topic 1 chem atomic structure and the periodic table
1/62
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
63 Terms
define relative isotopic mass
the weighted mean mass of an isotope relative to 1/12 of an atom of carbon-12
define relative atomic mass
the weighted mean mass of an atom relative to 1/12 of the mass of an tom of carbon-12
define relative molecular mass
the sum of the relative atomic masses of all atoms in its molecular formula
define isotopes
ATOMS with the same number of protons but different number of neutrons
what is the structure of a 1+ ion of the carbon-13 isotope?
six protons, seven neutrons, five electrons
the relative atomic mass of a sample of magnesium was found to be 24.3.
the % composition for 2 of the 3 isotopes is given below.
use these data to calculate the % composition of the third isotope and hence its relative isotopic mass.
RELATIVE ISOTOPIC MASS FOR (1): 25
PERCENTAGE ABUNDANCE FOR (1): 10
RELATIVE ISOTOPIC MASS FOR (2): 26
PERCENTAGE ABUNDANCE FOR (2): 11.01
% of third isotope: 100 - 10 - 11.01 = 78.99
equation: (78.99a + 25x10 + 26x11.01) / 100 = 24.3
so a = 24.0
state what is meant by the term 'orbital'
- a region within an atom
- that can hold up to two electrons (with opposite spins)
- where there is a high probability of finding an electron
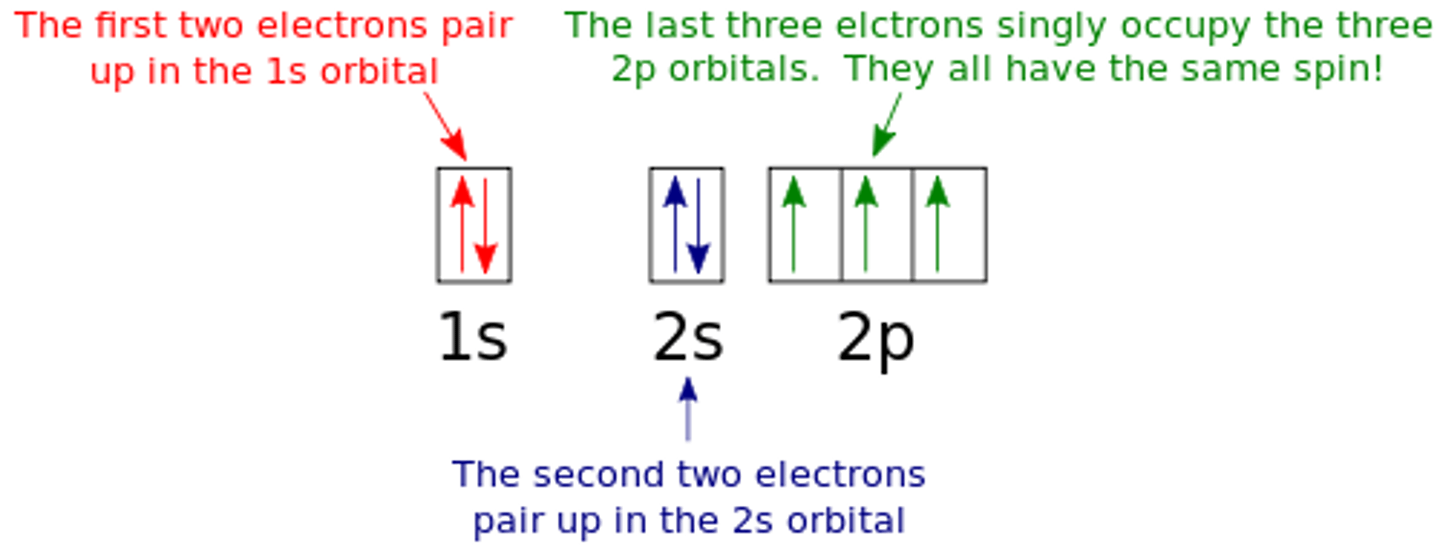
state what is meant by 2 arrows in the same box (1s2)
up and down arrows represents electrons that are in the same orbital with opposite spin
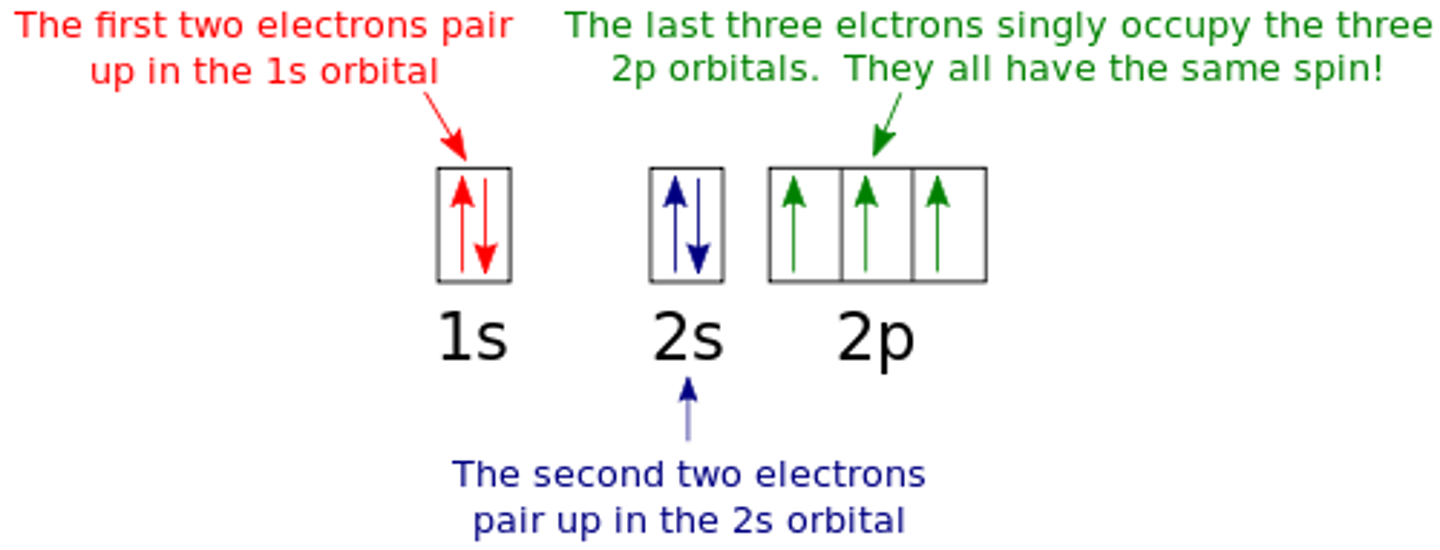
state why the arrows are all pointing in the same direction in the 2p boxes
3 electrons with the same direction of spin as they are all in different orbitals
state the shape of the 2s orbital of a silicon atom
spherical (NOT circular, its 3D)
state the difference between the 2 subatomic particles in a 2s orbital
contains electrons with opposite spin
state the shape of the p orbital and the maximum number of electrons each p orbital can hold
2
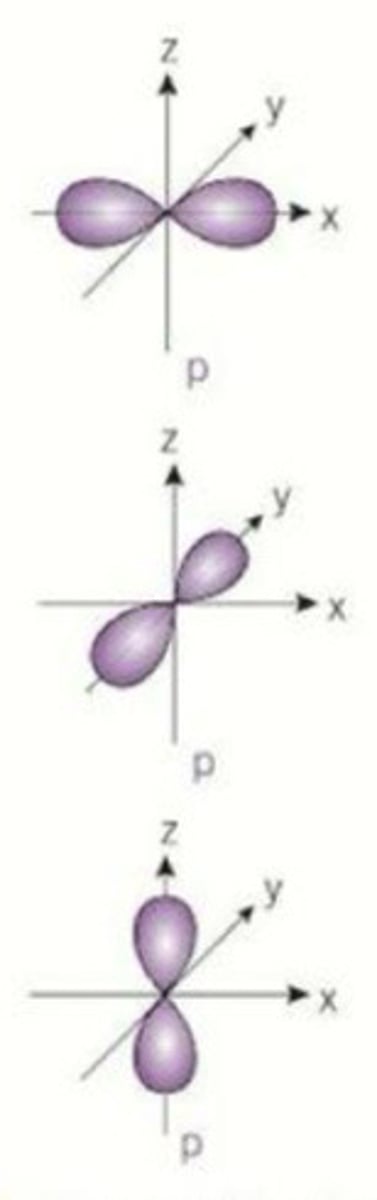
what is the electron configuration of chromium (24 electrons)
1s2 2s2 2p6 3s2 3p6 3d5 4s1
it prefers to have one electron in each orbital of the 3d subshell and just one it the 4s subshell - this gives it more stability.
