2) Cycloaddition Reactions & Woodward-Hoffmann Rules
1/37
Earn XP
Description and Tags
Topics included: General cycloaddition rxn info, WH rules, Diels-Alder reactions (+ substituent effects, regioselectivity, endo/exo selectivity, stereochemistry, heterodienophiles), additional cycloaddition rxns, 1,3-dipolar reagents
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
38 Terms
Explain nomenclature of cycloaddition reactions.
Square brackets: no. of electrons involved
Regular brackets: No. of atoms involved
Diene before dienophile
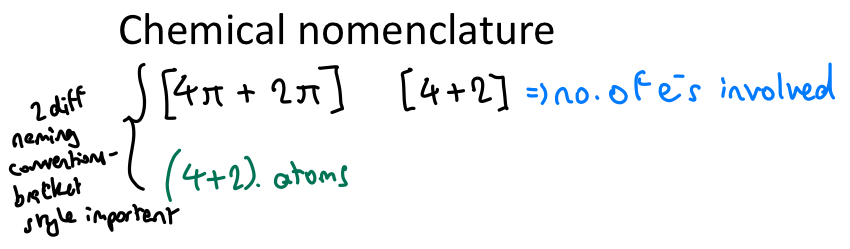
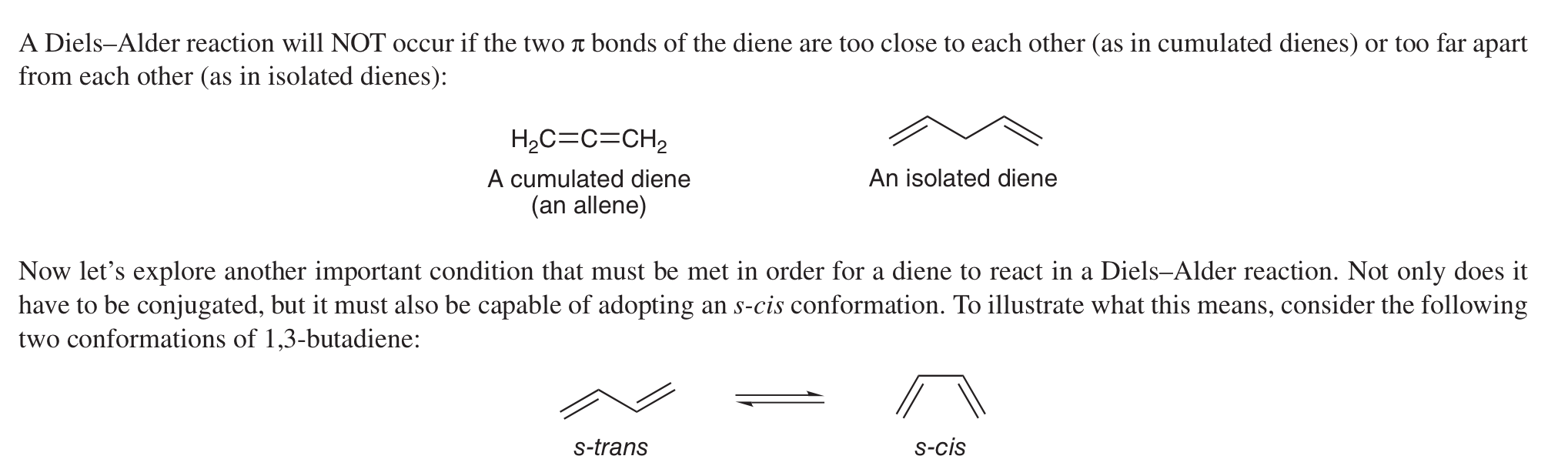
What’s a diene like in Diels-Alder reactions?
Conjugated diene w/ two pi bonds and must be able to form s-cis conformation.
Cannot be a cumulated diene (an allene) or an isolated diene.
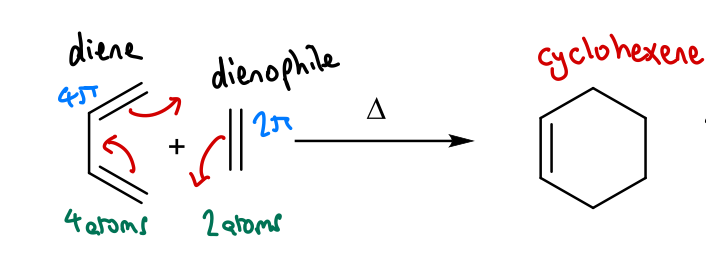
Show the general scheme of a Diels-Alder reaction.
What does FMO theory say about how a cycloaddition reaction can be successful?
Can only occur when overlap between HOMO of one reactant and LUMO of 2nd component enables correctly phased overlap.
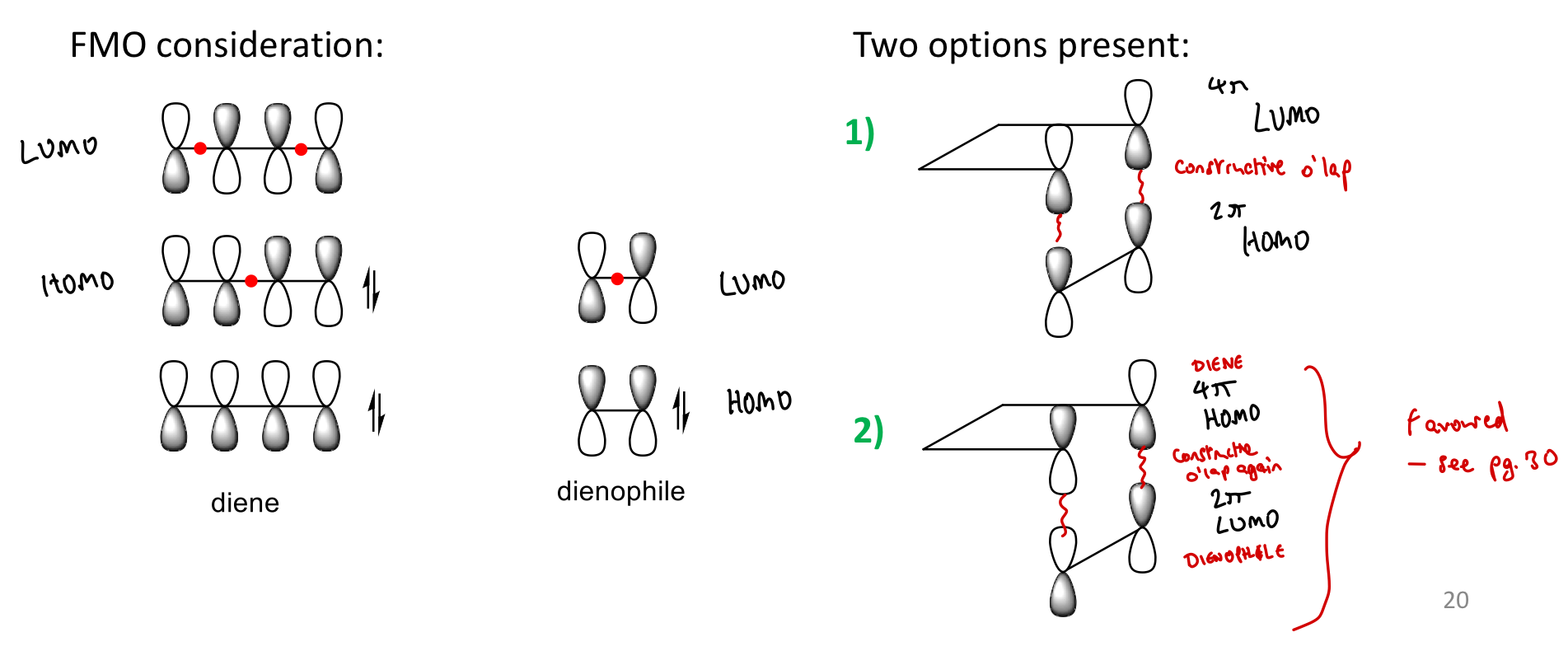
What is a suprafacial interaction?
Making interactions from lobes on same face of the molecule.
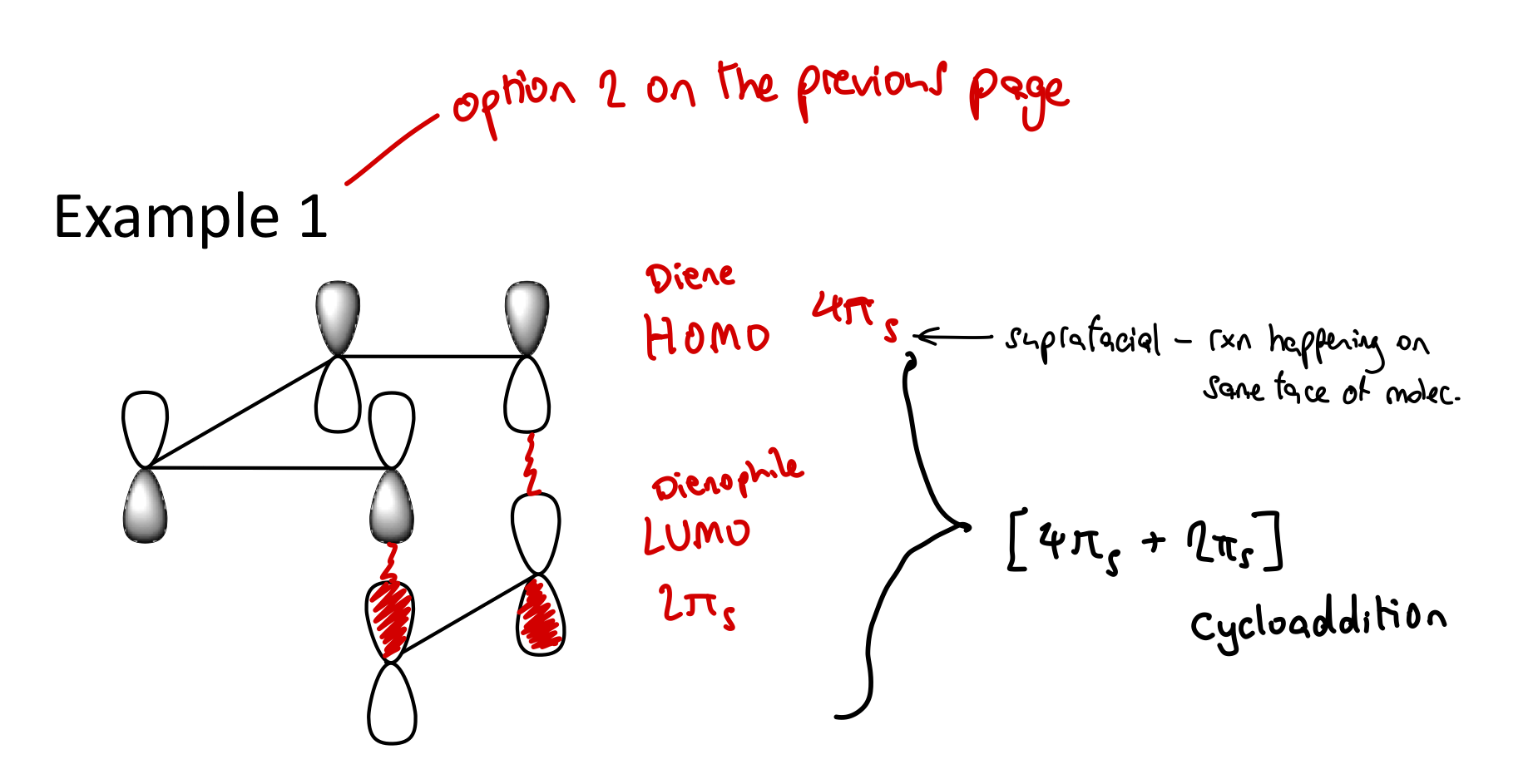
What’s an antarafacial interaction?
Making interactions from lobes on opposite faces.
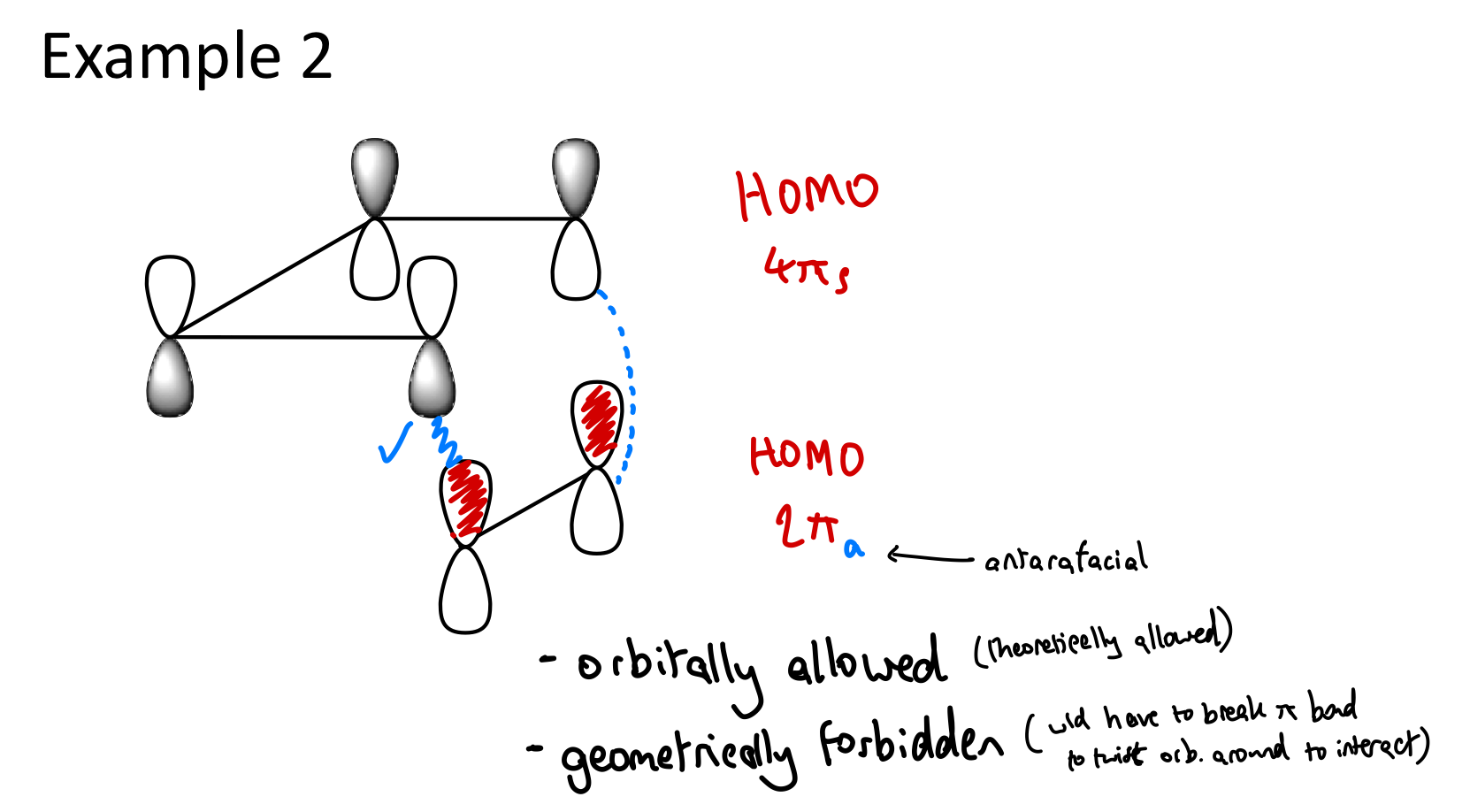
What are Woodward-Hoffman rules?
