Organic Chemistry 2 Final Exam Review
1/46
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
47 Terms
H2 (excess), Pd-c
reduces c=o to alcohols
reduces alkenes to alkanes
2 H2, Pd-c
reduced alkynes to alkanes
H2 (1 equiv), Pd-c
ONLY reduces alkenes to alkanes
1) LiAlH4 (LAH)
2) H2O
reduces everything except alkenes
reduces aldehydes to alcohols
reduces ketones to alcohols
reduces acid chlorides and esters to alcohols
reduces carboxylic acids to alcohols
reduces amides to amines
reduced alkyl halides(RX) to alkanes(RH)
opens epoxides at less substituted carbon (reduces epoxides to alcohols)
NaBH4, CH3OH
does not reduce alkenes to alkanes
ONLY reduced aldehydes and ketones to alcohols
very selective
Witting Reagents (Ph3P=CR2)
adds carbon-carbon double bond
selective to just aldehydes and ketones
converts c=o to c=c
H2O, H+ or -OH, delta
-turns CN to carboxylic acids
NaCN, HCl
adds CN as a nucleophile to a ketone or aldehyde to give a CN and an alcohol
does Sn2 nucleophilic attack
1) R”2CuLi 2)H2O (organocupurates)
does not react with ketones, aldehydes, or esters
reacts with acid chlorides to give a ketone product
reacts with alpha-beta unsaturated ketones at the beta carbon giving 1,4 addition
1) DIBAL-H 2)H2O
selective reducing agent
reduces acid chlorides and esters down to aldehydes
no rxn with carboxylic acids
1)R”MgBr 2)H2O (grinard)
reacts with aldehydes and ketones as Nu, creating an alcohol and adding carbons
reacts with acid chlorides and esters to give alcohols (2 equivs will give tertiary alcohols)
react with alpha-beta unsaturated ketones at the carbonyl carbon, giving 1,2 addition
1)R”Li 2)H2O (organolithiums)
-act as nucleophile with ketones and aldehydes, affording an alcohol and adding carbons
-turns acid chlorides and esters to alcohols and adds a carbon chain ( 2 equivs would give a tertiary alcohol)
reacts with alpha-beta unsaturated ketones at the carbonyl carbon to give 1,2 addition
Ag2O, NH4OH (tollens reagent)
-selective for turning aldehydes into carboxylic acids
CrO3, H2SO4, H2O (Jones)
-turns primary alcohols into carboxylic acids
-turns secondary alcohols into ketones
-turns aldehydes into carboxylic acids
*know mech for secondary alcohol to ketone
*know mech for primary alcohol to carboxylic acid (get to aldehyde then use chapter 18 to get to a hydrate then react with chromate again to get carboxylic acid)
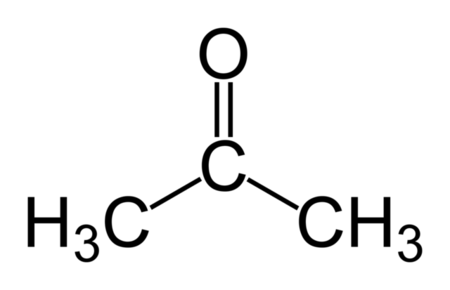
acetone
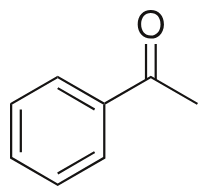
acetophenone
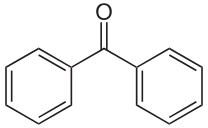
benzophenone
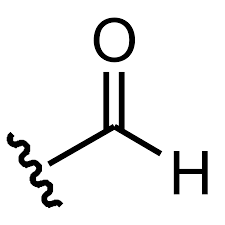
formyl substituent
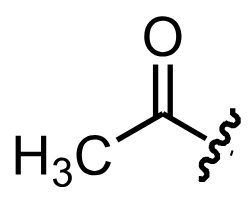
acetyl substituent
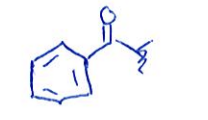
benzyl (Bz) substituent
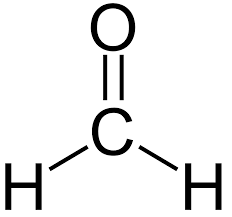
