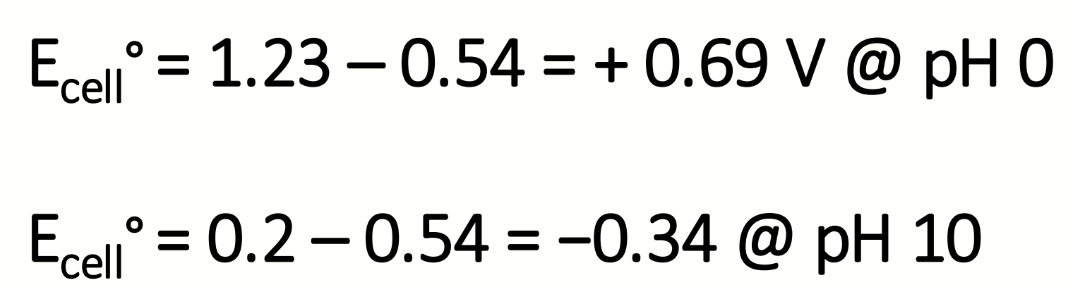

calculate the values of K at pH 0 and pH 10

chemical equation for the titration of iodine with thiosulfate
name the sulfur-containing ion formed
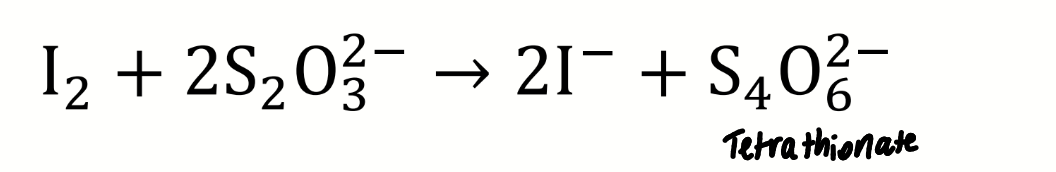
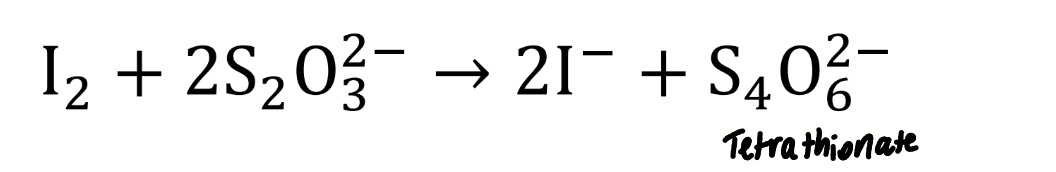
what are the reduction half-reactions
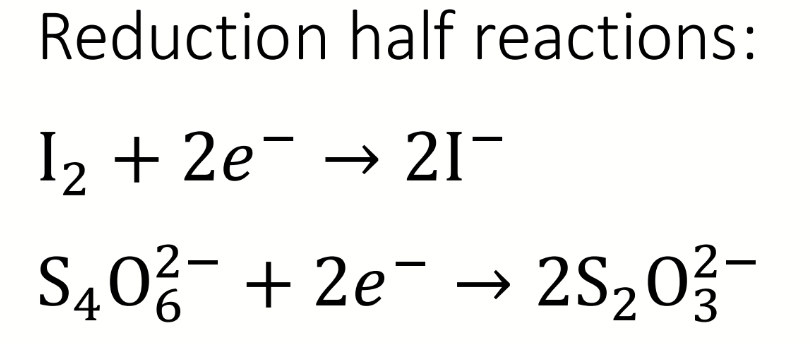
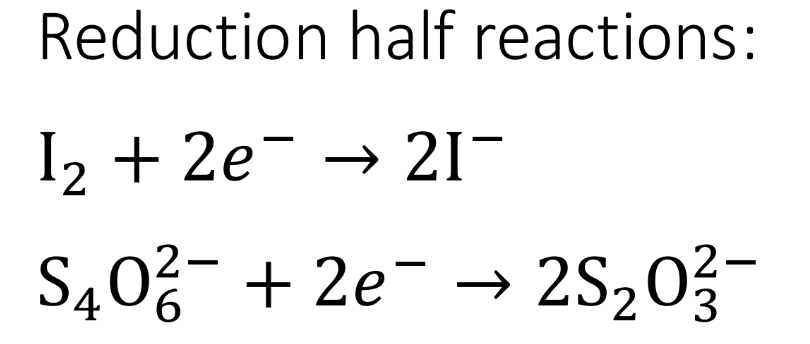
oxidising equivalents for step 3 of the Winkler reaction
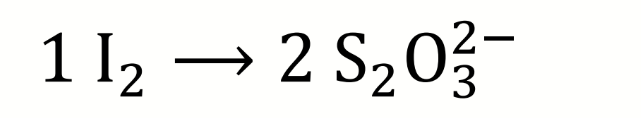
combine the oxidising equivalents for steps 1-3 of the Winkler titration
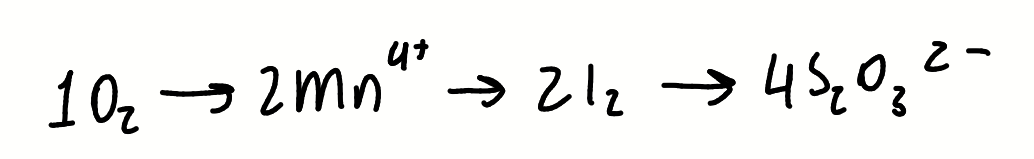
how is a potentiometric Winkler titration performed
