3.3.3.1 nucleophilic substitution
1/44
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
45 Terms
what are the halogenoalkanes?
the homologous series of compounds with the general formula CnH2n+1X (X is a halogen ie F, Cl, Br or I)
what 3 things can halogenoalkanes be classified as?
as primary, secondary or tertiary
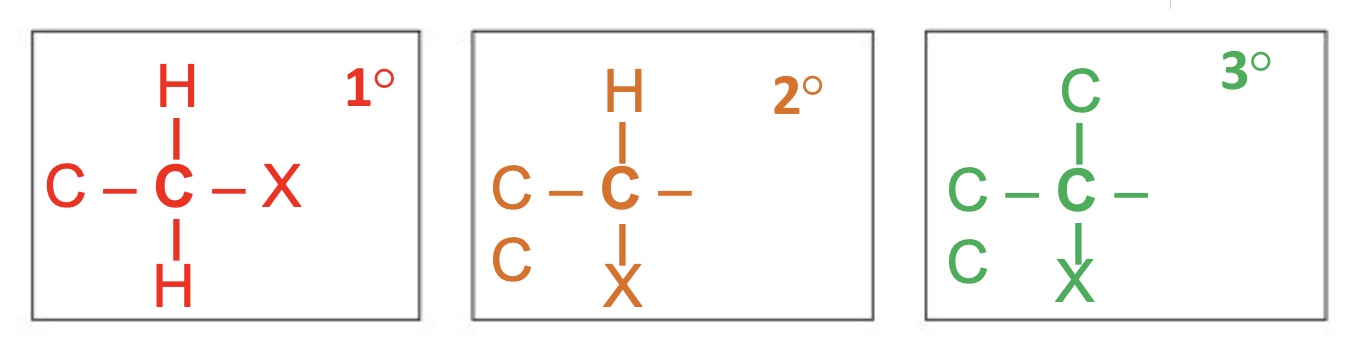
how are halogenoalkanes named?
as substituted alkanes with fluoro, chloro, bromo or iodo used as the prefix
what is a mechanism?
a pictorial representation of the movement of a pair of electrons during a chemical reaction
what will happen during a reaction?
some bonds will be broken + new bonds are formed
when will a pair of electrons move?
when bonds are broken + new bonds formed
what is the moving of electrons shown by?
by using a curly arrow
what do curly arrows show?
the movement of a pair of electrons in a mechanism
where are pairs of electrons present on a molecule?
they are present in two places on a molecule:
they are either present as a shared pair in a covalent bond or as a lone pair on an atom
therefore where must arrows in mechanisms start from?
they must start at a bond or a lone pair of electrons
curly arrows finish where?
in the new location of the electron pair
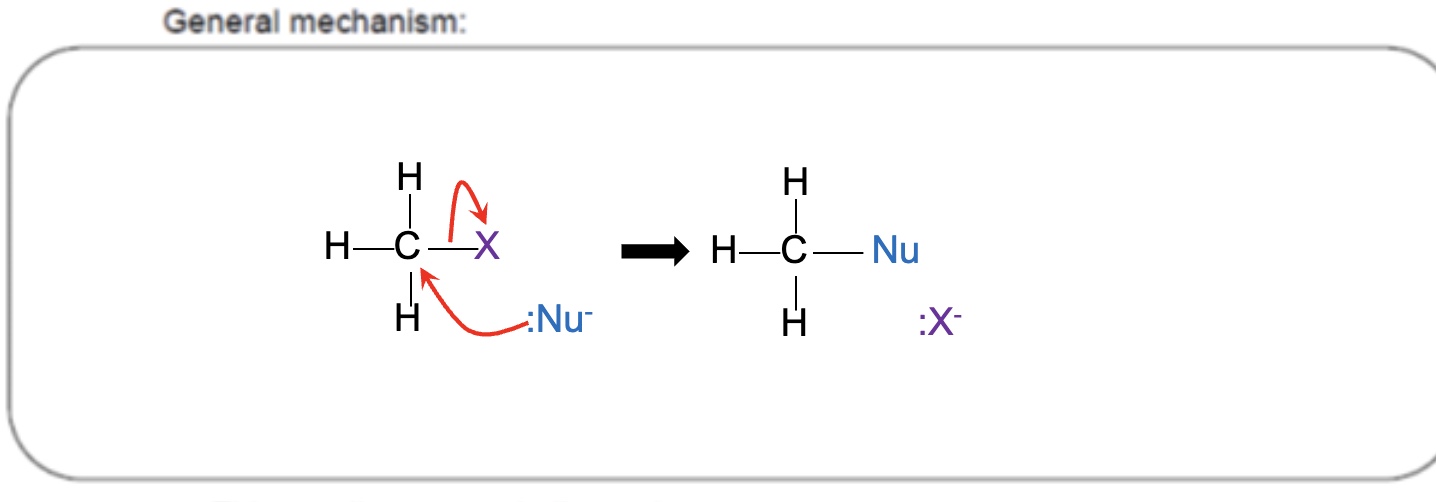
draw a mechanism arrow for the formation of a bond between :Y- and a C and a breaking of a bond between C and X

what is a nucleophile?