WH rules, a.k.a orbital-symmetry rules can be applied to pericyclic reactions to predict their likelihood of occurring (thermally or photochemically)
When the total number of [4q + 2]s and [4r]a components is odd:
thermal pericyclic reaction is allowed in the ground state of the reacting molecules(s).
If total of [4q + 2]s and [4r]a components is even:
the pericyclic reaction is allowed only in the excited state i.e., under photochemical conditions.
The terms q and r are integers denoting the number of electrons in a system.
![<p>WH rules, a.k.a orbital-symmetry rules can be applied to pericyclic reactions to predict their likelihood of occurring (thermally or photochemically)</p><p></p><ul><li><p>When the total number of [4q + 2]<sub>s</sub> and [4r]<sub>a</sub> components is <strong>odd:</strong></p><ul><li><p> <strong>thermal</strong> pericyclic reaction is allowed in the <strong>ground</strong> state of the reacting molecules(s).</p></li></ul></li></ul><p></p><ul><li><p>If total of [4q + 2]<sub>s</sub> and [4r]<sub>a</sub> components is <strong>even</strong>:</p><ul><li><p> the pericyclic reaction is allowed only in the <strong>excited</strong> state i.e., under <strong>photochemical</strong> conditions.</p></li></ul></li></ul><p></p><p>The terms q and r are integers denoting the number of electrons in a system.</p><p></p>](https://knowt-user-attachments.s3.amazonaws.com/f5b48ba3-c86c-4b42-a454-d9d2d18e0f4c.png)
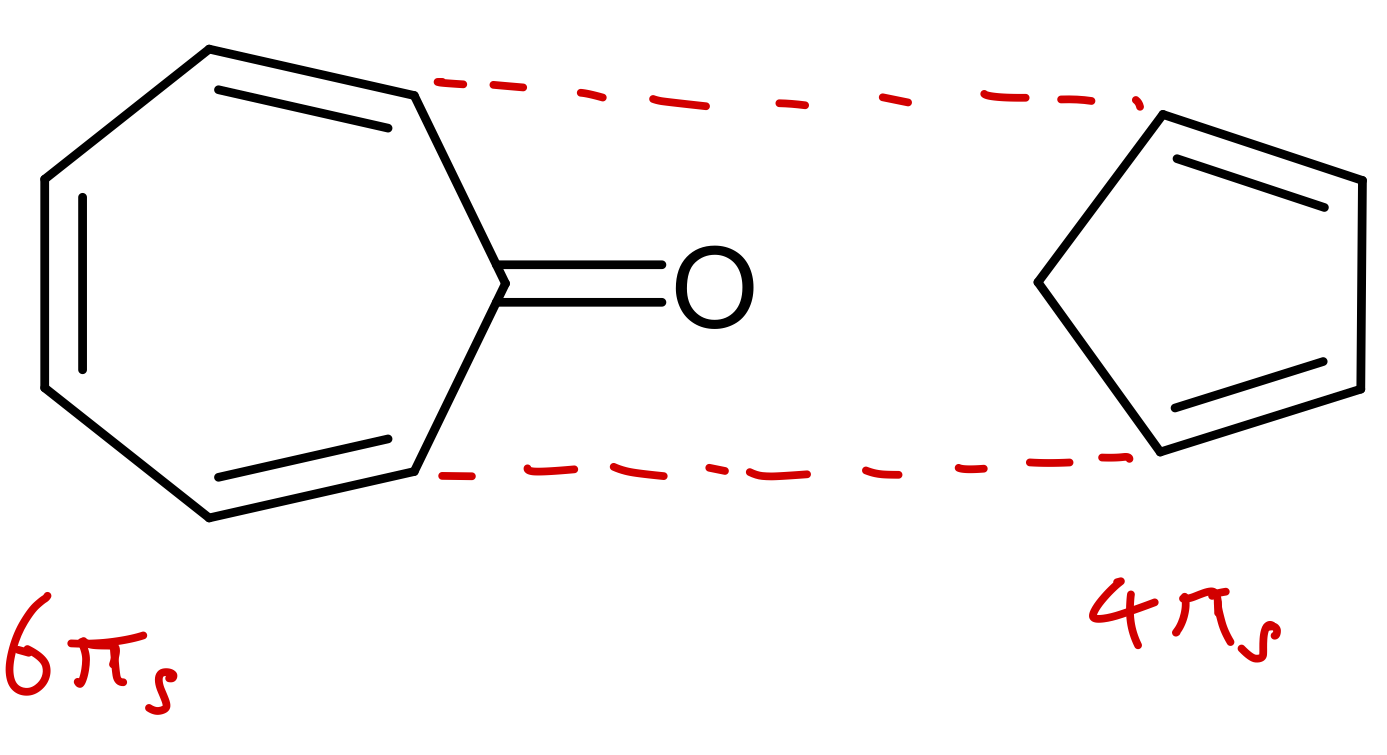
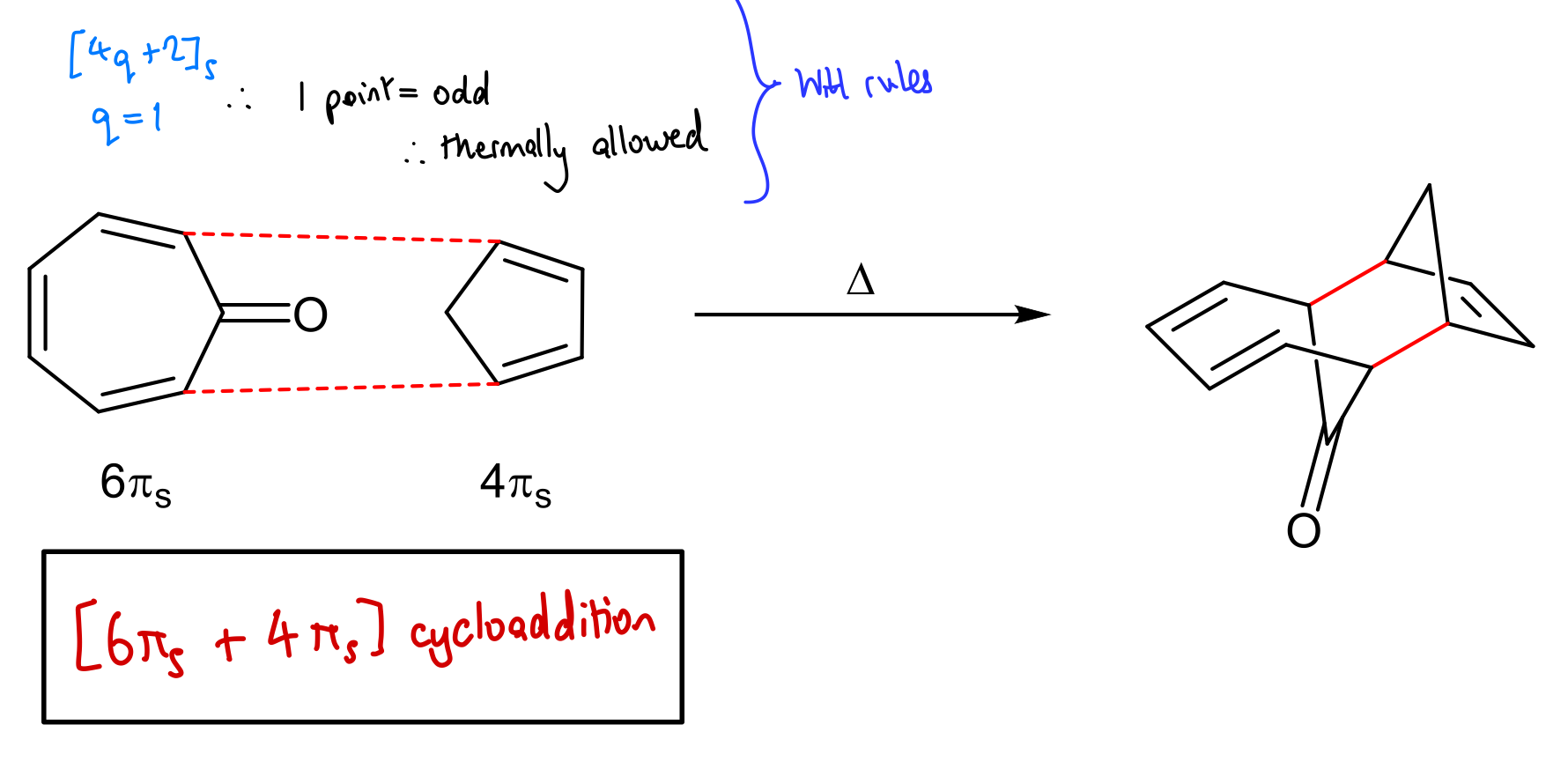
Use WH rules to predict outcome of this reaction:
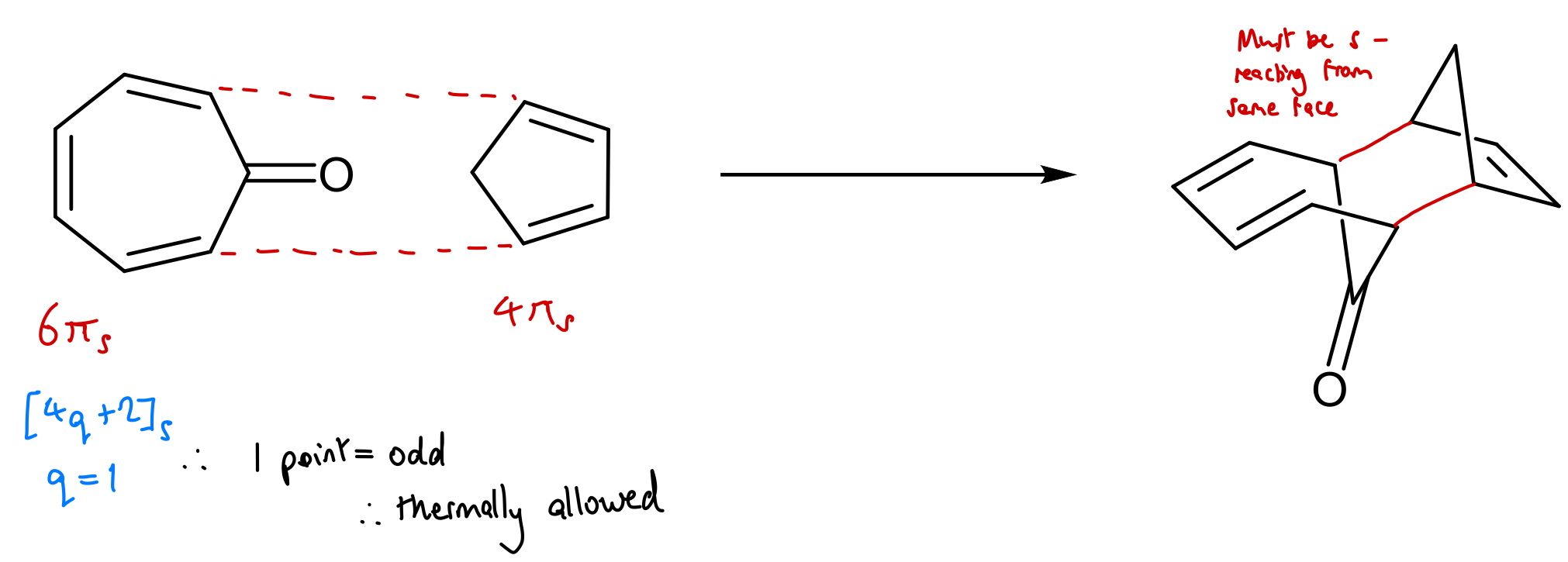
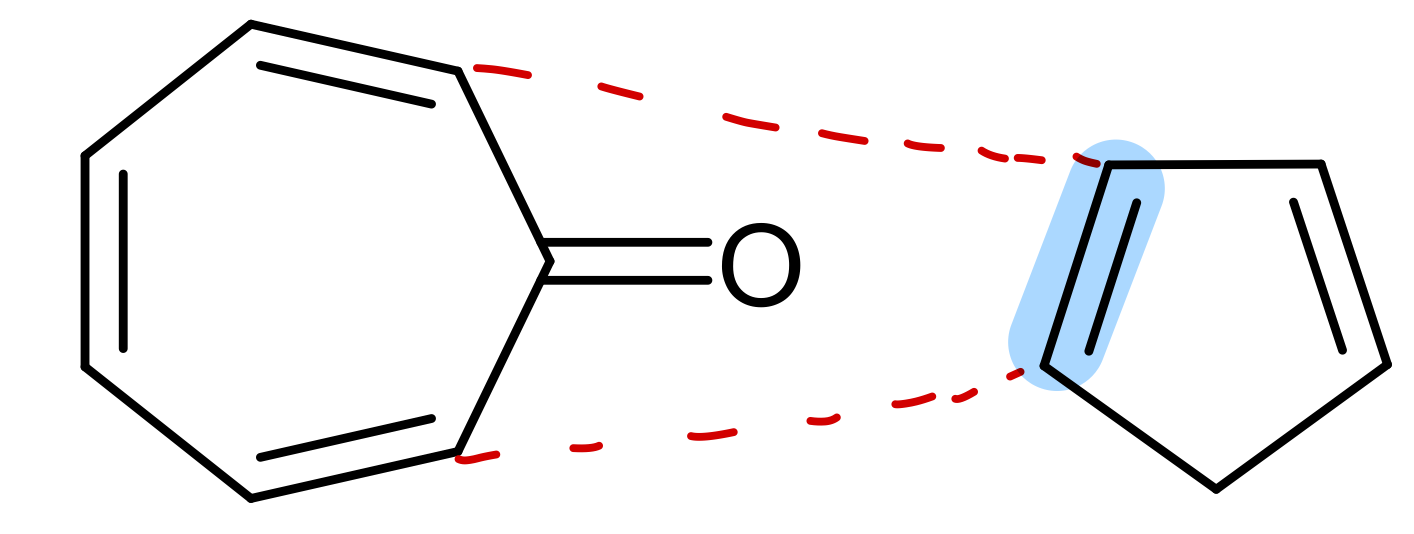
Use WH rules to predict outcome of this reaction:
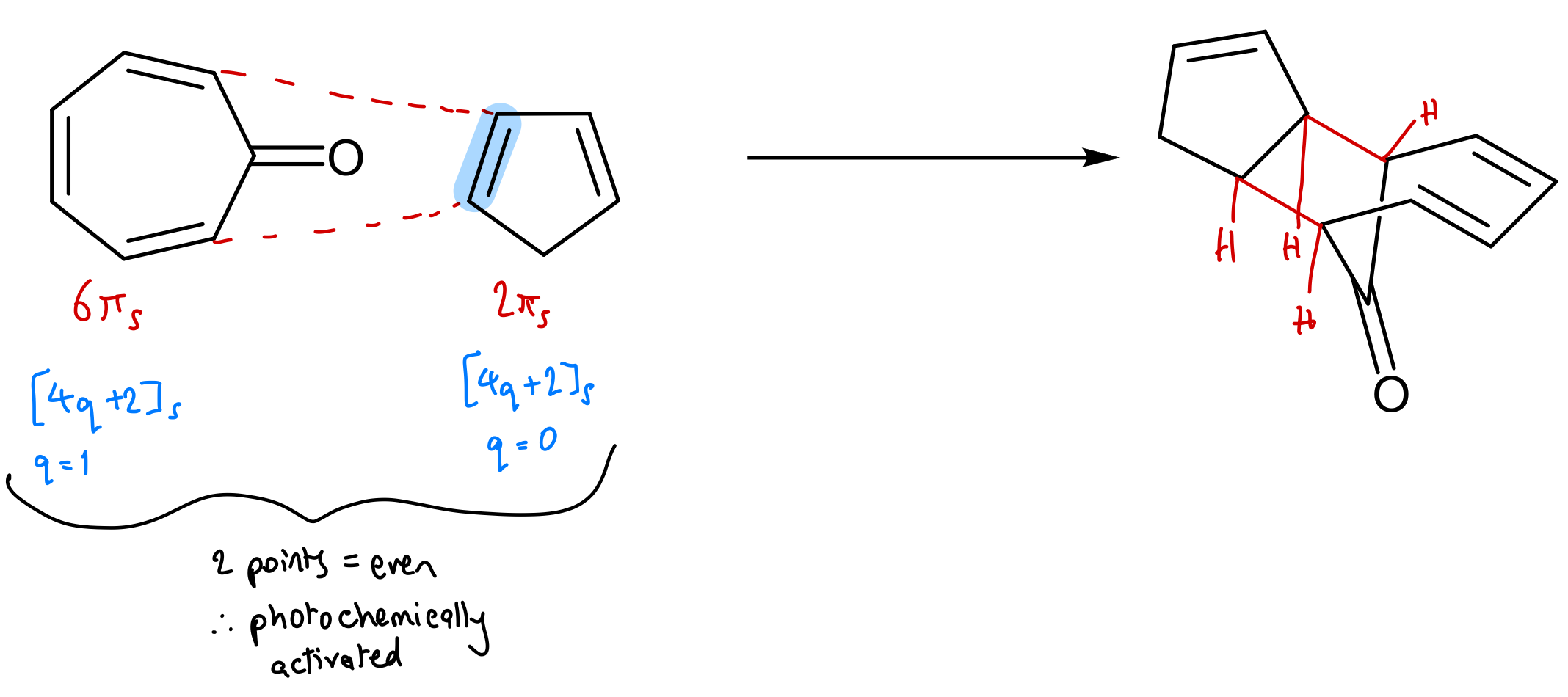
Show validation of WH rules prediction using FMO theory for this reaction:
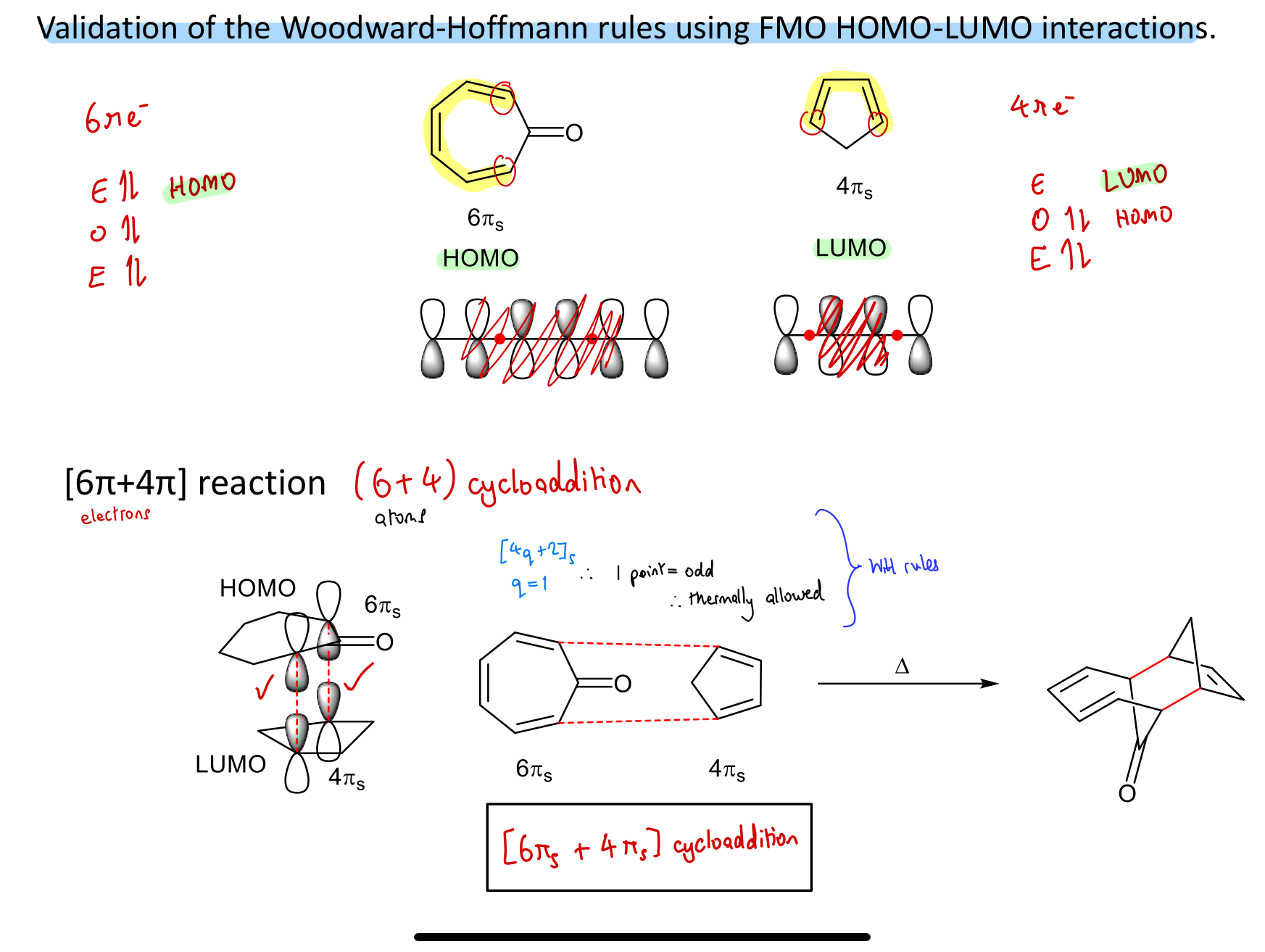
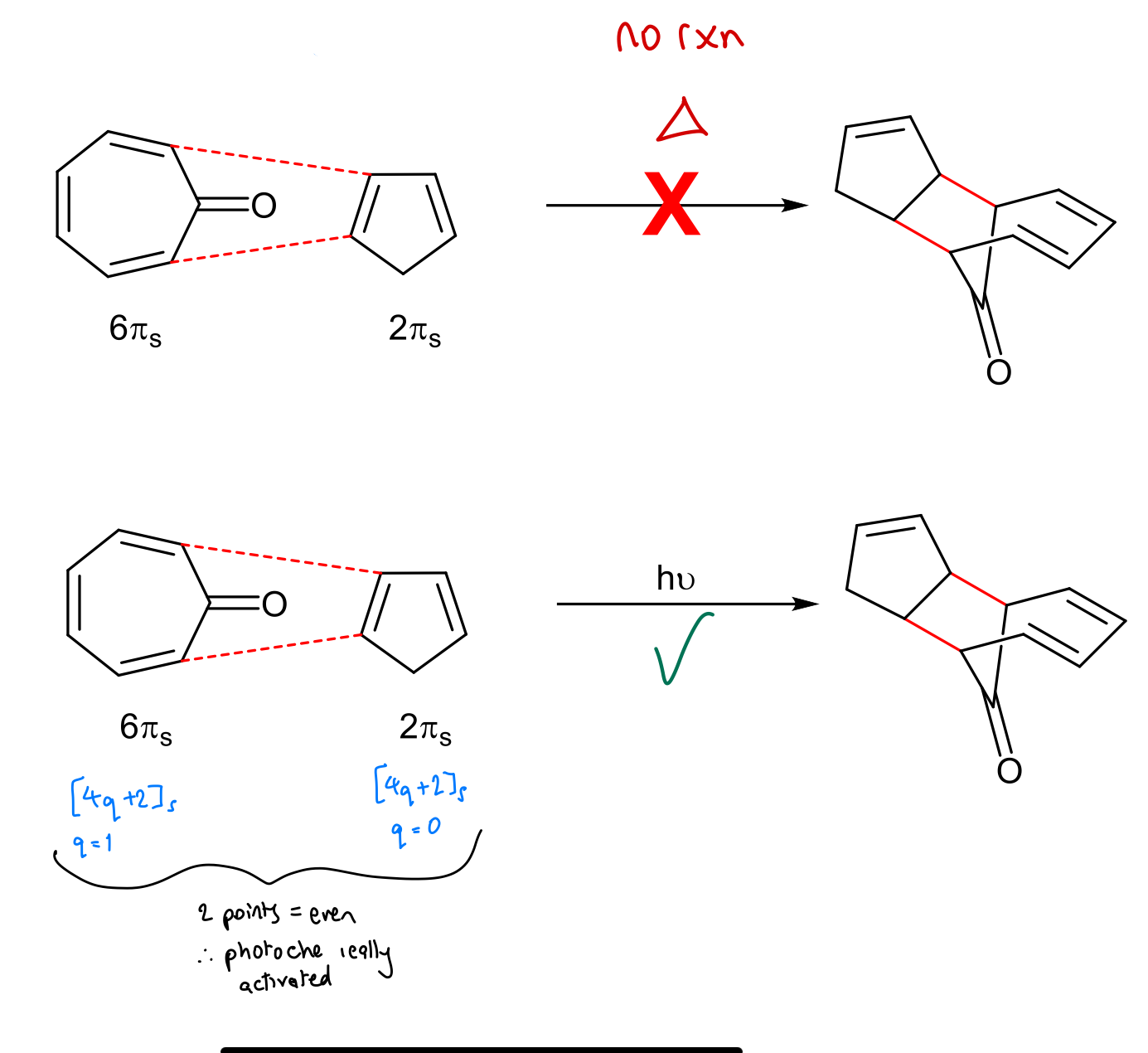
Show validation of WH rules prediction using FMO theory for this reaction:
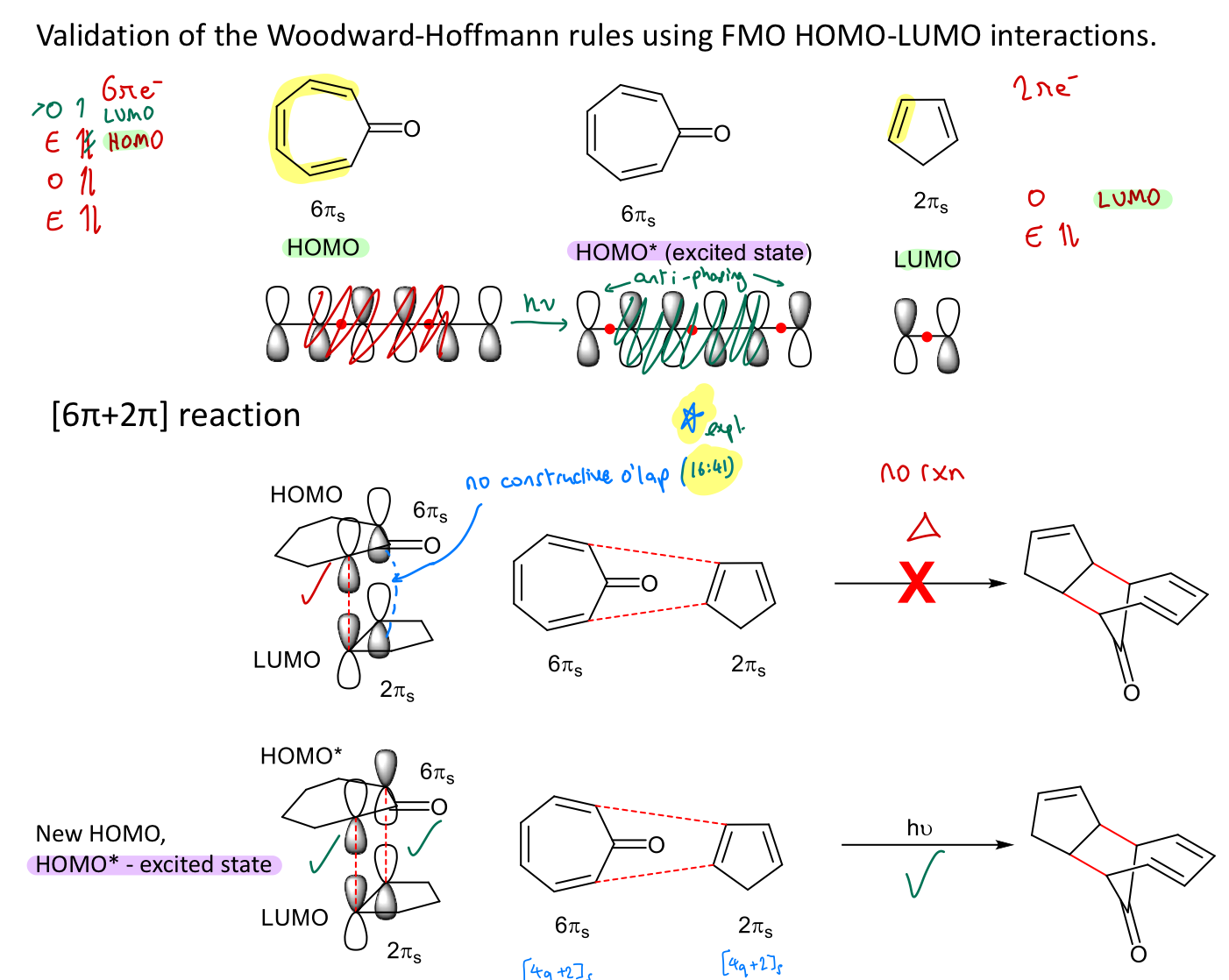
Are [2π + 2π] cycloaddition reactions thermally or photochemically allowed?