what is added and how
what is measured (physically how)
automated addition of S2O3-
potential difference measured between platinum and calomel electrodes
equation for Ecell in a potentiometric titration
Ecell = EPt - Ecal
for the Winkler titration, give the equation for EPt
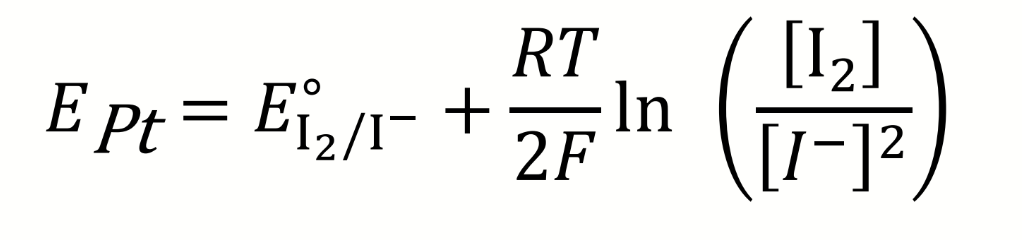
what does SCE stand for
Saturated Calomel Electrode
draw a diagram of an SCE and indicate what is happening at the other electrode
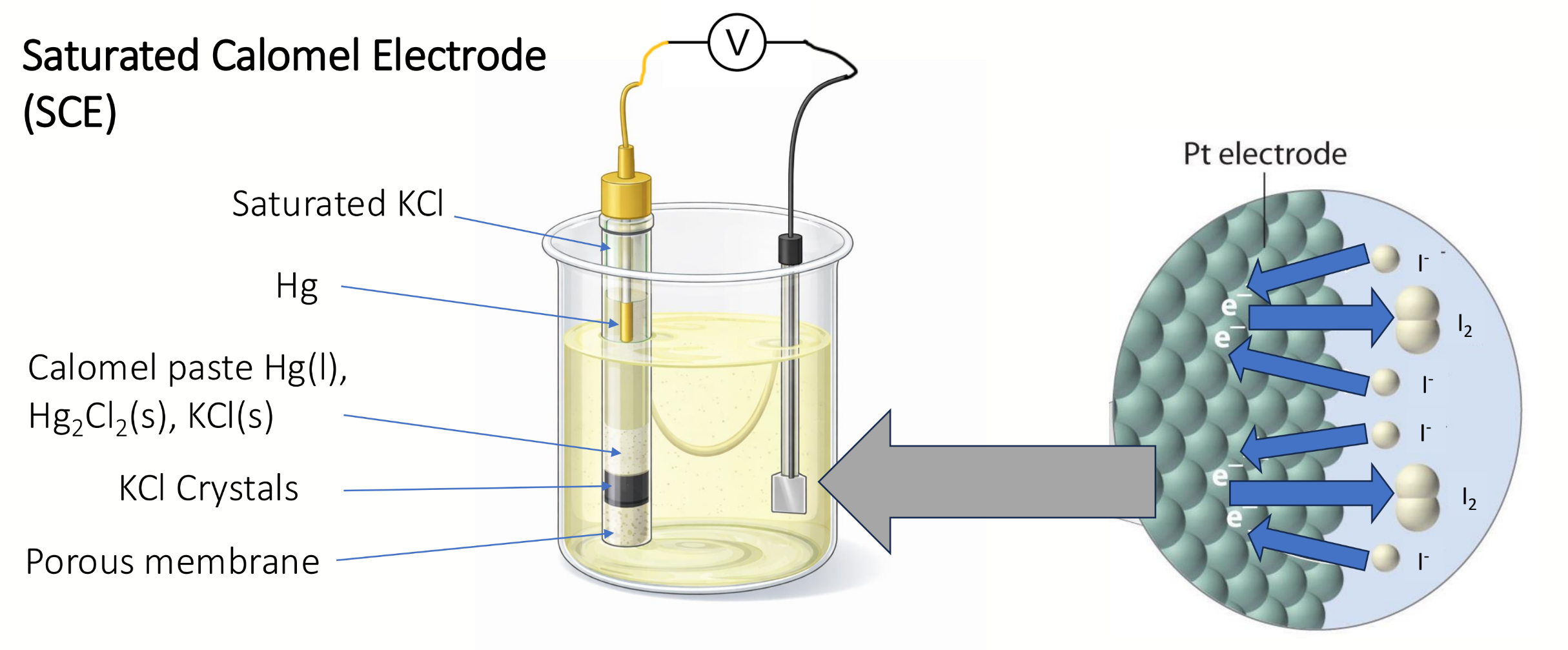
what is the calomel half-reaction
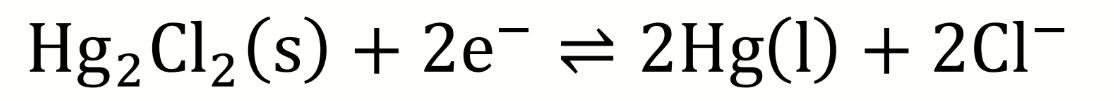
what is ESCE relative to SHE
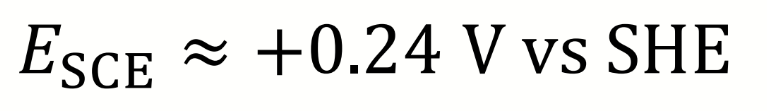
