3.2. Fundamentals of Organic Chemistry and Important Macromolecules
1/66
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
67 Terms
1. In positron emission tomography, 2-fluoro-2-deoxy-D-glucose labeled with the radionuclide ¹⁸F is used as a glucose analog.
How many nucleons does the nucleus of this radionuclide contain?
A. 7
B. 9
C. 18
D. 19
E. 27
C. 18
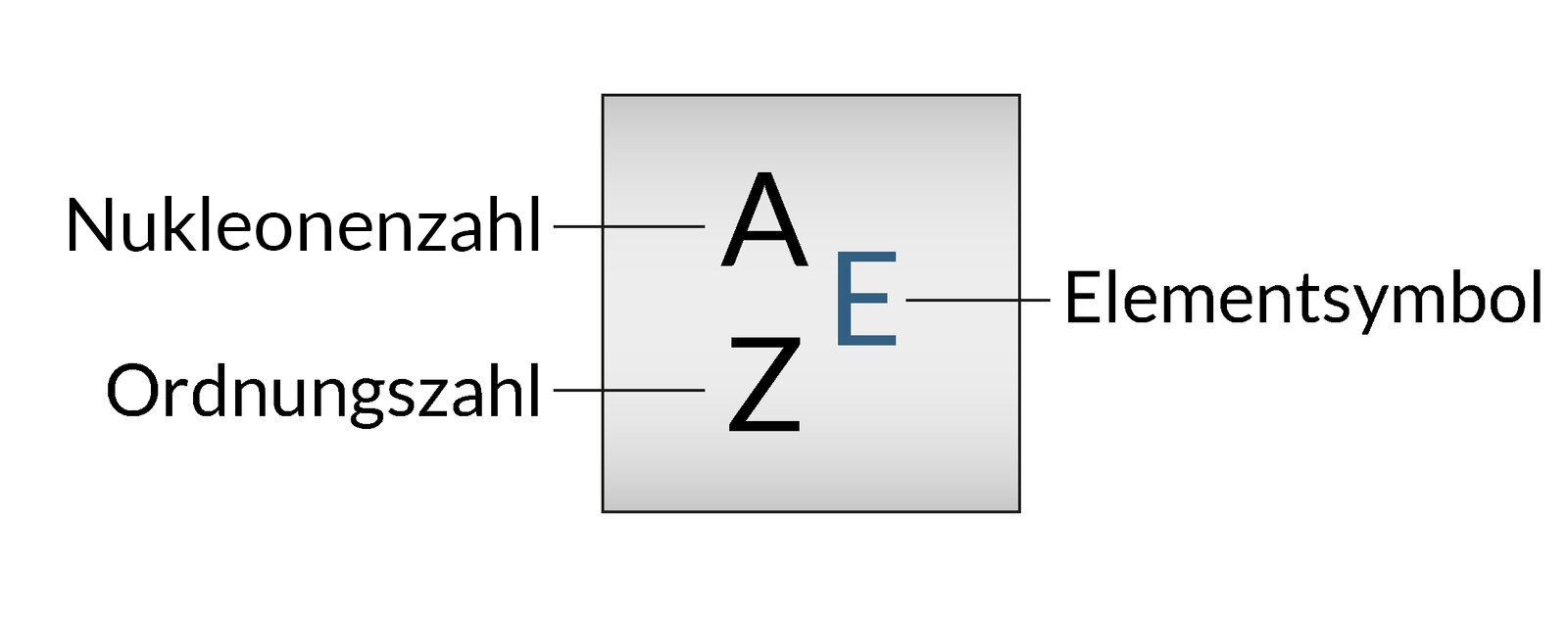
2. To prepare a solution for nasal irrigation, 2.5 g of sodium chloride are dissolved in water. The final solution has a mass of 250 g.
What is the concentration or salt content of this solution?
(M(Na) = 23 g/mol, M(Cl) = 35 g/mol)
A. 1-molal
B. 1-molar
C. 1-normal
D. 1-ppm
E. 1-percent
E. 1-percent
3. When barium sulfate is used as an X-ray contrast agent, contamination with the more soluble barium chloride (BaCl₂) can lead to serious adverse events.
What type of bond is present in solid barium chloride?
A. Ionic bond
B. Coordinate bond
C. Metallic bond
D. Polar covalent bond
E. Nonpolar covalent bond
A. Ionic bond
4. Photometry is used, among other things, for the quantitative determination of numerous blood values in clinical-chemical enzyme diagnostics. The solution of a substance with concentration c₁ shows an extinction of E₁ = 0.6 in the photometer. For the standard solution of the substance with concentration c₂ = 1.2 mg/mL, the extinction (at the same wavelength) is E₂ = 0.36.
What is the concentration c₁ of the test solution?
A. 0.6 mg/dL
B. 20 mg/dL
C. 2 mg/mL
D. 7.2 mg/mL
E. 20 mg/mL
C. 2 mg/mL
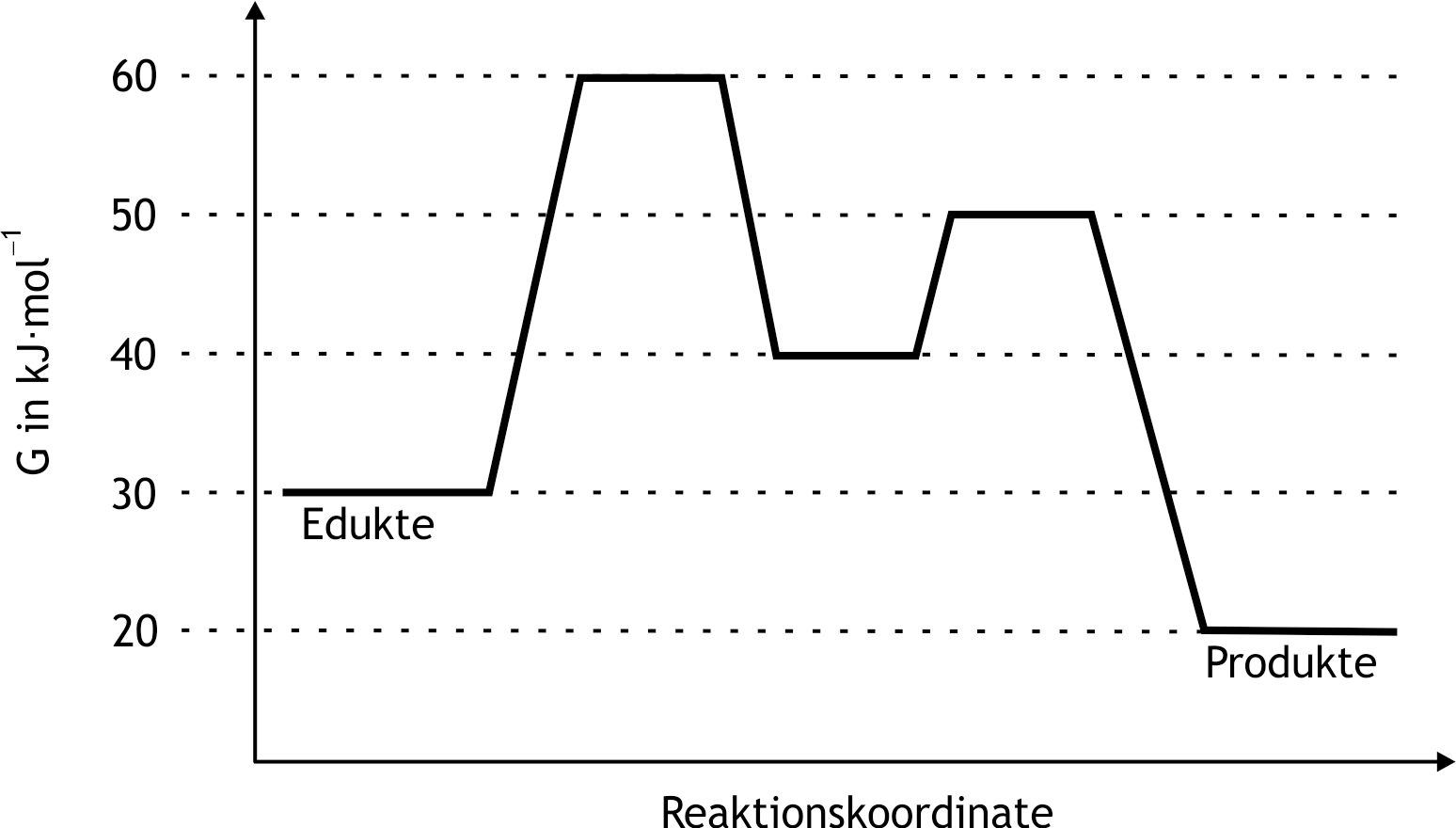
5. Enzymes catalyze the vast majority of biochemical reactions in biological systems. The following shows the energy diagram of a two-step enzyme-catalyzed reaction: What is the (Gibbs) activation energy that determines the overall reaction rate?
A. 10 kJ·mol⁻¹
B. 20 kJ·mol⁻¹
C. 30 kJ·mol⁻¹
D. 40 kJ·mol⁻¹
E. 50 kJ·mol⁻¹
C. 30 kJ·mol⁻¹
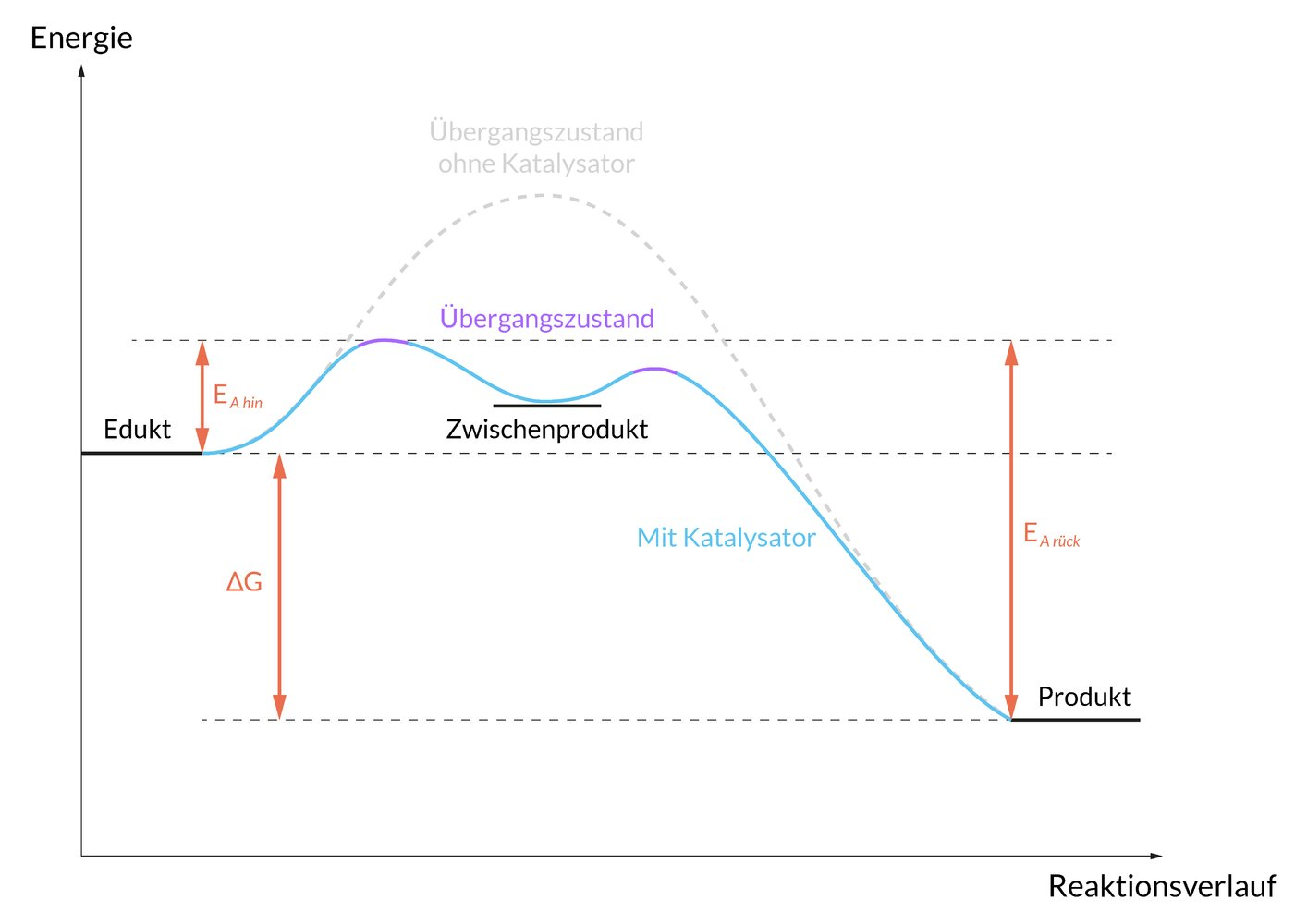
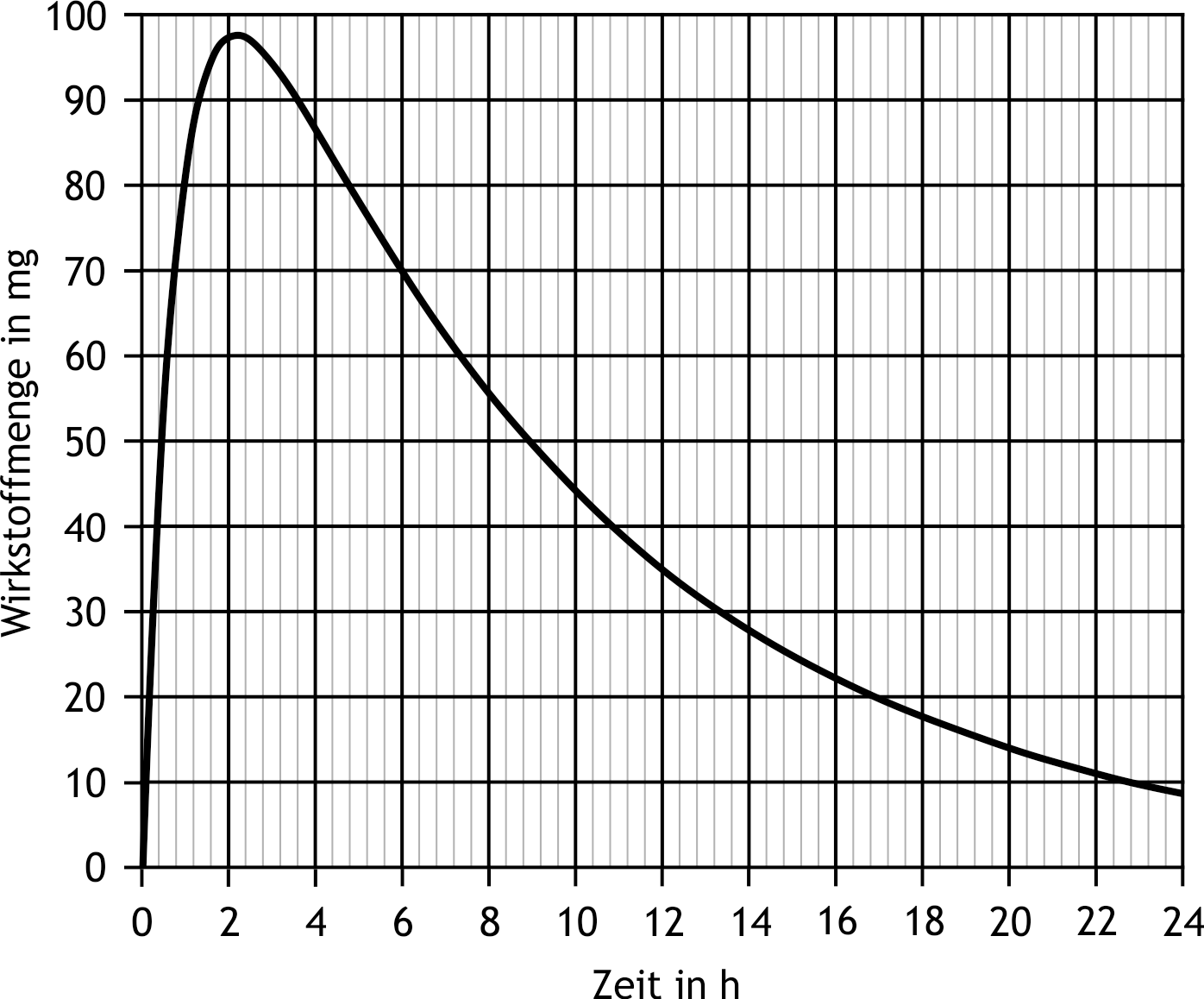
6. The absorption or elimination of a drug can be described by simple kinetic models. The following diagram idealizes the total amount of drug in the body as a function of time. It is assumed that absorption and elimination each follow a first-order reaction. What is the approximate half-life for the elimination of this drug?
A. 0.5 h
B. 2 h
C. 6 h
D. 9 h
E. 15 h
C. 6 h
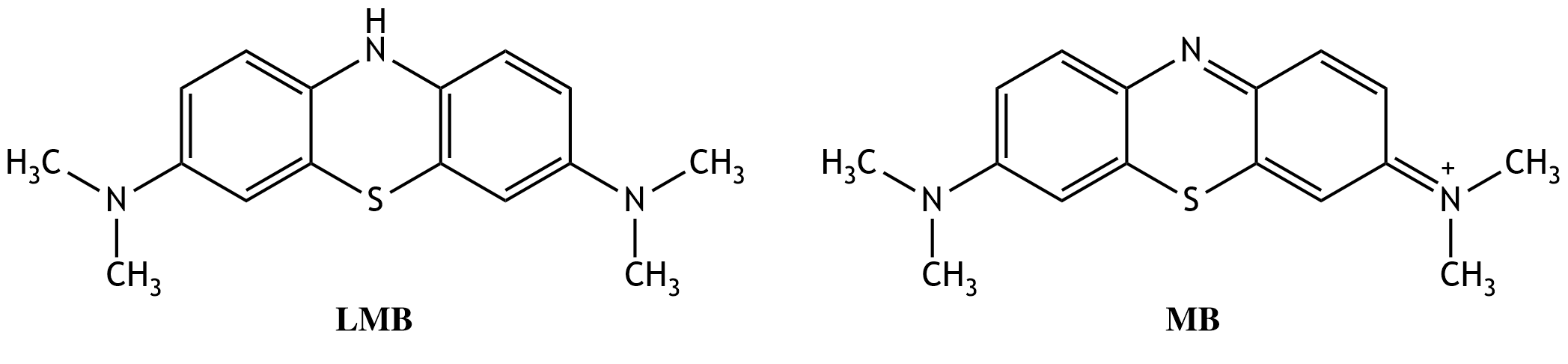
7. An increased methemoglobin concentration in the blood can be reduced by administration of methylene blue. In this process, methemoglobin is converted to hemoglobin by leukomethylene blue (LMB). During the reaction, leukomethylene blue is converted back into methylene blue (MB). Leukomethylene blue acts as a:
A. Base
B. Nucleophile
C. Oxidizing agent
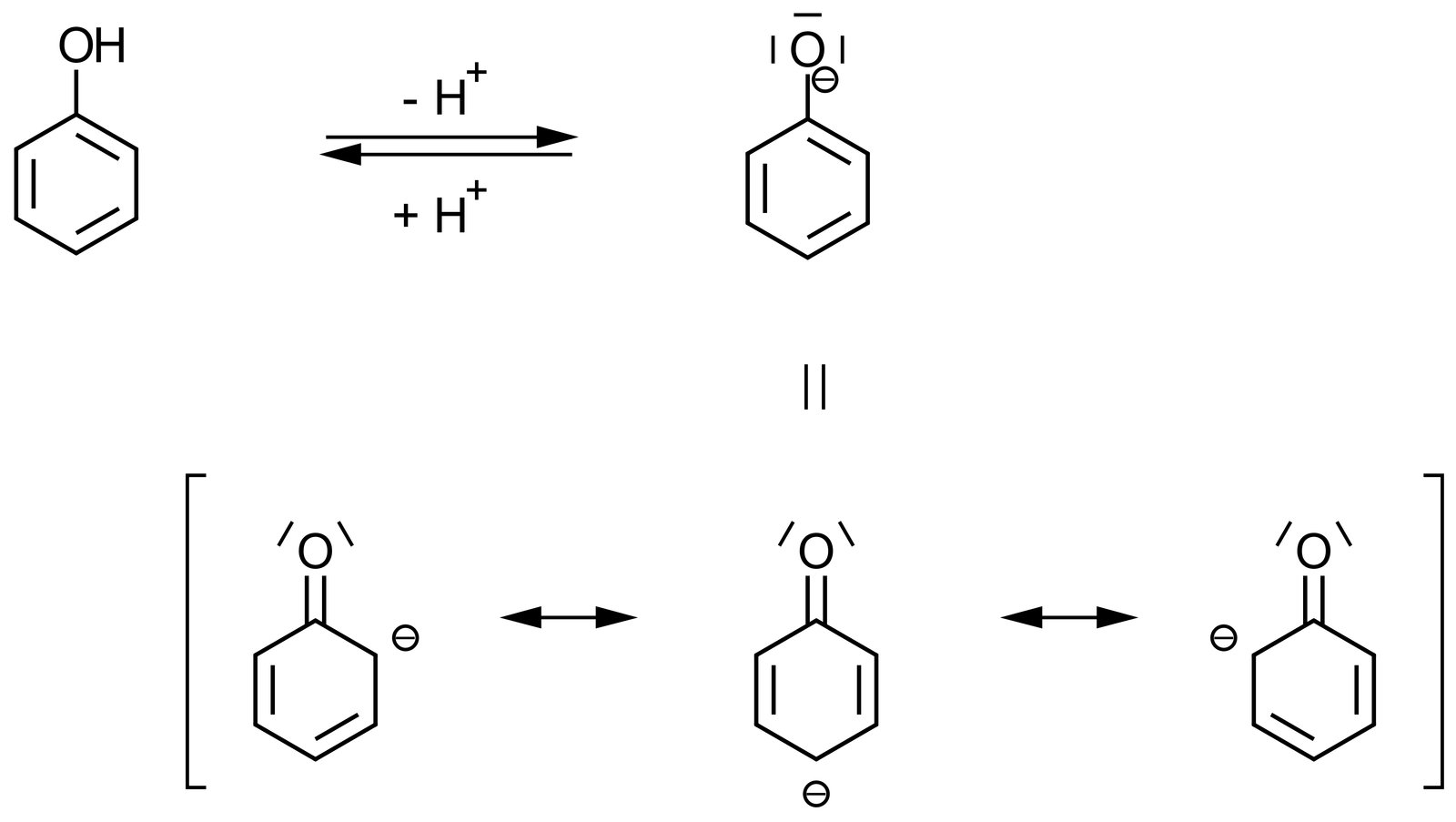
D. Reducing agent
E. Acid
D. Reducing agent
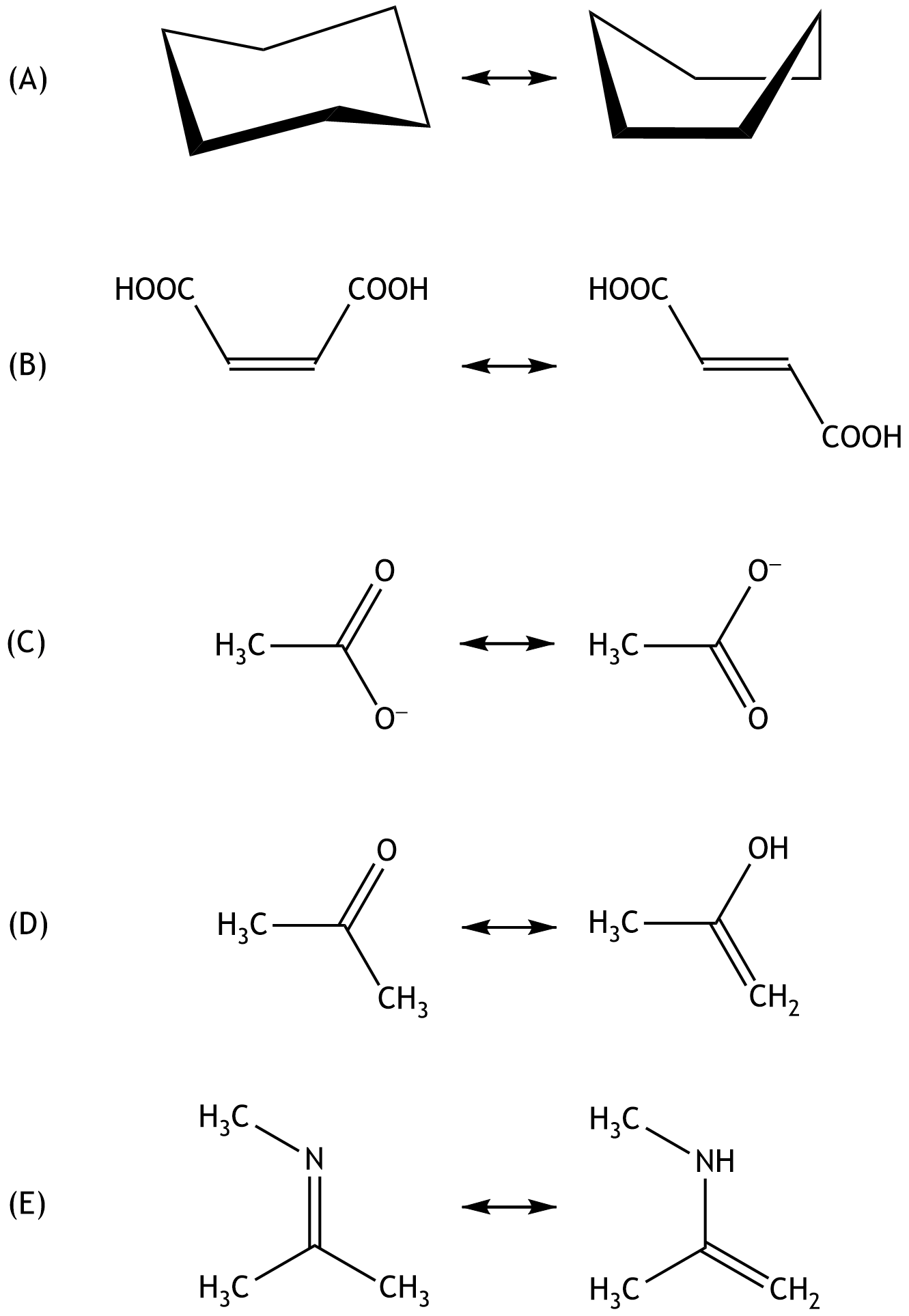
8. Structural formulas of chemical compounds provide information about how atoms in a molecule are connected to one another.