a species that contains a pair of electrons which it can donate to form a bond
the carbon-halogen bonds are ____
polar
explain how the carbon-halogen bonds are polar
the halogen atoms in halogenoalkanes are more electronegative than C
the electrons in the C-X bond are attracted towards the halogen atom which gains a slight negative charge,δ-, leaving the carbon atom electron deficient (with a slight positive charge), δ+
what happens to the electron deficient carbon atom?
it is readily attacked by species that contain a lone pair of electrons ie a nucleophile
describe what reaction will happen when the carbon atom is attacked by a nucleophile
the nucleophile will donate its lone pair of electrons to carbon, forming a bond
the carbon-halogen bond then breaks + a halide ion is released
this type of reaction is known as nucleophile substitution as the halide ion is substituted for a nucleophile
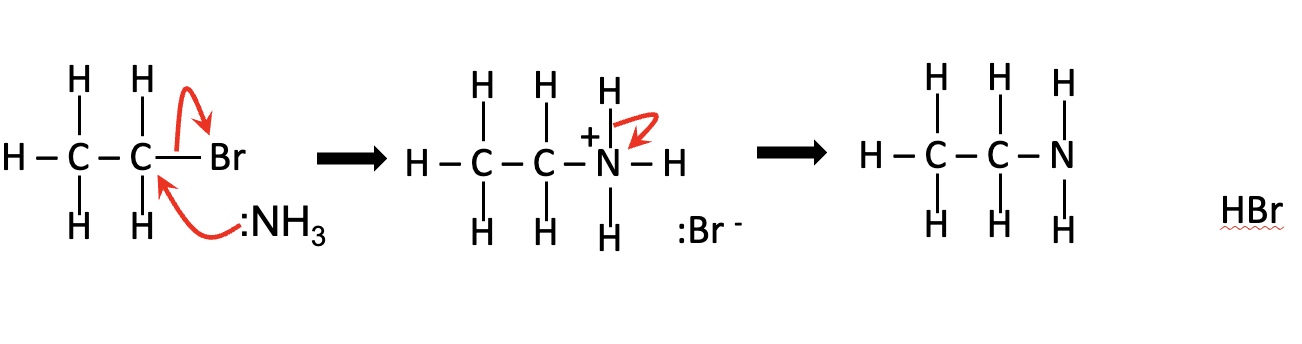
give the steps to drawing a mechanism for nucleophilic substitution
the lone pair on nucleophile forms a covalent bond with the δ+ carbon atom — this is shown using a curly arrow
the electrons in the C-X bond move onto the halogen atom, breaking the C-X bond — this is shown using a curly arrow
by gaining the electrons from the bond, the halogen forms a halide ion
give 3 nucleophilic substitution reactions of halogenoalkanes
hydroxide ions (OH-) forming alcohols
cyanide ions (CN-) forming nitriles
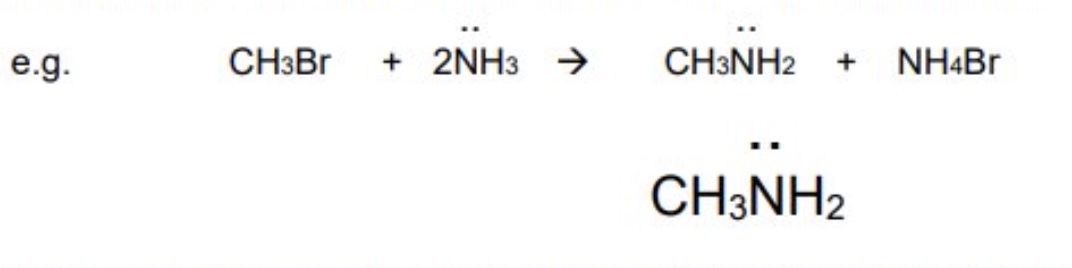
excess ammonia (NH₃) forming primary amines
give the reagent + conditions for nucleophilic substitution with hydroxide ions
reagent — NaOH
conditions — warm, aqueous
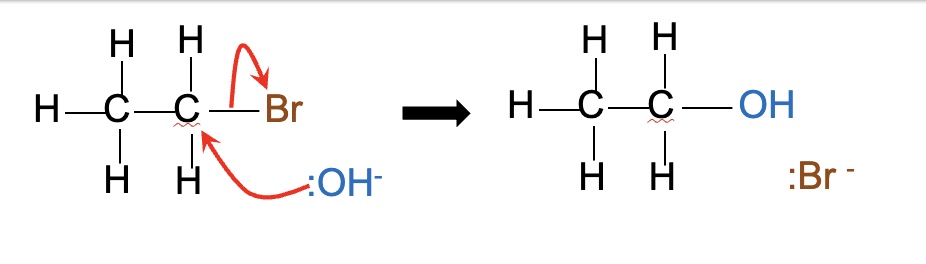
what happens when halogenoalkanes are warmed with aqueous sodium hydroxide (NaOH) or potassium hydroxide (KOH)?