what is the half reaction at the platinum electrode for a potentiometric Winkler titration
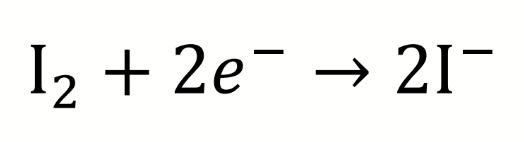

calculate E
what is it relative to?
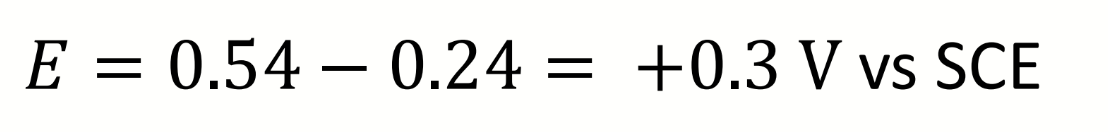
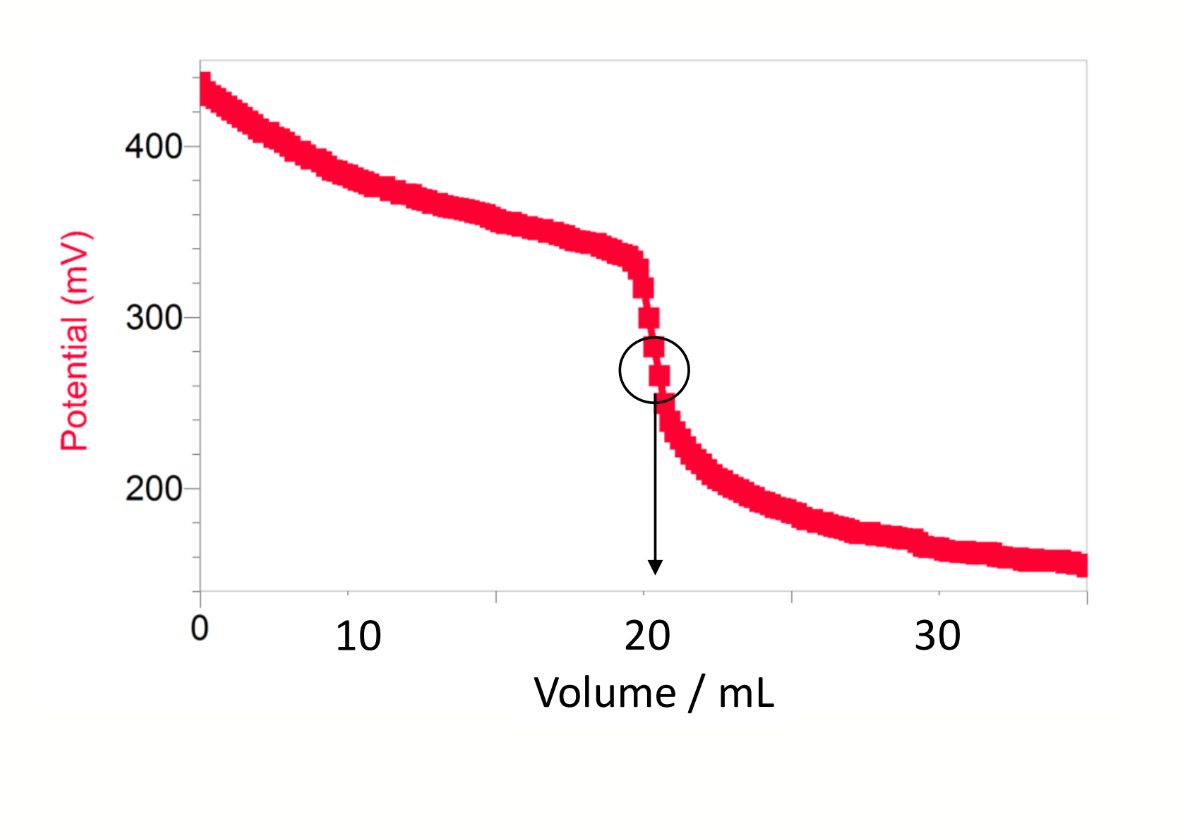
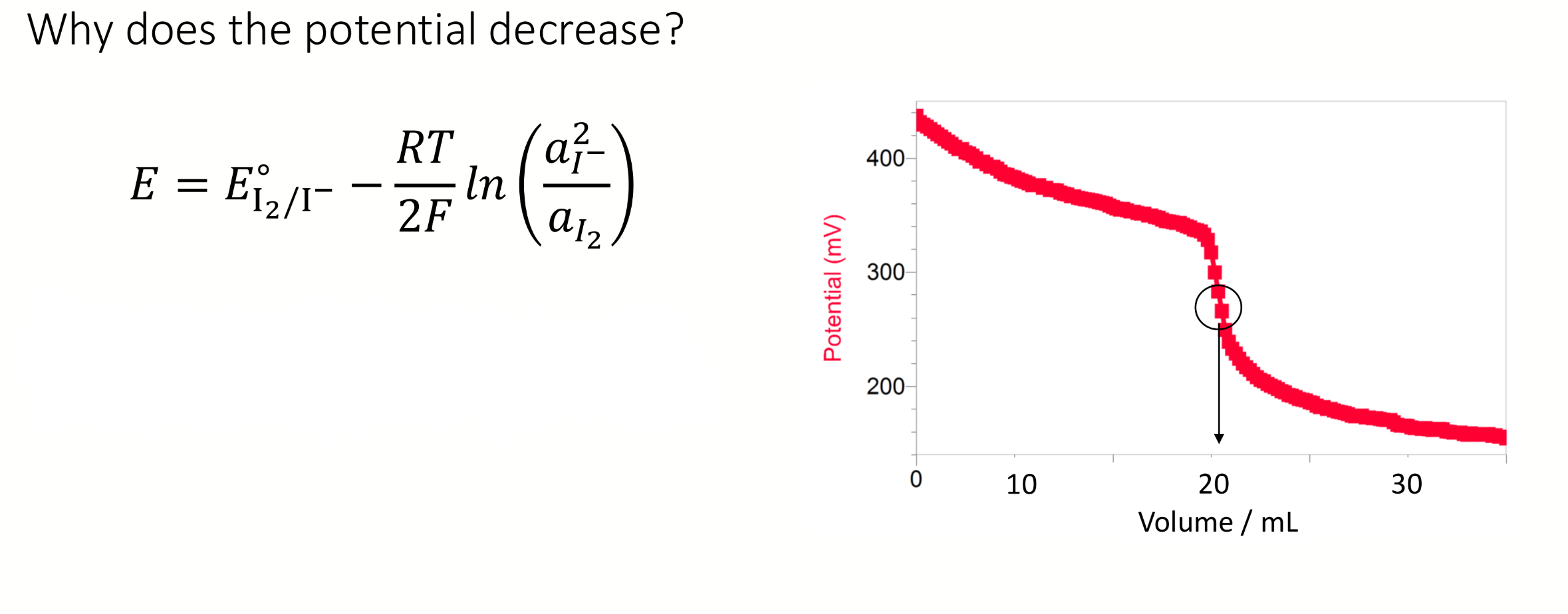
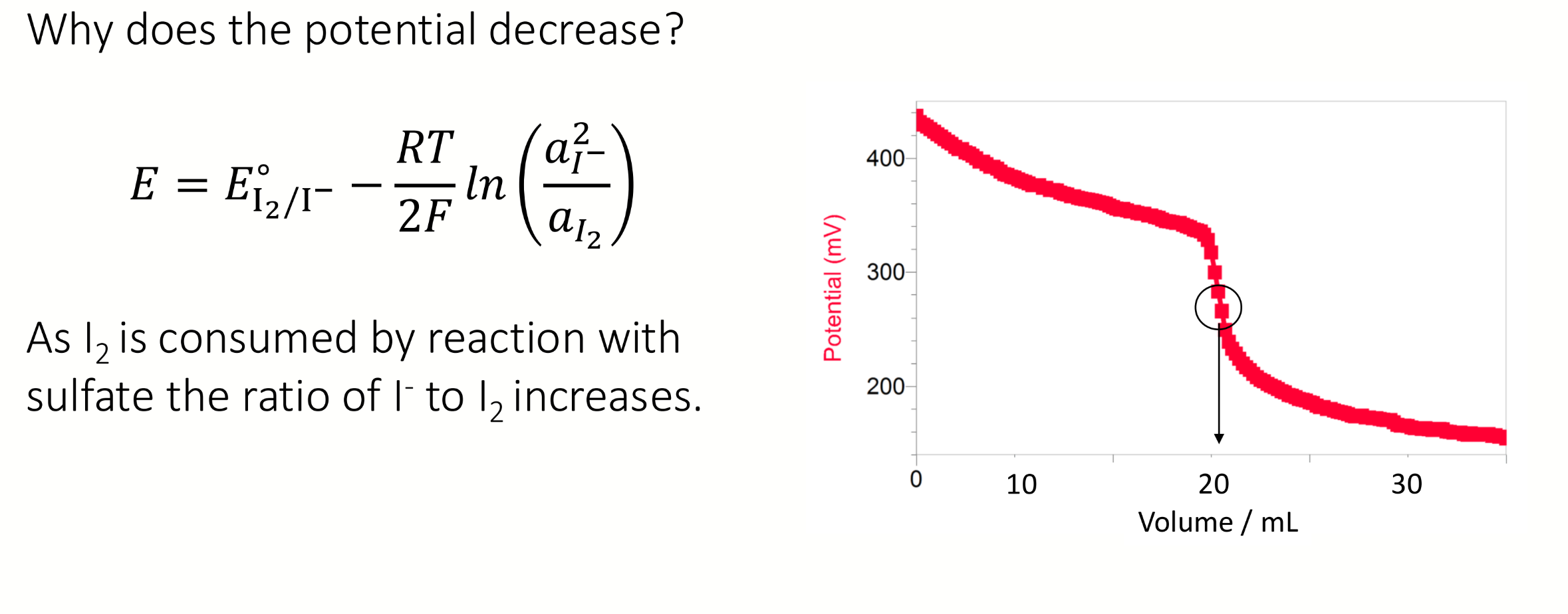
shape of a graph of potential (y) vs volume (x) for potentiometric Winkler
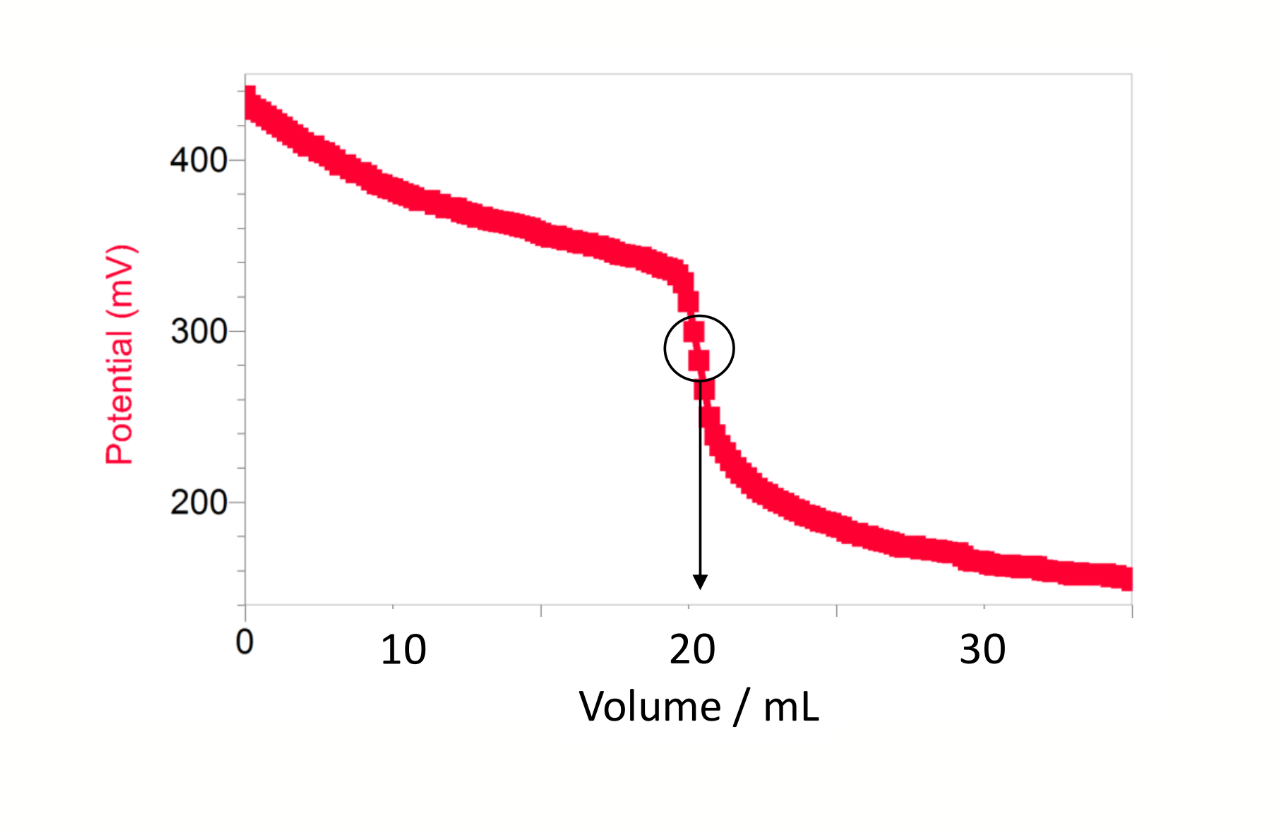
what is the equivalence point for potentiometric Winkler
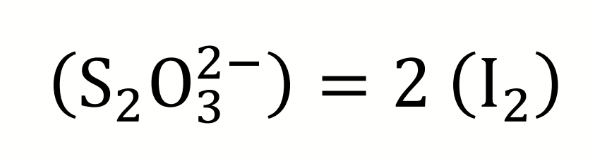
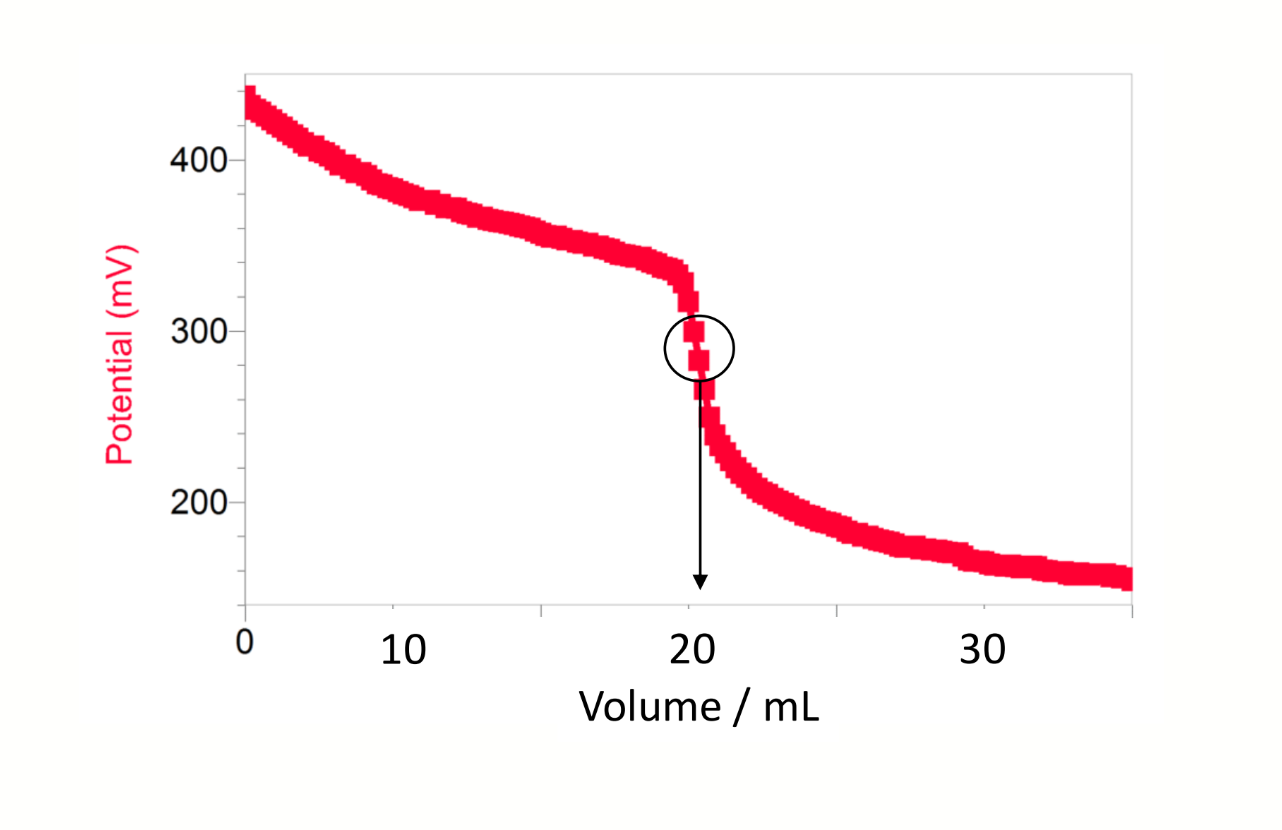
what is the end point for potentiometric Winkler