Which of the answer choices (A) to (E) represents resonance (mesomerism)?
A. (A)
B. (B)
C. (C)
D. (D)
E. (E)
C. (C)
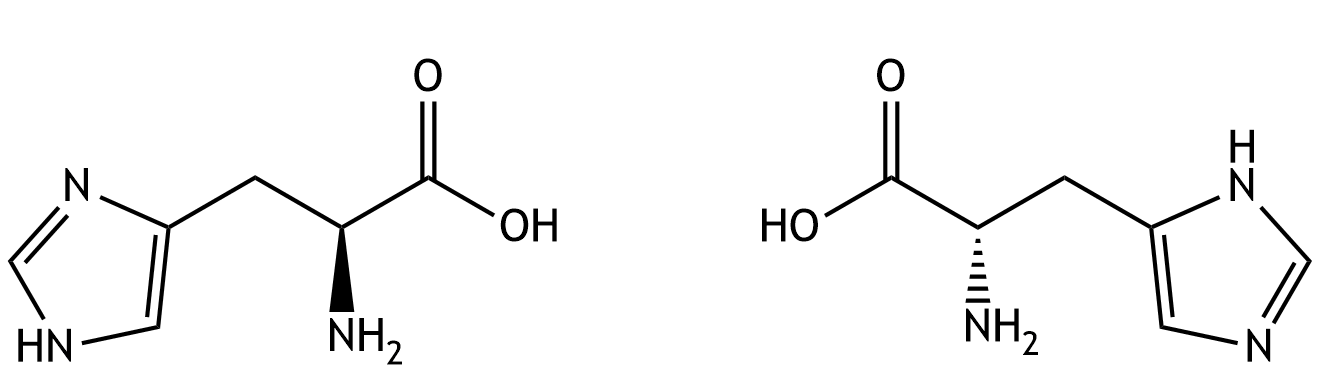
9. The amino acid histidine serves, among other things, as a direct precursor for the formation of histamine. In solution, histidine can in principle exist in two isomeric forms that can interconvert. What type of isomers are these?
A. Diastereomers
B. Enantiomers
C. Epimers
D. Conformers
E. Tautomers
E. Tautomers
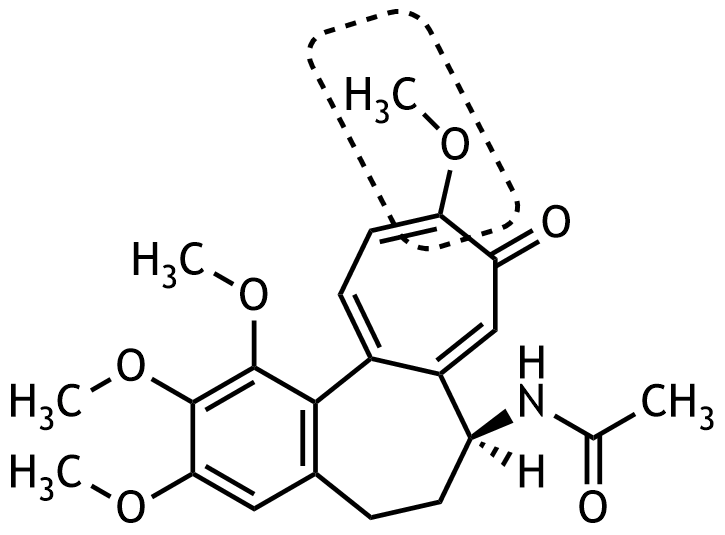
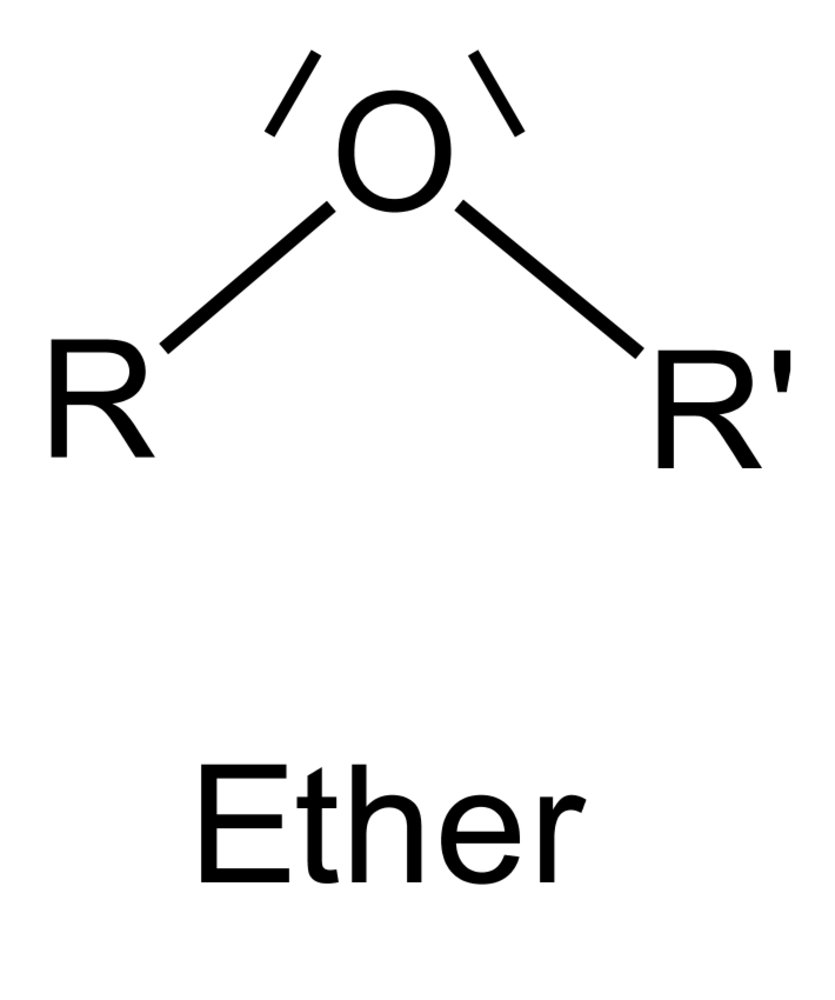
10. The alkaloid colchicine, a mitosis inhibitor, is found in the autumn crocus (Colchicum autumnale) and represents one of the treatment options for acute gout attacks. It has the following chemical structure:
Which class of compounds is shown in the shaded box?
A. Alcohol
B. Carboxylic acid anhydride
C. Ester
D. Ether
E. Hemiacetal
D. Ether
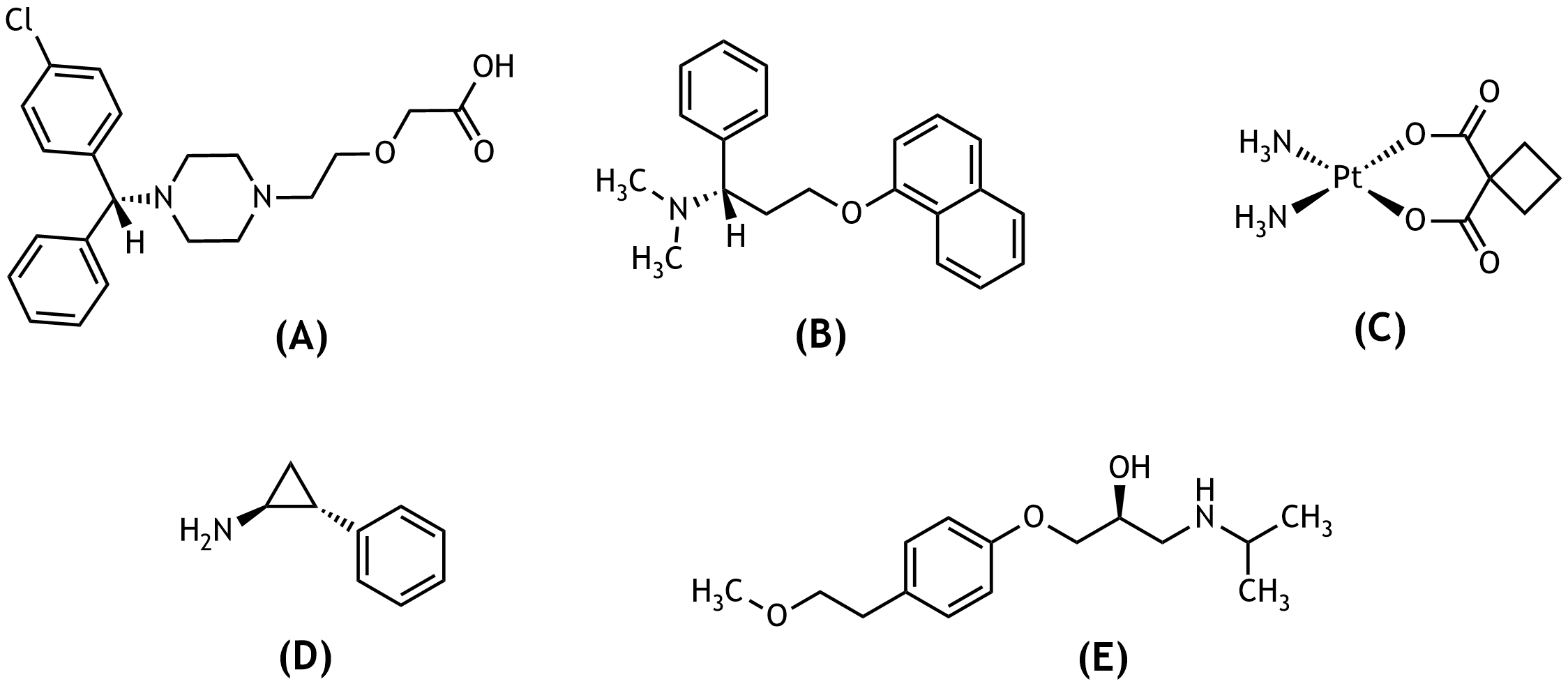
11. Amino groups are important functional groups in active pharmaceutical ingredients and can, among other effects, increase water solubility.
Which of the depicted active substances contains a secondary amino group?
A. (A)
B. (B)
C. (C)
D. (D)
E. (E)
E. (E)
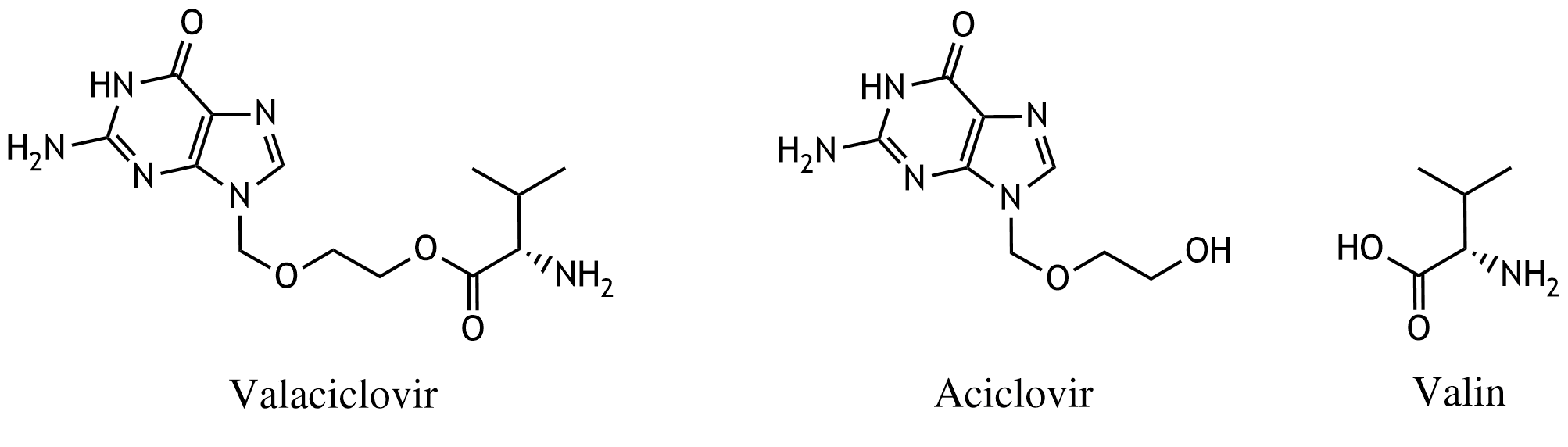
12. To increase the bioavailability of the antiviral drug acyclovir, which belongs to the group of nucleoside analogs, it is administered as the prodrug valacyclovir, a compound formed with the amino acid valine. In the human body, this compound is cleaved into acyclovir and valine. This reaction is a:
A. Elimination
B. Homolysis
C. Hydrolysis
D. Condensation
E. Esterification
C. Hydrolysis
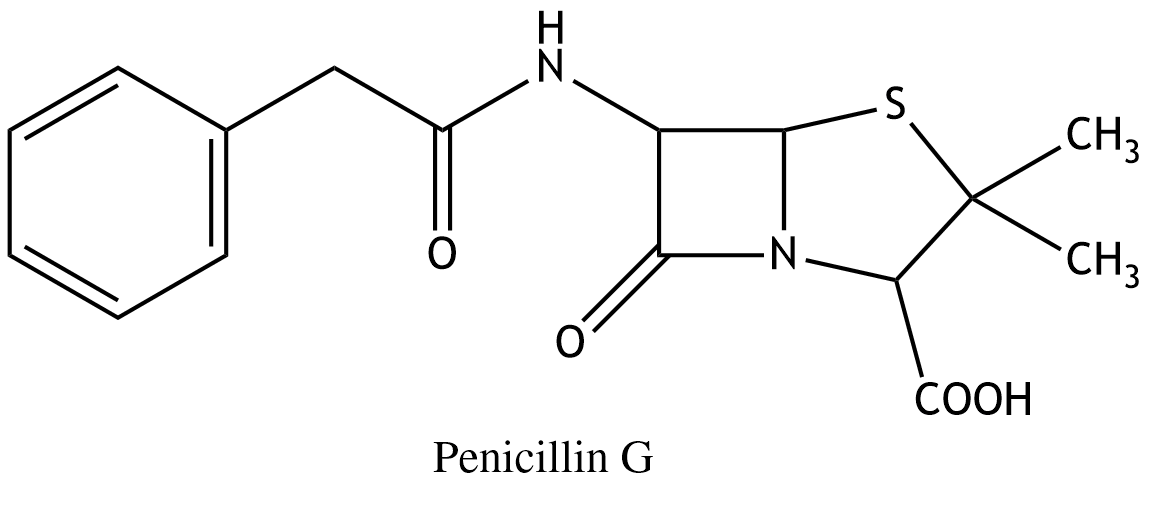
13. Penicillin G is used as an antibiotic against certain bacterial infections. The bacterial enzyme transpeptidase interacts with the antibiotic, and the four-membered ring structure (the β-lactam ring) plays an important role in this interaction. This four-membered ring is:
A. A cyclic carboxylic acid amide
B. A cyclic carboxylic acid anhydride
C. A cyclic ester
D. A cyclic ether
E. A cyclic thioester
A. A cyclic carboxylic acid amide

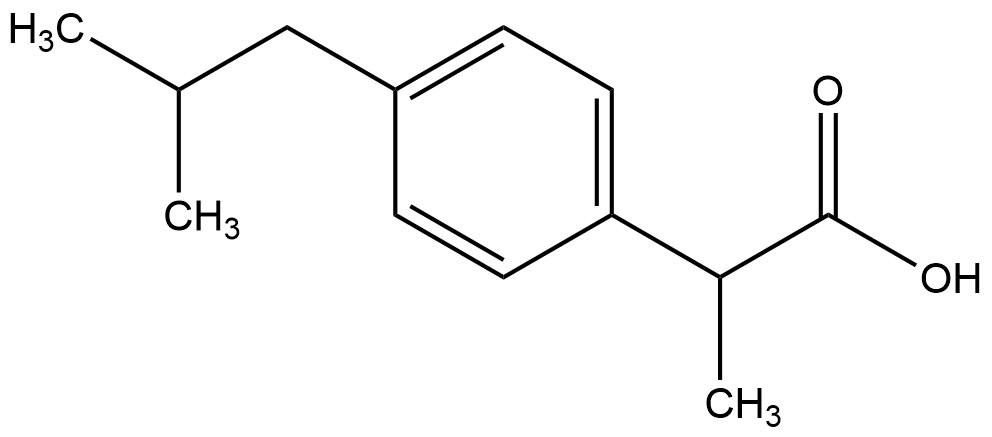
14. The following structural formula represents the non-selective cyclooxygenase inhibitor (COX inhibitor) ibuprofen, which is used to treat pain, inflammation, and fever. Ibuprofen occurs in two different isomeric forms.
With which nomenclature are these isomeric forms designated?
A. cis- and trans-
B. E- and Z-
C. erythro- and threo-
D. R- and S-
E. syn- and anti-
D. R- and S-
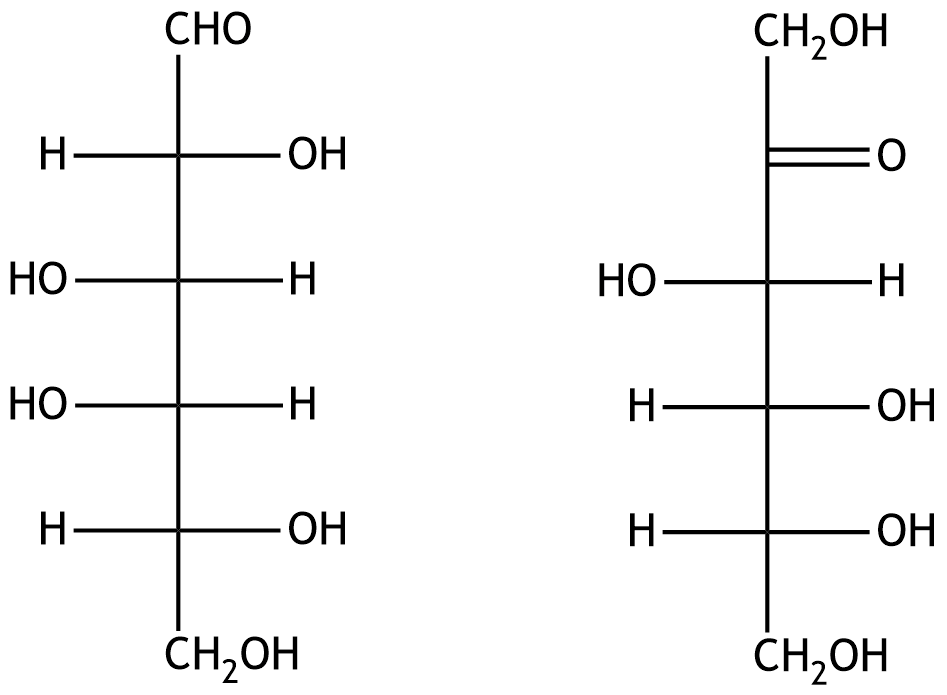
15. Monosaccharides fulfill important functions as energy carriers and as building blocks of cellular structures.
In what type of isomeric relationship do the two hexoses galactose and fructose (shown in the Fischer projection) stand to each other?
