MI 240 Unit 1
1/167
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
168 Terms
Radiation biology is the study of….
the effects of ionizing radiation on biological tissue
records of storage and disposal of residual and contaminated items must be kept for…
inspectors
atom is ionized =
deposit of energy in tissue
biological effects begin here
radiation interacts at the _____ level
atomic
x-ray energy deposited in the atom and ionization can affect the ____ holding atoms together causing the molecule to _____
bonds, break apart
cause of cancer or genetic mutations
cells begin to function improperly/ serious impairment
cause of radiation sickness
cells can cease to function/ cell death
cells respond differently depending on dose…
radiation is more liekly to kill a cell outright than to turn it into cancer cell
high but non-lethal doses of radiation to specific tissues can cause changes which can lead to certain types of cancer
when do early tissue interactions typically occur?
minutes
hours
days
weeks
examples of early tissues interactions
nausea
fatigue
diffuse redness of skin (erythema)
hair loss (epilation)
intestinal disorders
fever
blood disorders
shedding of outer layer of skin
early effects of radiation cause
vary depending on duration of exposure time to ionizing radiation
typically from high doses given acutely
acute radiation syndrome (radiation sickness) will most likely take place
early tissue interaction whole body exposure
hemopoietic (hematologic) syndrome
GI syndrome
CNS syndrome
early tissue interaction local tissue damage
skin
gonads
when do late tissue interactions occur?
months or years after exposure (down the line)
examples of late tissue interactions
cataracts
fibrosis
organ atrophy
loss of parenchymal cells
reduce fertility
sterility
late effects of radiation is caused by
lower doses given over long periods of time, but can also occur from higher acute exposures
late tissue effects local tissue damage
skin
cataracts
fetal effects of radiation is caused by
embryo/fetus exposed in utero to low or high levels of radiation
types of fetal effects of radiation
prenatal death
neonatal death
congenital malformation
childhood malingnancy
diminished growth and development
human populations in which radiation effects have been observed
early radiologist
radium watch dial painters, radium craze
Thorotrast contrast studies
certain conditions treated with radiation
uranium miners
atomic bomb survivors
Marshall islanders- bomb testing
radiation accident victims- Chernobyl, TMI, Fukushima
radiation therapy patients
what does coulomb/kg measure
exposure to ionizing radiation in air; electrical charge
measures the output of imaging systems and radiation monitors used to calibrate machines
amount of radiation that may strike an object when in the vicinity of a radiation source
applies only to x-rays and gamma rays
Air KERMA stands for
Air- Kinetic energy released in matter
Air kerma is measures in
gray (gy)
Air Kerma is
kinetic energy transferred from photons to electrons during ionizations/ excitiation
SI unit that can express how energy transfers from the beam to air
Air Kerma is mainly used in
fluoro (if shown ‘per min’ it is showing from fluoro)
OR
absorbed dose in air =
what is delivered to the patient
Air Kerma expresses
the radiation concentration delivered to a point, such as the entrance surface of a patient’s body
DAP meter stands for
dose area product
what does the DAP meter measure
measures the amount of energy delivered to the patient by the x-ray beam
total sum of air kerma over exposed area of patient’s surface
where is the DAP meter located
located between the collimator and patient (beyond the collimator)
equation for DAP meter
mGy x cm²
absorbed dose is measured/ expressed in
gray (gy)
cGy, mGy, uGy
absorbed dose is responsible for
biologic damage to the tissue that is exposed
Absorbed dose measures what
the quantity that express the concentration of radiation energy absorbed at a specific point within the body tissue
indicates the energy that the patient actually receives from an exposure to ionizing radiation
what unit is used to measure the biologic effects of radiation?
sievert
the concept of radiation weighting factor is used to do what?
takes into account the radiation type and energy range that may cause biologic damage
equivalent dose (sv) measures what
average dose in a tissue or organ in the human body and its associated radiation weighting factor
weighting factor (Wr) is what
takes into consideration that some radiation types may cause more biologic damage than other types depending on the dose
1 mGy =
1 mSv
equation for equivalent dose
EqD = D x Wr
equivalent dose is used to
compare biologic damage from different types of radiation
quantity of radiation recieved by radiation workers
the concept of tissue weighting factor is used to do what?
takes into account the radiosensitivity of the organ or tissue irradiated (specific organ)
effective dose (sv) is what
the sum of the weighted equivalent doses for all irradiated tissues or organs
equation for effective dose
EfD = dose x Wr x Wt
if more than one area of the body is exposed the effective dose is just the ….