what is the electron configuration of copper (29 electrons)
1s2 2s2 2p6 3s2 3p6 3d10 4s1
this is because Cu prefers to have a full 3d subshell and just one electron in 4s as its more stable this way
what is the electron configuration of Cu2+ (27 electrons)
1s2 2s2 2p6 3s2 3p6 3d9
what is the electron configuration of the atom of element which has the LOWEST first ionisation energy?
1) 1s2 2s2 3p6 3s2
2) 1s2 2s2 2p6 3s2 3p1
3) 1s2 2s2 2p6 3s2 3p2
4) 1s2 2s2 2p6 3s2 3p3
2) 1s2 2s2 2p6 3s2 3p1
- 1 is incorrect since it is 1 orbital closer to the nucleus than the rest so the nuclear pull is higher
- 3 and 4 is incorrect since it has a higher nuclear charge (proton no.) than 2, yet shielding effects are the same
give the meaning of the term 'periodicity'
illustrate your answer by referring to the atomic radii of the period 2 and period 3 elements.
- a trend of repeating physical / chemical properties with increasing atomic number
- atomic radii decrease from left to right
- pattern is the same in period 2 and 3
why is potassium placed after argon in the periodic table, even though it has a smaller relative atomic mass?
- periodic table depends upon atomic number
- Ar has less protons than K
hydrogen can be placed in several different positions in periodic tables.
one is immediately above lithium in group 1.
another is in the centre of the first row, as shown in the periodic table on the back cover.
criticise the position of hydrogen immediately above lithium by giving one reason in favour and two against.
- hydrogen is 1s1 as it has one electron in its s orbital
- rest of group 1 are alkali metals but hydrogen is not
- hydrogen does not react in the same way as the rest (different chemical properties)
- hydrogen forms an H- ion
explain why the Cl-Cl bond is shorter than the I-I bond
- iodine has a larger atomic radius
- greater shielding
- so less attraction between nucleus and bonding pair of e-
define ionisation energy
the energy required to remove one mole of electron from one mole of gaseous atoms to form one mole of gaseous 1+ ion
write the equation, including state symbols, to show the first ionisation energy of sulfur
S (g) => S+ (g) + e-
"first ionisation energy is the energy released when one mole of gaseous atoms loses one mole of electrons to form one mole of gaseous 1+ ions.
there is one incorrect word in the student's definition.
identify the word, giving reasons why its incorrect.