Why? Explain using W-H rules and FMO rationalisation.
Is 0 points in WH rules odd or even?
Even
How are Diels-Alder rxns a valuable source of 1,6-dicarbonyls in respect to retrosynthesis?
Obtained when used in conjunction with a second stage ozonolysis.
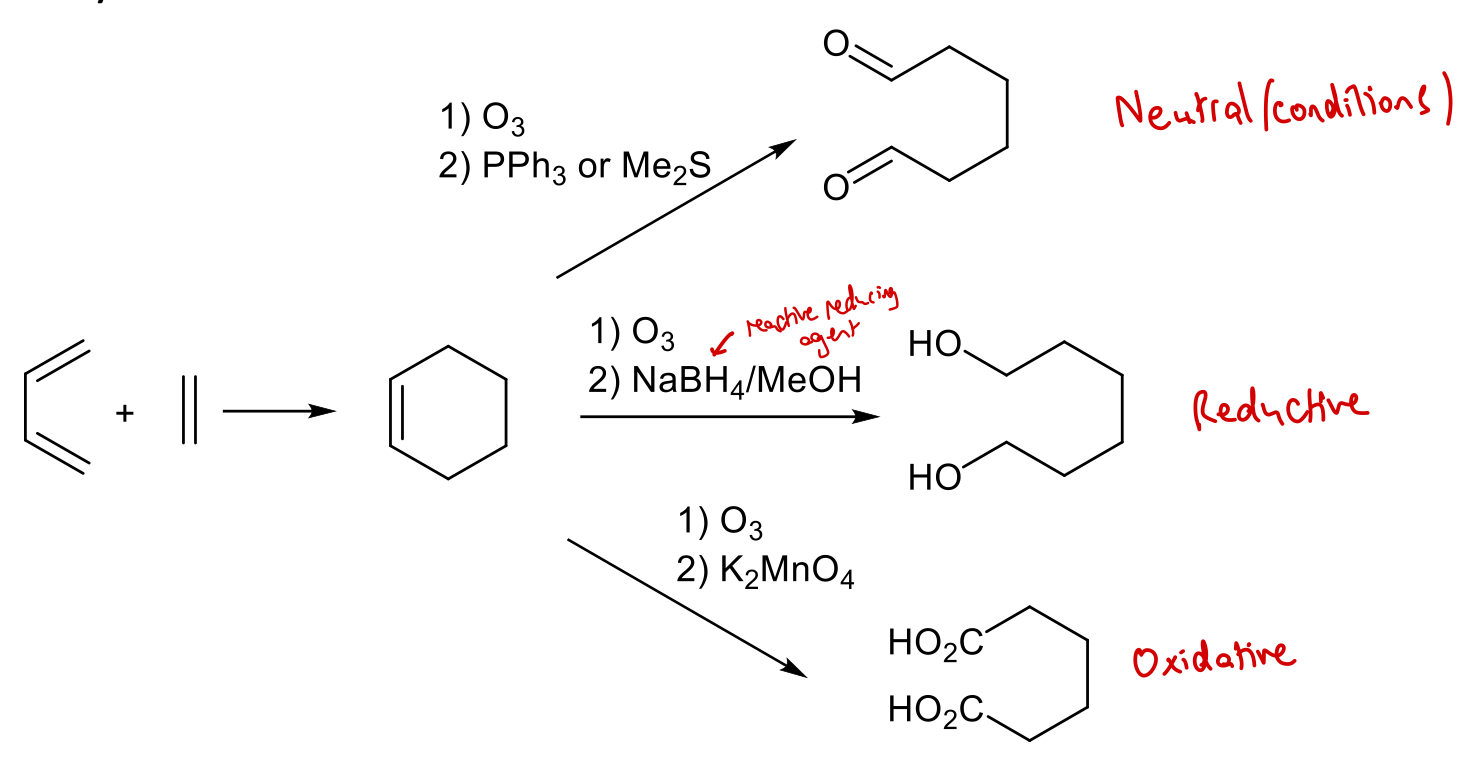
State reagents required to obtain 1,6-dicarbonyl under neutral conditions using Diels-Alder rxn followed by 2nd stage ozonolysis.
1) O3
2) PPh3 or Me2S
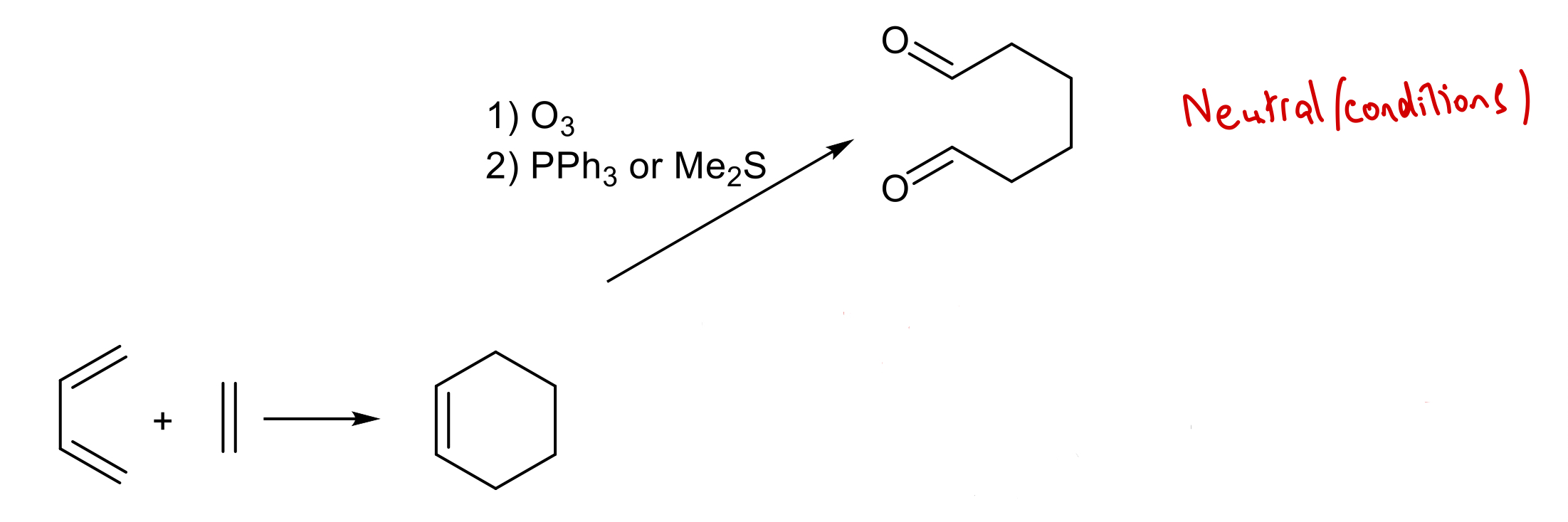
State reagents required to obtain 1,6-dicarbonyl under reductive conditions using Diels-Alder rxn followed by 2nd stage ozonolysis.
1) O3
2) NaBH4/MeOH
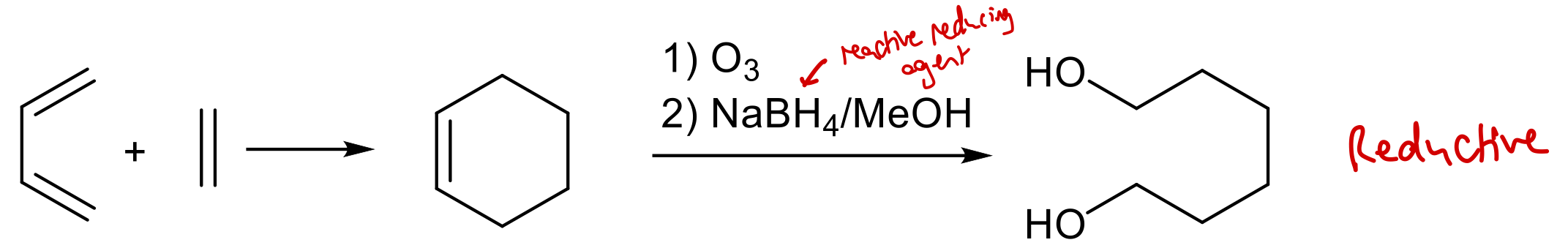
State reagents required to obtain 1,6-dicarbonyl under oxidative conditions using Diels-Alder rxn followed by 2nd stage ozonolysis.
1) O3
2) K2MnO4
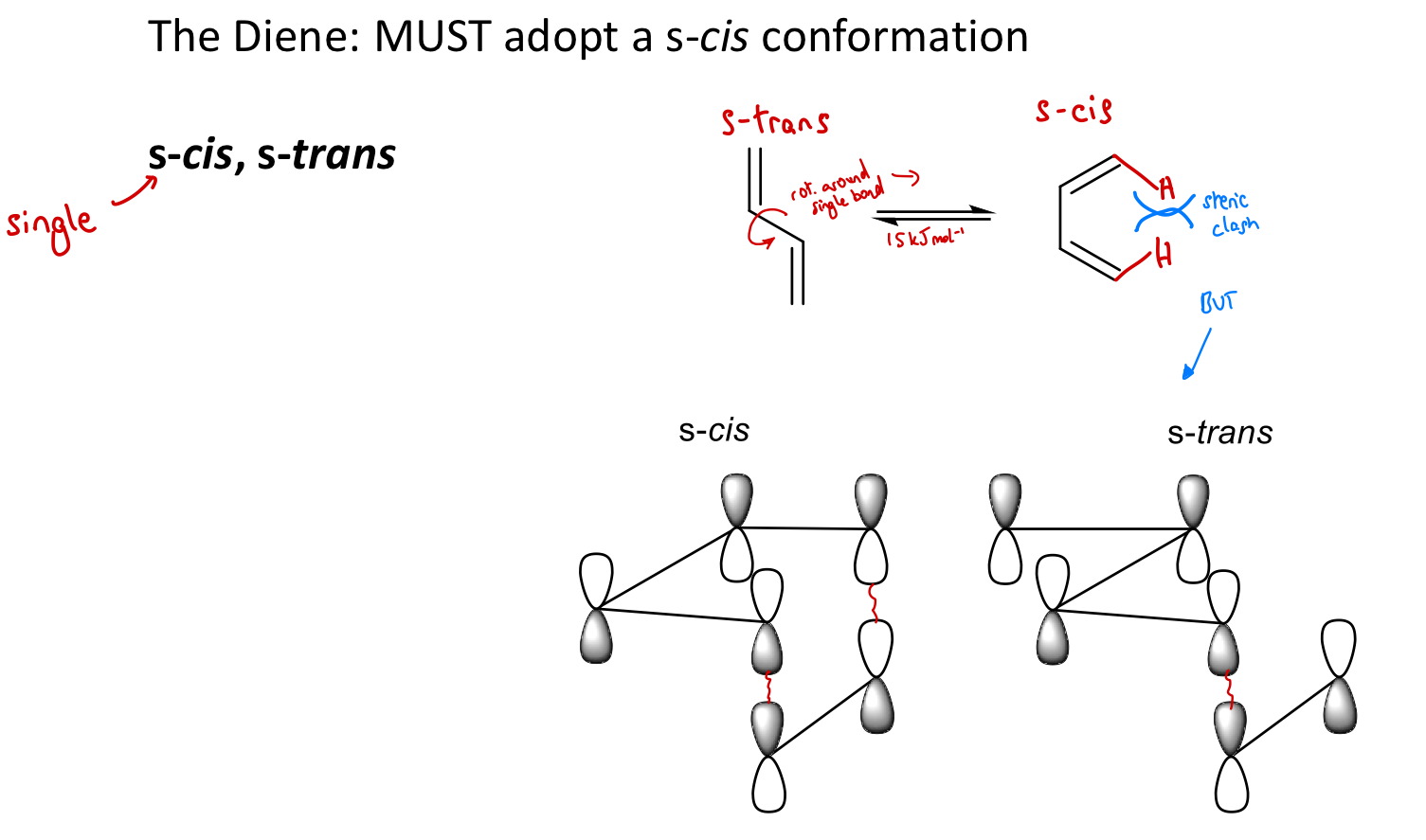
Why must the diene adopt an s-cis conformation in DA reactions, even though eqm favours diene s-trans conformation?