experimental observation of the equivalence point → point of inflection on the titration curve
how is the end point found for potentiometric titration
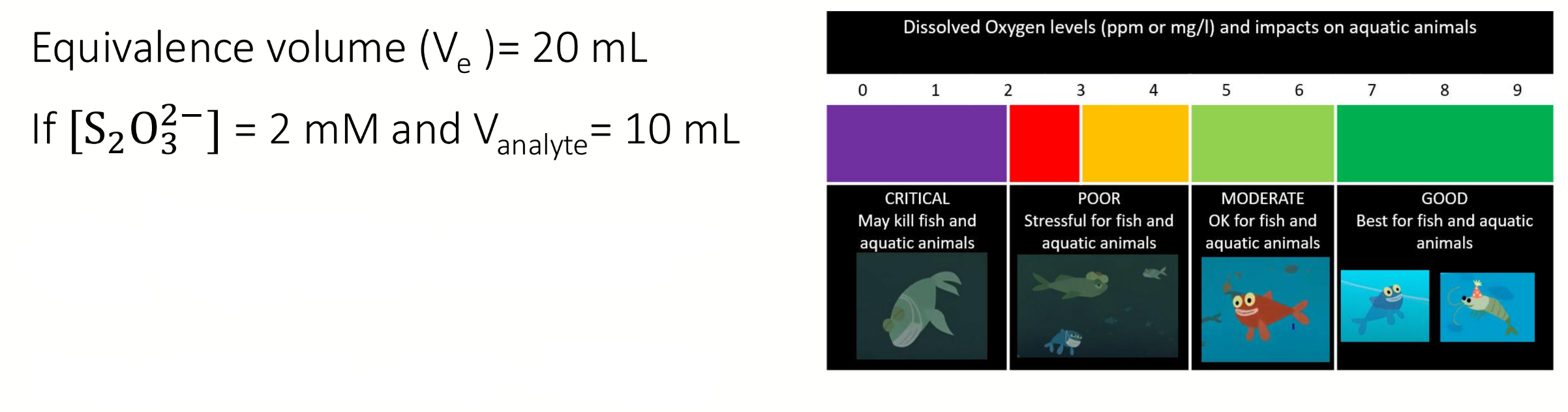
calculate and classify the dissolved oxygen level

how does complexation affect redox (pairs)
complexation stabilises two oxidation states in a redox pair differently - this affects the redox potential of the metal
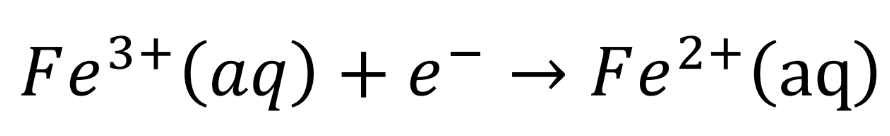
how to denote a reduction pair
Fe(III)/Fe(II)
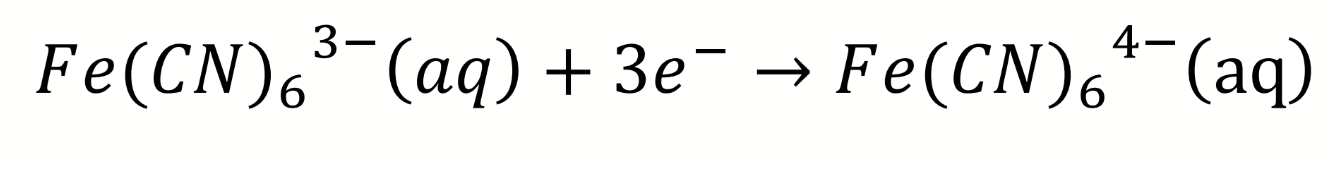
how is this redox pair denoted
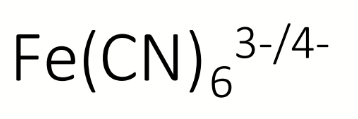
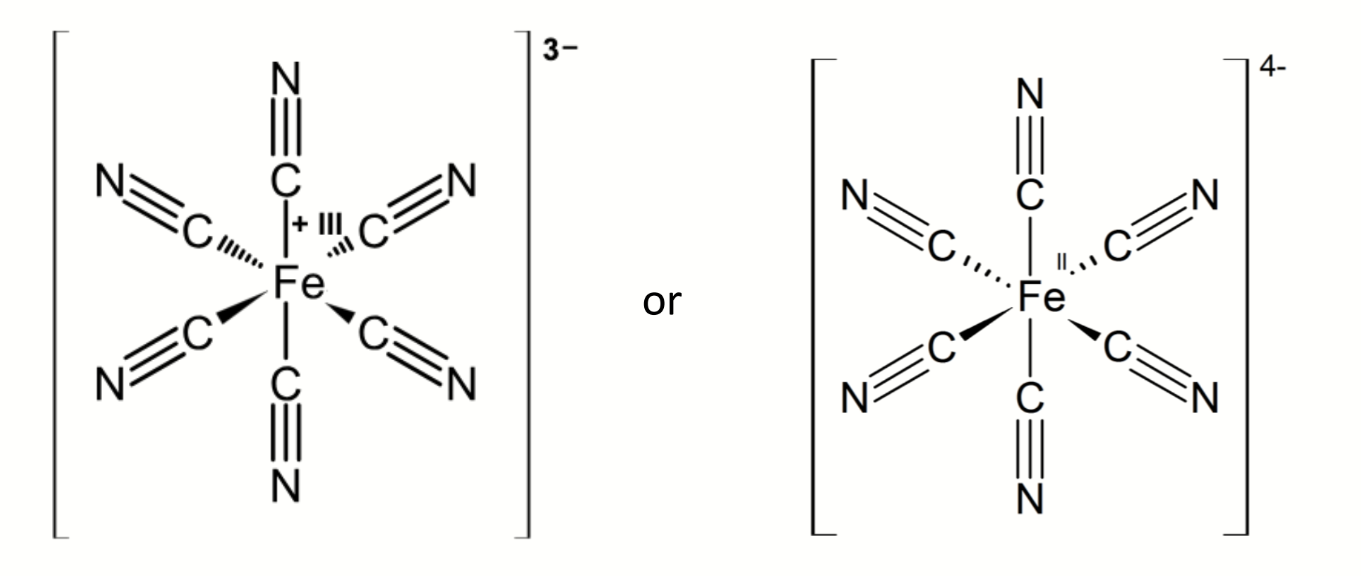