- energy is released
- energy is required to overcome the electrostatic forces of attraction from the nucleus to the electron
with reference to their electronic configuration, explain why chlorine and iodine have many similar chemical reactions
- both iodine and chlorine have 7 electrons in their outermost shell
- number of electrons in outer shell governs chemical reactions
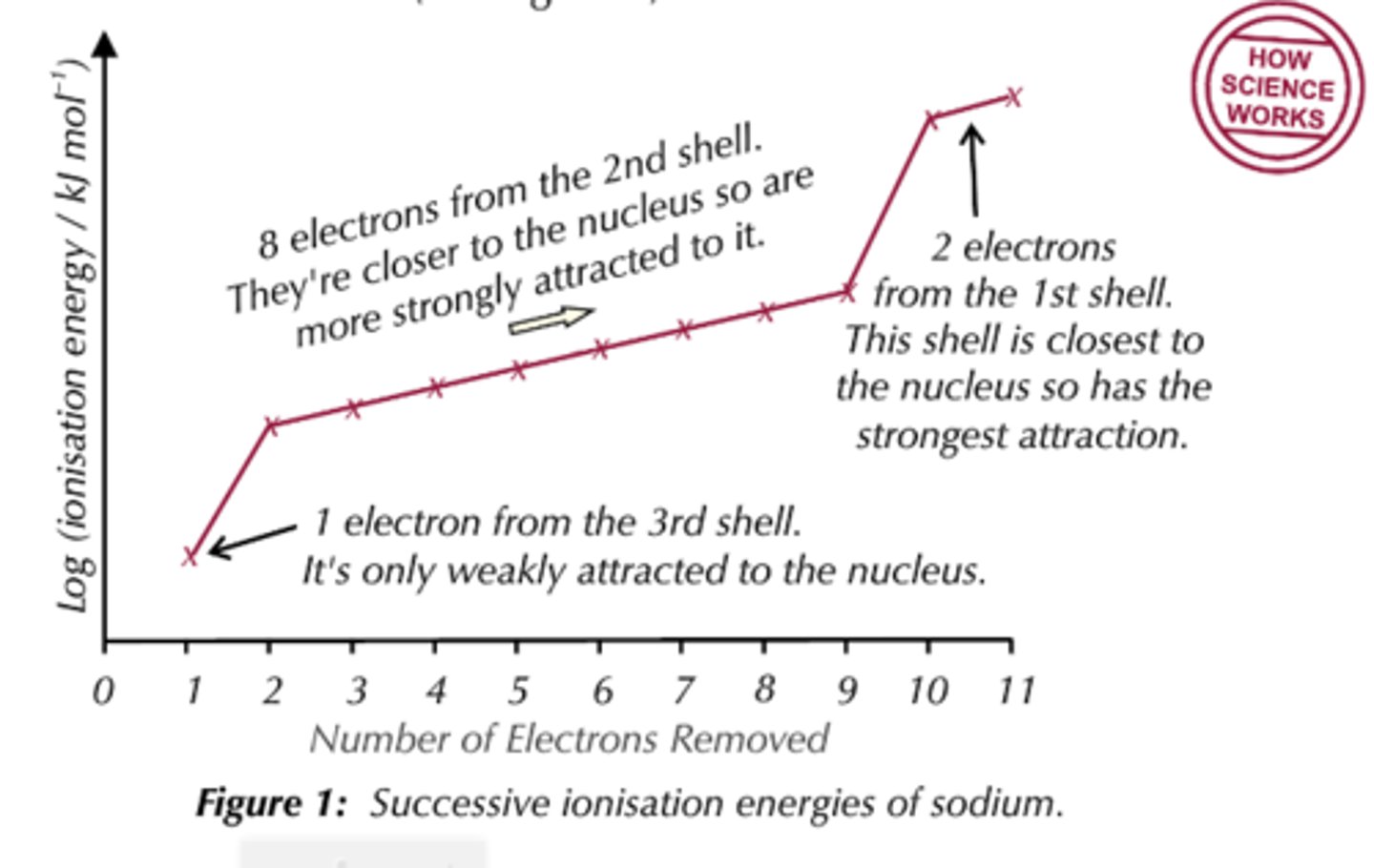