Ends of diene are too far apart to react w/ dienophile in s-trans conformation - poor overlap of MOs with those of the dienophile.
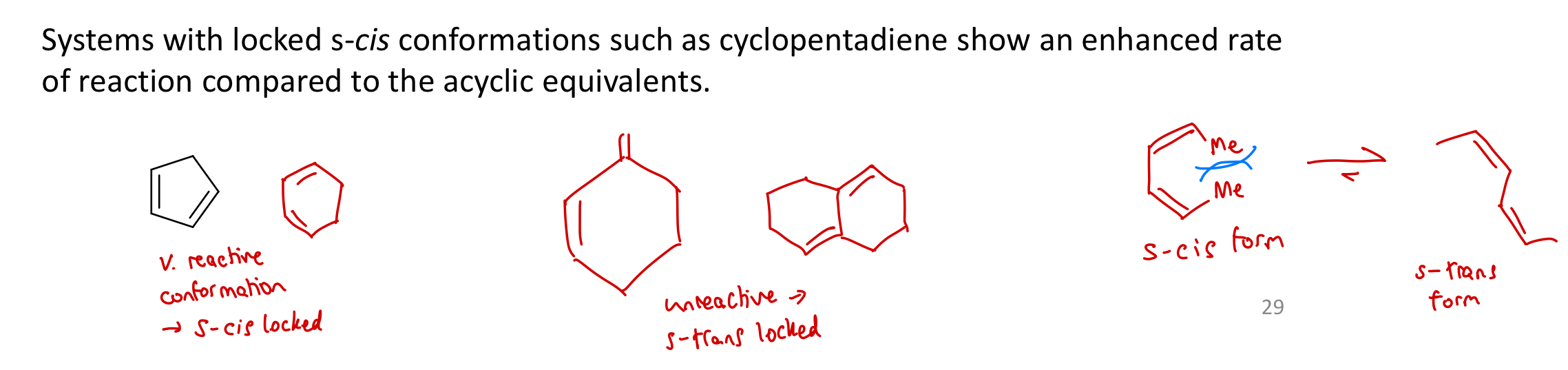
Which systems react at the fastest rate in DA reactions?
Systems with locked s-cis conformations, e.g. cyclopentadiene (compared to acyclic equivalents.
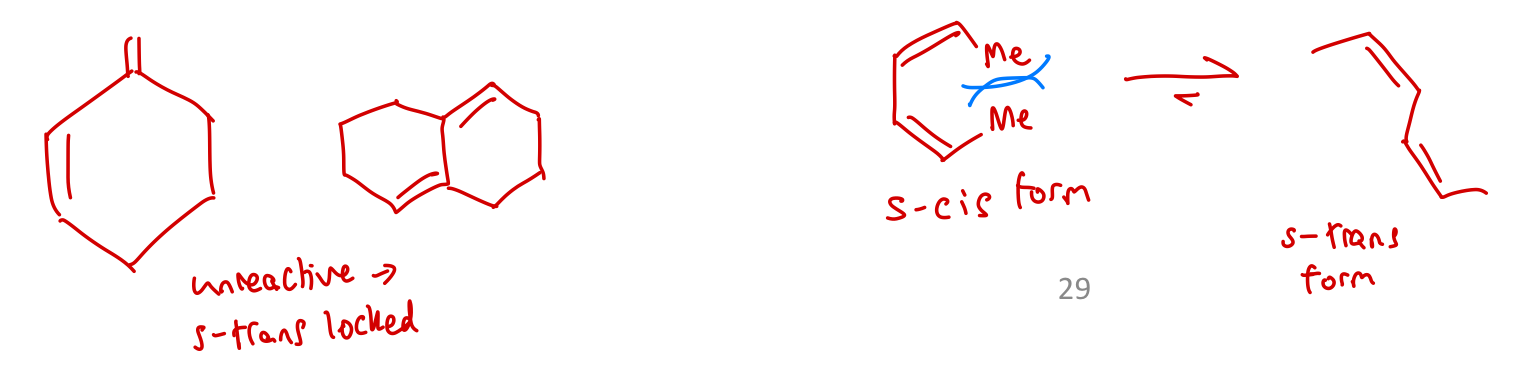
Will s-trans locked systems undergo DA reactions?
Nope!
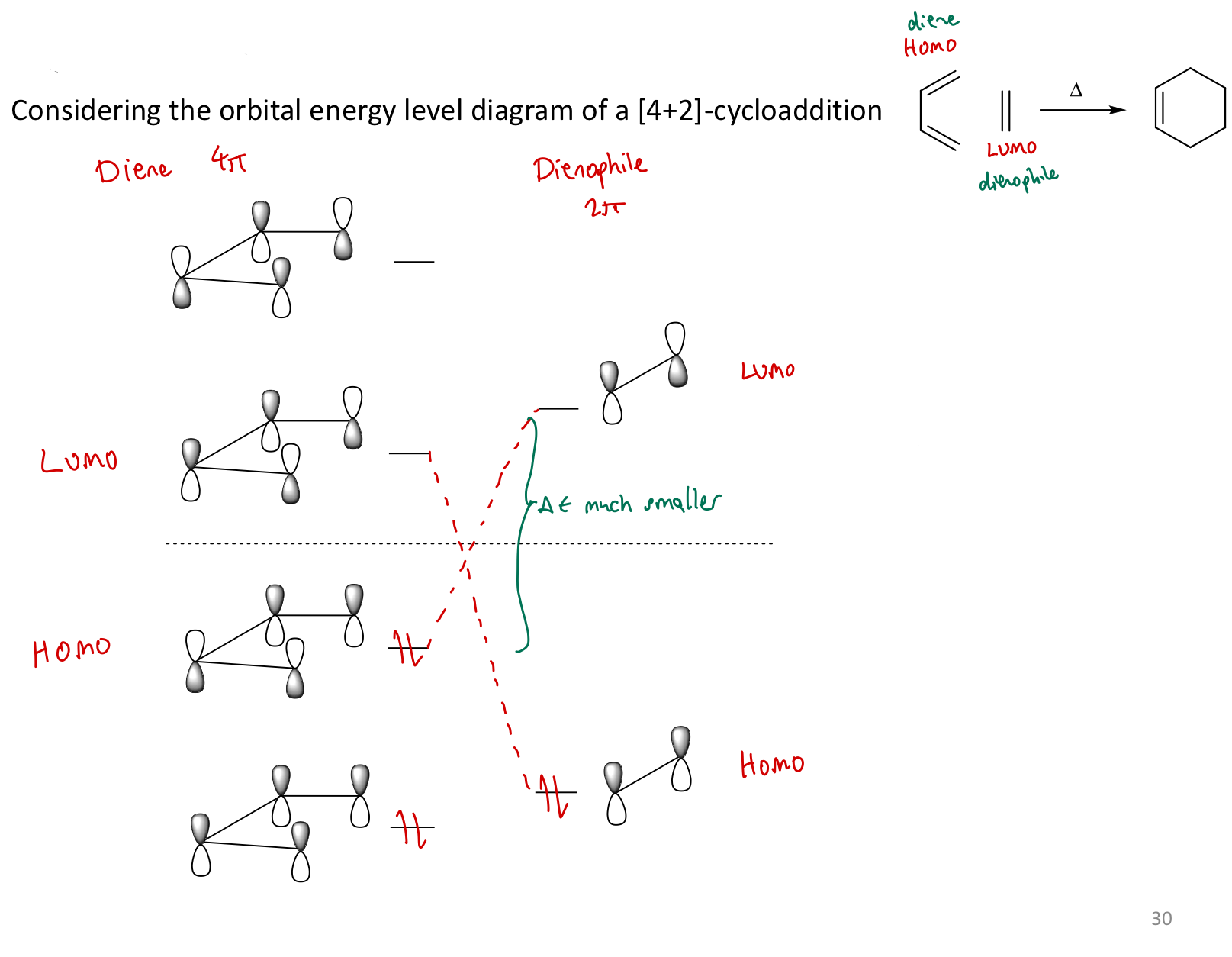
Which MOs do we consider on the diene and dienophile respectively, and why?
HOMO on diene
LUMO on dienophile
ΔE smaller than the other way round
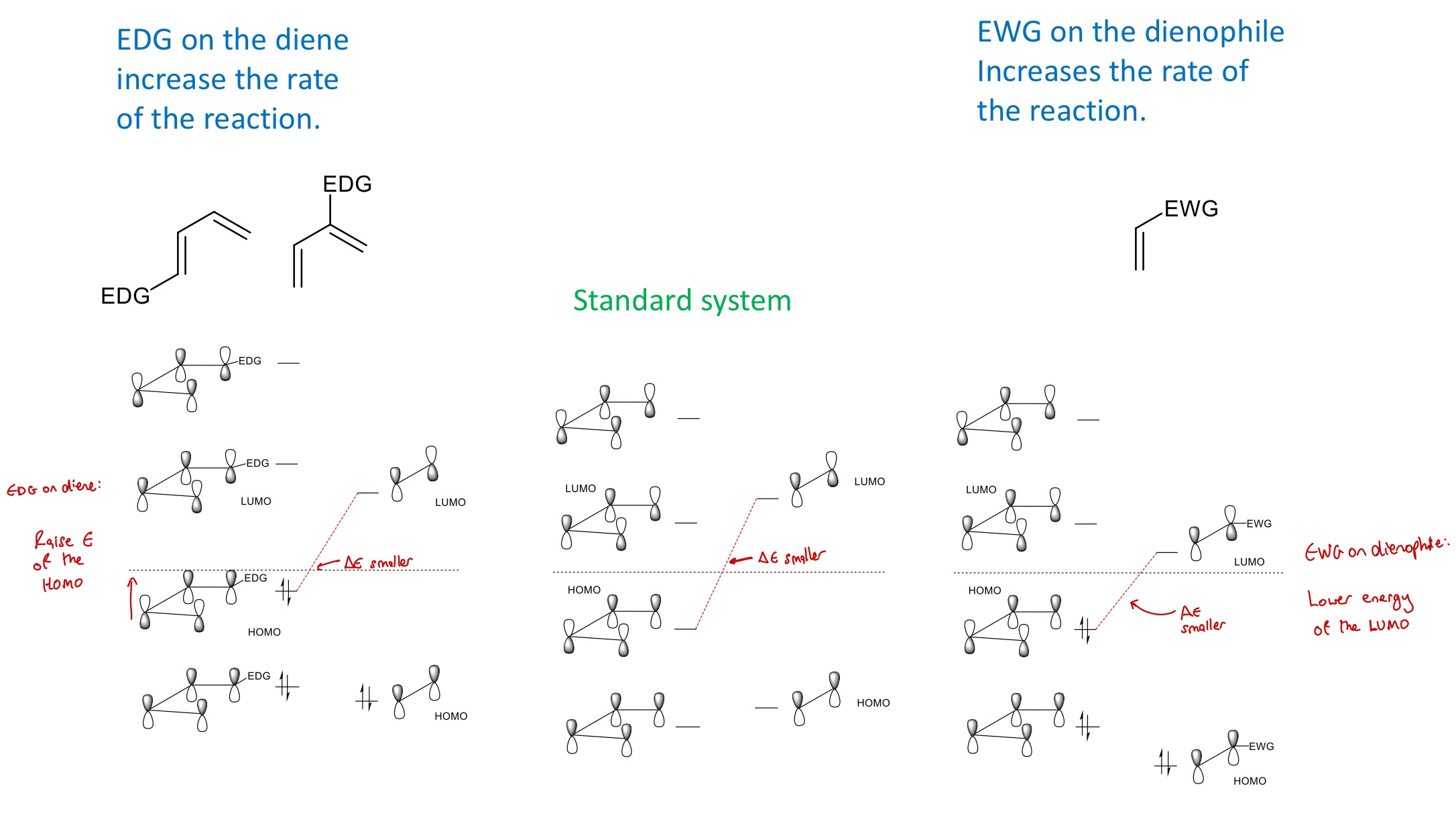
How can we increase ROR in DA reactions via substituent effects?
EDG on diene: raises E of HOMO
EWG on dienophile: lowers E of LUMO
Why is cyclopentadiene super reactive?
It’s s-cis locked!!
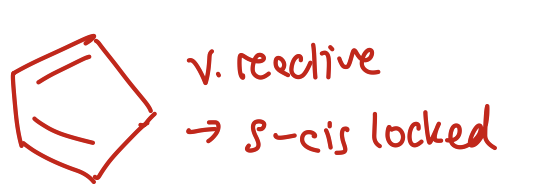
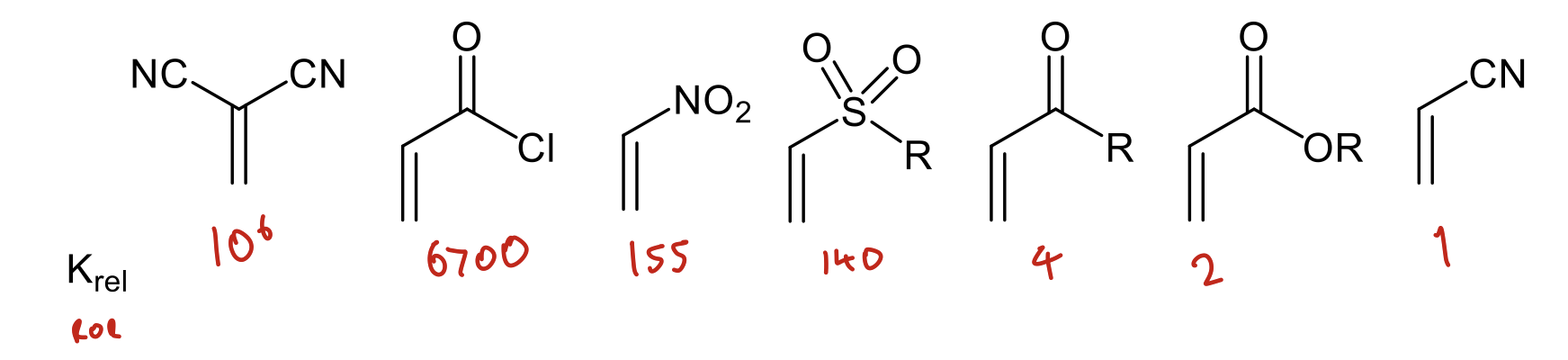
How does the ROR compare with different groups when reacting with cyclopentadiene in DA reaction?
