Chem 1b- Structures of Crystalline Solids
1/39
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
40 Terms
Van Der Waals Forces
Intermolecular Forces
Dipole-Dipole Forces
weak electrostatic between neighboring molecules
SN #2
Linear
SN #3
Trigonal Planar
SN #3 + 1 lone Pair
Bent
SN #4
Tetrahedral
SN #4 + 1 Lone Pair
Trigonal Pyramidal
SN #4 + 2 Lone Pair
Bent (p.2)
SN #5
Trigonal Bipyramidal
SN #5 + 1 Lone Pair
Seesaw
SN #5 + 2 Lone Pair
T-Shaped
SN #5 + 3 Lone Pair
Linear
SN #6
Octahedral
SN #6 + 1 Lone Pair
Square Pyramidal
SN #6 + 2 Lone Pair
Square Planar
Hydrogen Bond Donor
When H atom is covalently bonded to F, O, N atom
Hydrogen Bond Acceptor
The electronegative F, O, N atom in neighboring molecule
London Dispersion Forces
Weak electrostatic attractions between nonpolar covalent molecules/monatomic gases; when movement of electrons produces a temporary dipole
Crystalline Solid
solid material whose composition contains a highly ordered 3-D geometric shape and has a specific melting temperature
Amorphous Solid
Solid material whose composition are arranged in irregular manners; does not have well-defined shapes, and melts over a temperature range
Molecular Solids
Component units are molecules
