Determination of boric acid in boracic water by potentiometric titration
1/28
Earn XP
Description and Tags
Determination of boric acid in boracic water by potentiometric titration. Standardization of sodium hydroxide solution against potassium hydrogen phthalate by visual titration. Determination of boric acid in boric water by potentiometric titration. Subject: analytical chemistry laboratory exercise
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
29 Terms
Boric Acid (H3BO3)
A weak acid used in the experiment, whose concentration in boracic water is determined by potentiometric titration.
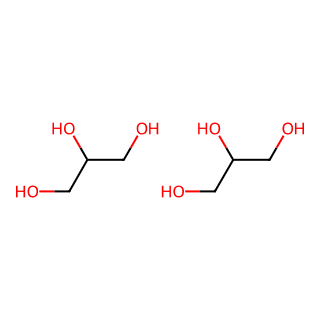
Glycerol
A substance added to the boric acid solution in the experiment to form a complex with the acid, making the titration more effective by increasing the acidity.
Boric Water
A dilute aqueous solution of boric acid, often used as an antiseptic or eyewash in medical applications.
Titration
A laboratory technique where a solution of known concentration is used to determine the concentration of an unknown solution.
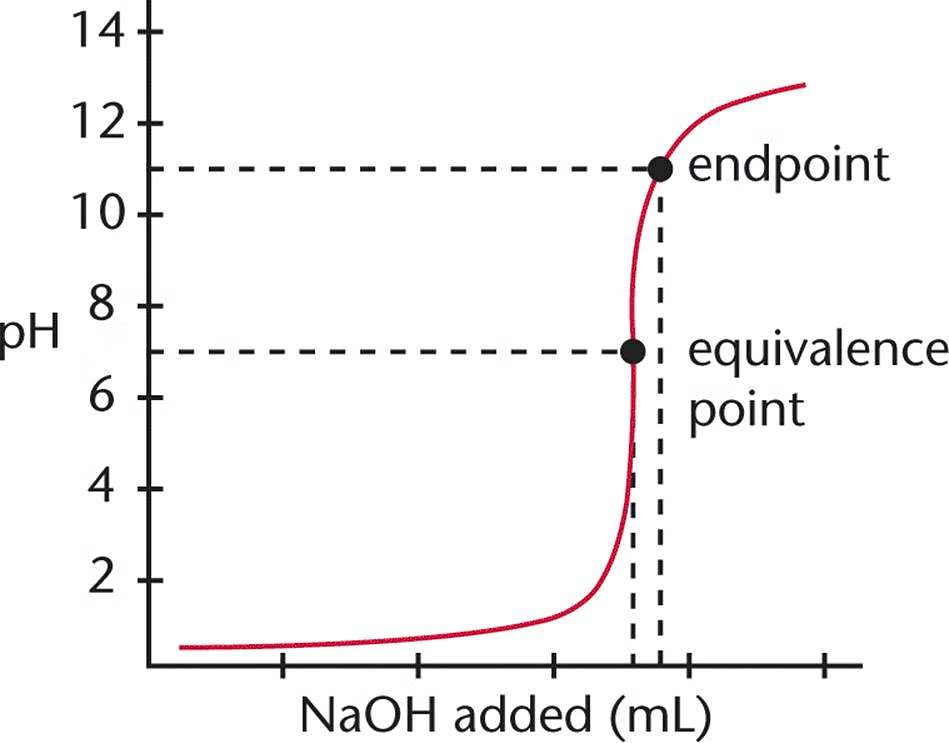
Equivalence Point
The point in a titration where the amount of titrant added is stoichiometrically equivalent to the amount of substance being titrated.