Increases with stronger EWGs
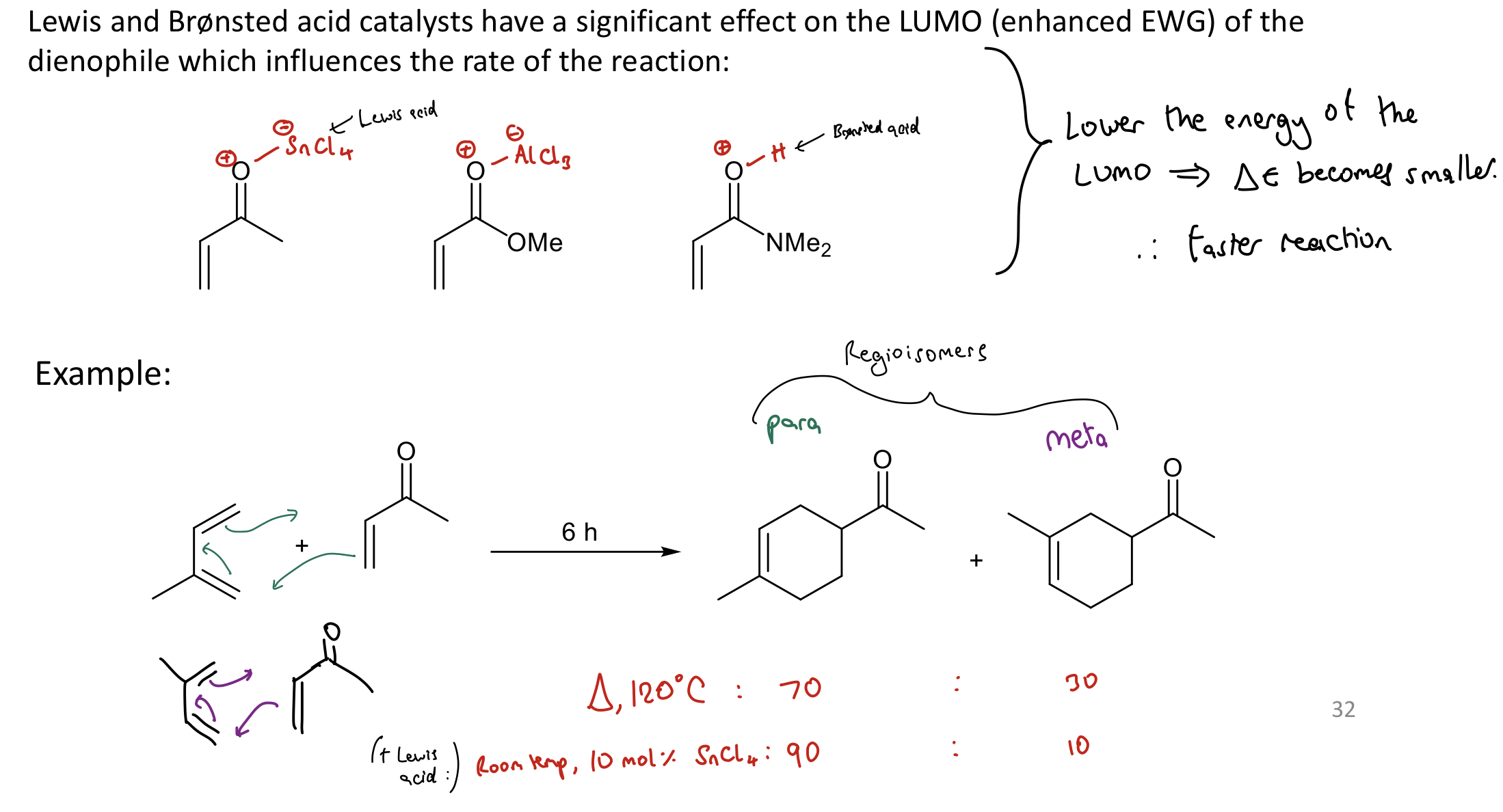
How do Lewis and Brønsted acid catalysts affect DA reactions?
Lower E of the dienophile LUMO
→ smaller ΔE → faster reaction
What’s the purpose of resonance descriptors for cycloaddition rxns?
Help predict regioselectivity by looking at charge separation.
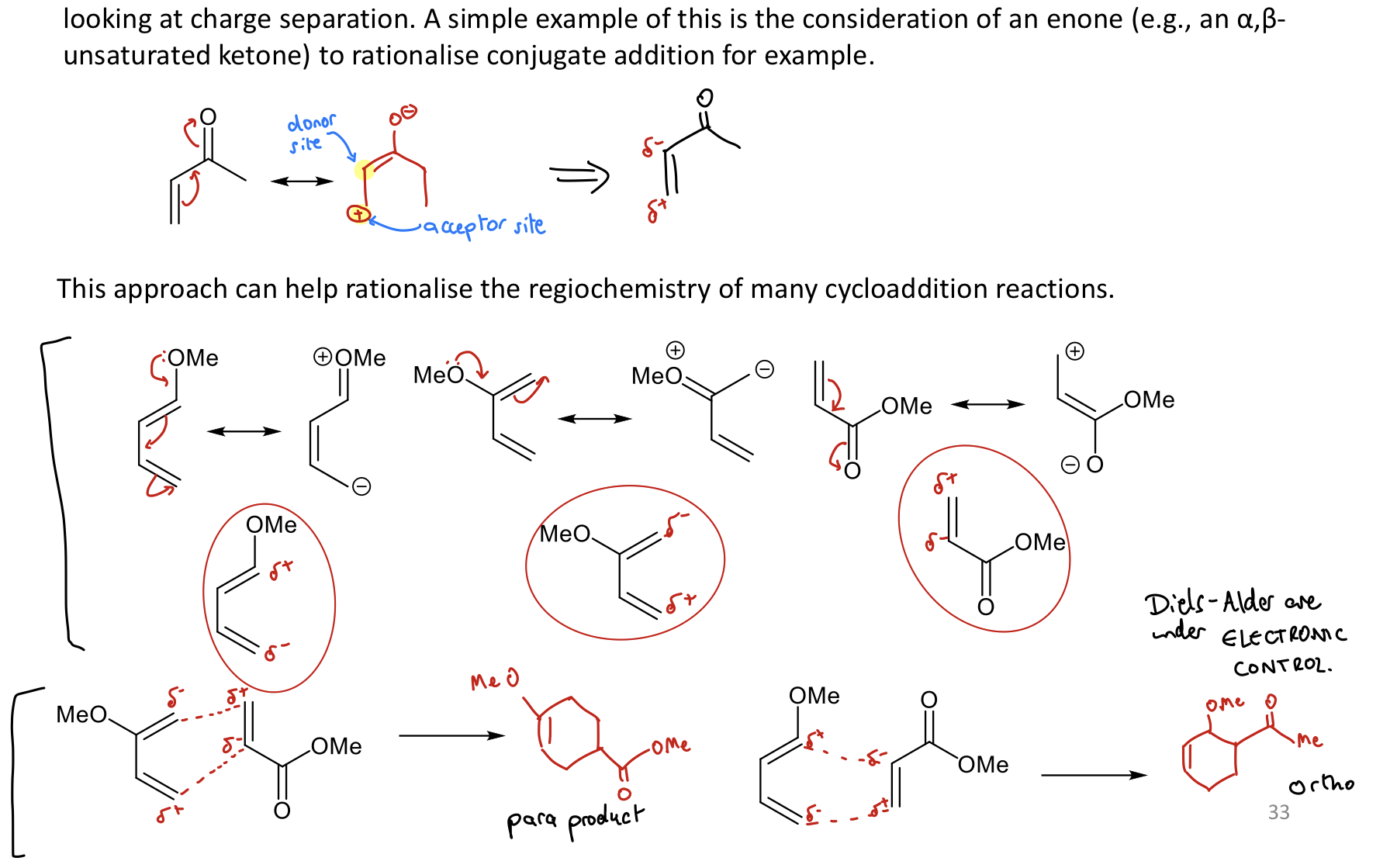
What do we use when resonance descriptors don’t work for DA reactions (10% of the time)?
MO coefficients of diene & dienophile
match up largest coefficients
Biggest MO coefficients = better overlap & bond formation
Can use regioselectivity tables to help
What does endo/exo selectivity represent in DA reactions?
Kinetic (endo) versus thermodynamic (exo) control
2 possible diastereoisomers formed (endo and exo)
Endo-cycloadduct often kinetically favoured
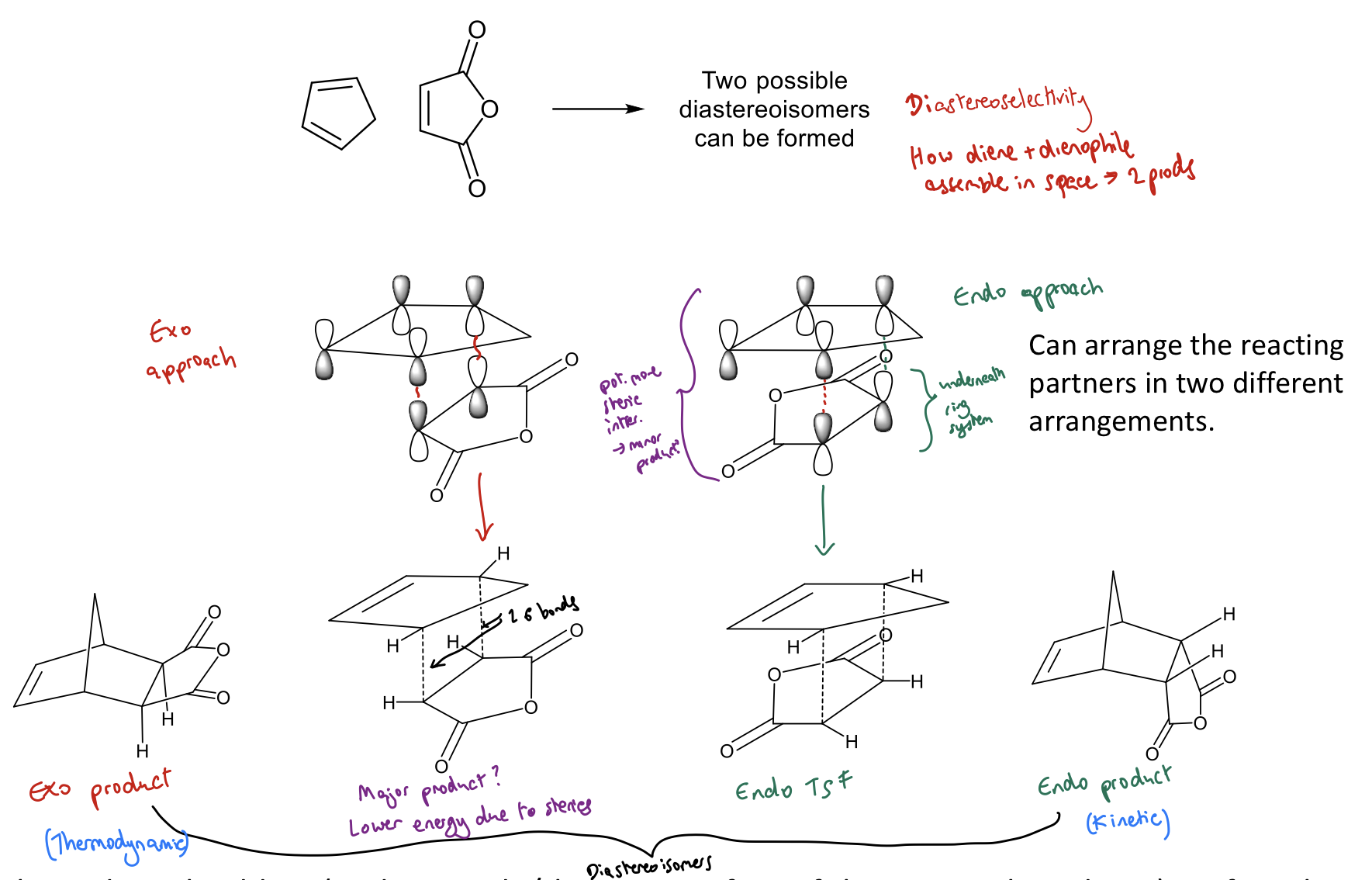
Why is the endo-cycloadduct favoured in DA reactions?