state what deductions can be made from a graph of successive ionisation energy of sodium to conform it is in Group 1 of the periodic table
- big increase between first and second electron removed suggests Na is a group 1 element
- big increase between ninth and tenth electron removed
- one electron in the outermost shell
- eight electron in the second shell
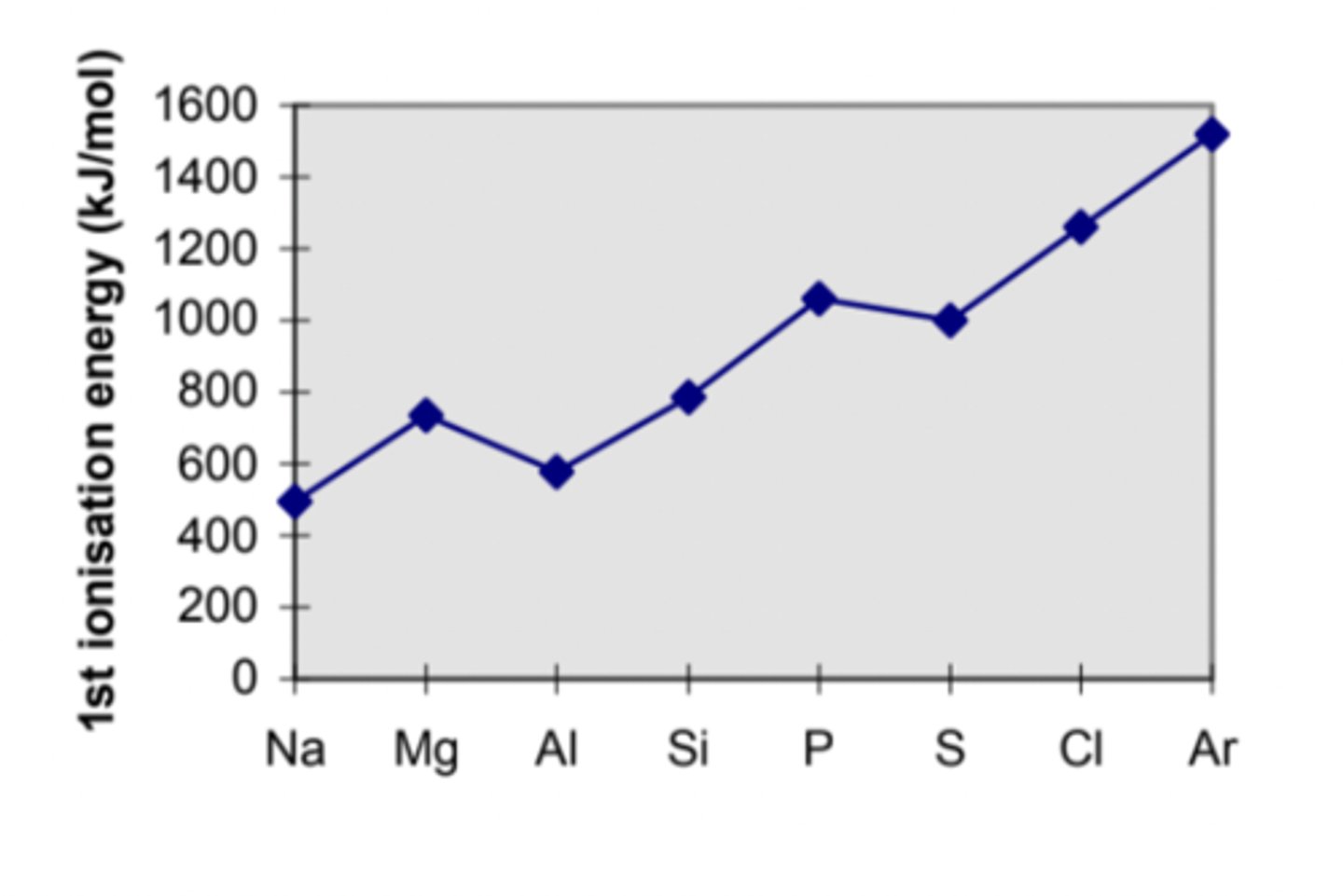
show how the first ionisation energies of the Period 3 elements change across the period on a graph.