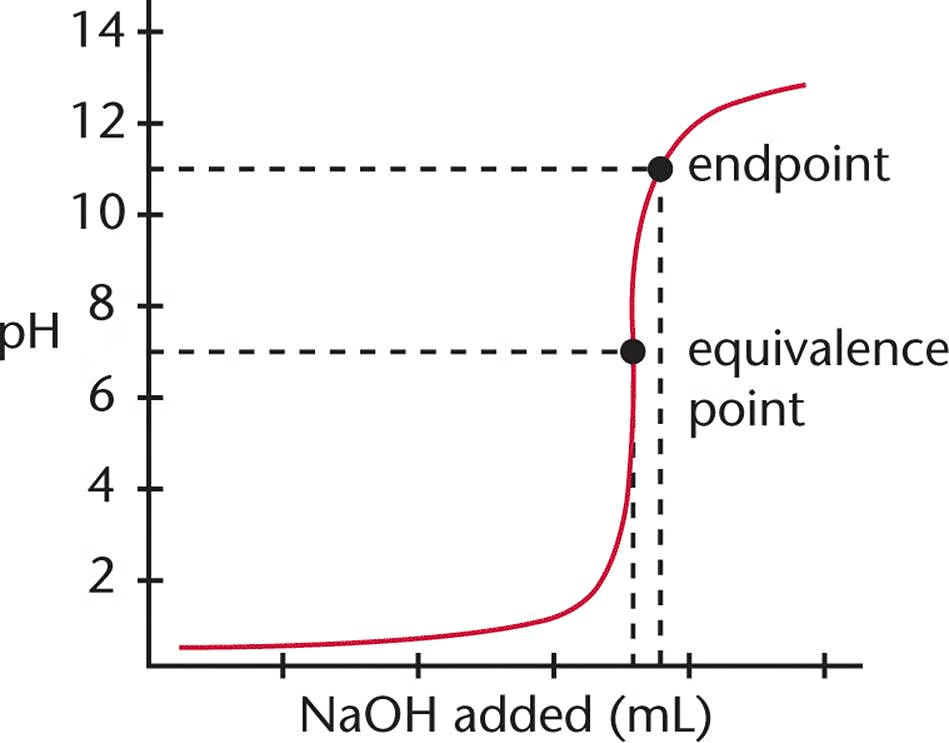
Endpoint
The point in a titration where the indicator changes color, signaling that the titration is complete, often near the equivalence point.
Volumetric solution
A solution of known concentration used in titrations. Its concentration is usually determined using a primary standard.
Sodium Hydroxide
A strong base used in titration experiments to neutralize acids and determine their concentration through pH measurement.
Standardization
The process of determining the exact concentration of a solution by titration with a standard solution of known concentration.
Primary Standard
A highly pure substance that can be used to determine the concentration of a solution in titration, due to its stability and known reactivity.
Potassium Hydrogen Phthalate
A primary standard substance used to standardize basic solutions like sodium hydroxide in titrations.
Indicator
A substance that changes color at a specific pH range, used to signal the end point of a titration.
Phenolphthalein
A chemical indicator that changes color from colorless to pink in basic solutions, often used to signal the endpoint in acid-base titrations.
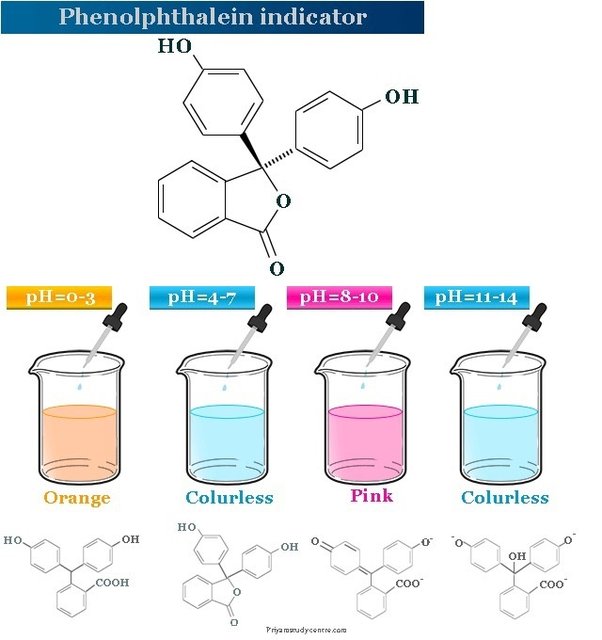
Standardization of Sodium Hydroxide Solution Against Potassium Hydrogen Phthalate
The process of determining the exact concentration of a sodium hydroxide solution by reacting it with a known quantity of potassium hydrogen phthalate.