first step to determine which species is most strongly complexed by CN?

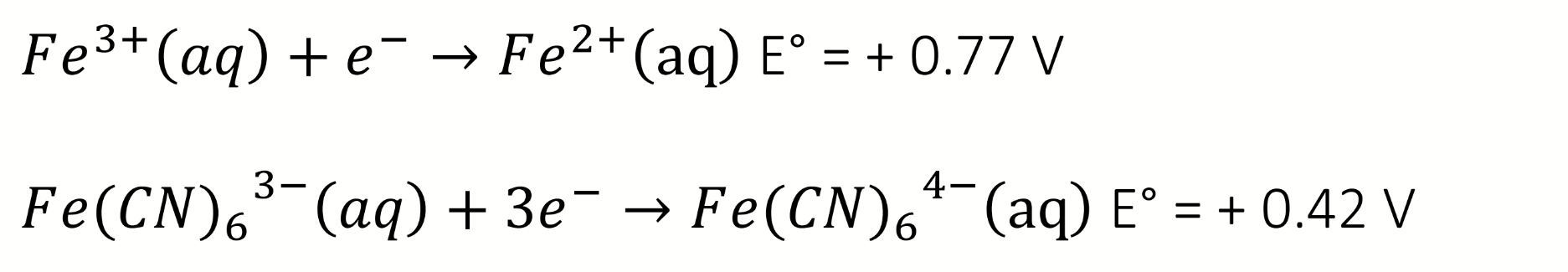
what can be deduced about the complexation of CN to Fe from these equations and potentials
Iron (III) is more difficult to reduce when complexed to cyanide, E° is smaller
CN is stabilising the Fe(III) oxidation state
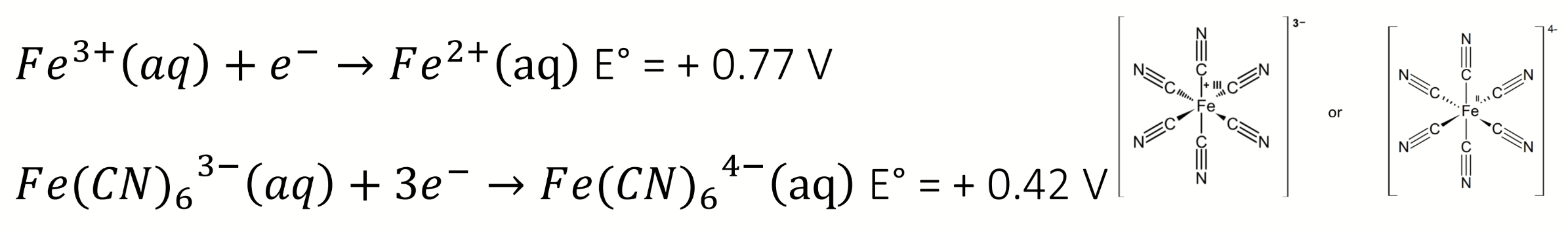
next step to determine which complex is most stable
find formation constants and ratio between them
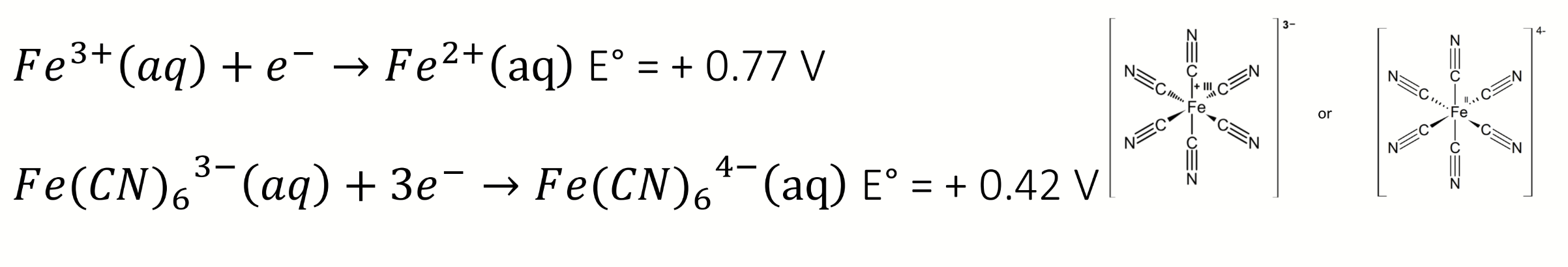
how to find ratio between formation constants

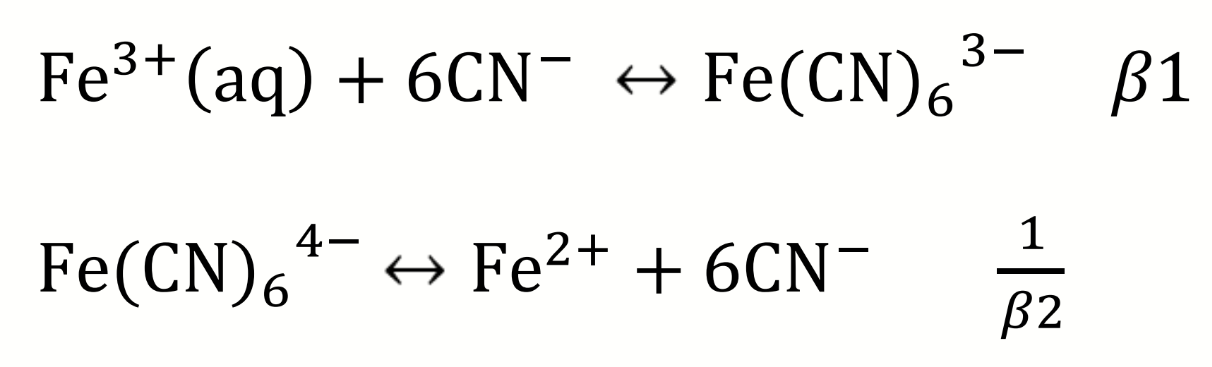
write the equilibrium for the coupled redox processes