formaldehyde
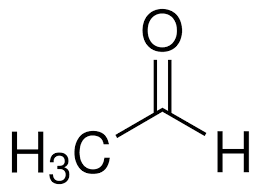
acetaldehyde
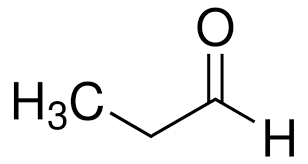
propionaldehyde
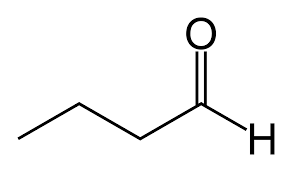
butyraldehyde
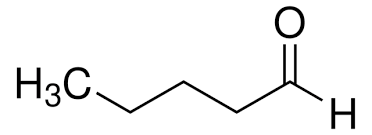
valeraldehyde
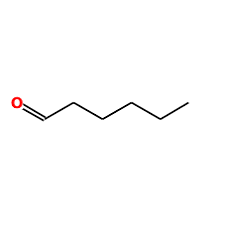
caproaldehyde
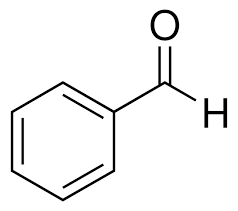
benzaldehyde
1) BH3 (R2BH) 2) H2O2, -OH
react with terminal alkynes to afford an aldehyde
H2O, H2SO4, HgSO4
react with terminal alkynes to afford a ketone
TBS-Cl, base
protecting group for OH, turns OH to OTBS
TBAF
deprotecting step for OH, turns OTBS back to OH
1) RNH2, mild acid 2)loss of H2O
reaction of primary amines to form an imine product
The intermediate is carbinolamine
this is reversible
*know mech
1) R2NH, mild acid 2) loss of H2O
reaction of secondary amines to form enamines
reversible reaction
intermediate is carbinoalmine
*know mech
H3O+ or H2O, H+
-does hydrolysis of imines back to a ketone/aldehyde and primary amine or enamines back to an aldehyde/ketone and secondary amine
-does hydrolysis of acetals back into a ketone/aldehyde and an alcohol (this process can also use H2SO4 and H2O)
H2O, H+ or -OH
-addition of H2O (hydration reaction)
-makes a gem-diol (hydrate) from a ketone or aldehyde
-can occur in basic or acidic condition
*Know both basic and acidic mech
1st step in basic conditions with hydration
nu attack at carbonyl
1st step in acidic conditions of hydration
Acid protonates the carbonyl oxygen
2 eq of an alcohol (or a diol) and H+ (usually in form of TsOH)
-addition of alcohol to form an acetal from a ketone or aldehyde
-an acetal is a molecule with a R group, a group that is a H or a R, and two OR groups
-removal of water or addition of excess alcohol drives this reaction to from the products
one equivalent of an alcohol with TsOH
-forms a hemiacetal from an aldehyde or ketone (very unstable)
-cycle hemiacetals that from 5 or 6 membered rings tho are very stable
HOCH2CH2OH and H+
acetals as protecting groups (turn ketones and aldehydes into an acetal to protect them from the desired reaction)
H+ and H2O
deprotecting step of acetals, turn them back into their OG aldehyde or ketone
Na, NH3
reduced alkynes to trans alkenes
H2, lindlar’s catalyst
reduced alkynes to cis alkenes
mCPBA or RCO3H
makes an epoxide from an alkene
*know this mech
1)mCPBA or RCO3H 2) H2O or -OH
anti-dihydroxylation (adds two OH groups across a double bond, trans to each other)
OsO4 w/ NMO
syn-dihydroxylation (adds two OH groups across a double bond cis to each other)
KMnO4
syn dihydroxylation (adds to OH groups across a double bond, cis too each other)
is a benzylic CH is present, KMnO4 converts alkyl benzenes into benzoic acids (cuts R group/chain off no matter how long and converts to carboxylic acid)