Due to secondary orbital interactions, which:
stabilise transition states → lower their energy → make them kinetically-favoured
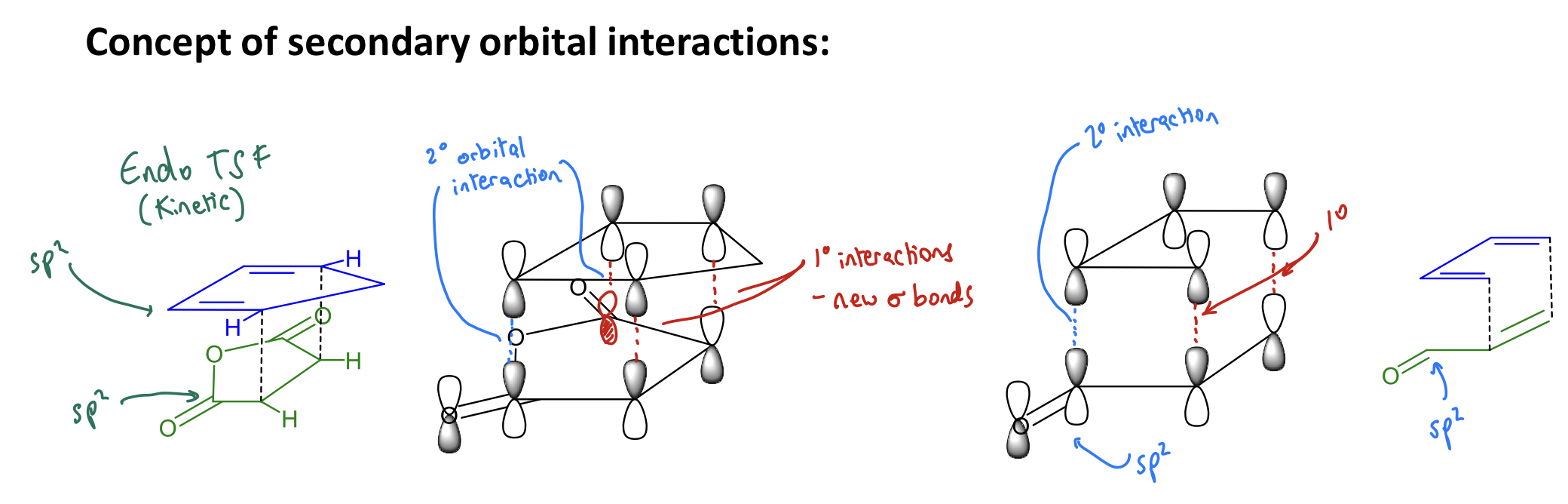
Show FMO evidence for 2ndary orbital interactions
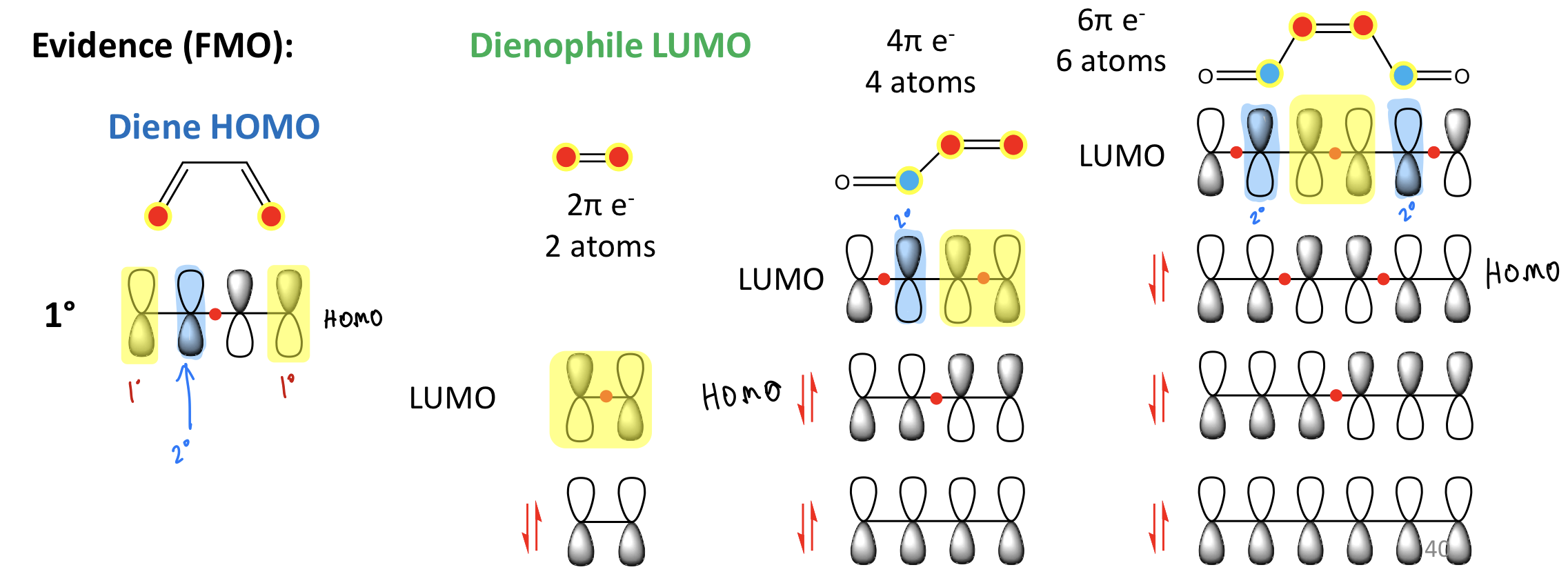
Drawing convention for determining stereochemical result of cycloaddition process
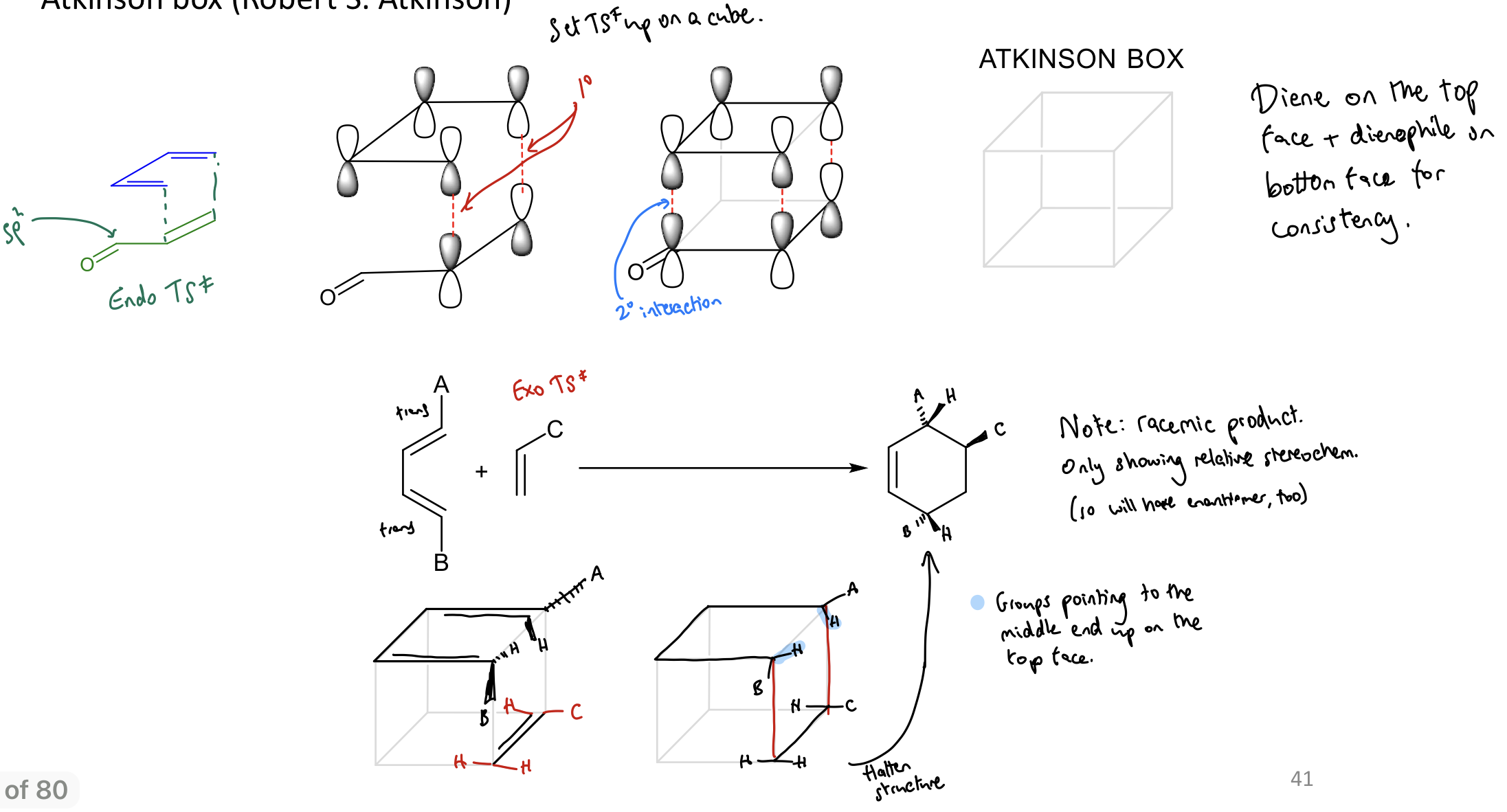
Use Atkinson box to draw TS
Diene on TOP; dienophile on bottom
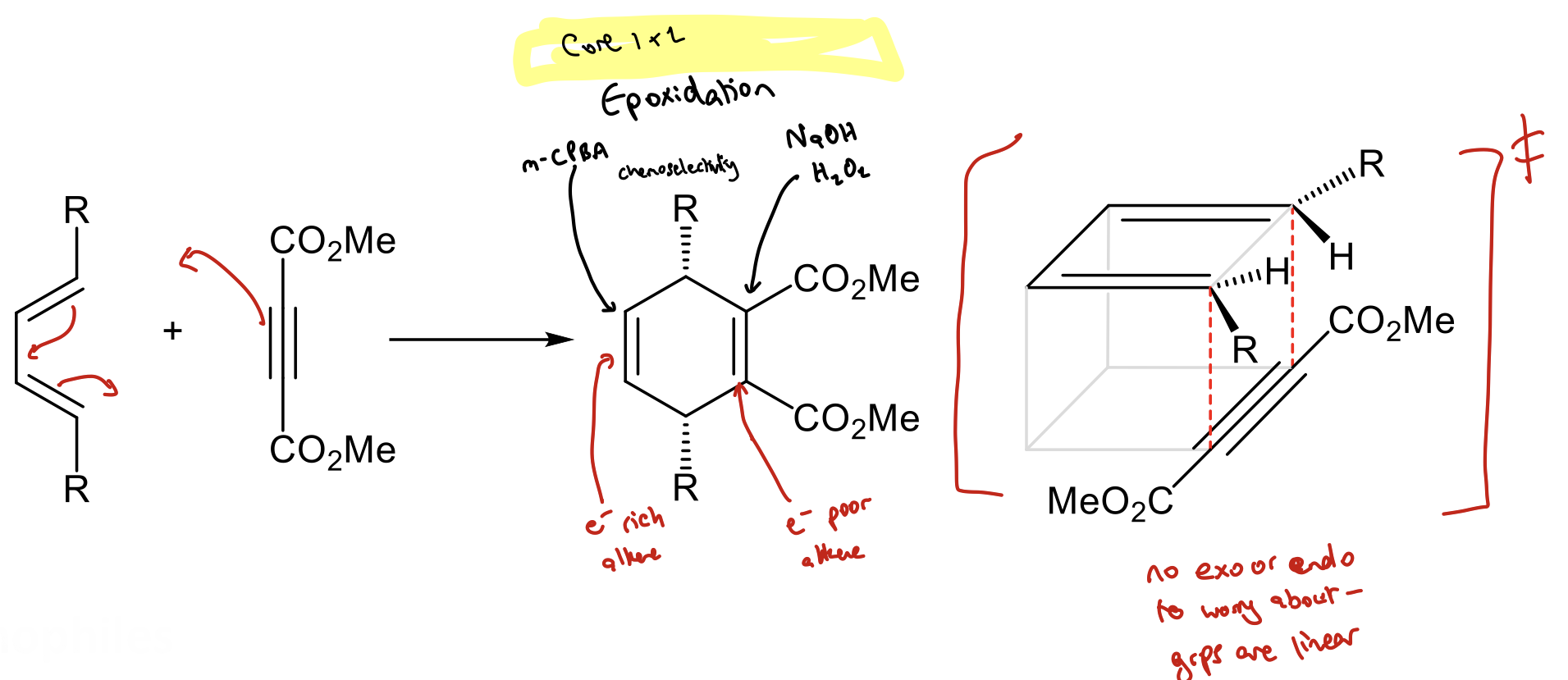
When do we not have to worry about endo/exo selectivity with cycloadditions?