A. Anomers
B. Diastereomers
C. Enantiomers
D. Epimers
E. Constitutional isomers
E. Constitutional isomers
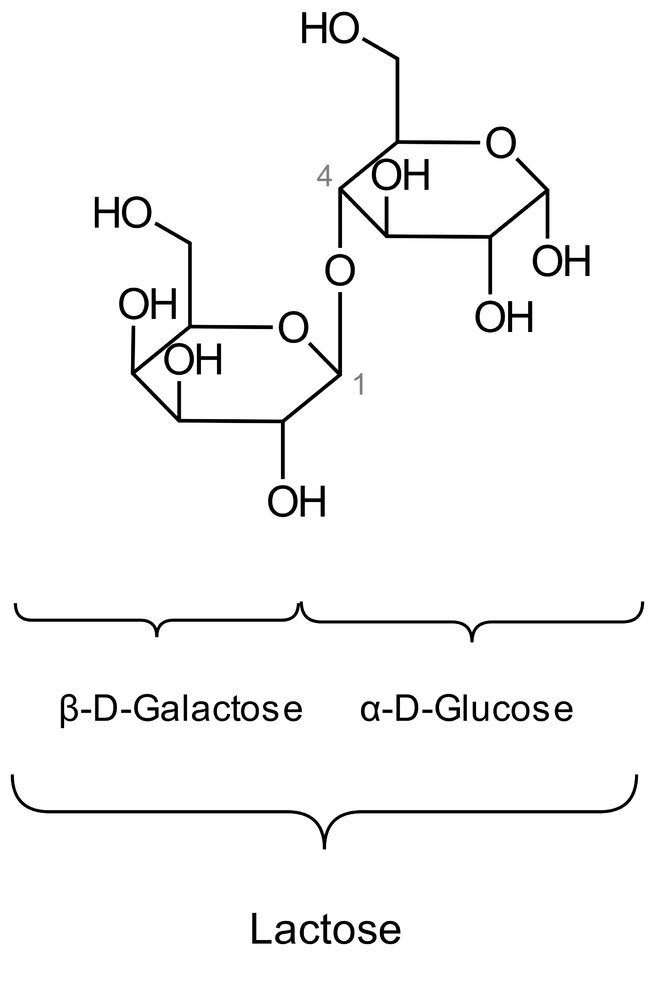
16. In lactose intolerance (milk sugar intolerance), the lactose ingested with food is not or only incompletely broken down. Symptoms include, among others, bloating and diarrhea. The cause is a lack or reduced activity of the digestive enzyme lactase.
What is produced, and in what amount, during the complete hydrolysis of 1 mol of lactose by lactase?
A. 1 mol fructose and 1 mol galactose
B. 1 mol glucose and 1 mol fructose
C. 1 mol glucose and 1 mol galactose
D. 2 mol galactose
E. 2 mol glucose
C. 1 mol glucose and 1 mol galactose
17. The ¹³C breath test is a noninvasive method used to determine bacterial urease activity in Helicobacter pylori infection. In this test, the patient orally ingests ¹³C-labeled urea, which is converted by urease into, among other products, ¹³CO₂. The ratio of ¹³C to ¹²C is then measured in the exhaled air.
How does the carbon isotope ¹³C differ from the carbon isotope ¹²C?
A. Contains one additional electron
B. Contains one additional neutron
C. Contains one additional proton
D. Has greater stability
E. Has a higher nuclear charge number
B. Contains one additional neutron
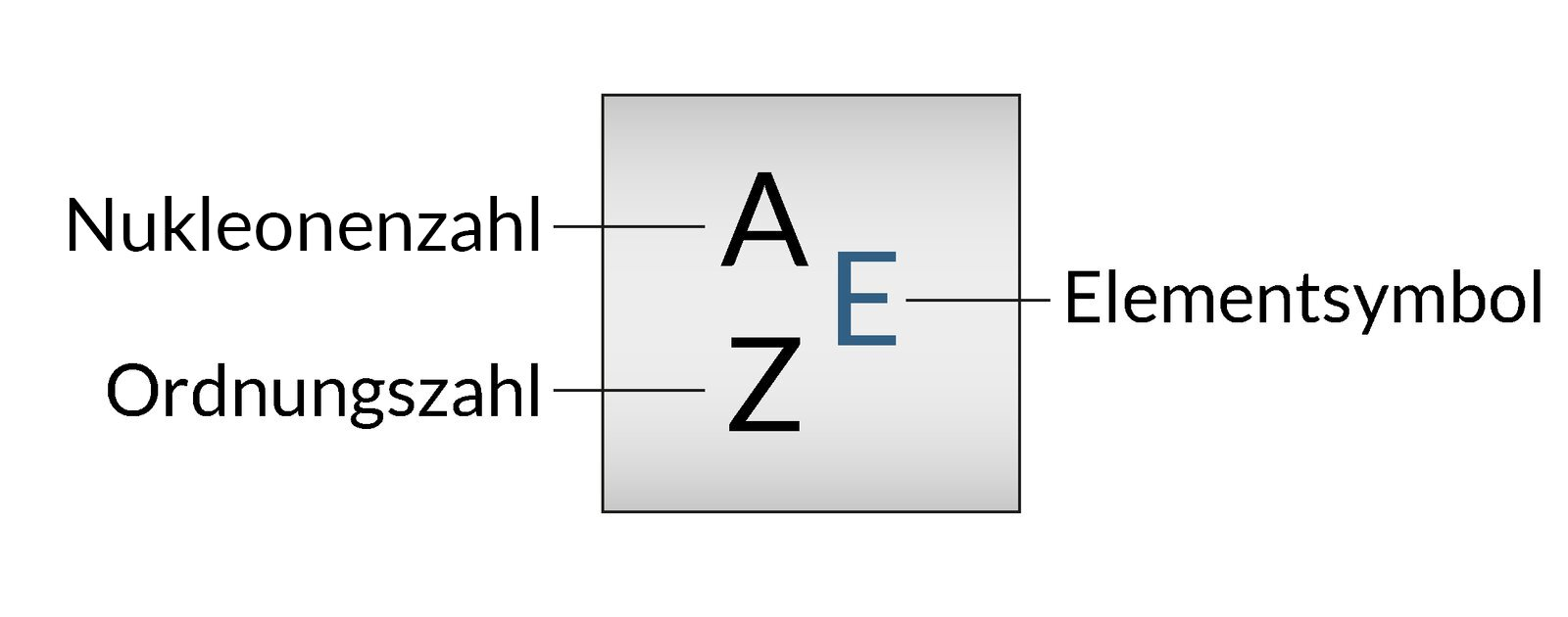
18. In metabolic intermediates, sulfur atoms can be present in functional groups similar to those containing oxygen atoms.
In what respect do the atoms of these two elements agree?
Sulfur and oxygen:
A. Contain the same total number of electrons
B. Contain the same total number of protons
C. Have the same electronegativity
D. Are in the same main group of the periodic table
E. Are in the same period of the periodic table
D. Are in the same main group of the periodic table
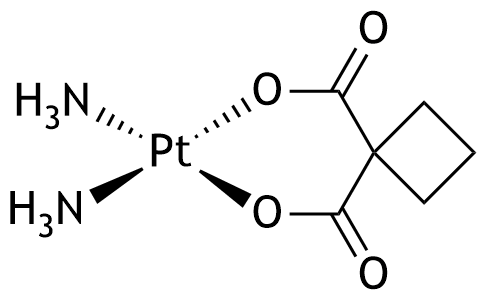
19. Carboplatin, with the following structural formula, is a cytostatic agent used in the treatment of malignant ovarian tumors. What is the coordination number of platinum in this compound?
A. 0
B. 1
C. 2
D. 3
E. 4
E. 4
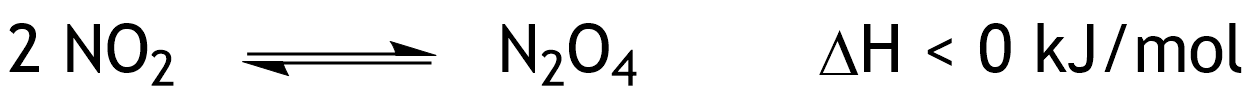
20. Nitrogen dioxide (NO₂) from car exhaust is associated with an increased incidence of cardiovascular diseases. In a closed system, the brownish-red gas nitrogen dioxide (NO₂) is in chemical equilibrium with its dimer, the colorless gas dinitrogen tetroxide (N₂O₄), as shown below.
How can this equilibrium be shifted toward N₂O₄?
A. By removing NO₂
B. By increasing the temperature
C. By increasing the pressure
D. By adding a catalyst
E. By adding N₂O₄
C. By increasing the pressure
21. The stomach contains hydrochloric acid, which serves, among other functions, to kill microorganisms ingested with food.
What is the pH value of an aqueous 0.01-molar hydrochloric acid (HCl) solution?
(Complete dissociation is assumed.)
A. 0.0
B. 1.0
C. 2.0
D. 3.0
E. 7.0
C. 2.0
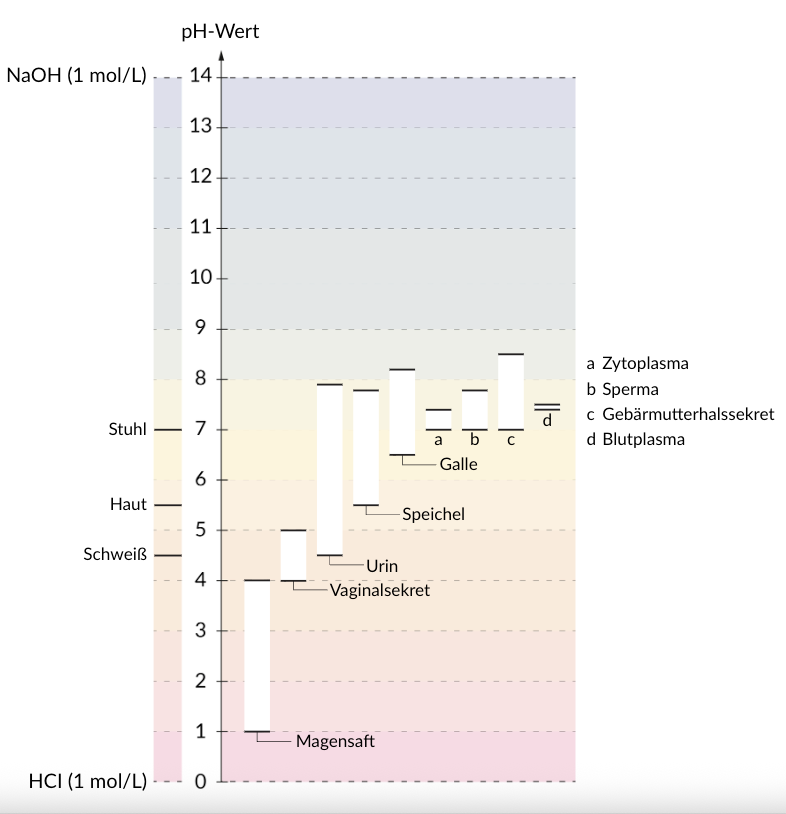
22. Oxidation numbers of carbon atoms can provide an indication of how much energy can be obtained from the oxidation of metabolites.
What is the oxidation number of the C₂ atom in acetic acid?
A. −3
B. −1
C. 0
D. +1
E. +3
A. −3
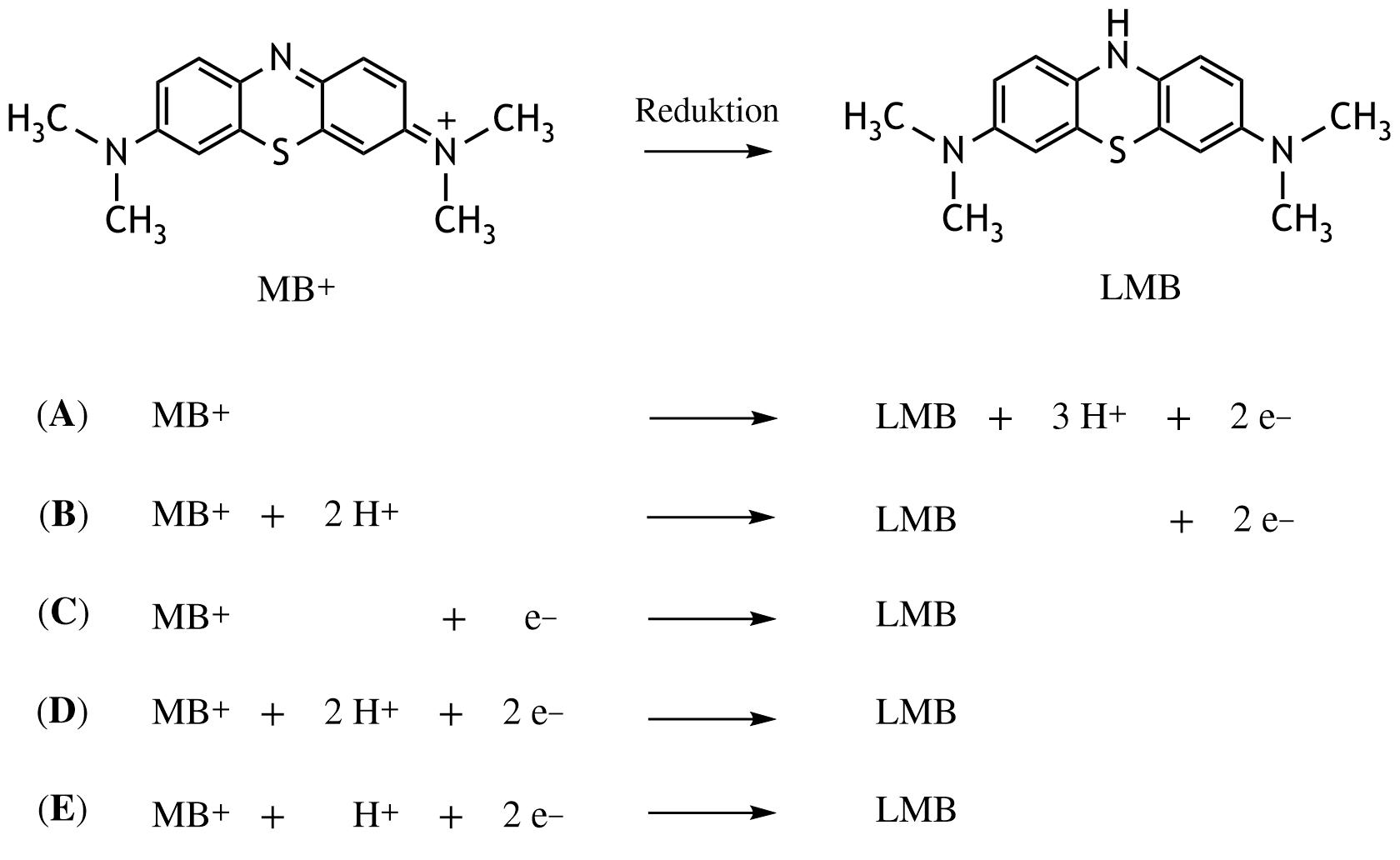
23. Methylene blue can be used, among other applications, as an antidote in cases of nitrite poisoning. In this process, methylene blue is enzymatically reduced to leukomethylene blue.
What is the correct reaction equation for the reduction of methylene blue (MB⁺) to leukomethylene blue (LMB)?
A. (A)
B. (B)
C. (C)
D. (D)
E. (E)
E. (E)
24. β-D-Glucopyranose is formed from open-chain D-glucose in water through an intramolecular nucleophilic addition.
From which oxygen atom of the open-chain D-glucose does this nucleophilic attack originate?
A. From the oxygen atom of the carbonyl group at C1
B. From the oxygen atom of the hydroxyl group at C2
C. From the oxygen atom of the hydroxyl group at C4
D. From the oxygen atom of the hydroxyl group at C5
E. From the oxygen atom of the hydroxyl group at C6
D. From the oxygen atom of the hydroxyl group at C5
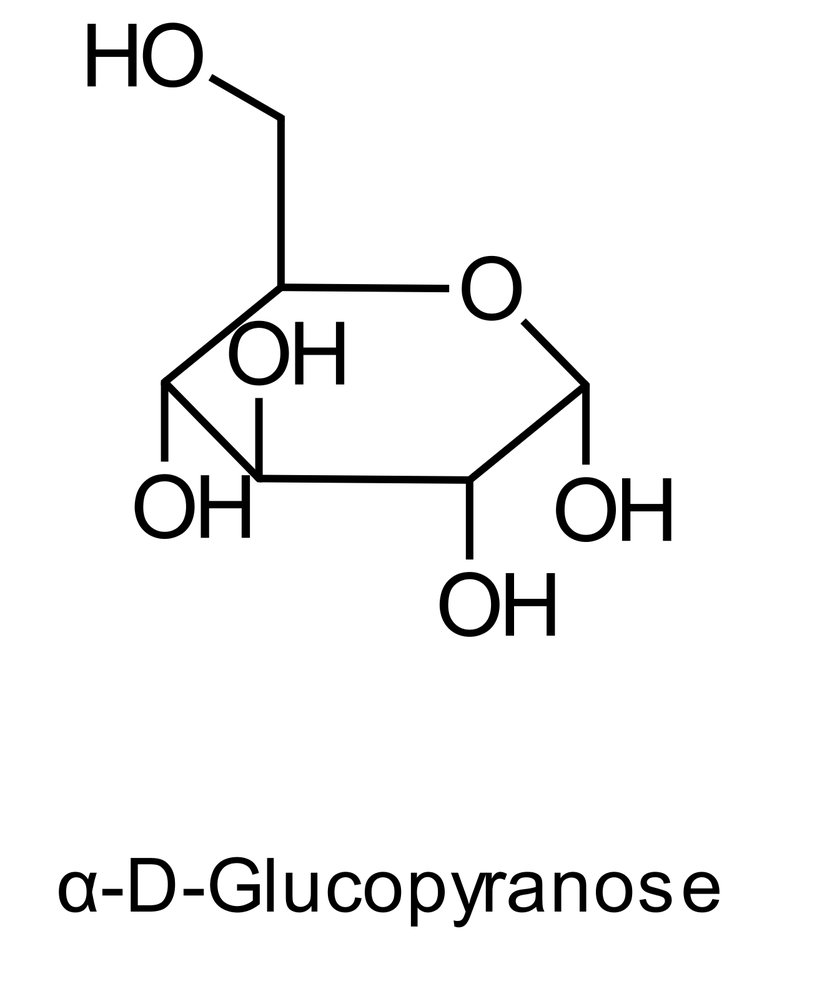
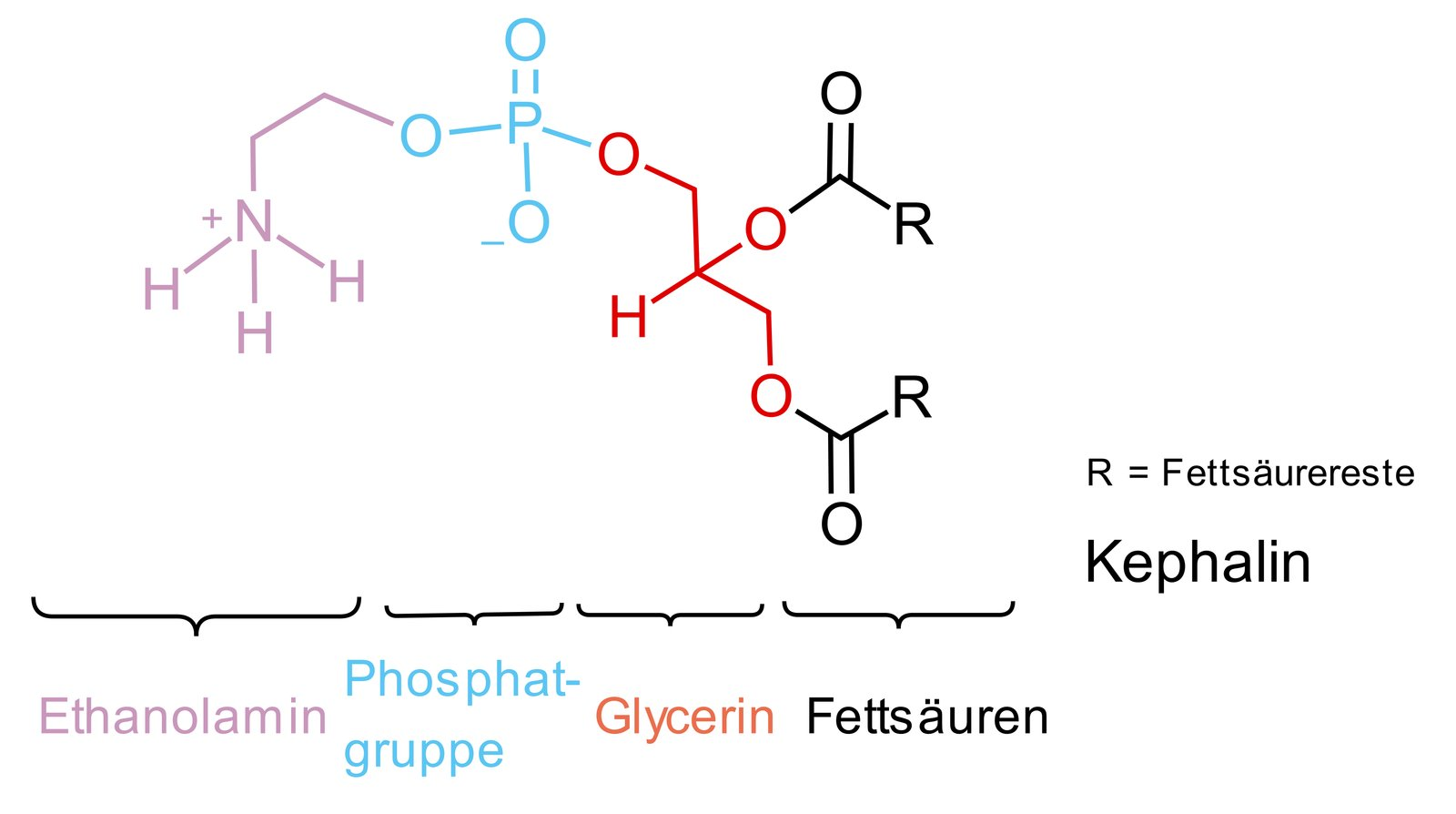
25. Nitrogen atoms present in biomolecules play a crucial role in their intra- and intermolecular interactions within the organism. The basicity of the various nitrogen atoms can differ.