Reaction of Potassium Hydrogen Phthalate with Sodium Hydroxide
An acid-base reaction where potassium hydrogen phthalate (an acid) reacts with sodium hydroxide (a base) to form potassium sodium phthalate and water. Reaction: KHC8H4O4 +NaOH → KNaC8H4O4 + H2O
Determination of Boric acid in Boric water
A titration process where boric acid concentration in boric water is determined by reacting with a strong base like sodium hydroxide. Glycerol is added to enhance the reaction by forming a more detectable borate complex.
Reaction of Boric acid with Sodium Hydroxide
The determination of boric acid can be done by alkalimetric titration after the addition of polyhydric alcohols or sugars, such as glycerol or sorbitol, with which it forms stronger complex acids. In this case, boric acid reacts with sodium hydroxide in a 1:1 ratio. Reaction to form a complex acid: H₃BO₃ + 2 C₃H₈O₃ → H[X]⁻ + H₃O⁺ + 2 H₂O
The titration reaction of glyceroboric acid with sodium hydroxide (simplified notation of the acid is H[X]): H[X] + NaOH → Na[X] + H₂O
Visual titration vs potentiometric titration
Visual titration relies on color changes of an indicator to determine the end point, while potentiometric titration uses a pH meter or electrode to measure changes in electrical potential.
Potentiometry
An analytical technique that measures the voltage of an electrochemical cell to determine the concentration of an analyte in a solution.
pH
A measure of the acidity or basicity of an aqueous solution, representing the concentration of hydrogen ions. pH = -log [H+]
pH Meter
A device used to measure the hydrogen ion concentration (pH) in a solution, providing information about its acidity or alkalinity.
pH meter VioLab 50

Ion-Selective Electrode (ISE)
An electrode that selectively responds to a specific ion in the presence of other ions, used in potentiometric measurements.
Glass Electrode
A type of pH electrode commonly used in potentiometric measurements that is sensitive to hydrogen ion concentration.
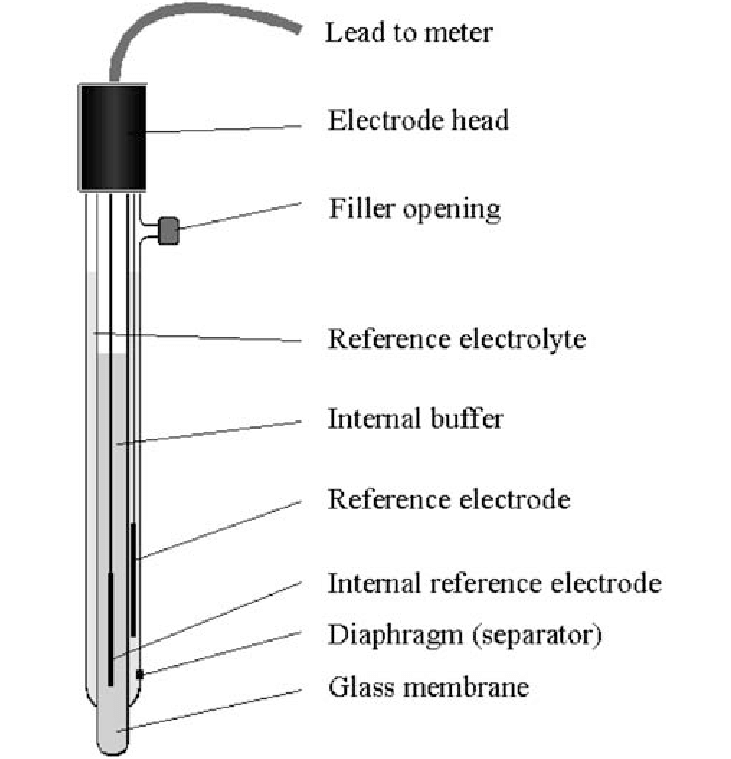
Electrode Calibration
The process of adjusting the readings of an electrode, typically a pH electrode, by using standard buffer solutions to ensure accurate measurements. 1. Prepare pH buffers (typically pH 4, pH 7, and pH 10). 2. Rinse electrode with distilled water. 3. Immerse in pH 7 buffer and calibrate. 4. Repeat for other buffers.

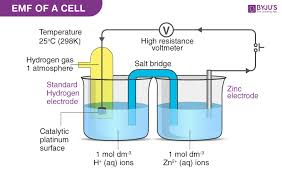
Galvanic Cell
An electrochemical cell that converts chemical energy into electrical energy through spontaneous redox reactions, often used in potentiometric measurements.