alcohols are formed → NaOH + KOH are ionic compounds containing the nucleophile :OH-

eg draw the mechanism for the reaction between:
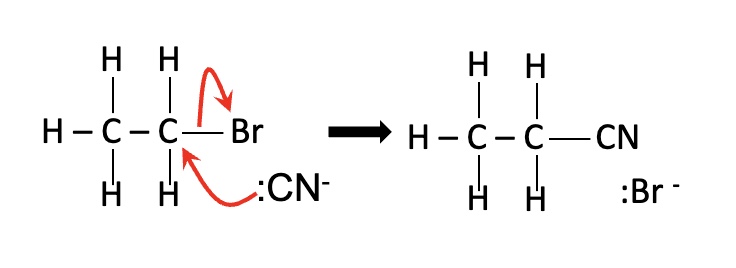
give the reagent + conditions for nucleophilic substitution with cyanide ions
reagent — KCN
conditions — aqueous ethanol
what happens when halogenoalkanes are warmed with an aqueous ethanol solution of potassium cyanide (KCN)?
nitriles are formed → this adds an extra carbon to the chain (the nucleophile in KCN is :CN-)
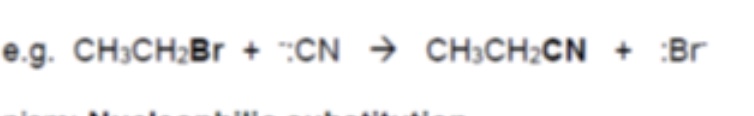
eg draw the mechanism for the reaction below:
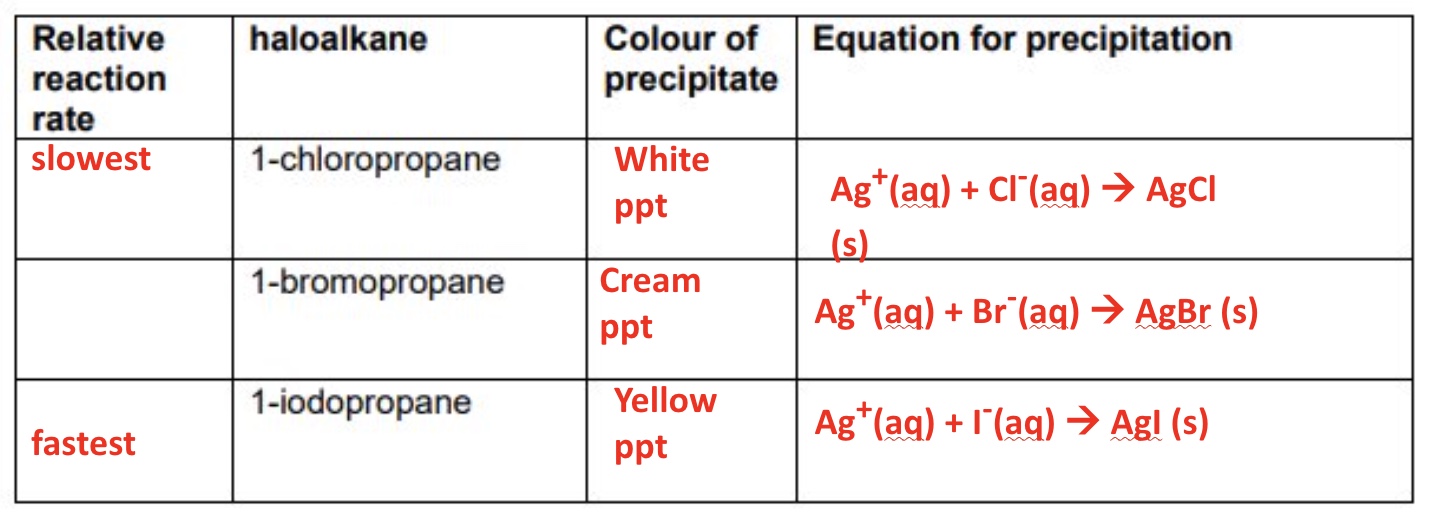
outline an experiment to show the rate of hydrolysis of the haloalkanes + strength of the C-X bond (bond enthalpies)

give the results of the experiment done to show the rate of hydrolysis of the haloalkanes using chloro/bromo/iodopropane → give the colour of precipitate formed, ionic equation
what happens to the C-X bond as you go down the halogens?