then calculate K and E°cell
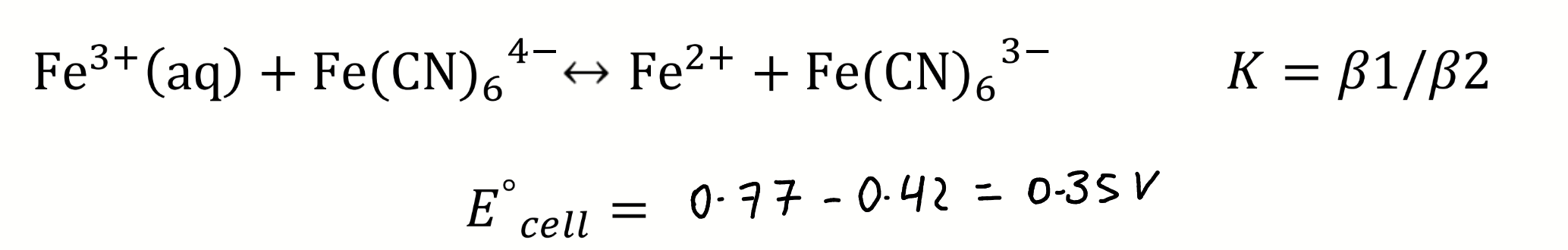
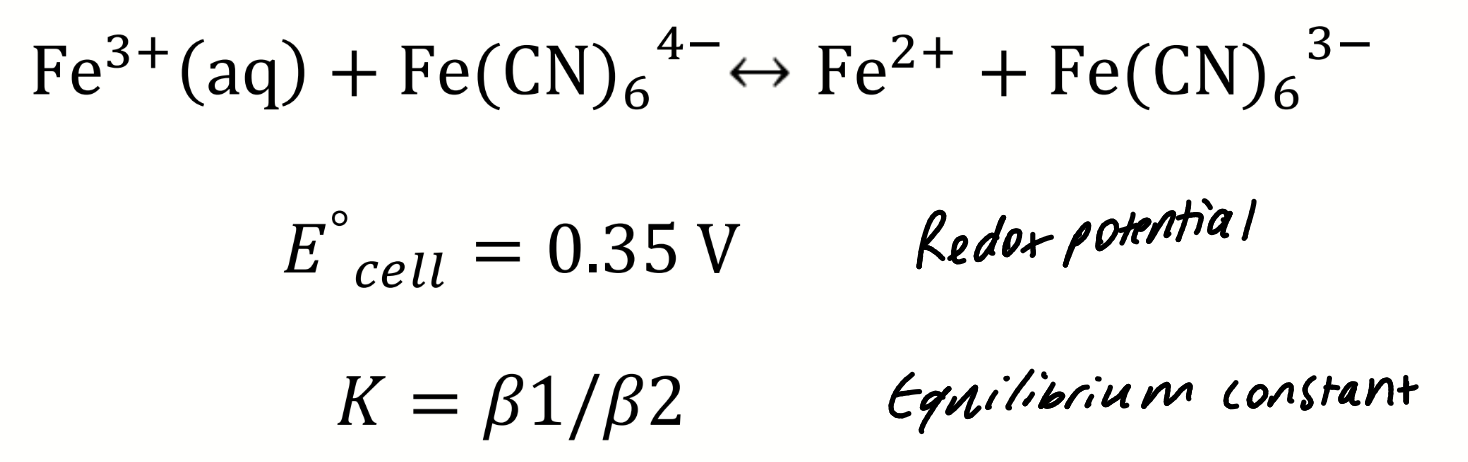
rearrange using the Nernst equation to find E
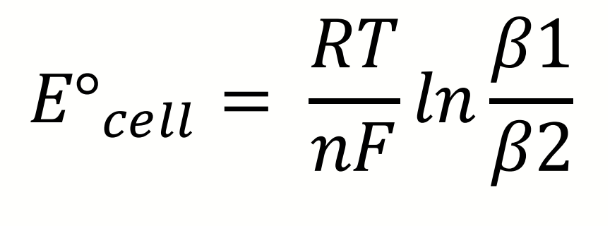
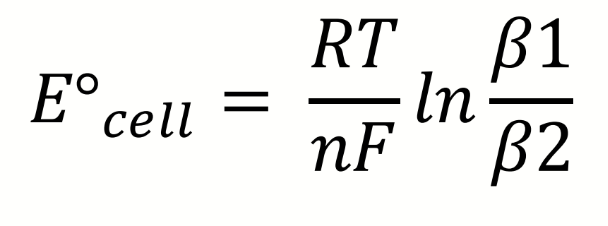
how can the ratio be found to determine relative stability of the complexes
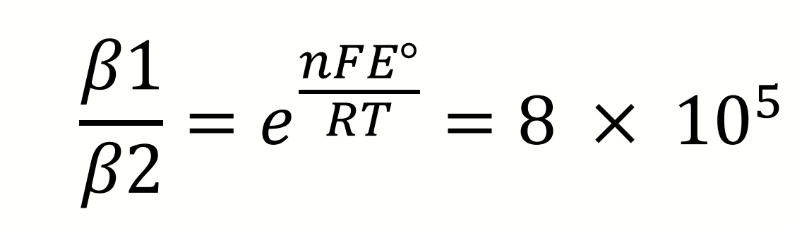
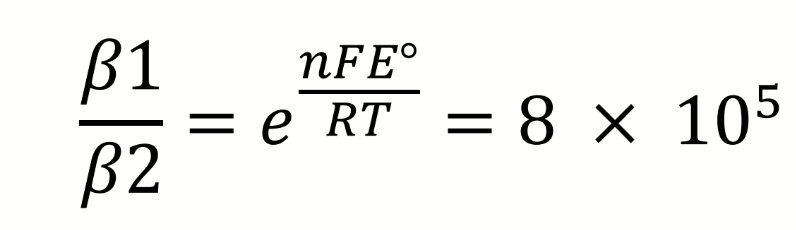
what does this say about the relative stabilities of the iron cyanide complexes
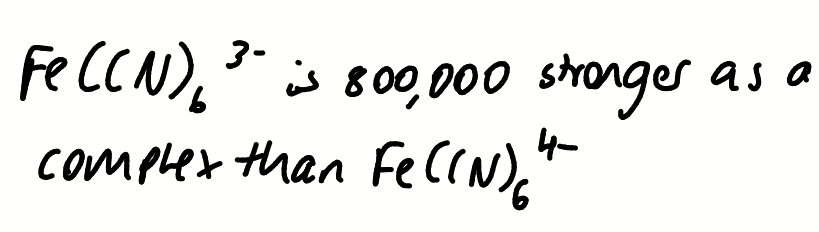
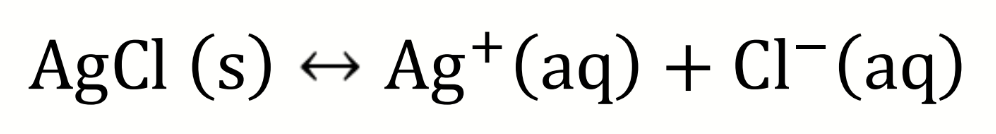
draw a diagram to show how the solubility of AgCl could be determined electrochemically