held together by intermolecular forces
Ionic Solids
Component units are cations and anions
Held together by electrostatic attractions
Atomic Solids
Component units are atoms
Metallic, Network, Group 8A solids fall under this category
Metallic Solids
Held together by metallic bonding
Network Solids
Held together by covalent bonding
Group 8A Solids
Held together by London dispersion forces
Simple Cubic Packing (l=2r)
CN: 6
Efficiency: 52%
Ave. : 1
Body Centered Cubic (l=4r/sqr3)
CN: 8
Efficiency: 68%
Ave.: 2
CCP/FCC (l= sqr8 r)
CN: 12
Efficiency: 74%
Ave.: 4
Hexagonal Close-Packed (HCP)
CN: 12
Efficiency: 72%
Ave.: 6
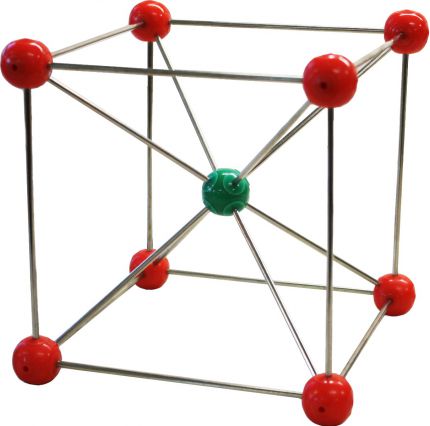
Picture: Cesium Chloride
Cesium Chloride
Cat/Anion: sc
CN: 8
Geometries: Cubic
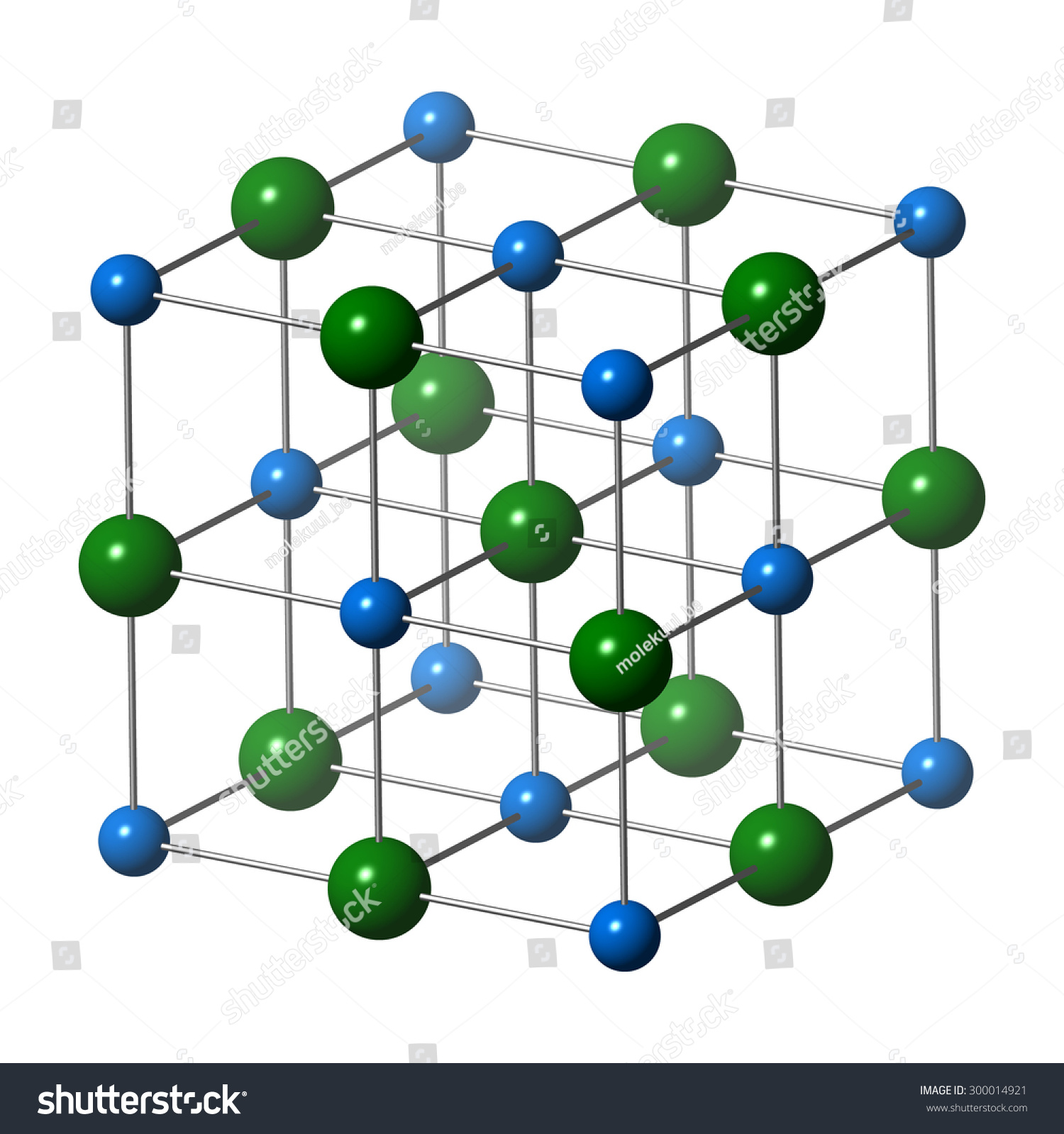
Rock Salt
Cat/Anion: fcc
CN: 6
Geometries: octahedral
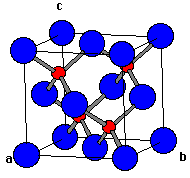
Zinc Blende
Cat/Anion: fcc
CN: 4
Geometries: tetrahedral
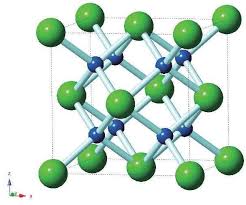
Fluorite
Cat/Anion: fcc. sc
CN: 8, 4
Geometries: cubic, tetrahedral
Antifluorite
Cat/Anion: sc, fcc
CN: 4, 8
Geometries: tetrahedral, cubic
Rock Salt Structure
Zinc Blende Structure
Fluorite Structure
AntiFluorite Structure