it gets weaker (based on bond enthalpies)
explain why the C-X bond gets weaker down the group
fluorine is the smallest atom of the halogens + the shared electrons in the C-F bond are strongly attached to the fluorine nucleus → this makes a strong bond
as we go down the group, the shared electrons in the C-X bond get further + further away from the halogen nucleus so the bond becomes weaker
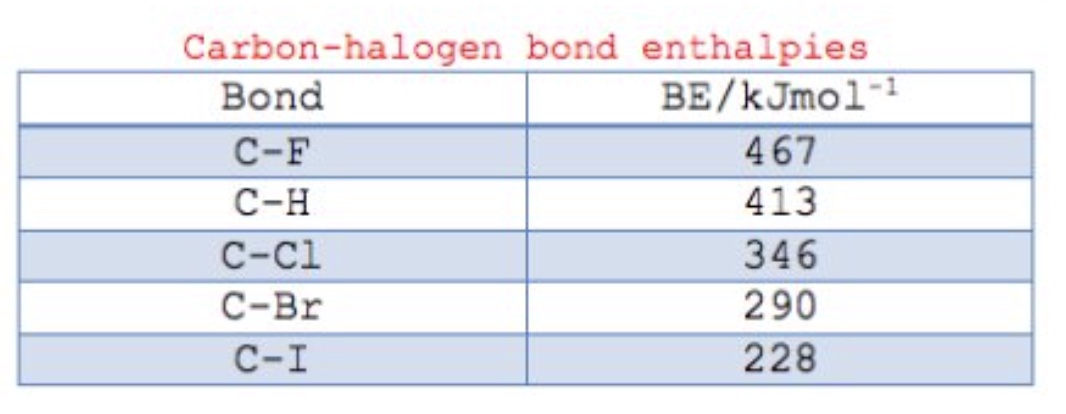
what do the bond enthalpies predict about haloalkanes?
that iodo-compunds, with the weakest bonds, are the most reactive and fluroro-compunds, with the strongest bonds, are the least reactive
therefore ______ increases as you go down group 7
reactivity
give the reagent + conditions for nucleophilic substitution with excess ammonia
reagent — NH₃
conditions — excess NH₃
when are primary amines formed? (2 hydrogens attached to the nitrogen)
when haloalkanes are warmed with an excess of ammonia in a sealed container → the nucleophile is ammonia in which the nitrogen atom has the lone pair of electrons
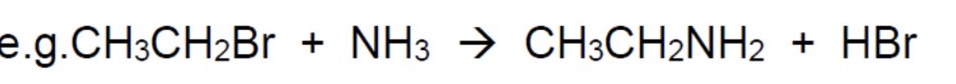
because this reaction produces HBr, what is the actual overall equation + why?
HBr is an acid + therefore will react with the excess base NH₃ to give NH₄Br
(NH₃ + HBr → NH₄Br)
the overall equation is shown in the image + its the one you write in the exam
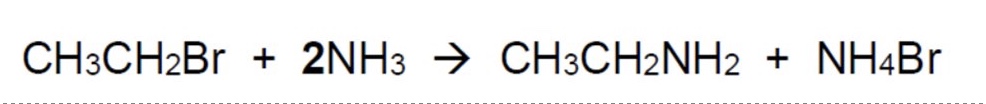
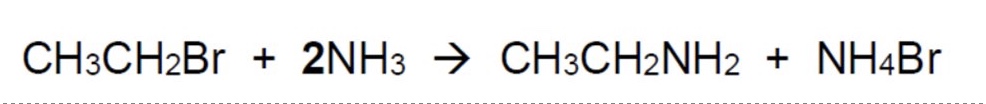
draw the mechanism for this reaction in the image (nucleophilic substitution with excess ammonia)
the primary amine that is formed in these reactions is also a _________
nucleophile
because the primary amine is also a nucleophile what does this mean can happen?
the primary amine can donate their lone pair to a δ+ carbon atom on another haloalkane → this leads to additional carbon groups bonded to the central nitrogen
how many further substitutions of primary amines can take place?