Which of the following compounds contains a nitrogen atom that is predominantly protonated under physiological conditions (pH 7.4)?
A. Acetylcholine
B. Adenosine triphosphate
C. Urea
D. Phosphatidylethanolamine
E. Uracil triphosphate
D. Phosphatidylethanolamine
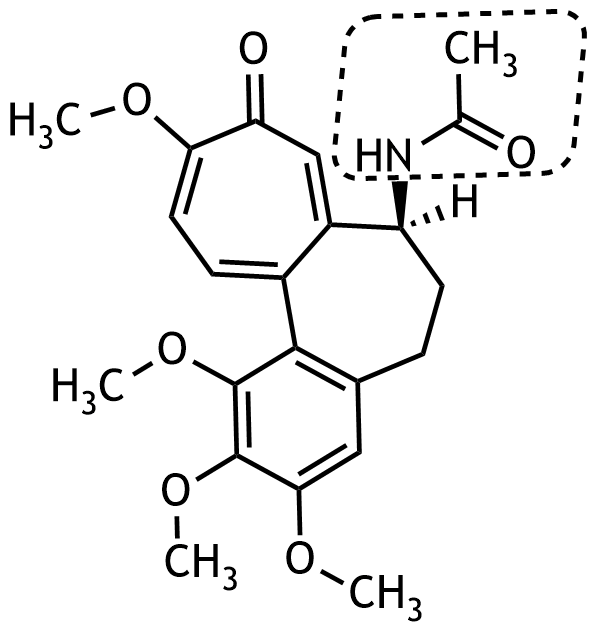
26. The alkaloid colchicine, a mitosis inhibitor, is found in the autumn crocus (Colchicum autumnale) and is one of the therapeutic options for acute gout attacks. It has the following chemical structure:
Which class of compounds is shown in the dashed box?
A. Amide
B. Imide
C. Imine
D. Primary amine
E. Tertiary amine
A. Amide
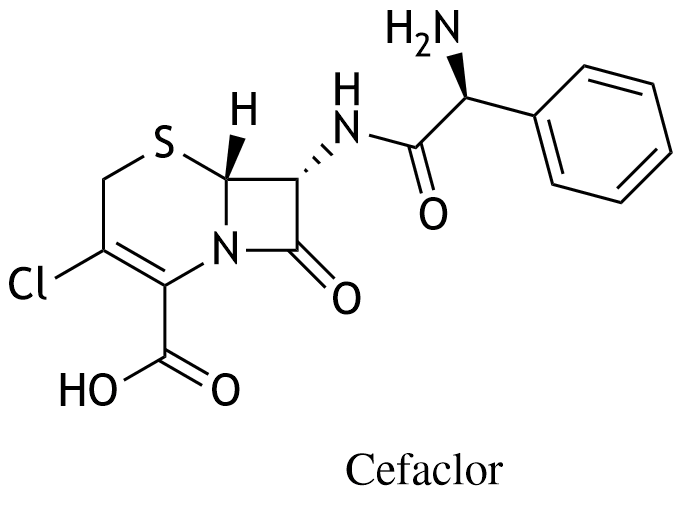
27. Cefaclor is an orally administered cephalosporin antibiotic.
Cefaclor is a:
A. Aldehyde
B. Ketone
C. Lactam
D. Lactate
E. Lactone
C. Lactam

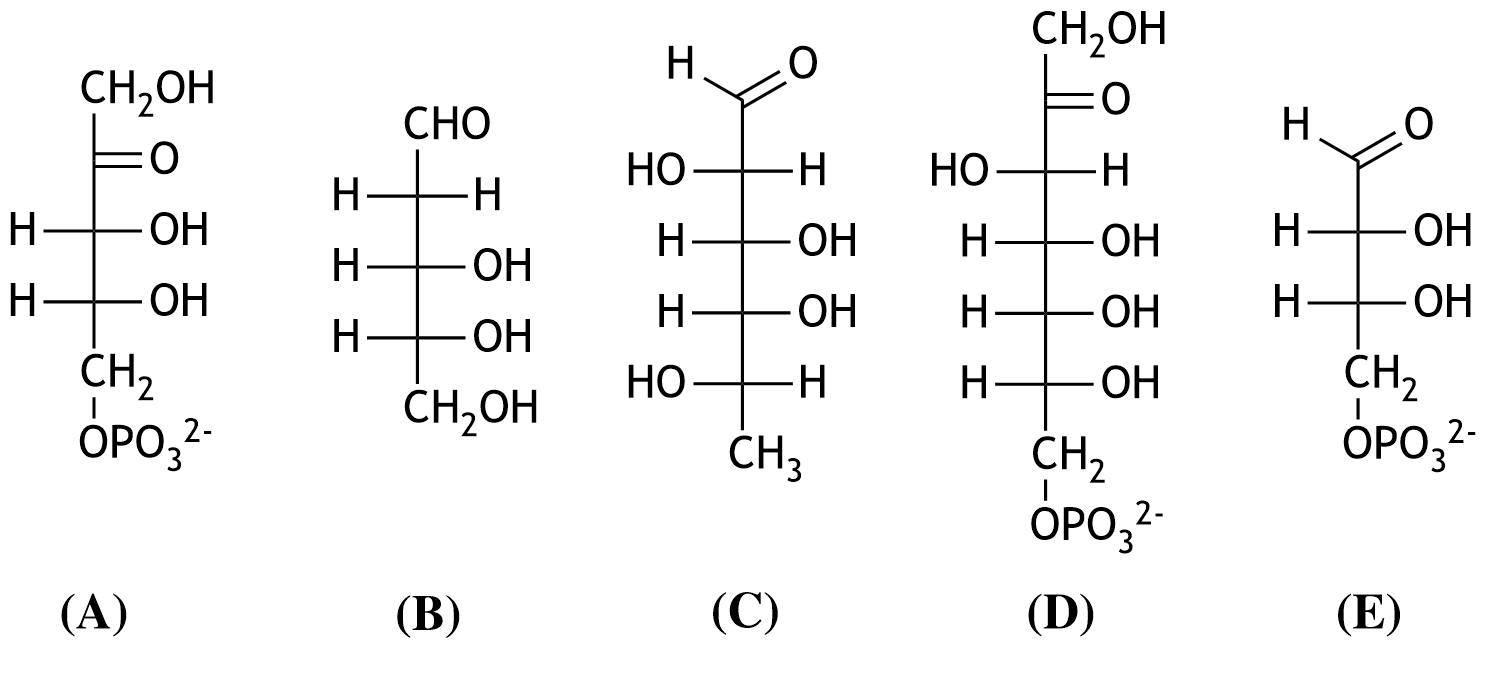
28. Stereoisomers of monosaccharides can occur in either D- or L-configuration.
Which of the shown monosaccharides (A–E) is in the L-configuration?
A. (A)
B. (B)
C. (C)
D. (D)
E. (E)
C. (C)
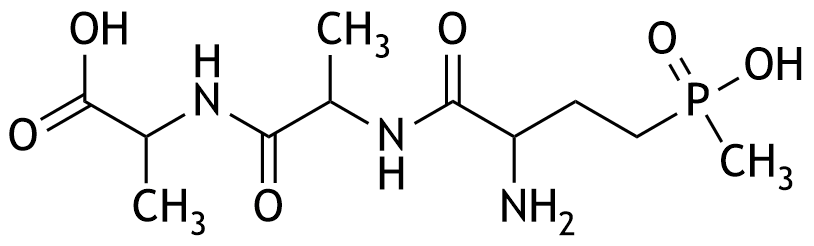
29. Poisoning with the natural herbicide bialaphos, as well as with herbicides that can be synthesized from bialaphos, can lead to severe complications such as respiratory arrest and metabolic acidosis. Bialaphos has the following chemical structure. How many chiral centers are present in this structural formula?
A. 1
B. 2
C. 3
D. 4
E. 5
C. 3
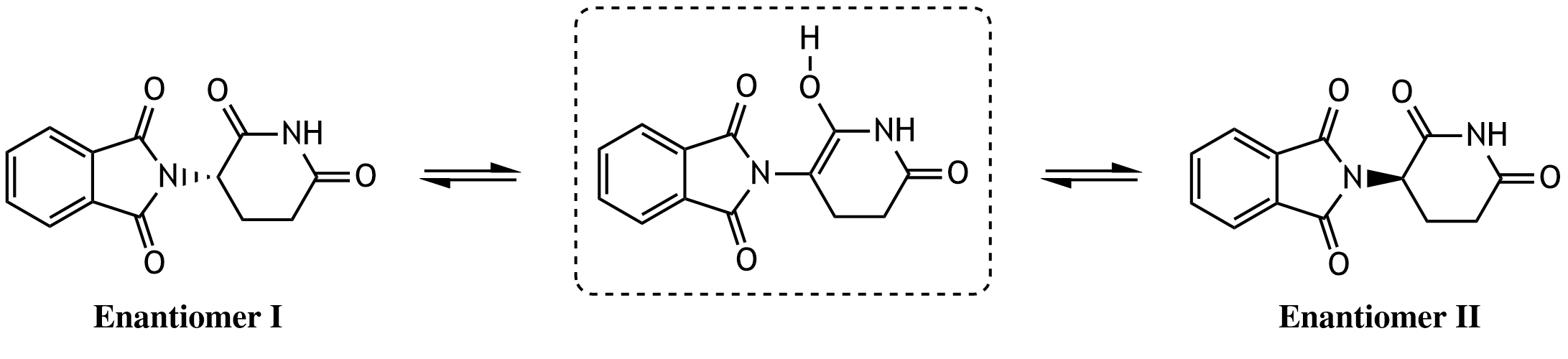
30. The drug thalidomide, originally marketed as a sleep and sedative medication, forms two enantiomers that interconvert in the human body. This process, known as racemization, occurs via a common achiral intermediate (shown in the dashed box). In what relationship does this intermediate stand to the two enantiomers I and II?
A. Diastereomer
B. Epimer
C. Conformer
D. Resonance structure (mesomeric form)
E. Tautomer
E. Tautomer
31. Isomers of drugs can exhibit fundamentally different modes of action and properties in the body.
By which method can a drug be distinguished from its enantiomer?
A. Agarose gel electrophoresis
B. Size-exclusion chromatography
C. Infrared spectroscopy
D. Mass spectrometry
E. Polarimetry
E. Polarimetry
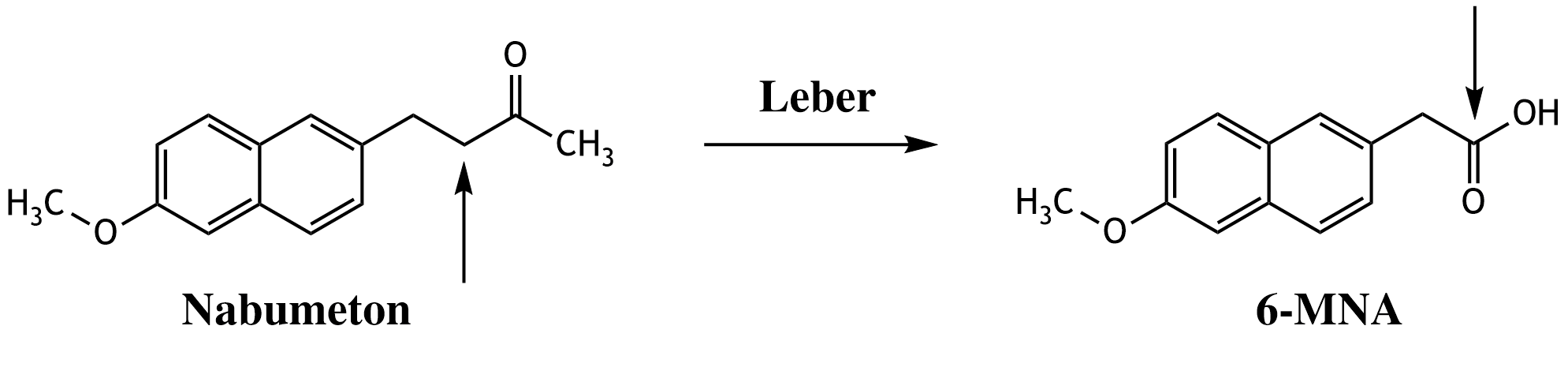
32. The cyclooxygenase inhibitor (COX inhibitor) nabumetone is metabolized in the liver to the active compound 6-methoxy-2-naphthylacetic acid (6-MNA).
Which type of chemical reaction has occurred at the carbon atom indicated by the arrow during this metabolism?
A. Elimination
B. Hydrolysis
C. Oxidation
D. Reduction
E. Esterification
C. Oxidation
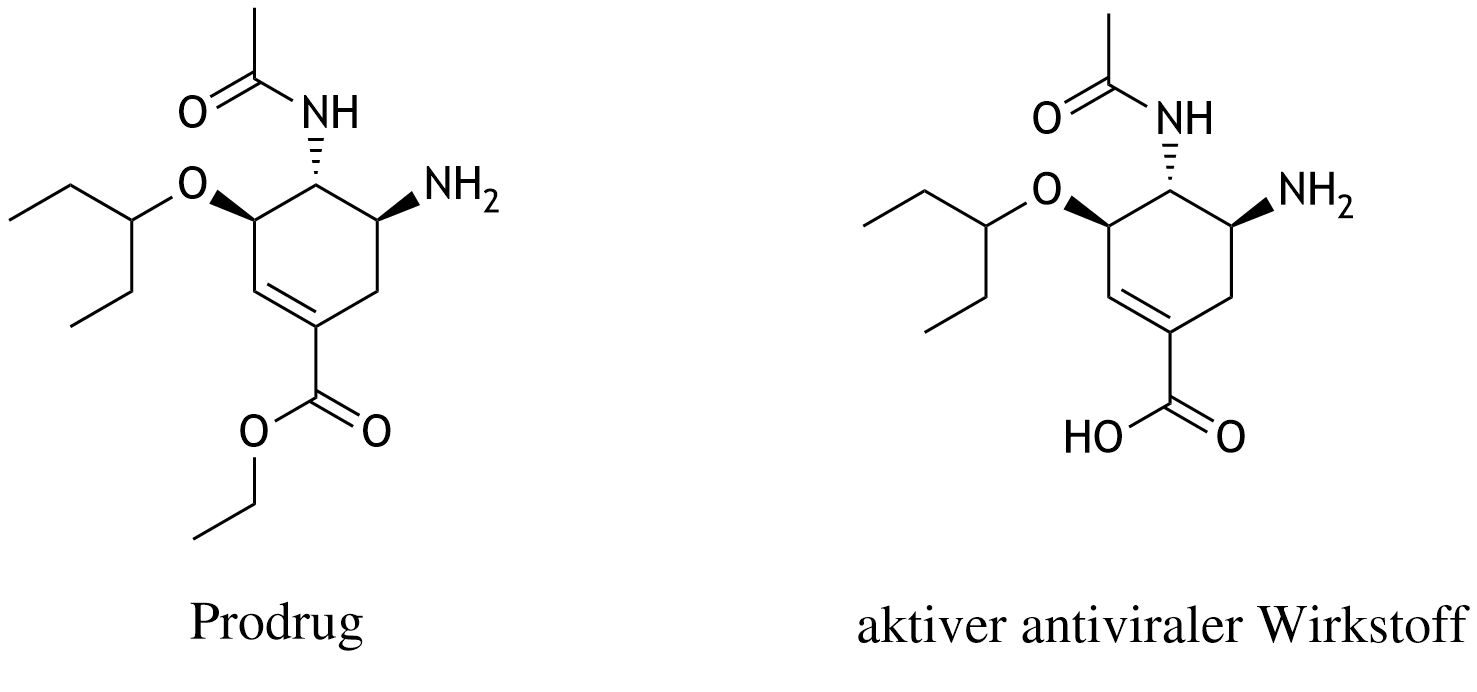
33. Oseltamivir is a drug from the group of neuraminidase inhibitors that can be used for antiviral therapy in influenza. Oseltamivir is a prodrug that, after oral administration, is enzymatically converted in the liver into the active antiviral compound. What type of reaction is involved in this conversion?
A. Dehydration
B. Elimination
C. Hydrolysis
D. Condensation
E. Esterification
C. Hydrolysis
34. Sucrase-isomaltase catalyzes the hydrolytic cleavage of various oligo-, tri-, and disaccharides. This reaction step is essential for the digestion of carbohydrates. The enzyme is found in humans in the small intestine and, to a lesser extent, in the large intestine on the surface of the microvilli.
What is produced, and in what amount, during the complete hydrolysis of 1 mol of isomaltose by sucrase-isomaltase?
A. 1 mol fructose and 1 mol galactose
B. 1 mol glucose and 1 mol galactose
C. 1 mol glucose and 1 mol mannose
D. 2 mol glucose
E. 2 mol mannose
D. 2 mol glucose
35. An antacid used to neutralize excess stomach acid contains, per dose, 7 mmol of magnesium hydroxide (Mg(OH)₂) and 4 mmol of aluminum hydroxide (Al(OH)₃).
What amount of stomach acid (= hydrochloric acid, HCl) can therefore be neutralized per dose at maximum?
A. 9 mmol
B. 11 mmol
C. 16 mmol
D. 20 mmol
E. 26 mmol
E. 26 mmol
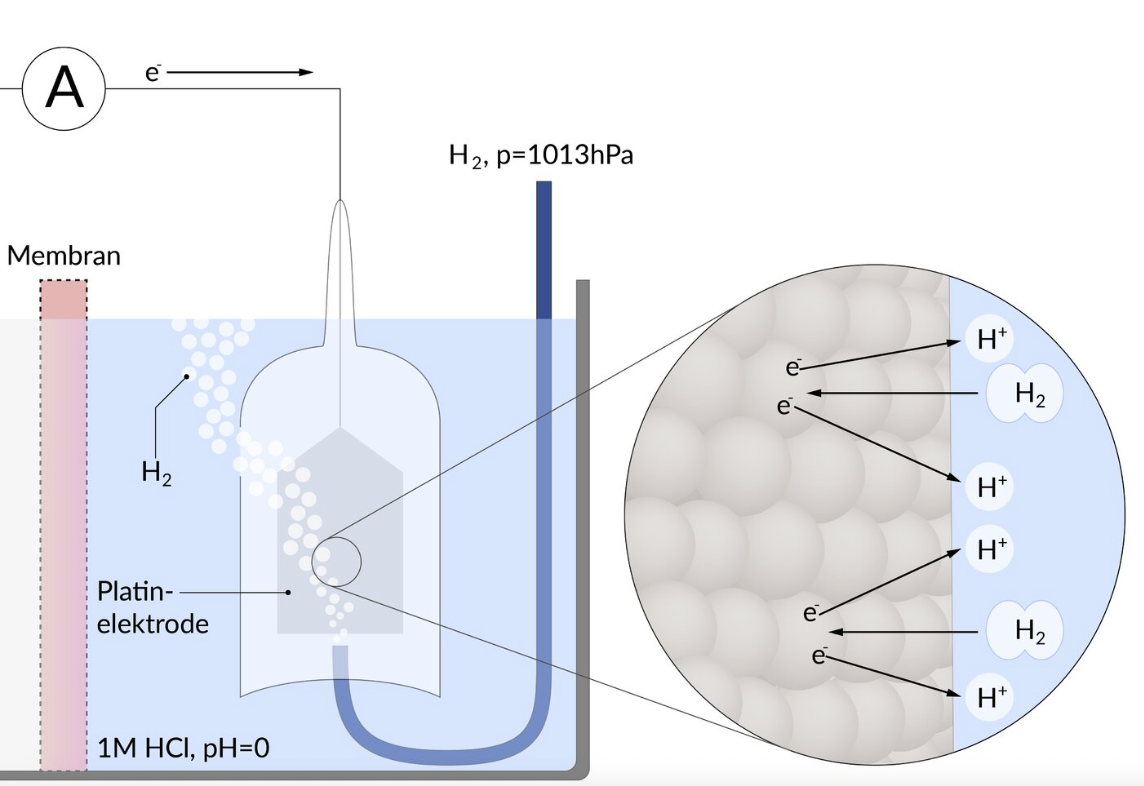
36. The direction of electron flow in redox reactions is determined by the redox potentials (E⁰) of the participating redox pairs. E⁰ is the potential measured under standard conditions relative to a specific reference electrode.
Which redox pair is characteristic of this reference electrode?