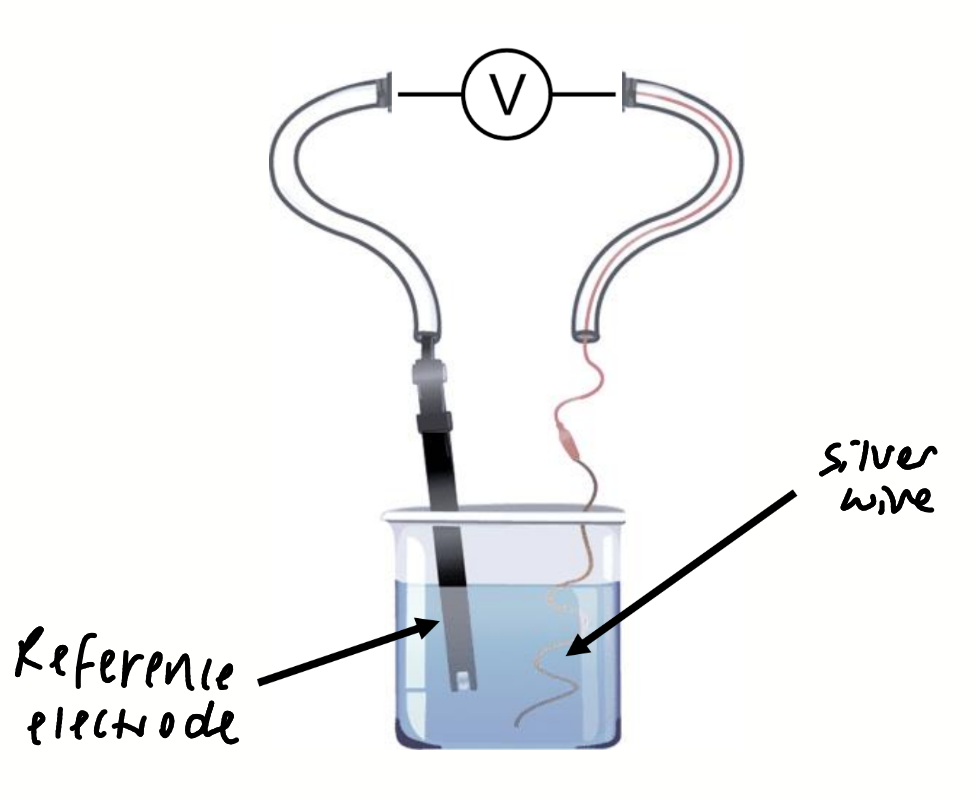
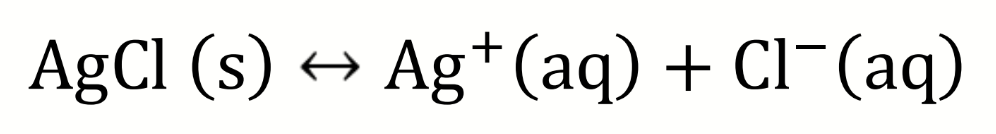
for this potentiometric titration
what is added into what
what are the concentrations at the equivalence point
silver nitrate added to NaCl solution
at the equivalence point [Ag+] = [Cl-]
example of a reference electrode used in potentiometric titrations? draw diagram
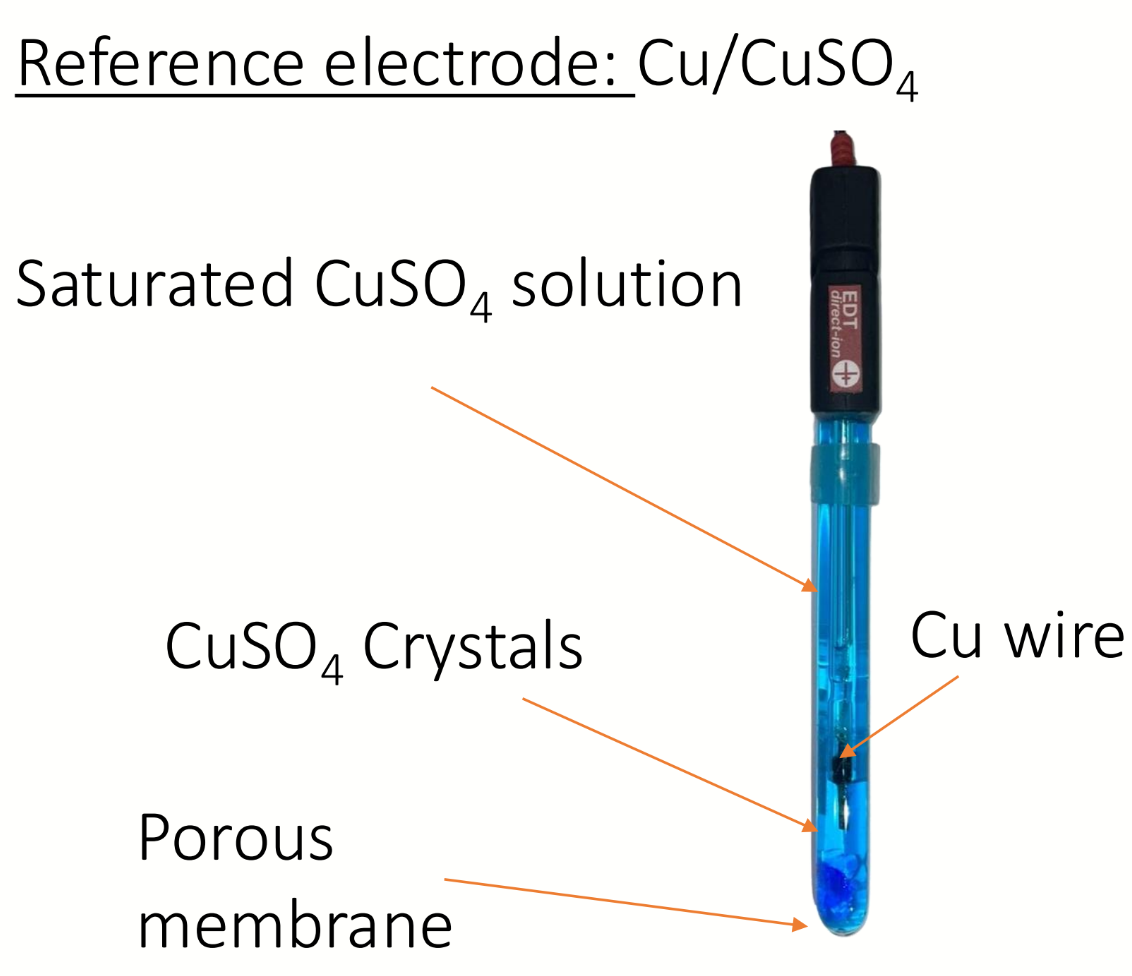
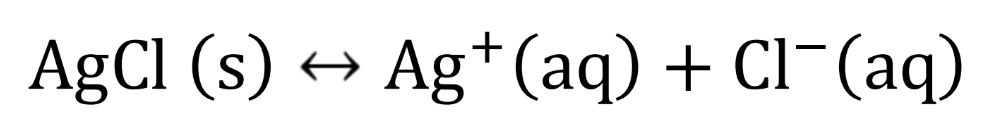
write a balanced equation for potentiometric titration of this with Cu/CuSO4 reference electrode
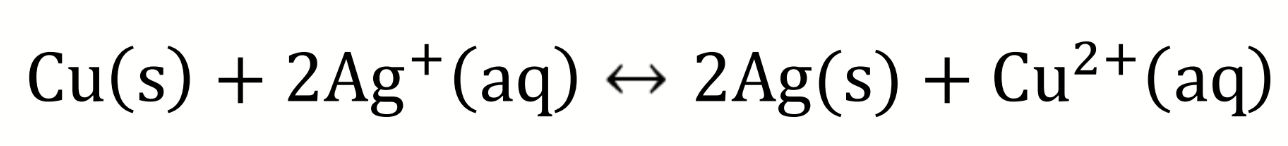
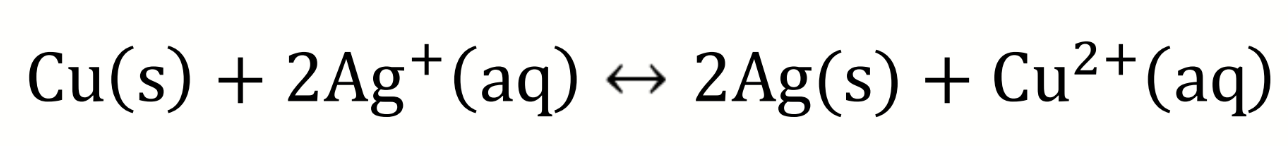
what are the half-equations?