When dienophile is linear
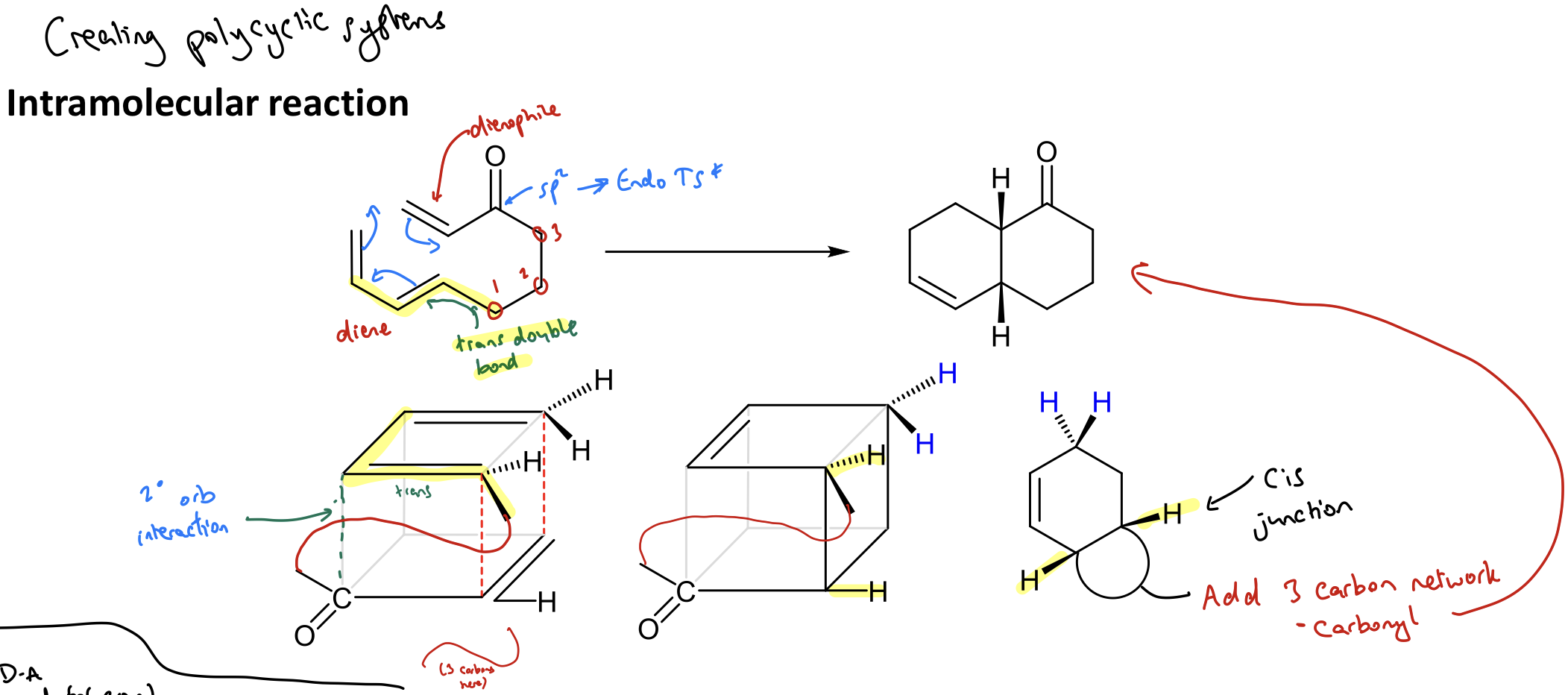
Show an example of creating a polycyclic system using an intramolecular reaction
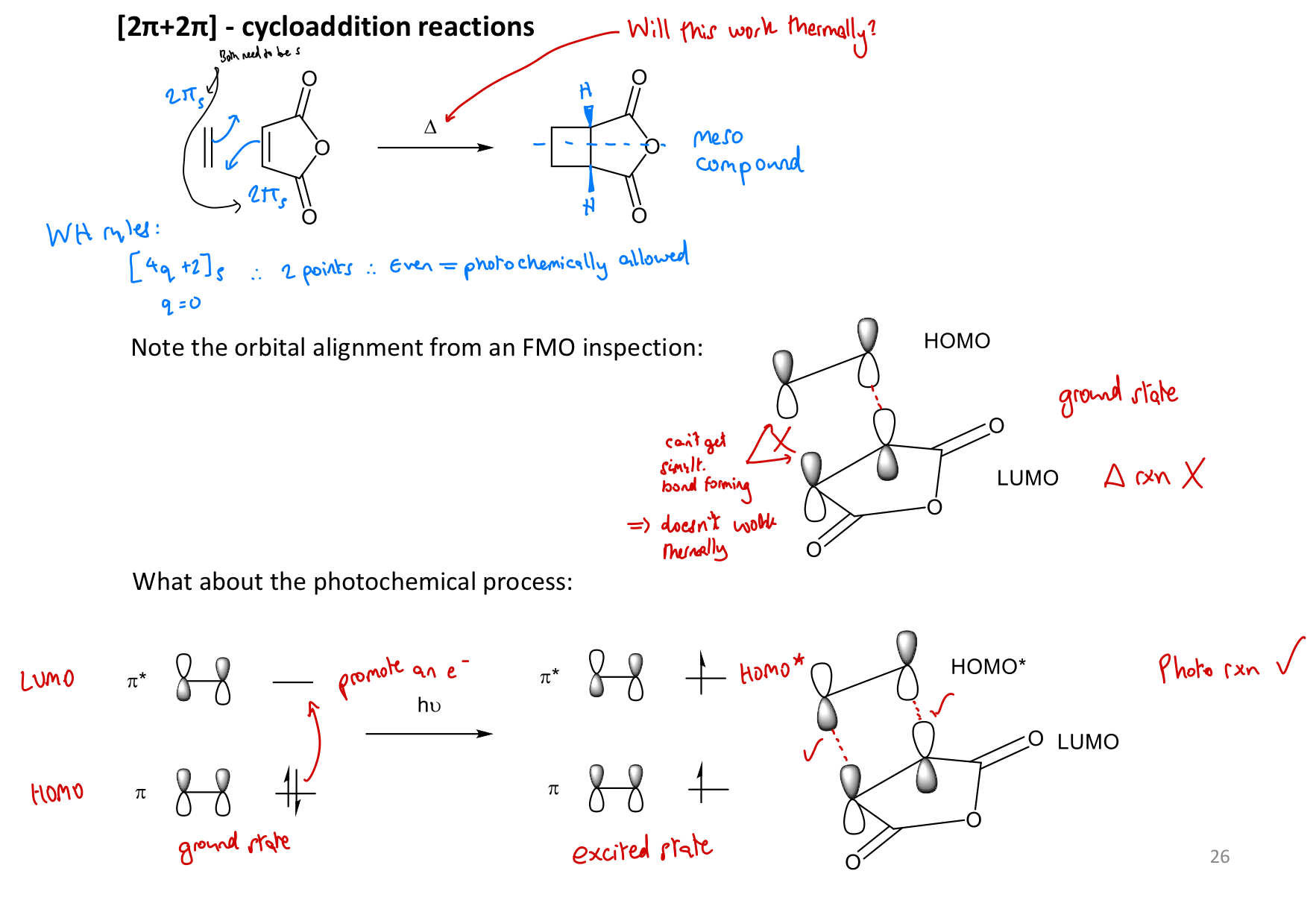
Why is a [2+2] cycloaddition thermally disallowed?
Mismatch of orbitals in HOMO-LUMO interactions
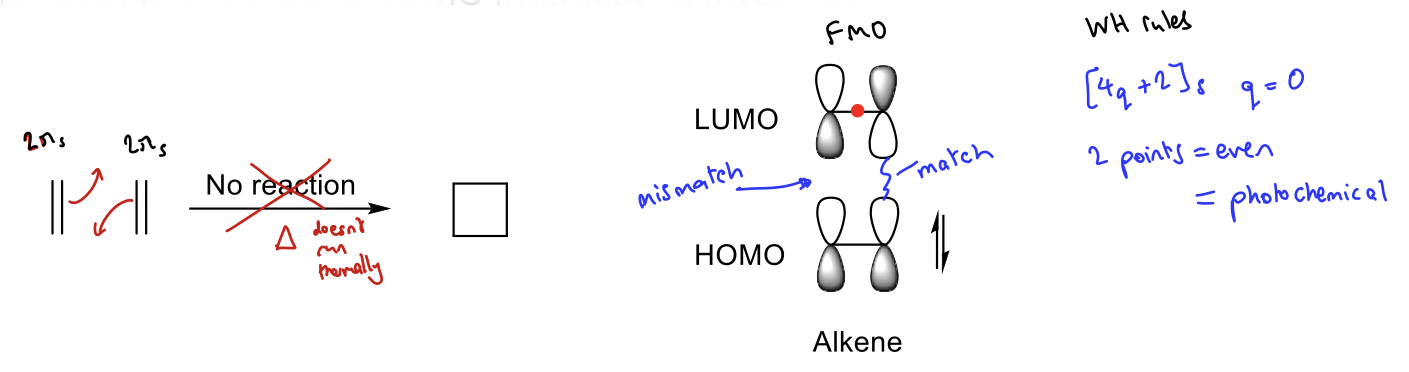
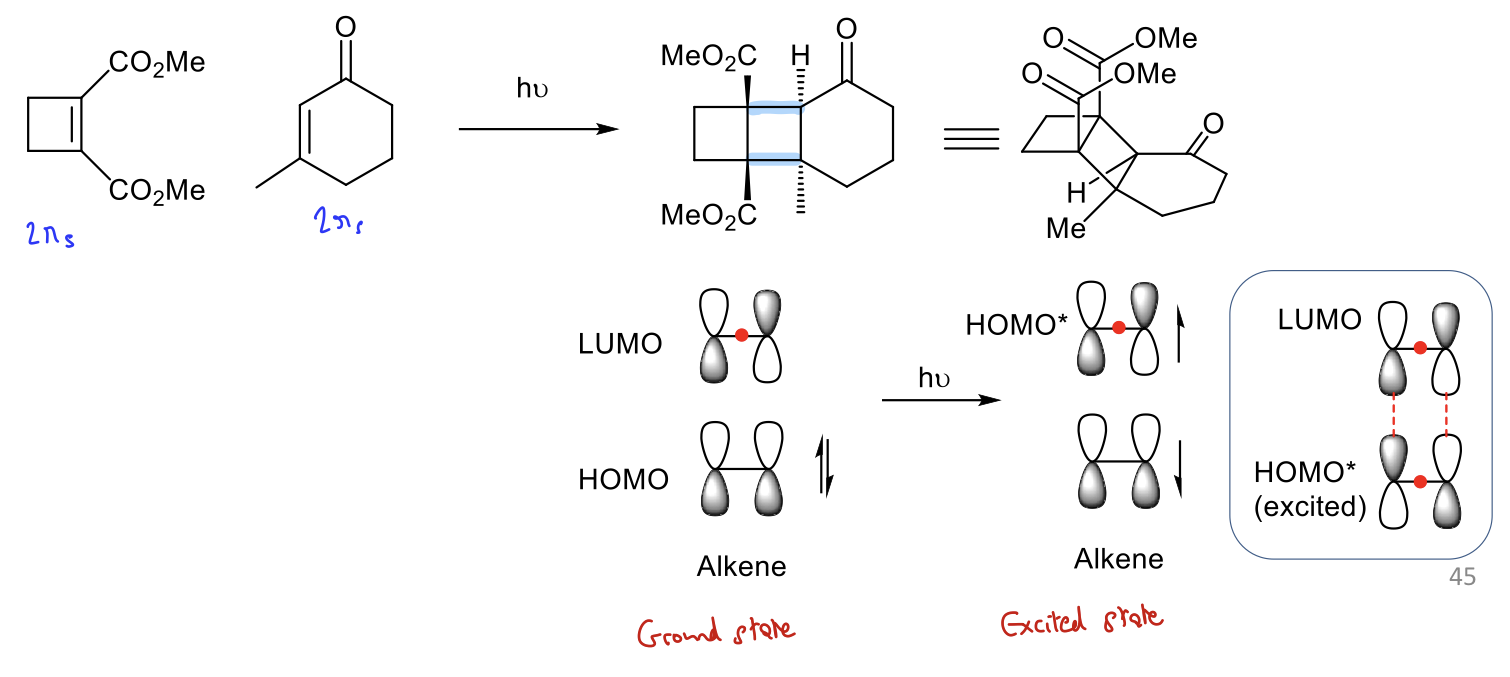
Show how [2+2] cyclisation reactions can be promoted photochemically. (FMO)
Which [2+2] cycloadditions are thermally allowed, and why?
Ones involving systems with cumulated double bonds, such as ketenes.
(See pg 47 for 2 more examples of ketene [2+2] cycloadditions.]
![<p>Ones involving systems with <strong>cumulated double bonds</strong>, such as <strong>ketenes</strong>.</p><p>(See pg 47 for 2 more examples of ketene [2+2] cycloadditions.]</p>](https://assets.knowt.com/user-attachments/8e4de6fa-54c6-415c-b482-c1372d42e37f.png)
Which other species can undergo similar [2+2] cycloadditions to the ketene ones?
Species which resemble ketenes e.g. isocyanates
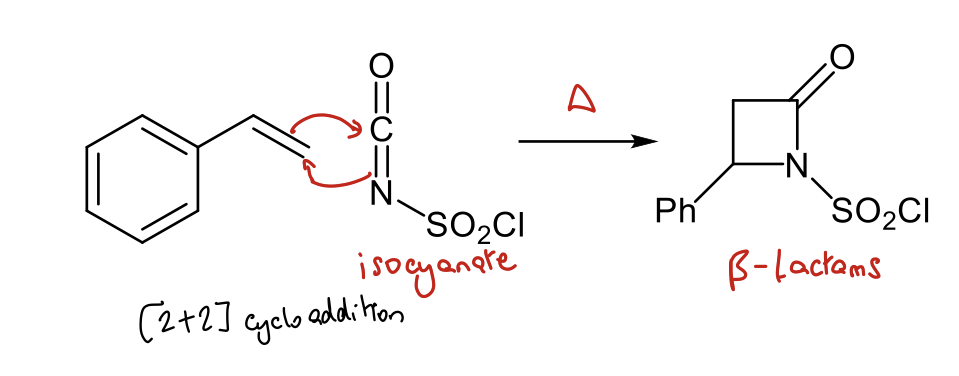
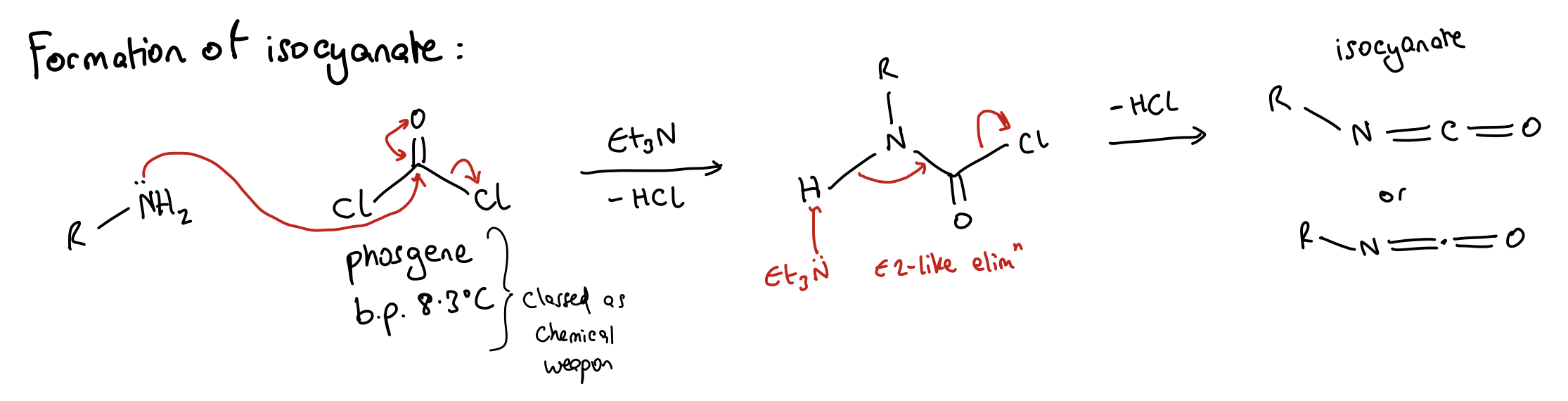
How is isocyanate formed?
Amine + phosgene with Et3N