A. Au³⁺/Au
B. Cu²⁺/Cu
C. 2 H⁺/H₂
D. Pt²⁺/Pt
E. Zn²⁺/Zn
C. 2 H⁺/H₂
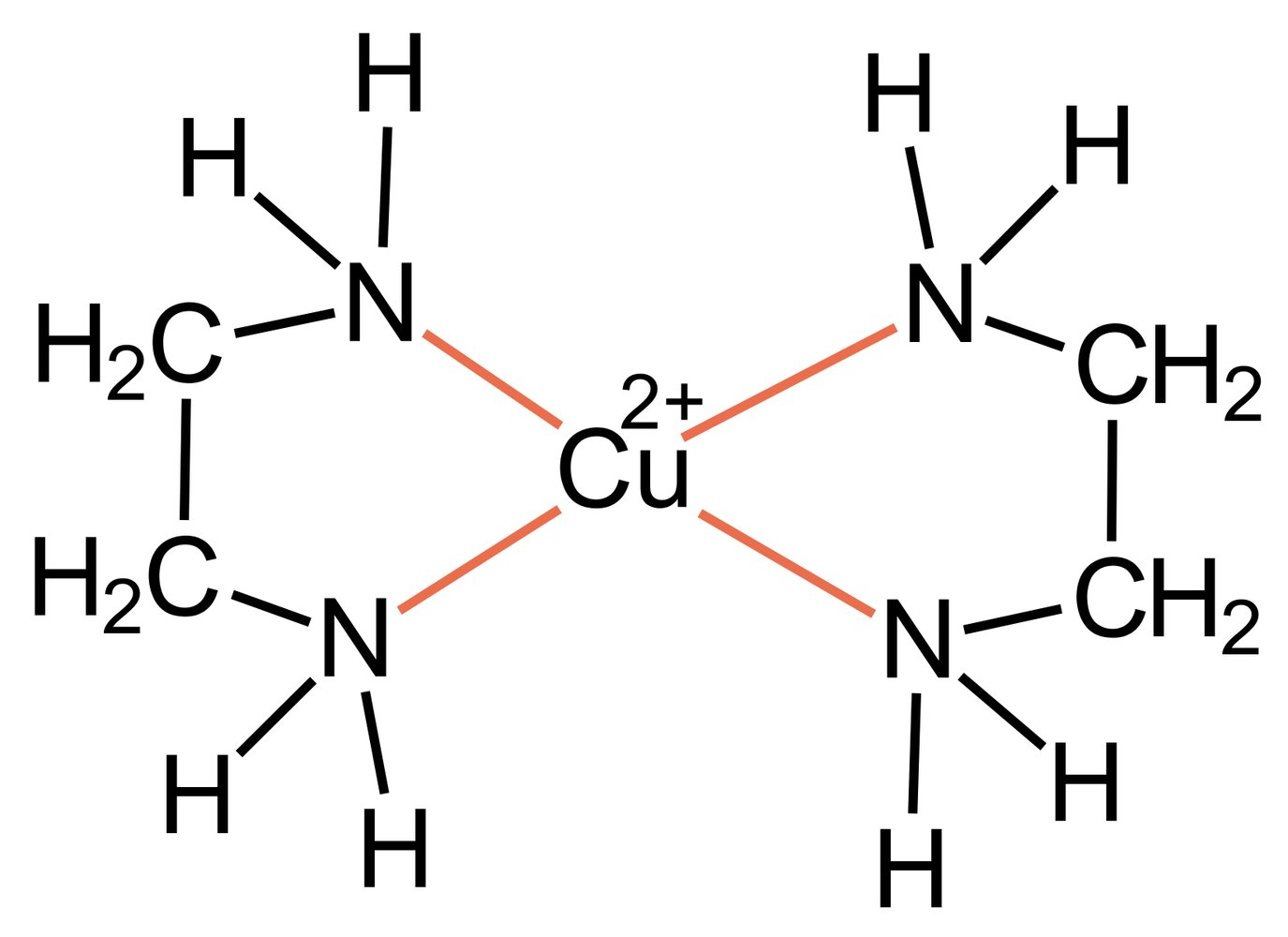
37. Wilson’s disease is a metabolic disorder in which the excretion of Cu²⁺ ions is impaired. As a result, Cu²⁺ ions accumulate in various tissues, which can manifest clinically as chronic hepatitis and extrapyramidal motor disturbances. For therapy, D-penicillamine is used as a chelating ligand.
What is the most likely basis of action of the chelating ligand used?
A. The Cu²⁺ ion forms a complex with the chelating ligand that has a low dissociation constant.
B. The Cu²⁺ ion donates its free electron pairs to the chelating ligand, forming a stable complex.
C. The chelating ligand binds to the Cu²⁺ ion exclusively via a hydrogen bond.
D. The chelating ligand reacts with the Cu²⁺ ion in a redox reaction, thereby rendering it harmless.
E. The chelating ligand acts as an efficient adsorbent for the Cu²⁺ ion.
A. The Cu²⁺ ion forms a complex with the chelating ligand that has a low dissociation constant.
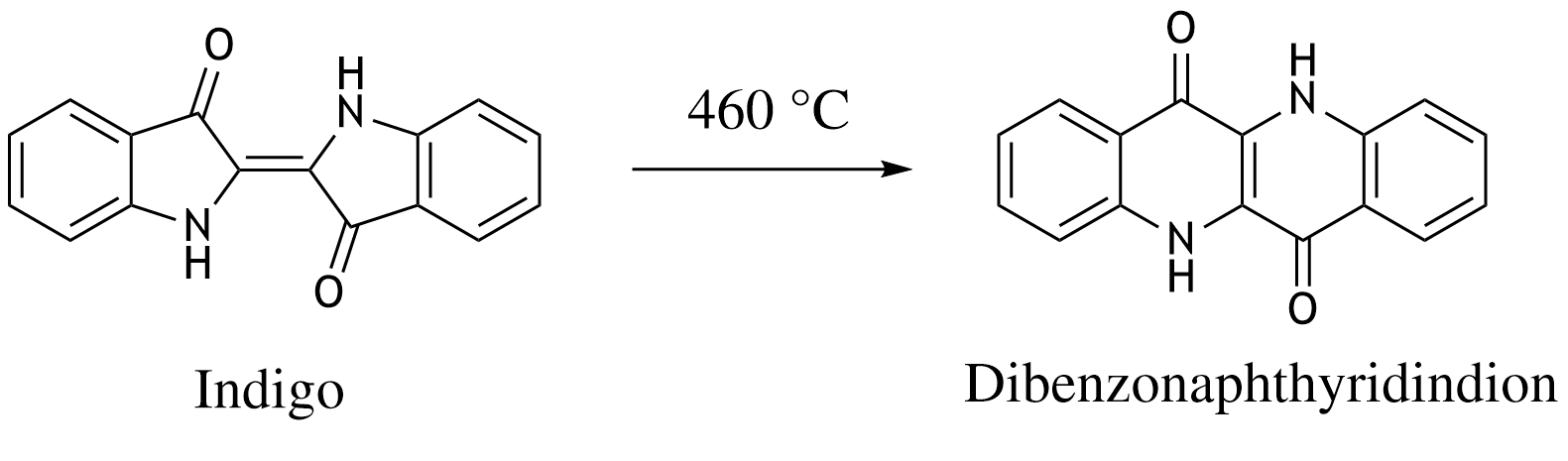
38. The blue dye indigo is converted to dibenzonaphthyridinedione upon heating.
What type of chemical process does this conversion represent?
A. Dehydration
B. Dehydrogenation
C. Hydrolysis
D. Racemization
E. Rearrangement
E. Rearrangement
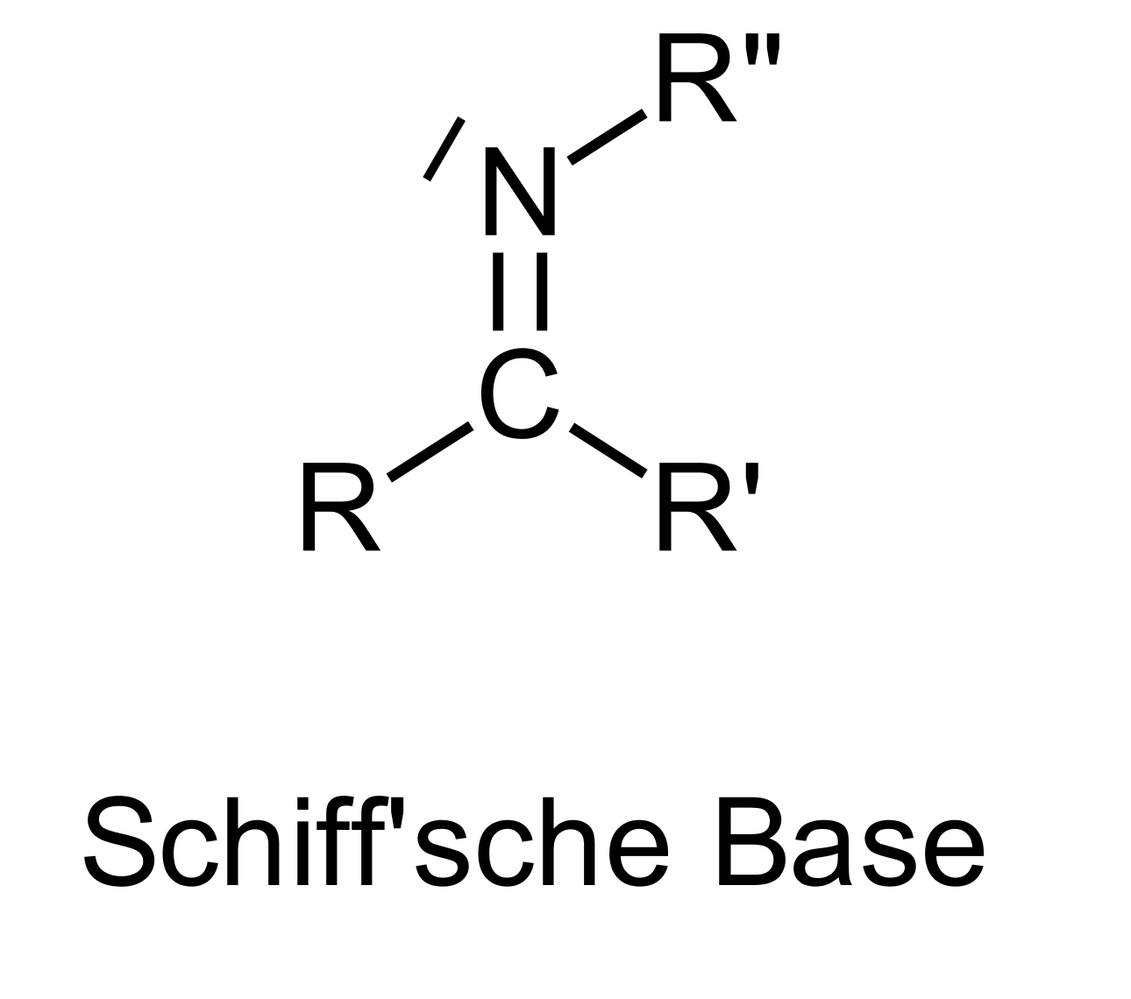
39. In rhodopsin, the chromophore is covalently bound to opsin via an imine linkage.
Which of the following functional groups must the chromophore possess in order to react with a lysine side chain in opsin to form this covalent bond?
A. Aldehyde
B. Alcohol
C. Carboxylic acid anhydride
D. Ester
E. Lactone
A. Aldehyde
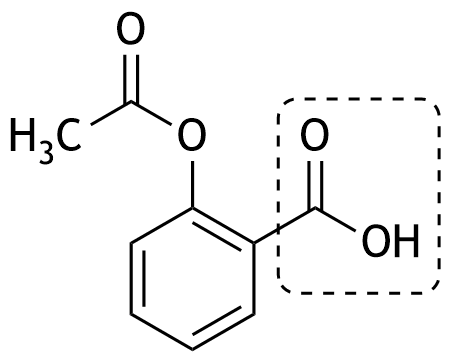
40. The structural formula of acetylsalicylic acid is shown, which is used clinically, among other purposes, for the treatment of pain and fever.
Which functional group is shown in the dashed box?
A. Amino group
B. Carbonyl group of an aldehyde
C. Carbonyl group of a ketone
D. Carboxyl group
E. Methyl group
D. Carboxyl group
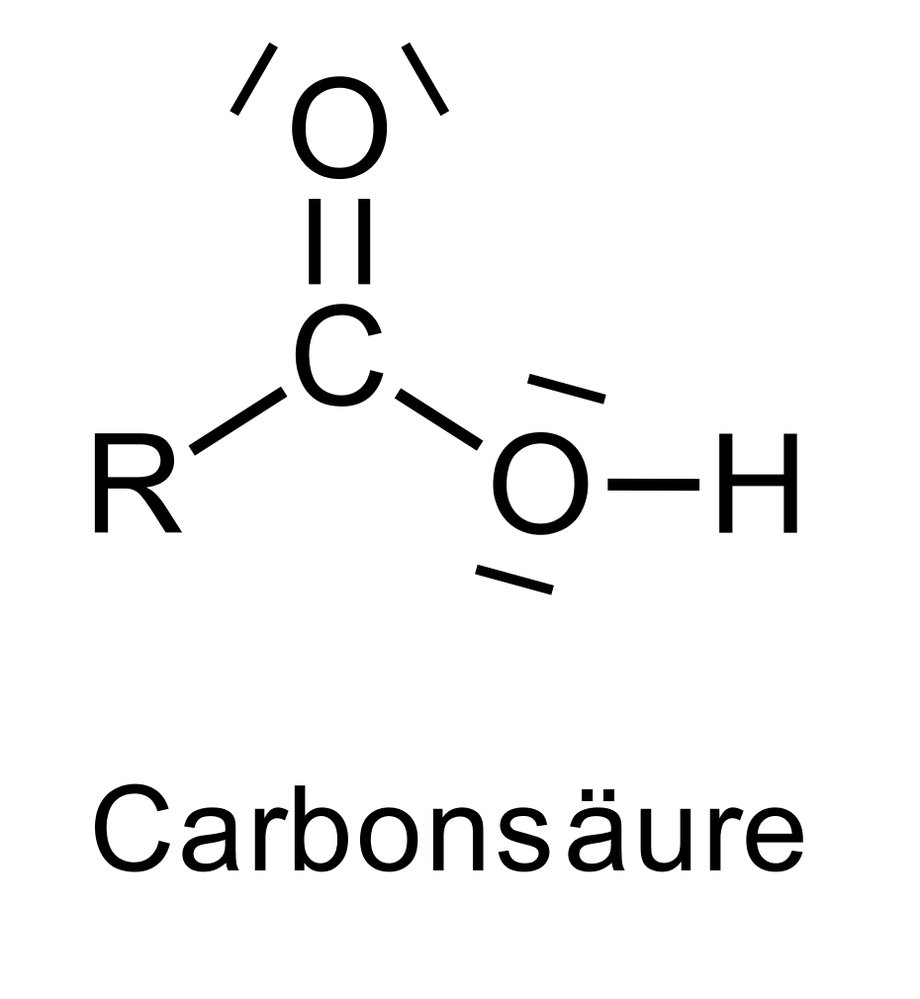
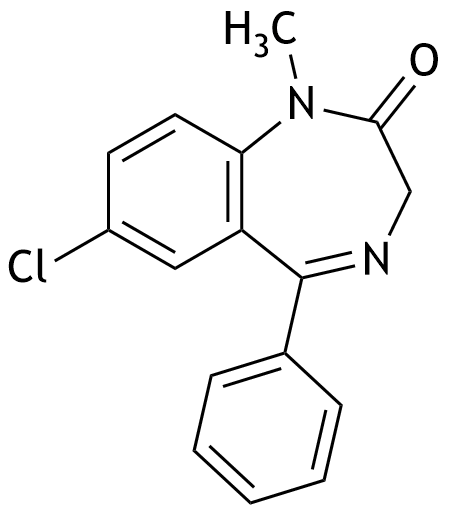
41. The psychoactive drug diazepam, belonging to the group of benzodiazepines, has the following chemical structure: It is a:
A. Aldehyde
B. Amide
C. Amine
D. Enamine
E. Ketone
B. Amide

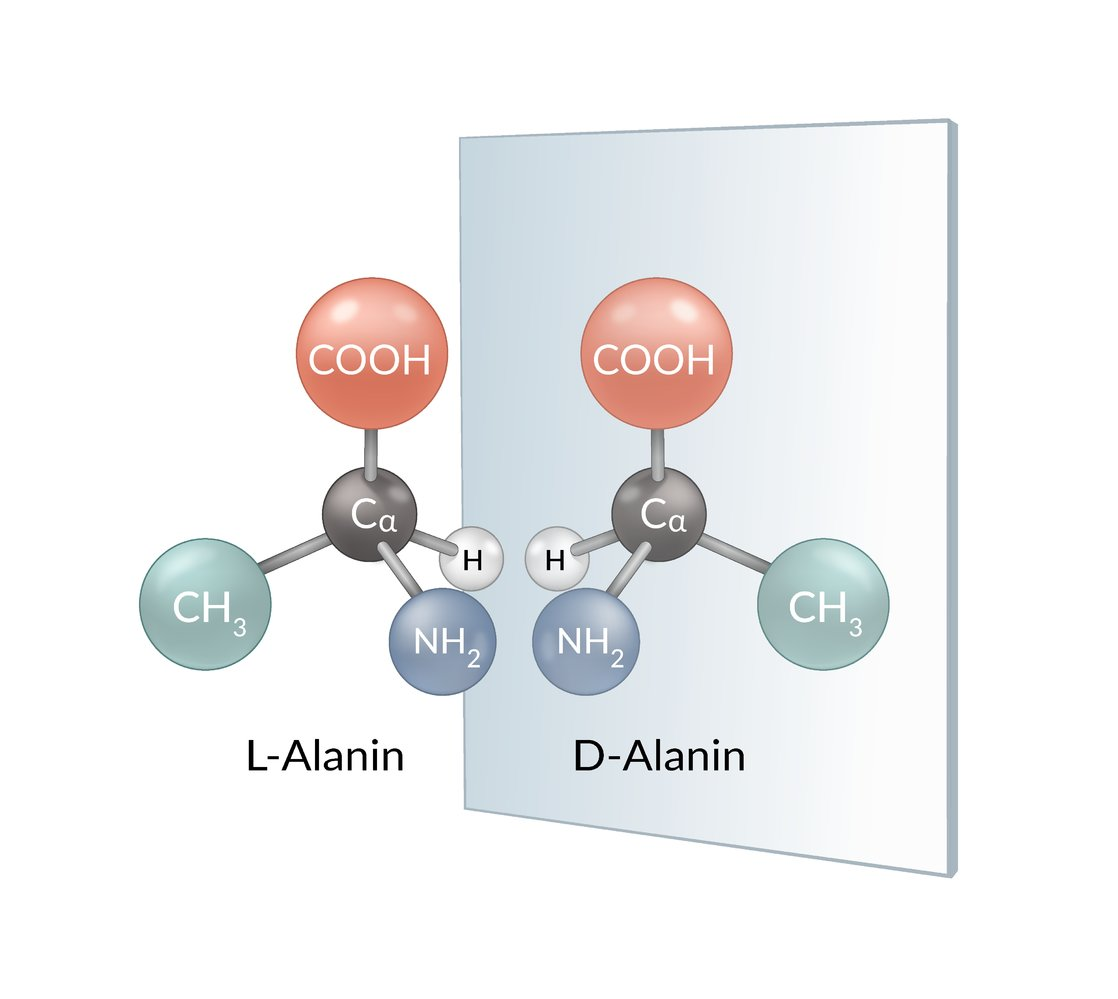
42. Many bacteria produce D-alanine, which is then incorporated into the peptidoglycan of their cell walls.
What type of isomerism exists between D-alanine and L-alanine?
They are:
A. Diastereomers
B. Enantiomers
C. Conformers
D. Constitutional isomers
E. Tautomers
B. Enantiomers
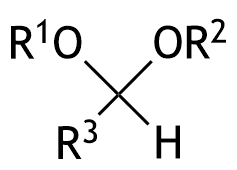
43. The following functional group with the organic residues R₁–R₃ is a central component of carbohydrate chains. What is this functional group called?
A. Acetal
B. Enamine
C. Hemiacetal
D. Hydrate
E. Imine
A. Acetal
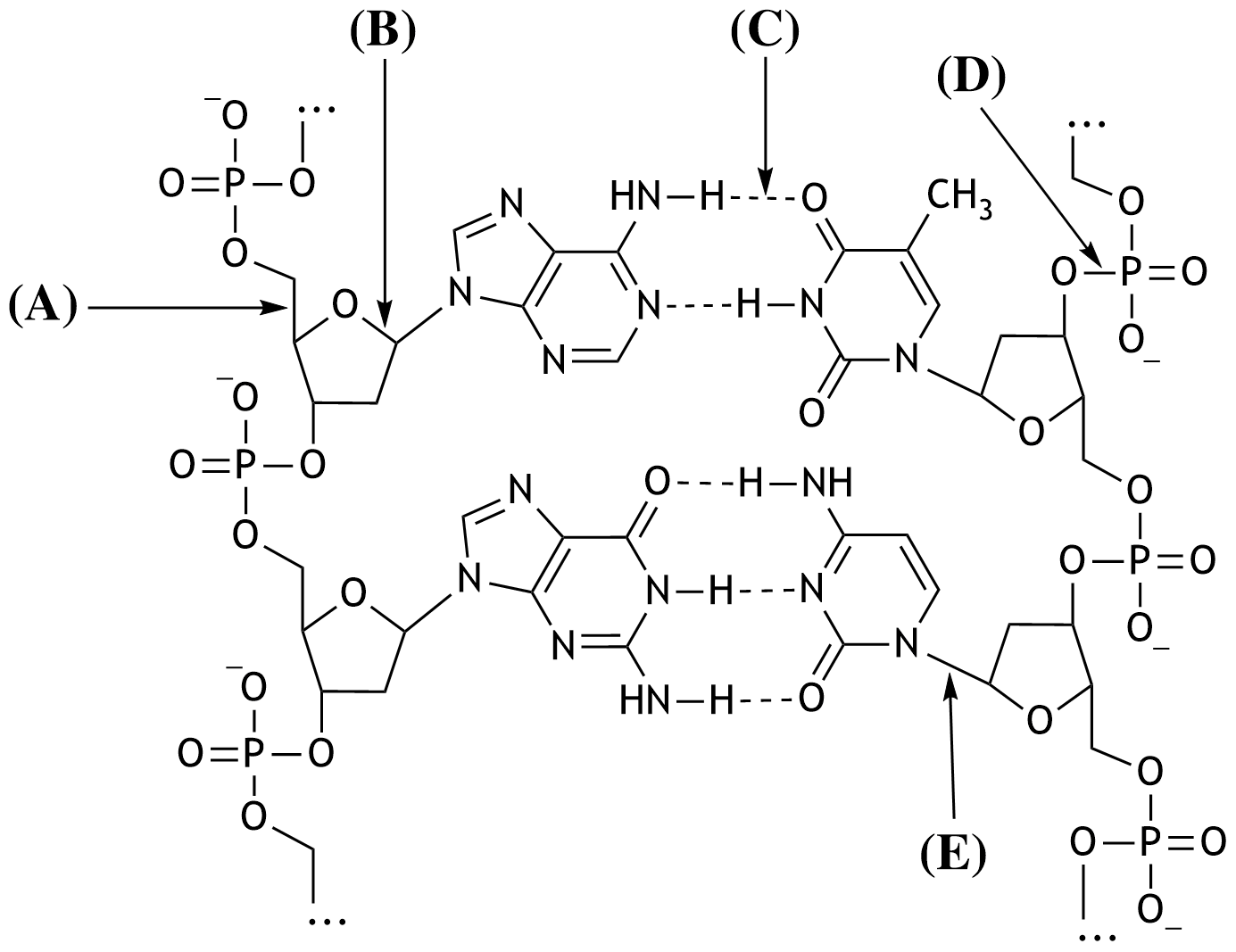
44. The building blocks of deoxyribonucleic acid (DNA) are linked together through the formation of ester bonds. Shown below is a section of the structural formula of a double-stranded DNA molecule.