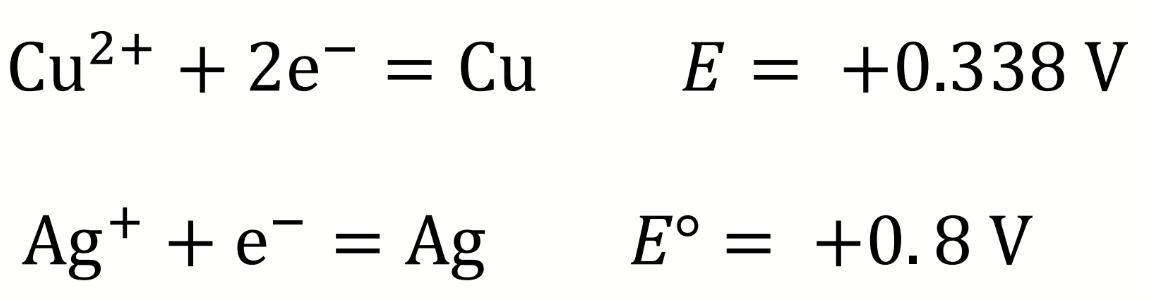
equation for Emeas (potentiometric titration)

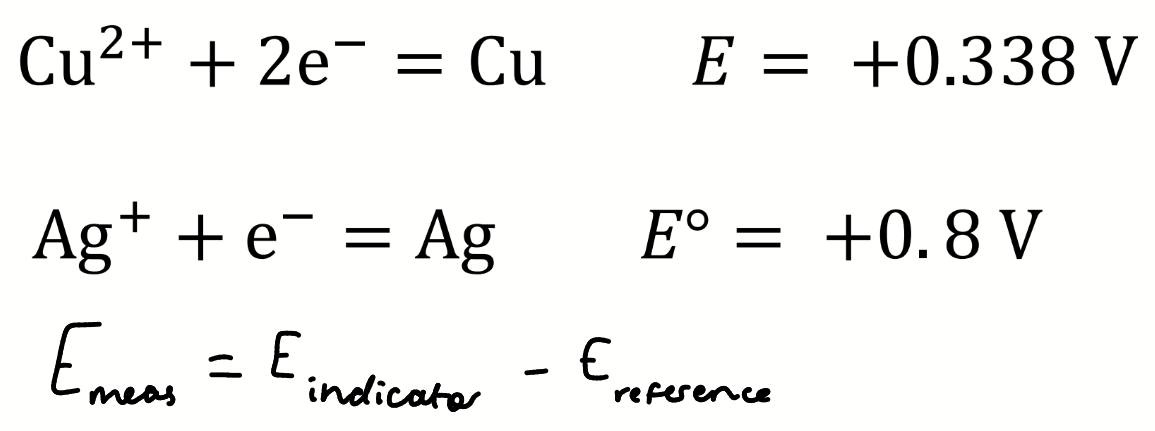
calculate Emeas
against?
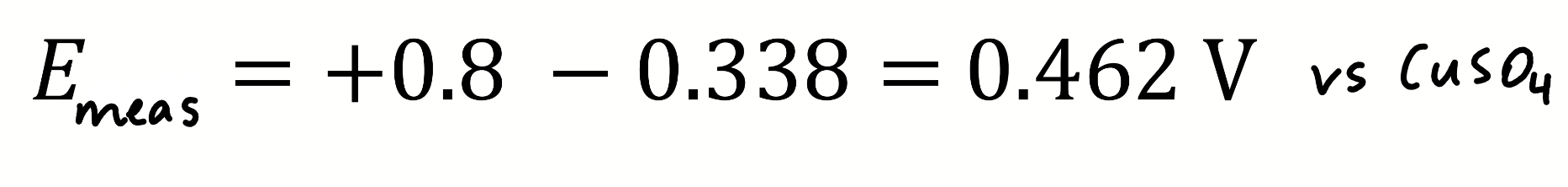
if at the equivalence point [Ag+] = [Cl-], what is Ksp?
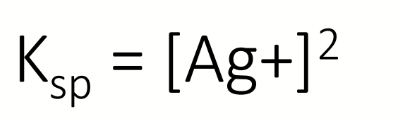
equation for E° involving E°cell and parts of Nernst
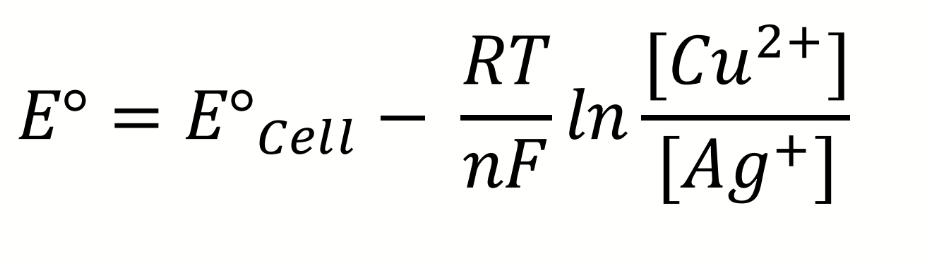
![<p>calculate K<sub>sp</sub></p><ul><li><p>E = 0.19 V</p></li><li><p>E<sub>Cu/Cu<sup>2+</sup></sub> = 0.462V</p></li><li><p>[Cu<sup>2+</sup>] in electrode = 0.1M</p></li></ul><p></p>](https://assets.knowt.com/user-attachments/2b1251e7-b8ba-415f-9b99-8e8025b70f55.png)
calculate Ksp
E = 0.19 V
ECu/Cu2+ = 0.462V
[Cu2+] in electrode = 0.1M
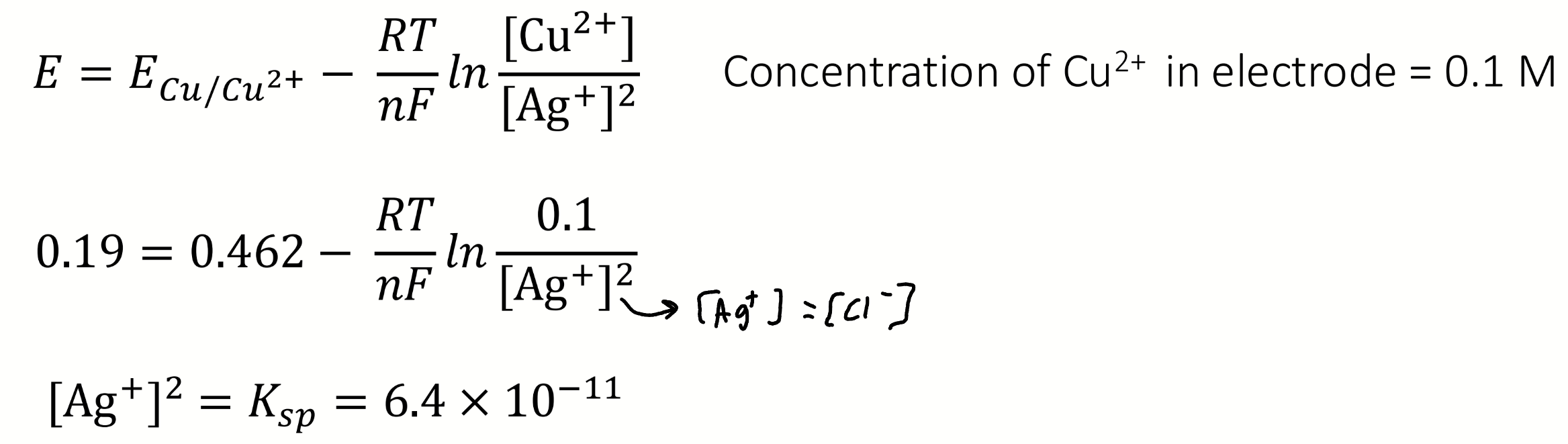
what is an advantage of using potentiometric titration to determine solubility
very low Ksp values can be accessed through equilibrium potentials rather than direct detection of solutes