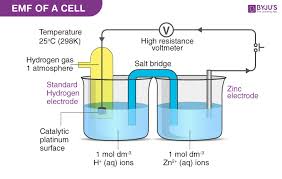
EMF (Electromotive Force)
The voltage generated by an electrochemical cell, which drives the movement of electrons and can be measured in potentiometry.
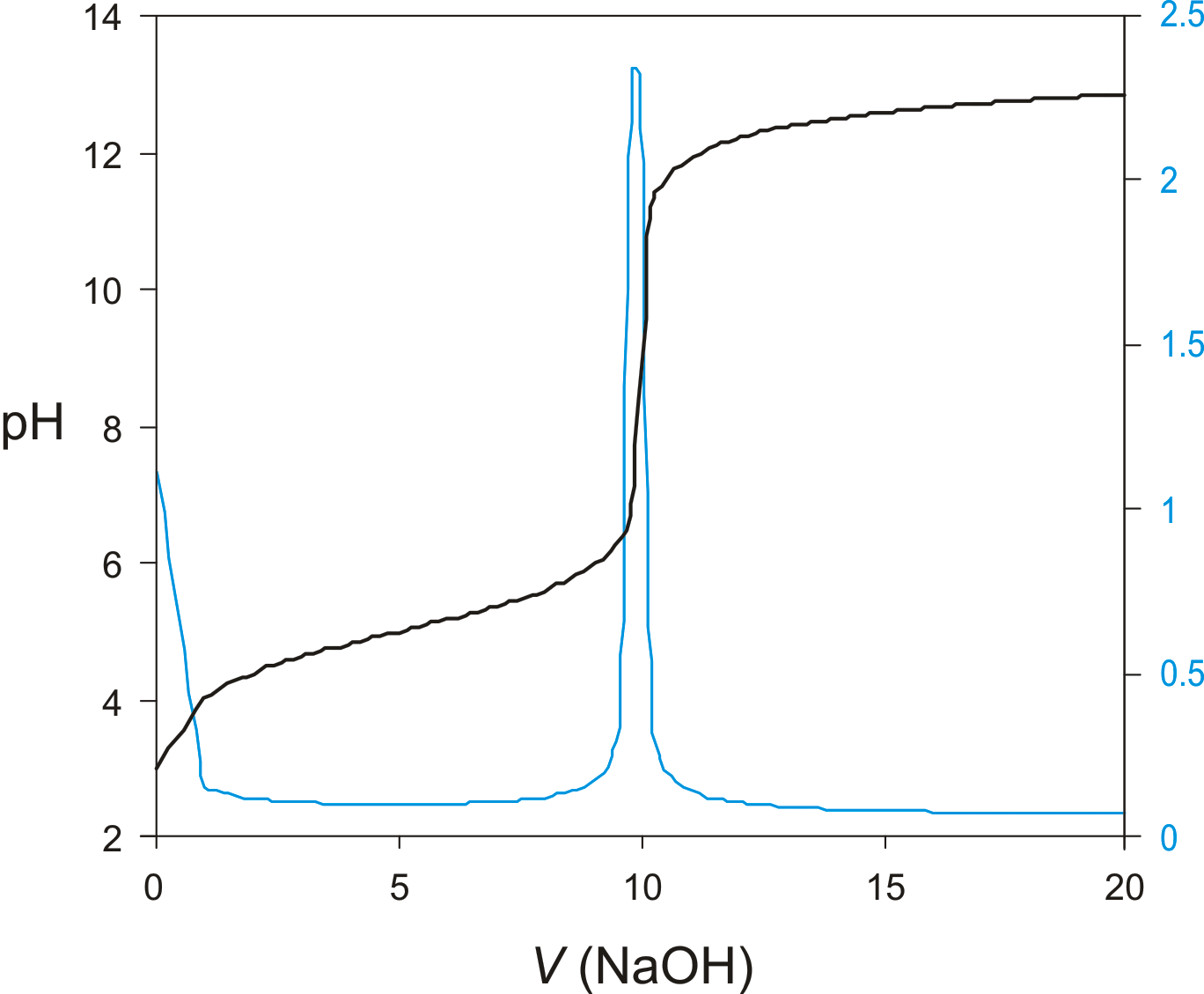
Titration Curve
A graph that shows the change in a solution's pH or potential as a