Which of the arrows (A–E) points to an ester bond?
A. (A)
B. (B)
C. (C)
D. (D)
E. (E)
D. (D)
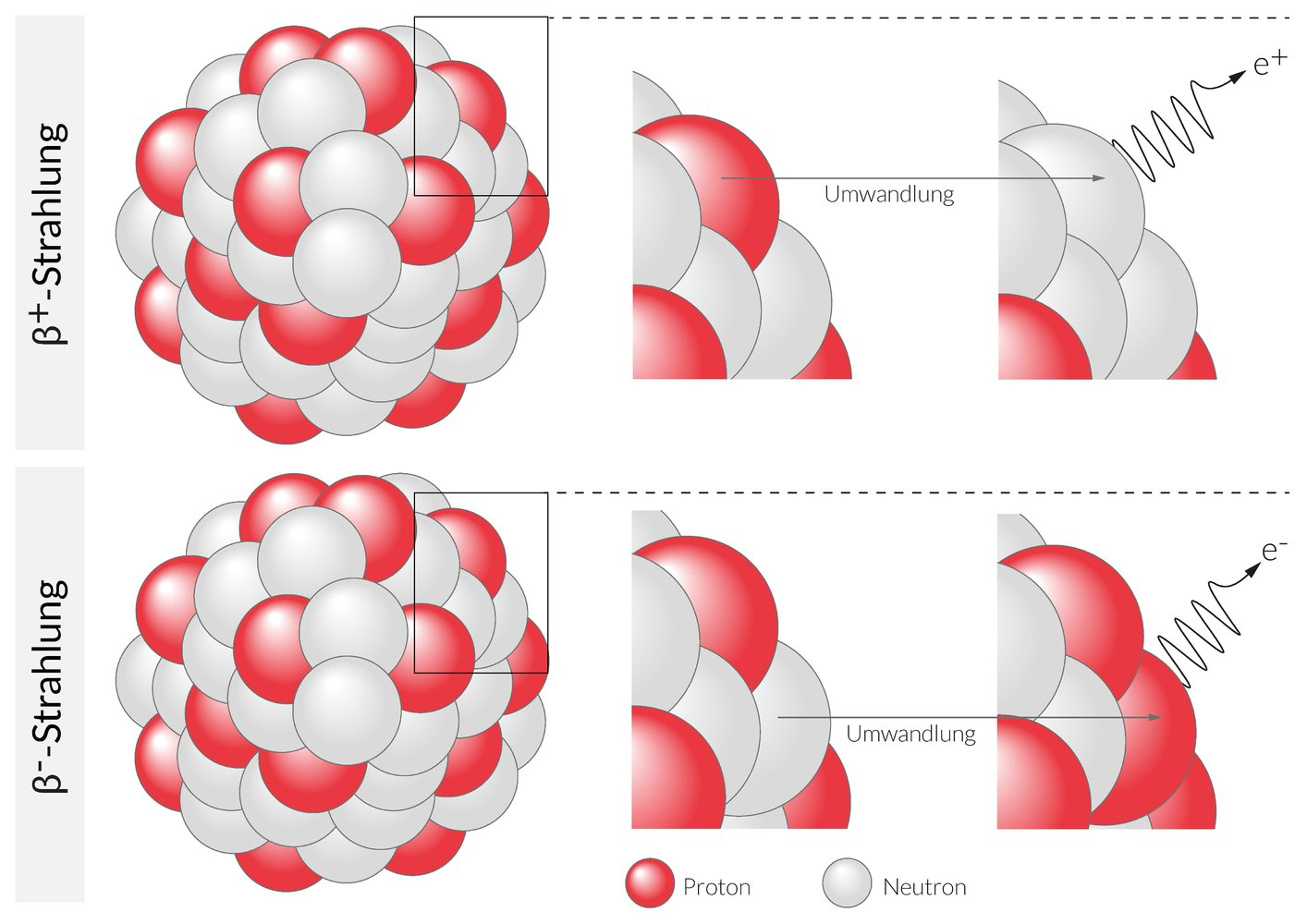
45. In positron emission tomography (PET), 2-fluoro-2-deoxy-D-glucose is used as a glucose analog labeled with the radionuclide ¹⁸⁹F.
Which nuclide is formed when ¹⁸⁹F decays by emitting a positron?
A. ¹⁹⁹F
B. ¹⁴⁷N
C. ²²¹¹Na
D. ¹⁸¹⁰Ne
E. ¹⁸⁸O
E. ¹⁸⁸O
46. Medical oxygen is supplied in gas cylinders with an internal pressure typically of 200 bar. Manufacturers specify that these cylinders must not be heated above 50 °C and must be stored accordingly.
By approximately what percentage does the internal pressure increase if such a gas cylinder is heated from 30 °C to 90 °C?
A. 3 %
B. 20 %
C. 40 %
D. 60 %
E. 300 %
B. 20 %
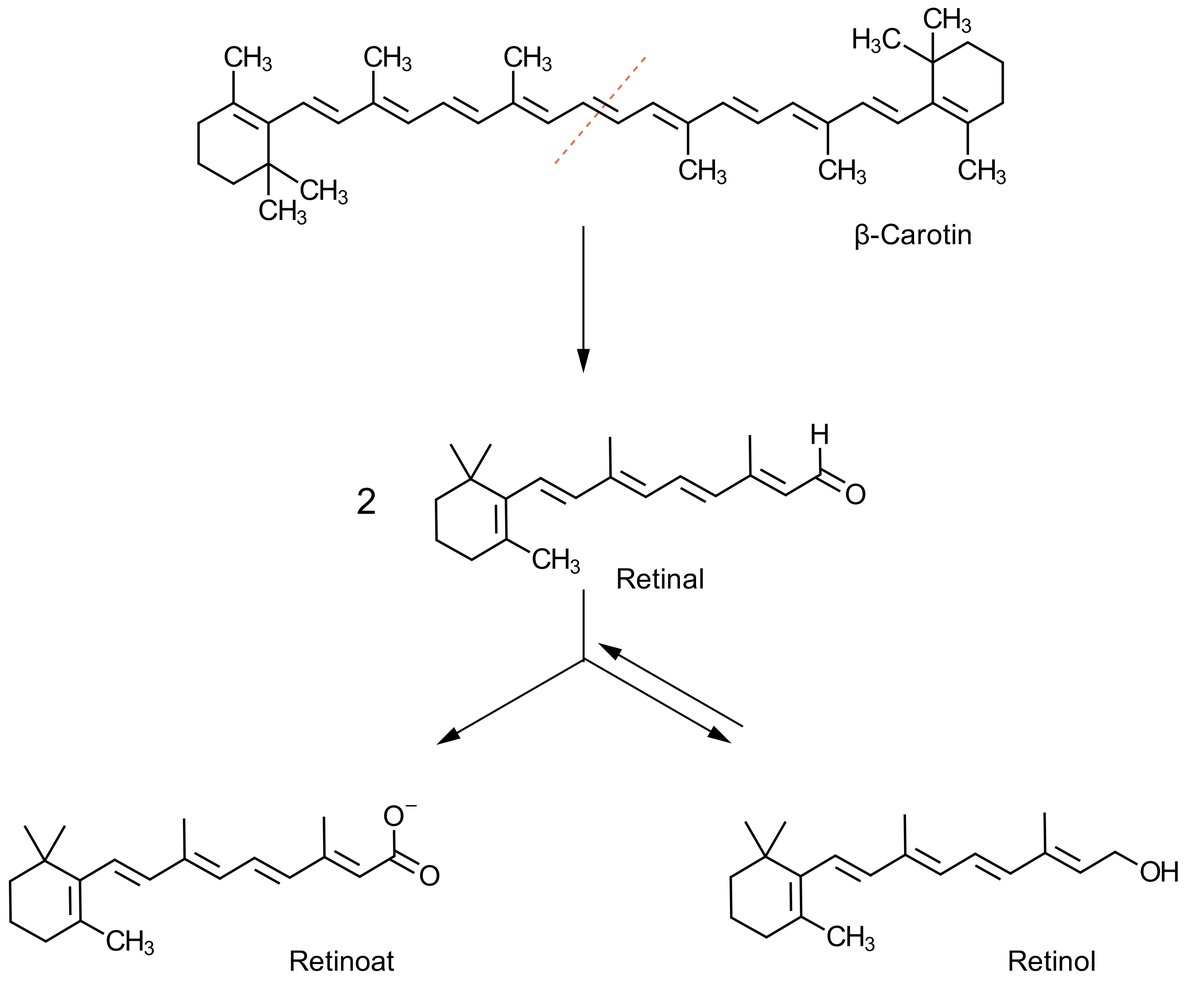
47. β-Carotene obtained from food can be metabolized to retinol. The UV/Vis spectrum of β-carotene shows an absorption maximum at approximately 425 nm, whereas the UV/Vis spectrum of retinol shows an absorption maximum at approximately 325 nm.
How can this shift in absorption maxima in the UV/Vis spectrum best be explained?
Compared with retinol:
A. β-Carotene has the lower molar mass
B. β-Carotene has the higher water solubility
C. β-Carotene is negatively charged
D. cis-configured double bonds in β-carotene improve its ability to absorb light
E. β-Carotene possesses the more extensive system of conjugated double bonds
E. β-Carotene possesses the more extensive system of conjugated double bonds
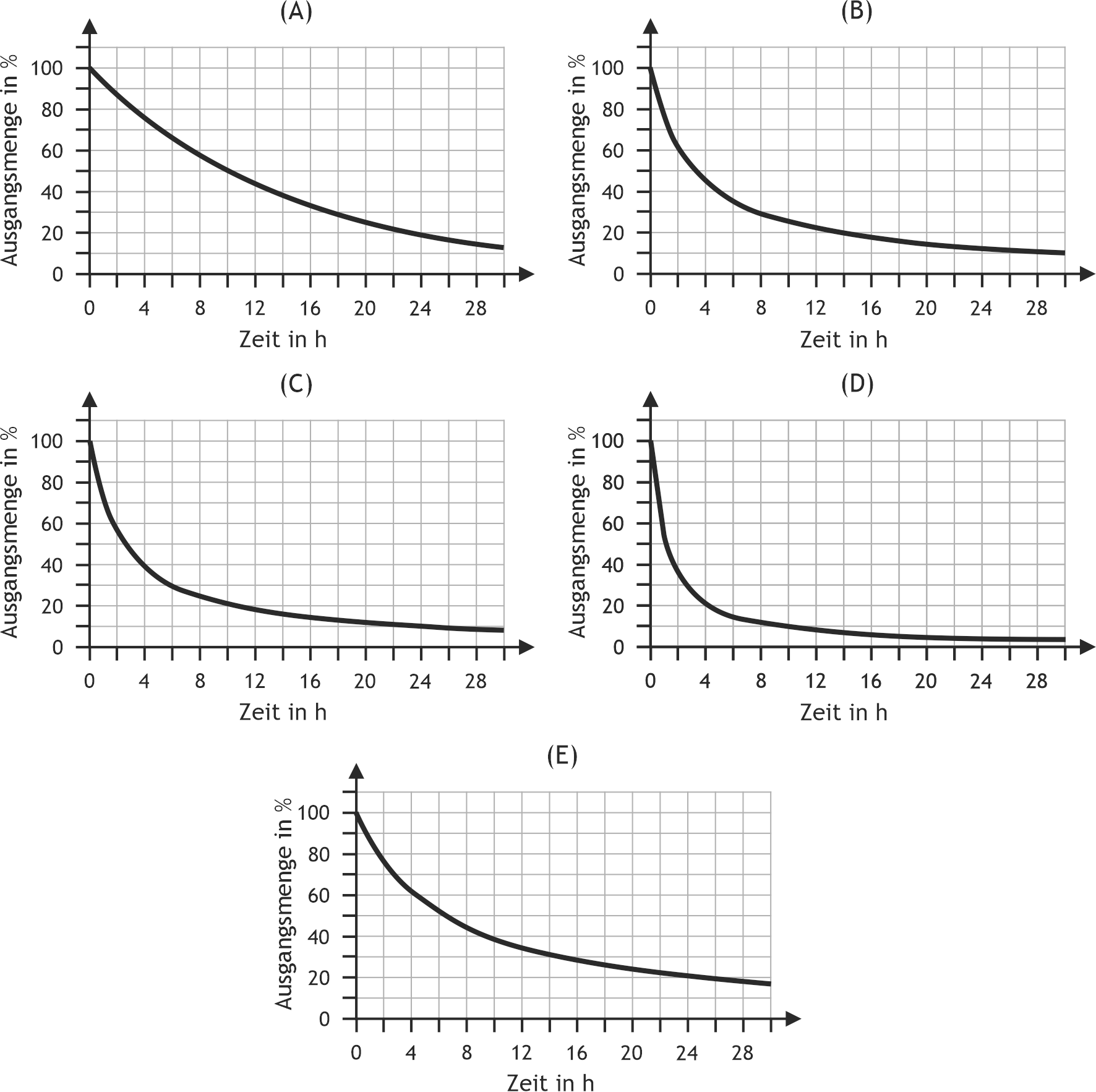
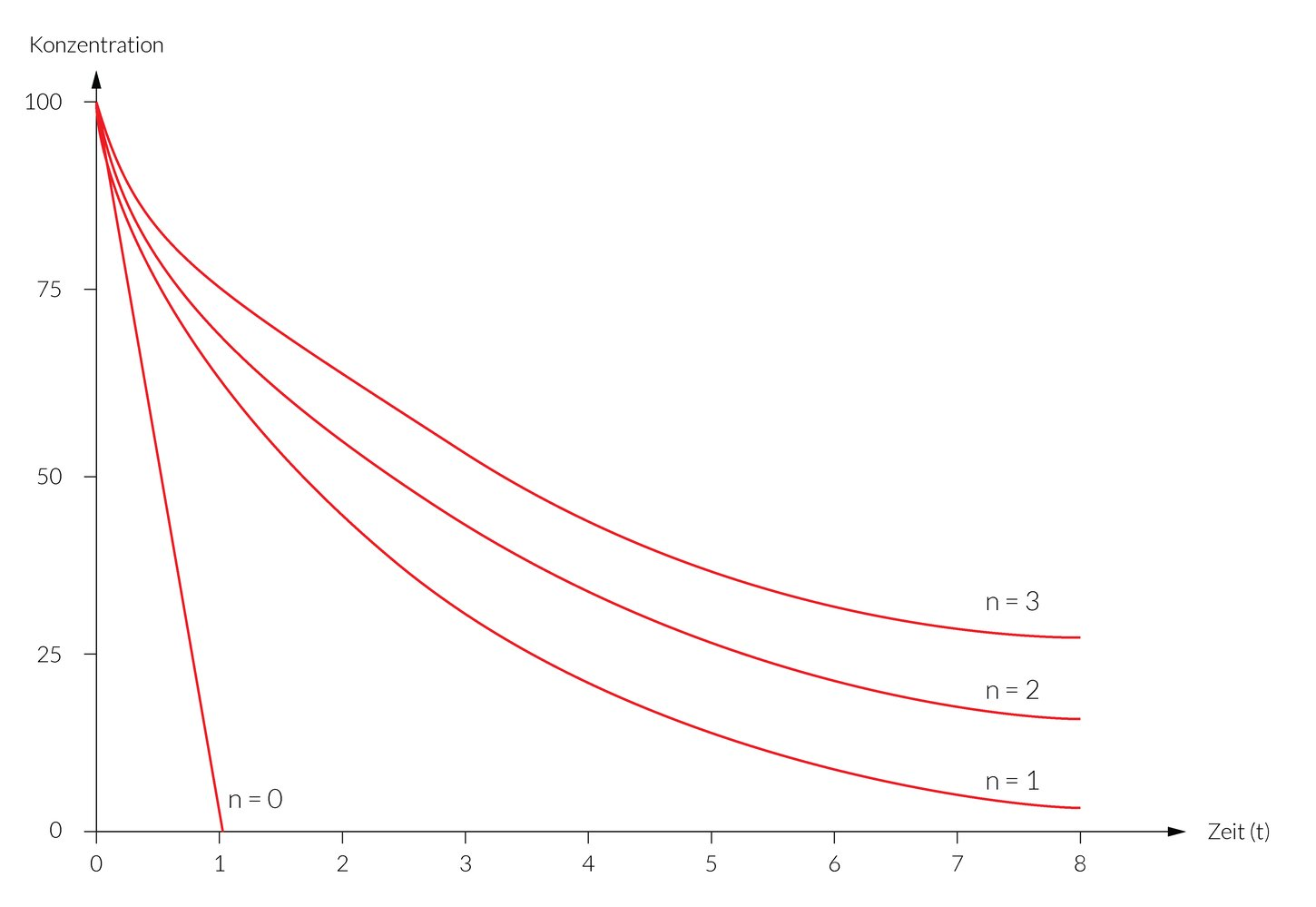
48. Some biochemical processes can be described by first-order kinetics.
Which of the following reaction progress curves most closely corresponds to first-order kinetics?
A. (A)
B. (B)
C. (C)
D. (D)
E. (E)
A. (A)
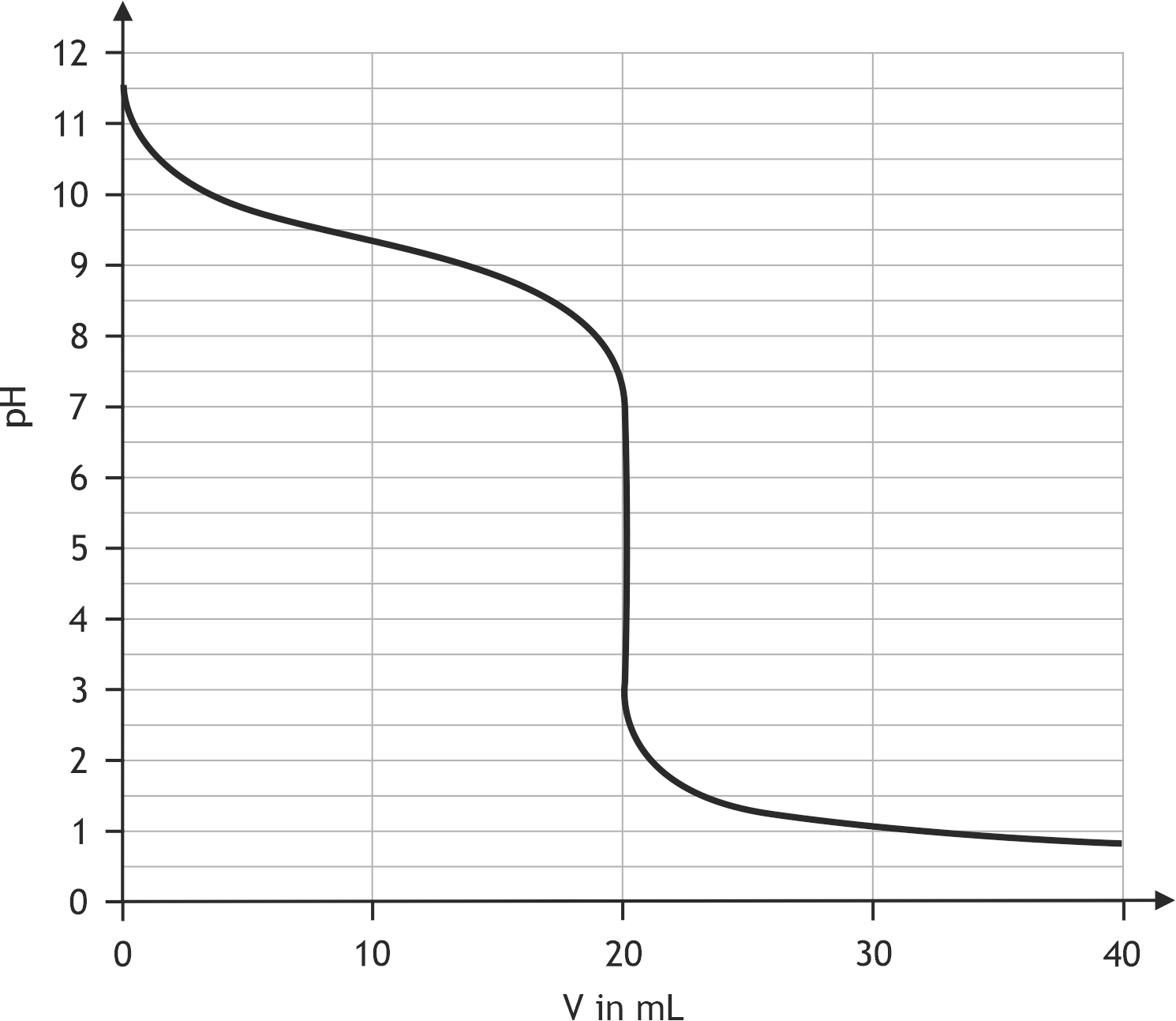
49. The following diagram shows the titration of a weak base (B) with a strong acid. What is the approximate pKₛ value of the corresponding acid (HB⁺)?
A. 1.0
B. 4.7
C. 5.0
D. 8.8
E. 9.3
E. 9.3
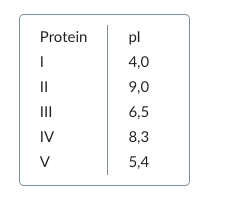
50. A native electrophoresis is used to separate a protein mixture consisting of five proteins at a pH value of 6.5.
The isoelectric points (pI) of the proteins are as follows: How many proteins migrate toward the anode?