three
outline the first further substitution of primary amines + give the equation
the primary amine produced can also react with another haloalkane to produce a secondary amine
haloalkane + primary amine → secondary amine + HX
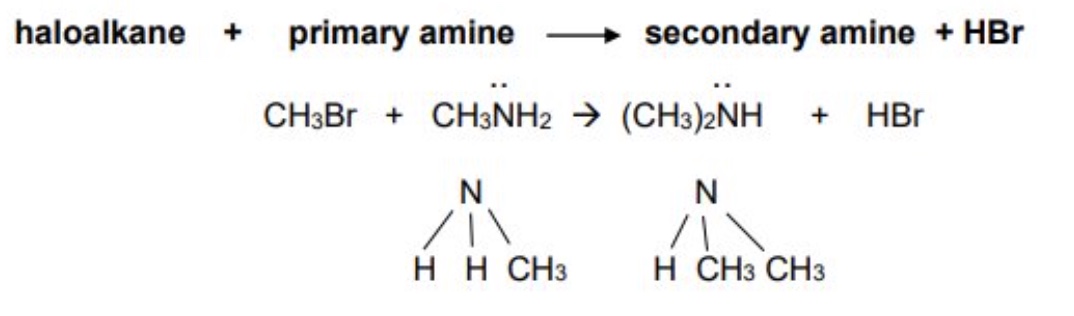
outline the second further substitution of primary amines + give the equation
the secondary amine produced can also react with another halogenoalkane to produce a tertiary amine
haloalkane + secondary amine → tertiary amine + HX
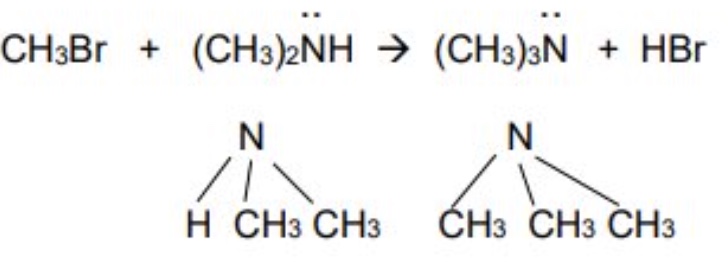
outline the third further substitution of primary amines + give the equation
the tertiary amine produced can also react with another haloalkane to produce a quaternary ammonium salt
haloalkane + tertiary amine → quaternary ammonium salt
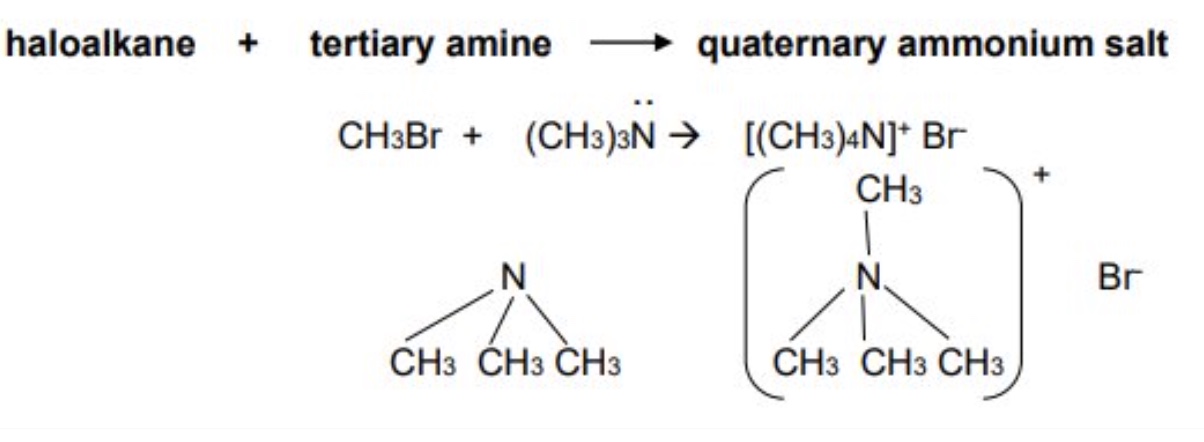
what products are usually obtained from nucleophilic substitution with excess ammonia?
mixtures of primary, secondary, tertiary + quaternary ammonium salts are usually obtained
a large excess of halogenoalkane leads to what?
to a high yield of quaternary ammonium salt
what leads to a better yield of primary amine?
a large excess of ammonia
why is there no further substitution beyond the third further one?
the quaternary ammonium salt formed contains no lone pairs on the nitrogen