titrant is added, used to identify the equivalence point in a titration. (Blue curve = derivative curve)
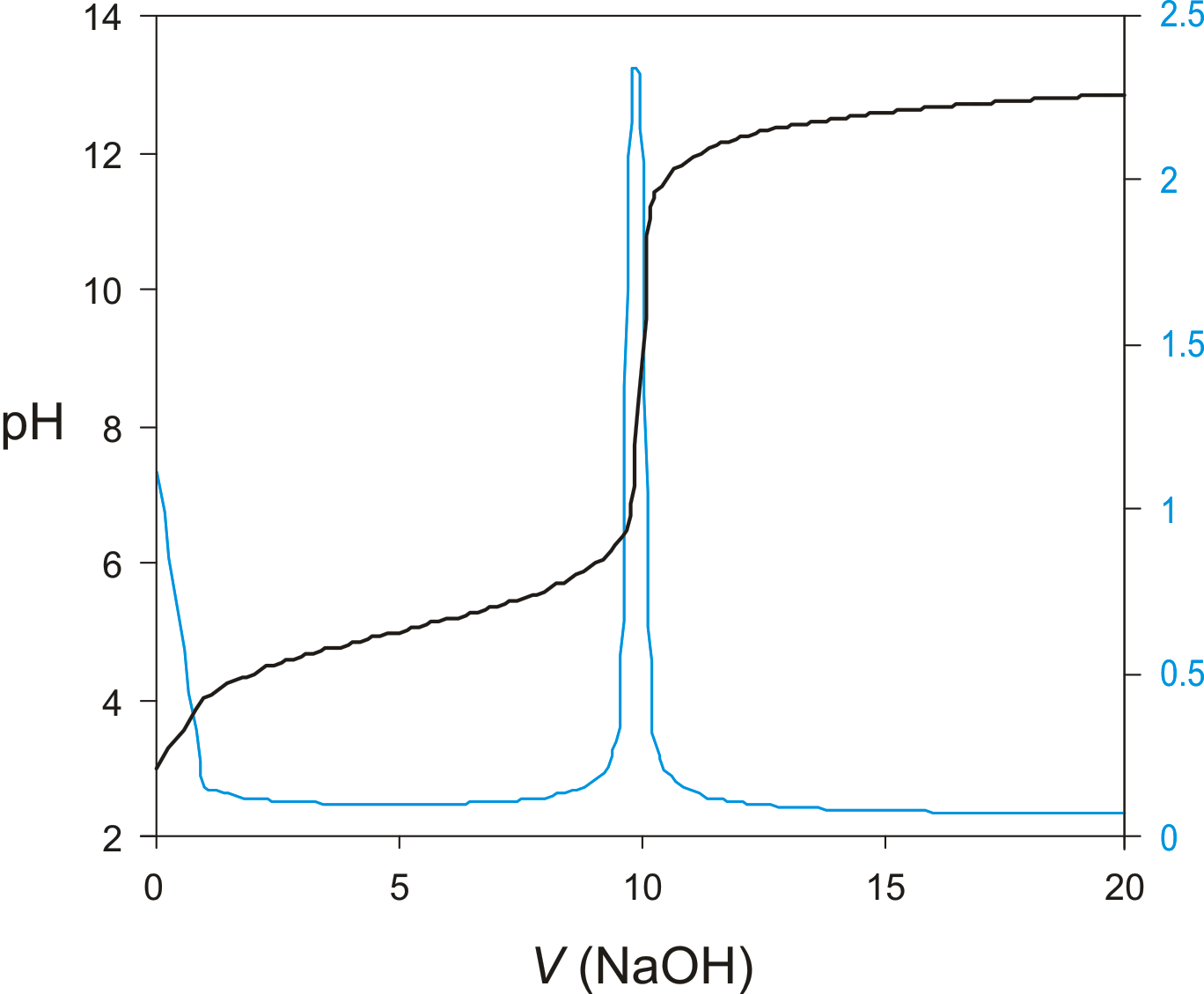
Derivative Curve
A graph of the first derivative of a titration curve, used to more accurately determine the equivalence point of a titration. (Blue curve = derivative curve)