A. 1
B. 2
C. 3
D. 4
E. 5
B. 2
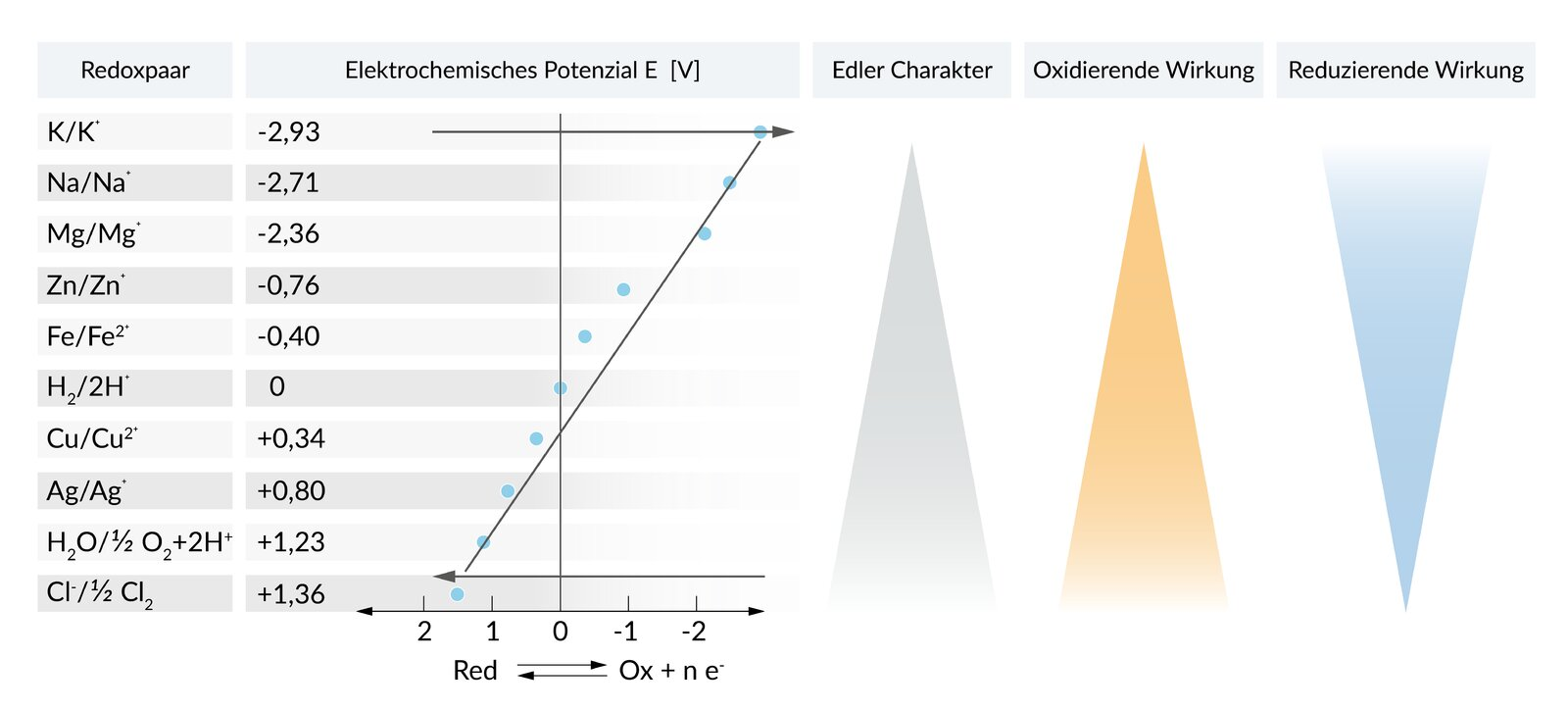
51. To predict the course of a redox reaction, for example between two metals, the standard potential (E⁰) of the involved metals according to the electrochemical series plays a decisive role. The addition of a metal M to an aqueous CuSO₄ solution leads to the deposition of copper. The addition of the same metal M to an aqueous ZnCl₂ solution leads to the deposition of zinc.
Which sequence of metals according to the electrochemical series is therefore correct, with the metal having the most positive standard potential (E⁰) placed on the far right?
A. Cu / M / Zn
B. Cu / Zn / M
C. M / Zn / Cu
D. Zn / Cu / M
E. Zn / M / Cu
C. M / Zn / Cu
52. Barium sulfate is used as an oral X-ray contrast agent for imaging the gastrointestinal tract because of its low water solubility and high X-ray absorption capacity.
What is the unit of the solubility product of barium sulfate?
A. Dimensionless
B. mol·L⁻¹
C. mol⁻¹·L⁻¹
D. mol·L⁻²
E. mol²·L⁻²
E. mol²·L⁻²

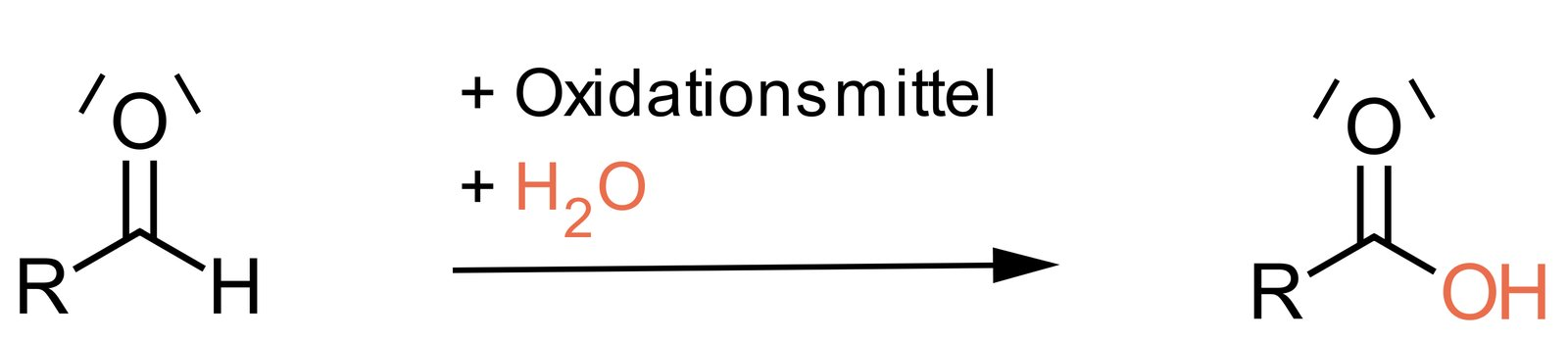
53. In a positive Tollens’ test, elemental silver is formed, which usually deposits as a silver mirror on the walls of the reaction vessel. The following reaction takes place.
What type of reaction occurs with respect to the carbonyl carbon shown?
A. Hydration
B. Hydrogenation
C. Hydrolysis
D. Oxidation
E. Reduction
D. Oxidation
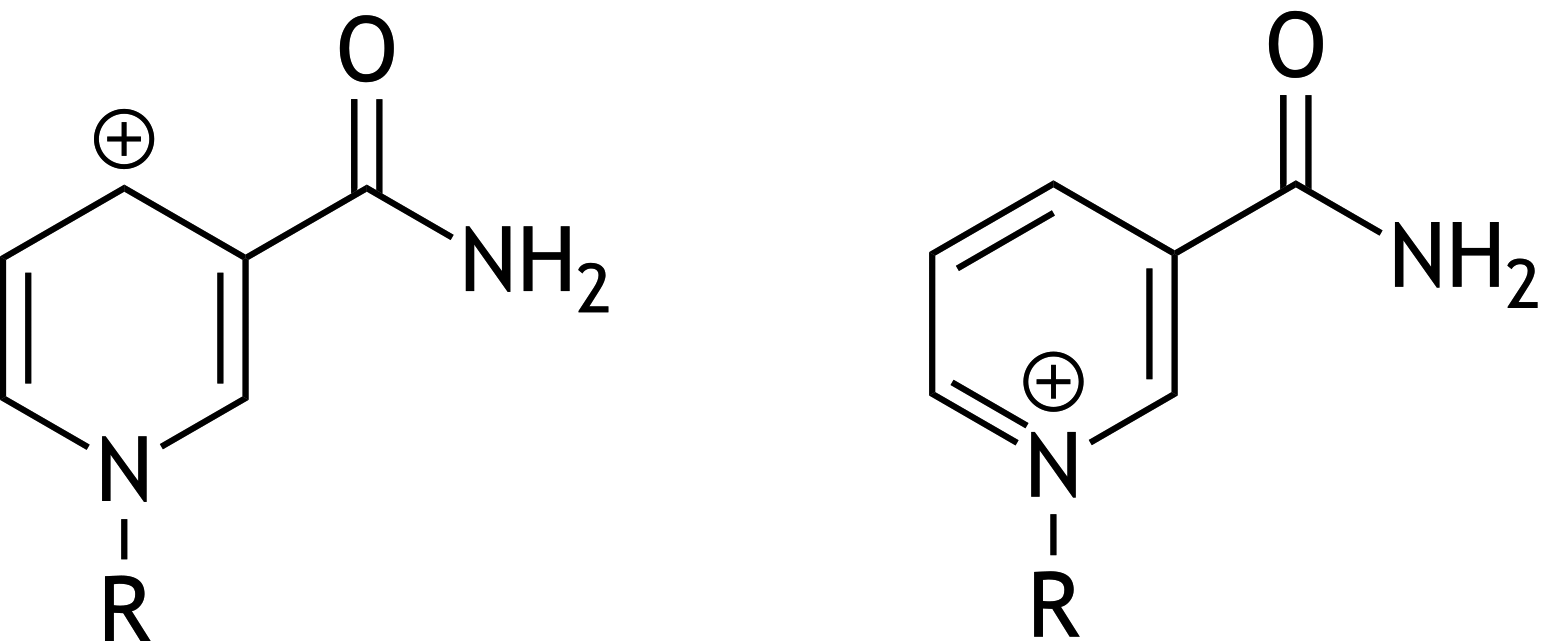
54. Oxidoreductases often require NADH as a coenzyme when catalyzing a reduction reaction. During the reduction, NADH donates a hydride ion, forming NAD⁺.
What is the relationship between the two forms of NAD⁺ shown below?
A. Conformational isomers
B. Constitutional isomers
C. Resonance structures (mesomeric forms)
D. Stereoisomers
E. Two compounds in equilibrium
C. Resonance structures (mesomeric forms)
55. Isotopes of different elements play an important role in biomedical research.
In what respect do the following hydrogen isotopes differ?
¹¹H ²¹H ³¹H
A. Number of electrons
B. Nuclear charge number
C. Number of neutrons
D. Atomic number
E. Number of protons
C. Number of neutrons
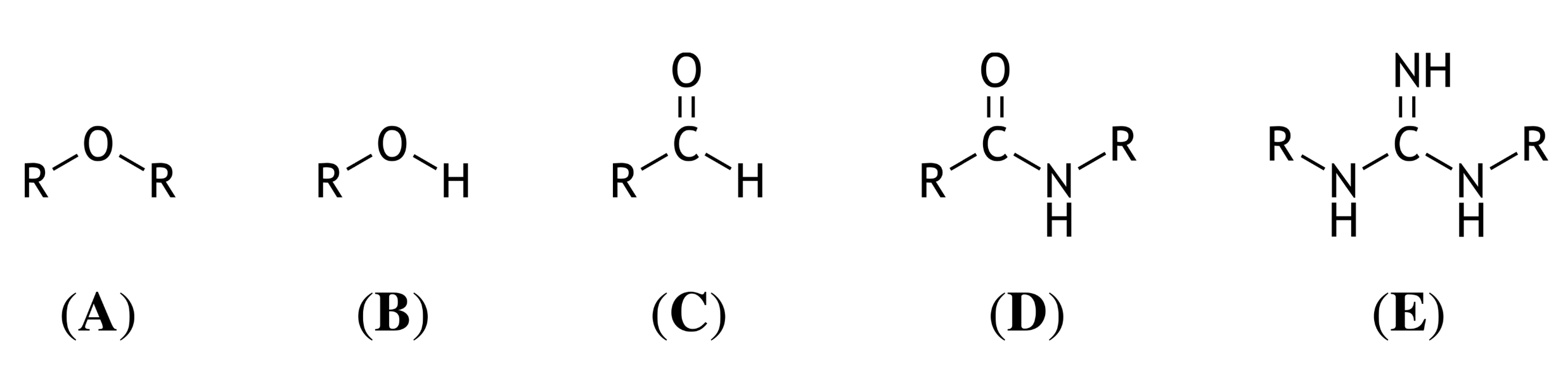
56. Hydrogen bonds are structural, noncovalent interactions that contribute to the formation and stabilization of an enzyme’s native conformation. Shown below are several chemical structures, where “R” represents an aliphatic residue.
Which of these chemical structures (A–E) can theoretically form the greatest number of intermolecular hydrogen bonds?
A. (A)
B. (B)
C. (C)
D. (D)
E. (E)
E. (E)
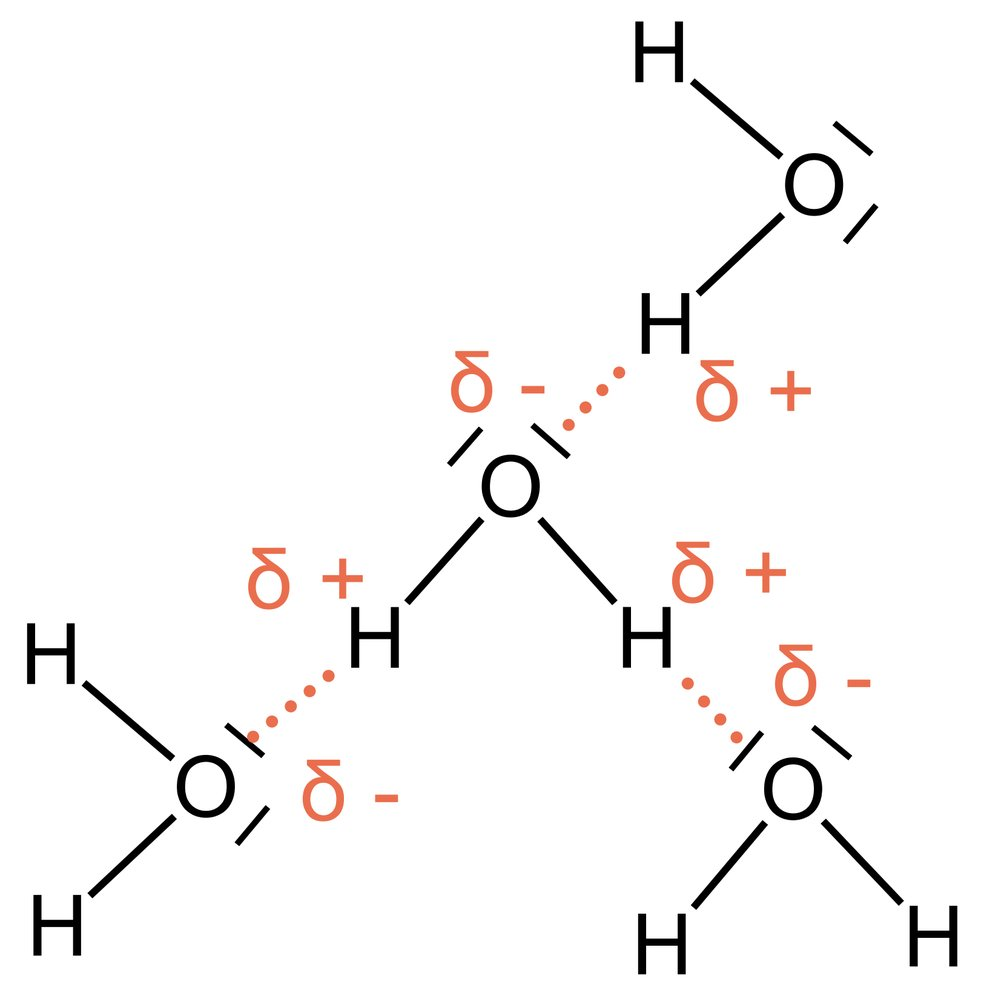
57. Calcium is an element of group 2 in the periodic table. The salt calcium phosphate can be used as a ceramic material for surgical bone replacement due to its low solubility. The solubility product quantitatively describes this property.
What is the unit of the solubility product of calcium phosphate?
A. mol·L⁻¹
B. mol·L⁻²
C. mol²·L⁻²
D. mol³·L⁻³
E. mol⁵·L⁻⁵
E. mol⁵·L⁻⁵
58. The reaction entropy (ΔS) describes the change in entropy during the course of a chemical reaction.
For which of the following reactions does ΔS > 0 apply?
A. CH₄(g) + 2 O₂(g) → CO₂(g) + 2 H₂O(g)
B. C₂H₄(g) + H₂(g) → C₂H₆(g)
C. NH₄Cl(s) → NH₃(g) + HCl(g)
D. N₂(g) + 3 H₂(g) → 2 NH₃(g)
E. 2 SO₂(g) + O₂(g) → 2 SO₃(g)
C. NH₄Cl(s) → NH₃(g) + HCl(g)
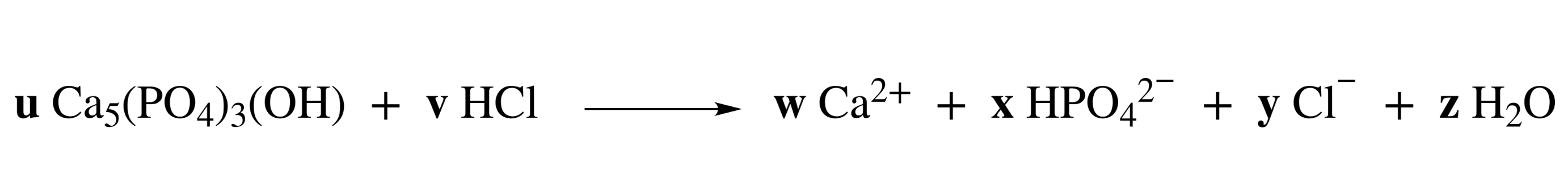
59. Hydroxyapatite in the extracellular bone matrix is dissolved by hydrochloric acid secreted by osteoclasts according to the following assumed reaction equation:
(Reaction not shown)
What is the value of v after stoichiometrically balancing this reaction equation?
A. 1
B. 2
C. 3
D. 4
E. 5
D. 4
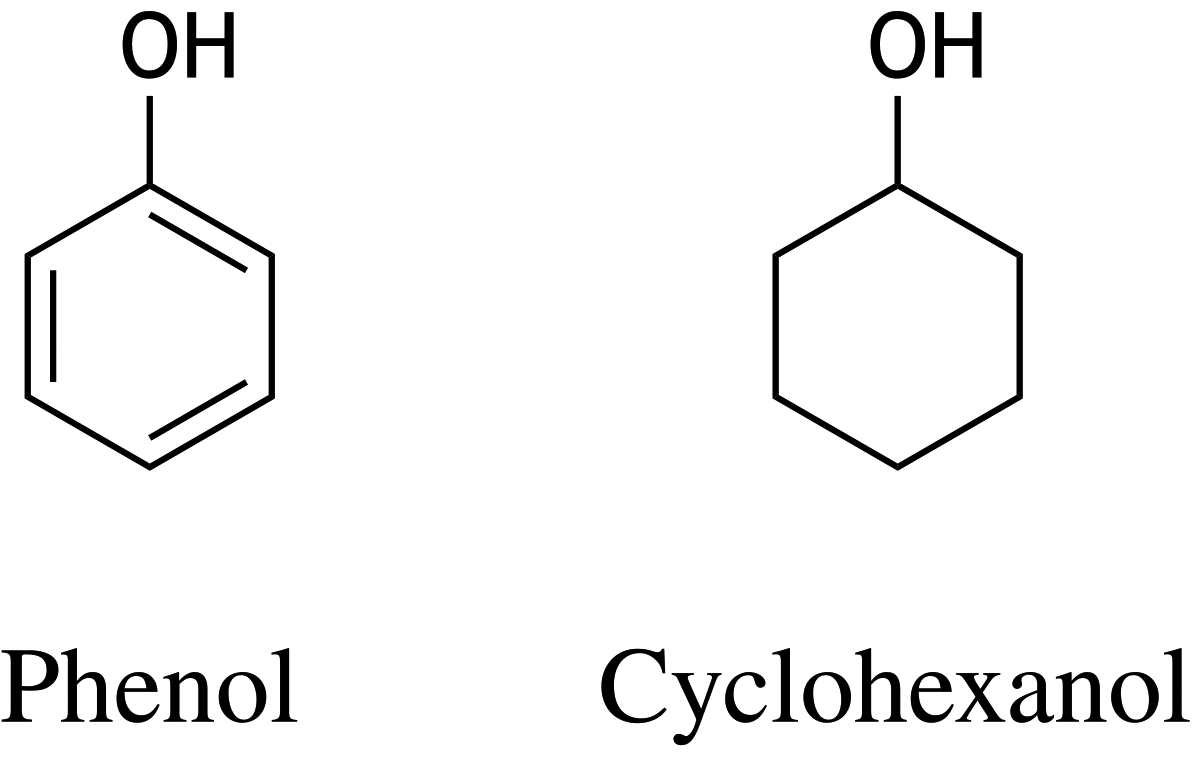
60. Phenol is used, among other things, as a starting material in drug synthesis. The hydroxyl group of phenol has a pKₛ value of 10, while that of cyclohexanol has a pKₛ value of 16. Phenol and cyclohexanol have the following chemical structures:
What accounts for this difference in pKₛ values?
A. The negatively charged conjugate base formed by deprotonation of phenol is stabilized by resonance.
B. The negatively charged conjugate acid formed by deprotonation of phenol is stabilized by resonance.
C. The positively charged conjugate base formed by deprotonation of phenol is stabilized by resonance.
D. The positively charged conjugate acid formed by deprotonation of phenol is stabilized by resonance.
E. The negatively charged conjugate acid formed by protonation of phenol is stabilized by resonance.
A. The negatively charged conjugate base formed by deprotonation of phenol is stabilized by resonance.
61. Hydrogen peroxide belongs to the group of “reactive oxygen species (ROS)” and can oxidize methionine in a single step. In the oxidation product, the sulfur atom has an oxidation number of 0. The amount of this oxidation product increases with cellular age and serves as an indicator of oxidative stress.
To which of the following classes of compounds does this oxidation product belong?