sun of the effective doses for each area exposed
effective dose takes into account-
the effect of the type of radiation used and the radiosensitivity of the organ or tissue irradiated
best measure to overall risk of exposure to humans from ionizing radiation
conversion help
1 Gy = 1 Sv
1000 mGy = 1 Gy
1 Gy = 1000 mGy
cumulative lifetime limit x age
10 mSv x age or 0.01 Sv x age
annual limit
50 mSv or 0.05 Sv
lens of the eye
150 mSv or 0.15 Sv
skin, extremities
500 mSv or 0.5 Sv
embyro/fetus dose total gestation period
5 mSv or 0.005 Sv
embyro/fetus dose 1 month
0.5 mSv or 0.0005 Sv
general public annual limit- continuous
1 mSv or 0.001 Sv
general public annual limit - infrequent
5 mSv or 0.005 Sv
lens of eye for public
50 mSv or 0.05 Sv
skin, extremities for public
50 mSv or 0.05 Sv
an atom is comprised of
nucleus
electron shells
what is the nucleus
center of an atom
what is the nucleus composed of
protons
neutrons
where are the electron shells
they orbit the nucleus
what are the electron shells occupied by
electrons
what is the maximum number of electrons per shell
2n²
closer an e- shell is to the nucleus, the ____ the binding energy
higher
arrangement of shells determine what
how an atom reacts chemically
the outer shell can have a maximum number of how many electrons
8 electrons (octet rule)
valence electrons are what
electrons in the outmost shell of an atom
atoms tend to gain, lose, or share valence electrons to achieve filled outer shell
what is the octet rule?
electrons in outermost shell can have no more than 8 electrons
an atom in the neutral state is
electrically neutral, balanced
# of electrons in shells is equal to # of protons in nucleus
ionization is what?
the adding or removing of an orbital electron from an atom
the larger to atom the ___ energy is needed to ionize
more
compton interaction ionizes an _____ electron
outer shell
photoelectric interaction ionizes an _____ electron
inner shell
ion pairs are…
the fundamental result of ionizing radiation interacting with matter
the first step in how x-rays create images or damage cells is what
ion pairs
how to make an ion pair
the atom is neutral
an x-ray photon strikes the atom with enough energy to knwock an electron out of its orbit
result in ion pair
anion
cation
a negative electron (anion) is
a newly freed, negatively charged electron (sometimes called a photoelectron)
a positive ion (cation) is
the rest of the atom, which lost a negative electron and is now positively charged
molecules are made up of
two or more atoms bonded together
molecules are formed by
ionic and covalent bonding
what is an ionic bond
it is the attraction between positively charged ions and negatively charged ions that forms after electrons are transferred from one atom to another
giving/ tranferring
what is a covalent bond
when two atoms share one or more pairs of electrons so that both atoms can fill their outer (valence) shells
stays connected/ ‘holding hands’
mass number (A) is
the number of protons plus the number of neutrons in a nuclide
atomic number (Z)
the number of protons in a nuclide
isotopes are
atoms with the same number of protons and electrons
but have a DIFFERENT number of neutrons
mass changes but the element stays the same
how many protons does barium have
56
the most abundant atom of barium has an atomic mass of
138
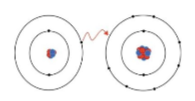
what is this an image of
ionic bond
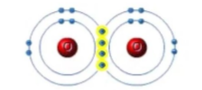
what is this an image of
covalent bond
another name for coherent scattering
classical scattering
coherent (classical) scattering is what
x-ray interact with an atom and excited it
x-ray scatters in different direction when it is released
no change in energy or wavelength
no energy transfer, no ionization
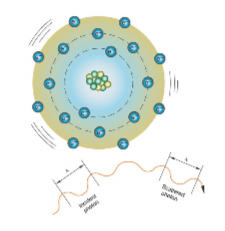
this is an example of
coherent (classical) scattering
coherent scattering happens at
below 10 kVp
compton scattering is
ionization of out shell electrons
out shell electron ejected
incident photon will scatter in a different direction
compoton or scattered photon
the scattered photon in compton scattering has
less energy
longer wavelength
lower frequency
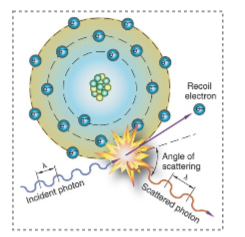
what is this an example of
compton scatter
compton scatter mainly affects
the technologists (us)
can increase occupation dose
compton scatter occurs at
higher kVp
Compton scatter produces large amount of scatter in the ____
patient
the process of photoelectric absorption is an interaction between
x-ray photon
inner shell electron