A. Disulfides
B. Sulfates
C. Sulfonates
D. Sulfoxides
E. Thiols
D. Sulfoxides
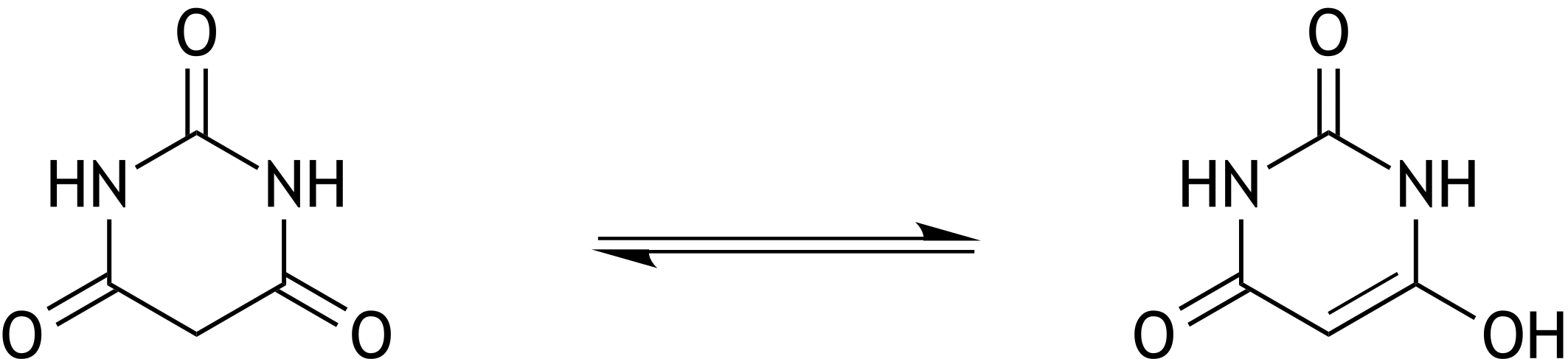
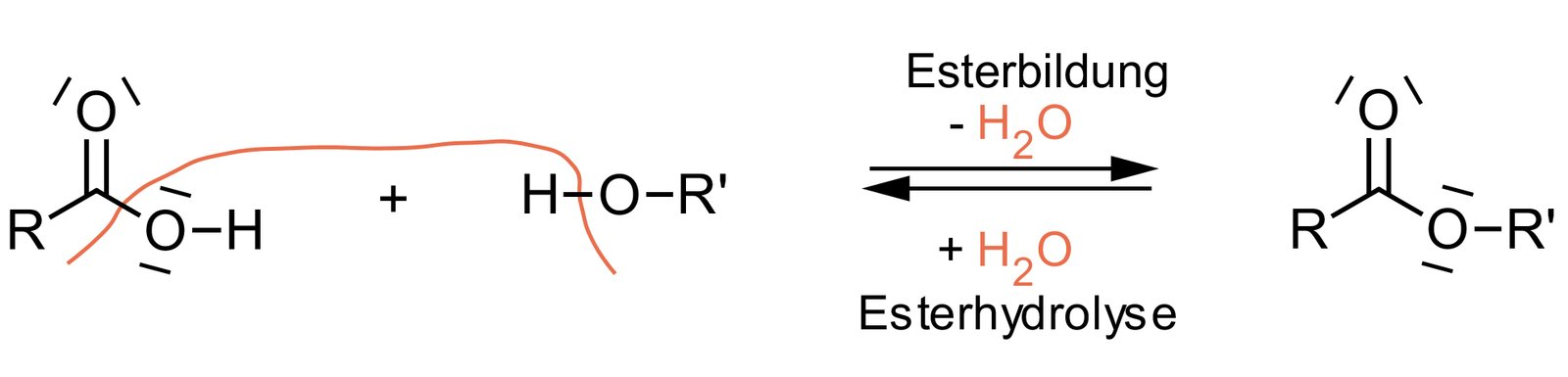
62. Barbiturates are drugs that, depending on the dose, have sedative, hypnotic, or narcotic effects. The basic structure of barbiturates is barbituric acid, which exists in solution in two different forms. The interconversion of these two forms represents an equilibrium reaction.
What is the relationship between these two forms?
A. Diastereomers
B. Enantiomers
C. Epimers
D. Conformers
E. Tautomers
E. Tautomers
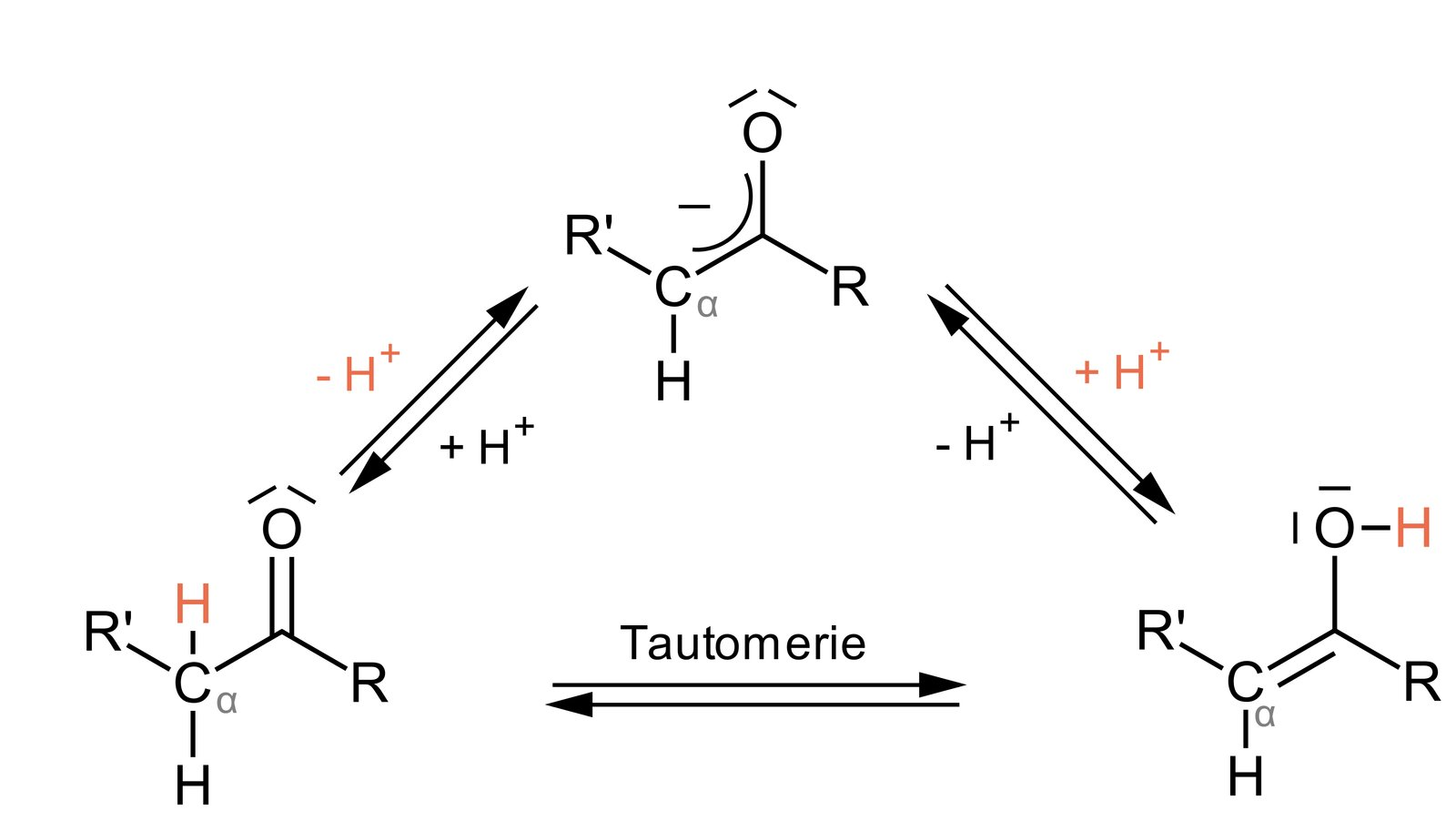
63. ACE inhibitors are commonly used to treat hypertension. They act as specific inhibitors of the angiotensin-converting enzyme (ACE). Under physiological conditions, this enzyme catalyzes the conversion of inactive angiotensin I into active angiotensin II.
What type of chemical bond is cleaved during this conversion?
A. Amide bond
B. Anhydride bond
C. Ester bond
D. Covalent C–C single bond
E. N-glycosidic bond
A. Amide bond
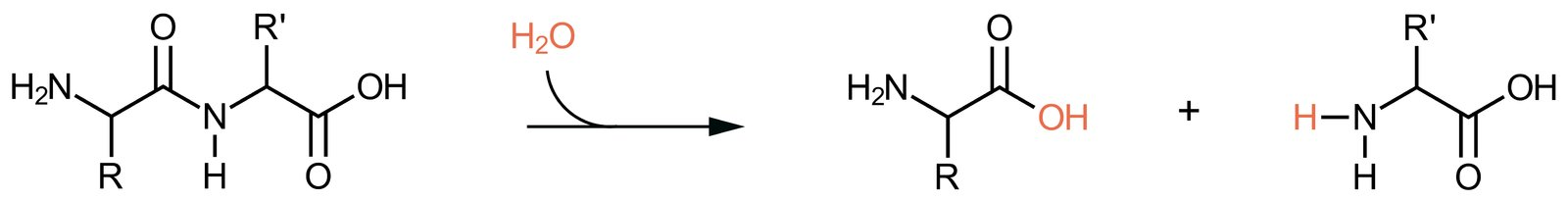
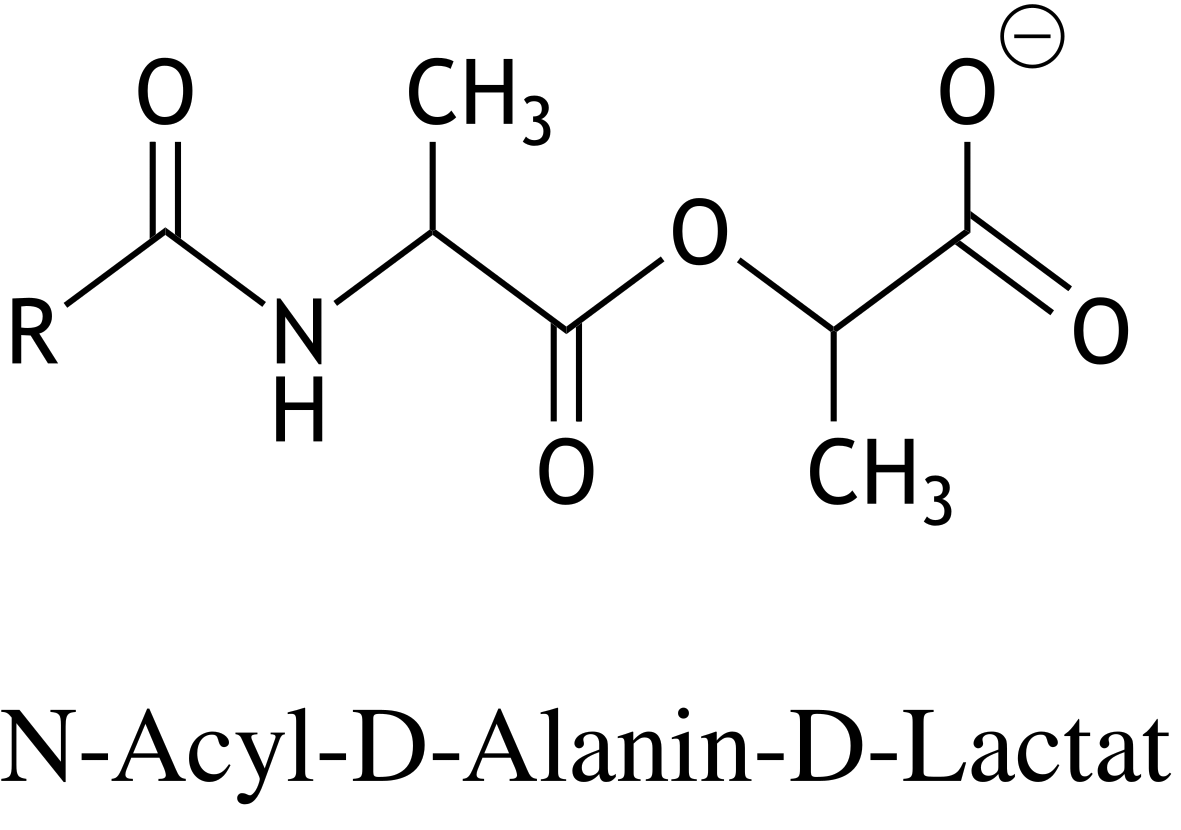
64. A mutation-induced replacement of the C-terminal D-alanine with D-lactate in the D-alanine–D-alanine groups of peptidoglycan precursors in bacterial cell walls can lead to resistance of Gram-positive bacteria against the antibiotic vancomycin. The structural formula of this D-alanine–D-lactate group is shown below.
Through which type of chemical bond is D-lactate linked to D-alanine?
A. Amide bond
B. Anhydride bond
C. Ester bond
D. Isopeptide bond
E. Peptide bond
C. Ester bond
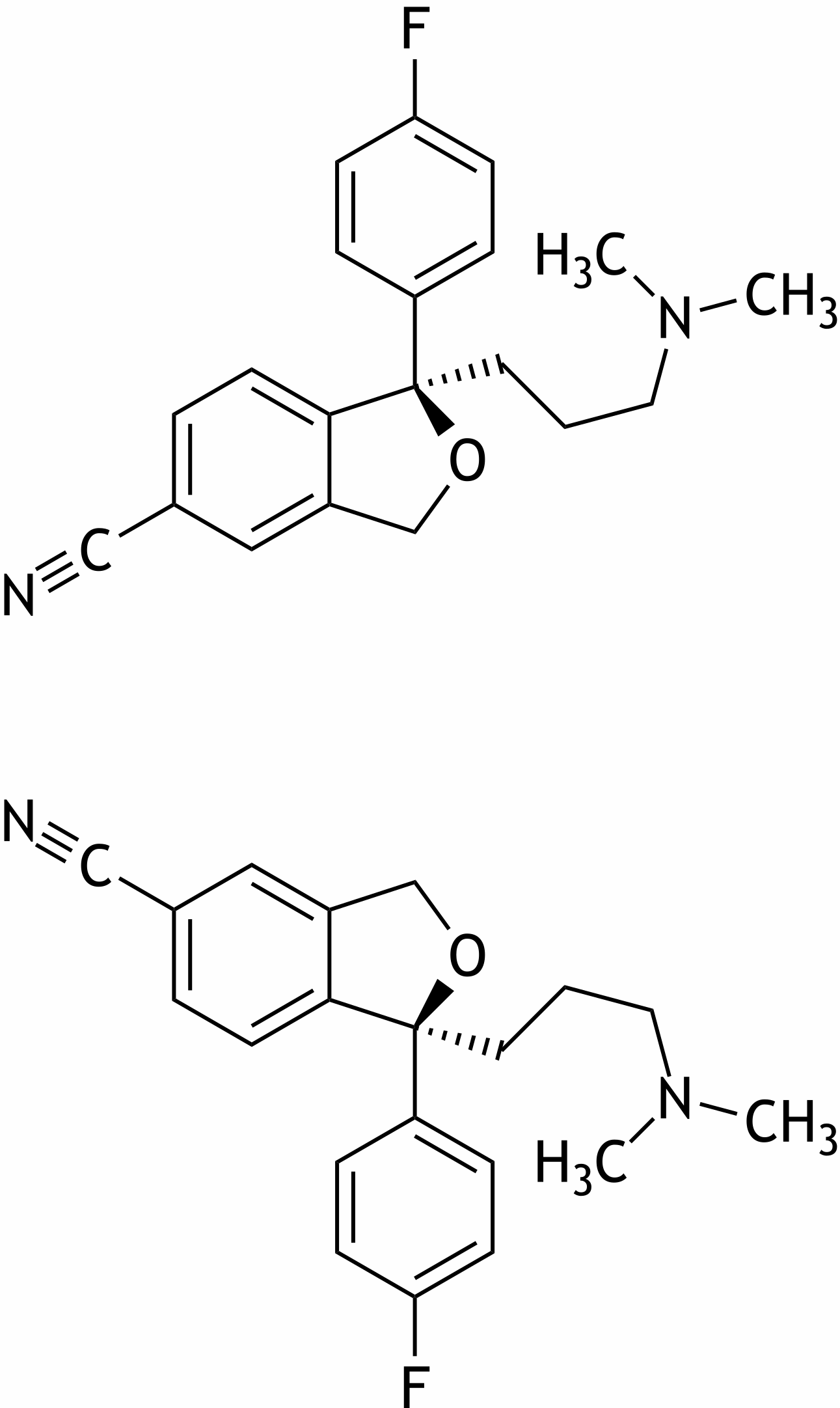
65. Citalopram is an antidepressant from the group of selective serotonin reuptake inhibitors (SSRIs). It is a mixture of the two isomers shown below.
What is the relationship between these two isomers?
A. Diastereomers
B. Enantiomers
C. Epimers
D. Conformational isomers
E. Constitutional isomers
B. Enantiomers
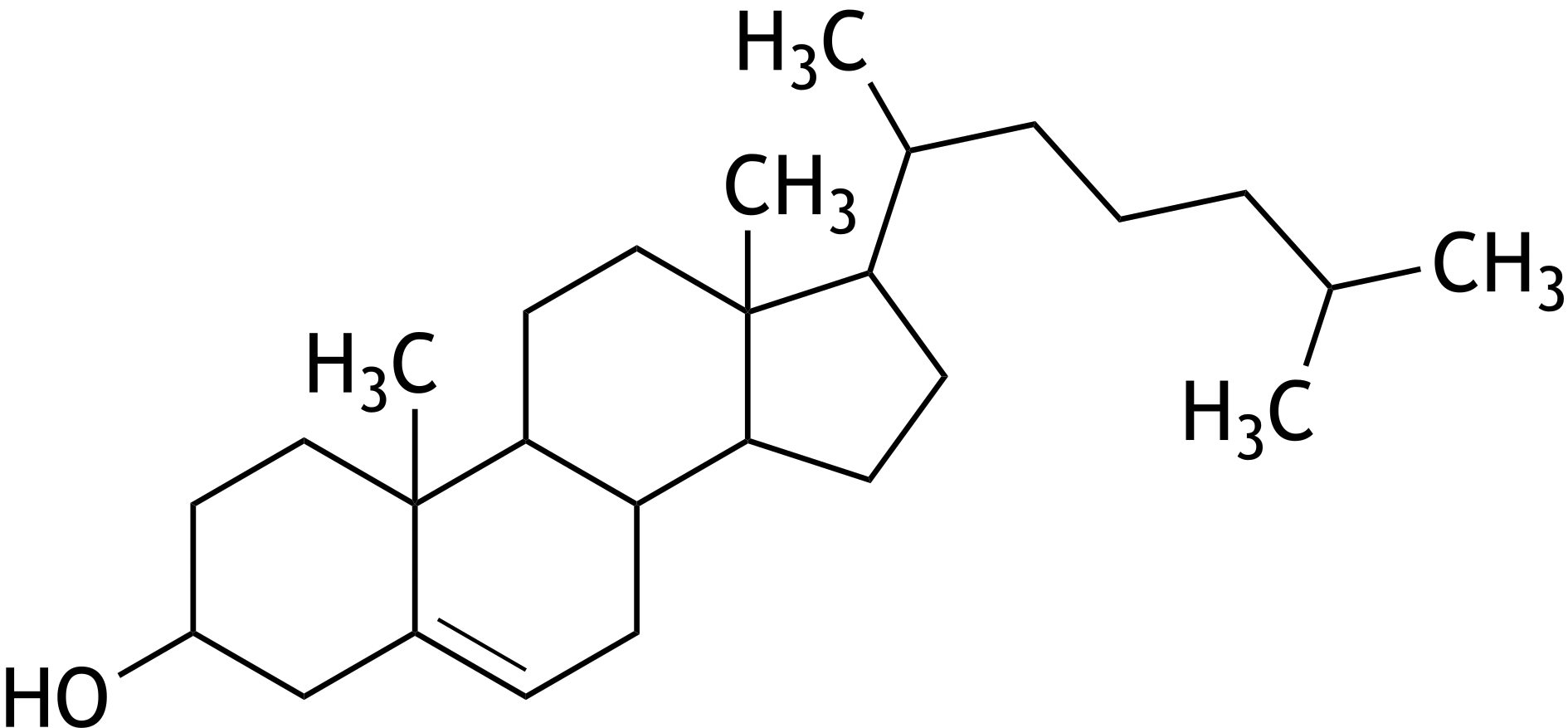
66. Biosynthetically produced cholesterol exists in only one isomeric form, even though cholesterol contains several chiral centers.
How many chiral centers does the cholesterol molecule shown below contain in total?
A. 4
B. 6
C. 8
D. 10
E. 12
C. 8
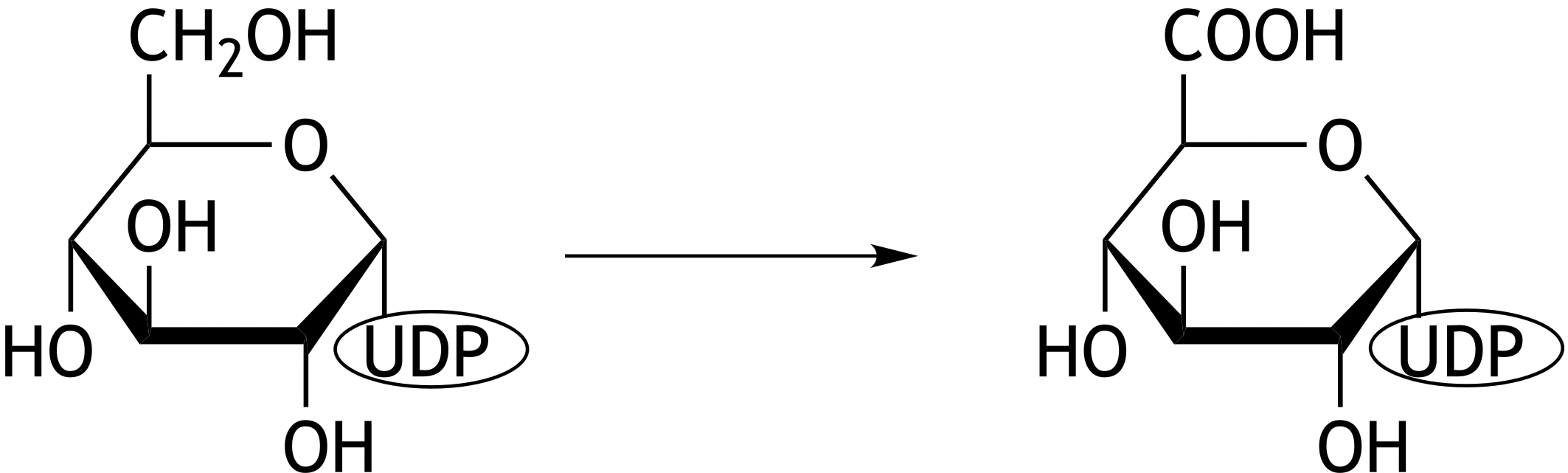
67. UDP-glucuronic acid is used in a phase II biotransformation reaction and can be synthesized from UDP-glucose. The reaction equation for the biosynthesis of UDP-glucuronic acid from UDP-glucose is shown below (not stoichiometrically balanced).
Which of the following reactions occurs in this process?
A. Carboxylation
B. Hydrolysis
C. Isomerization
D. Oxidation
E. Reduction
D. Oxidation