MP1 - Al below Mg but above Na
MP2 - rise from Al to Si / rise from S to Cl to Ar
MP3 - S is below P but above Si
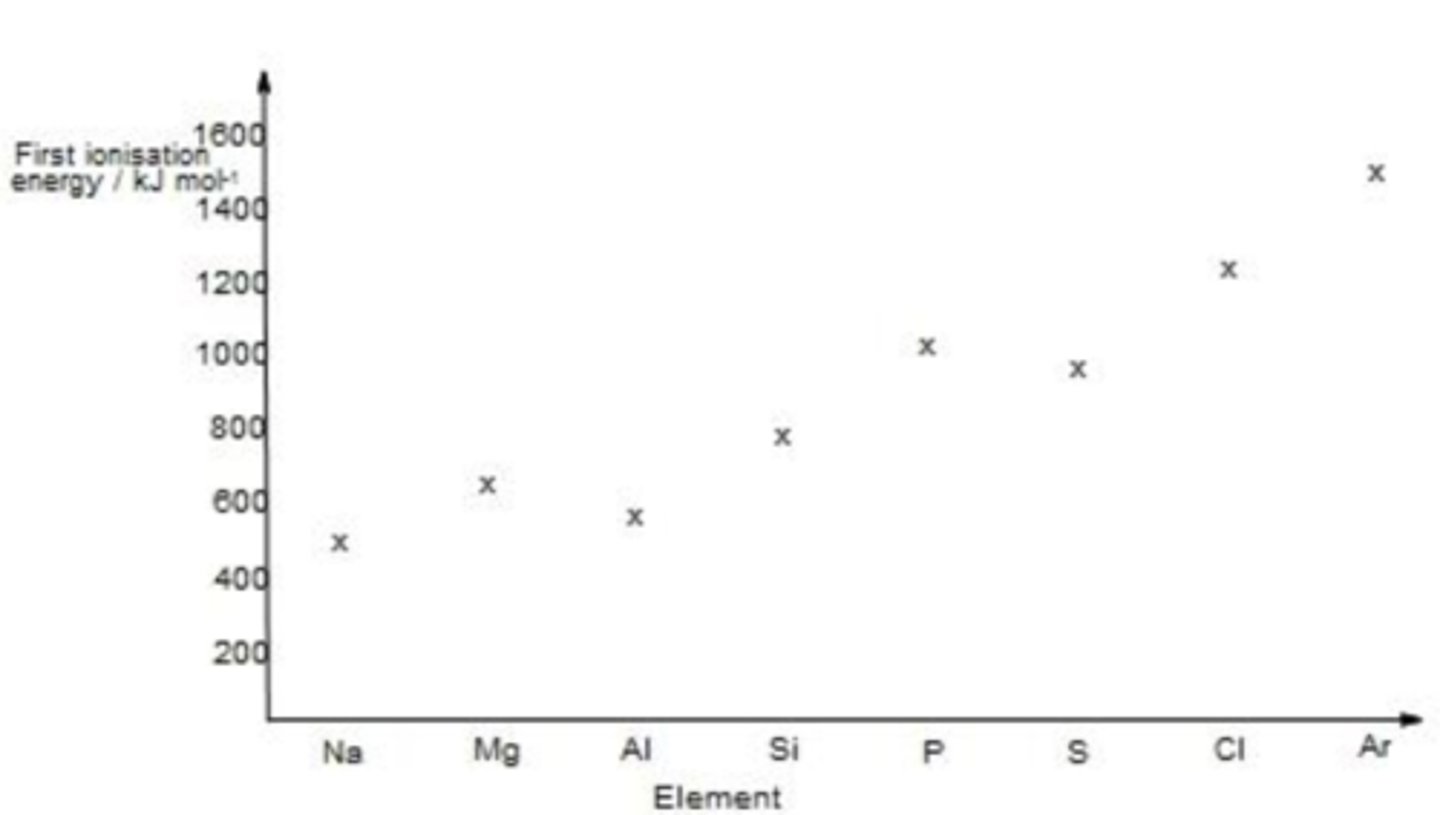
explain why first ionisation energy increase across a period
- the electron being removed is in the same shell with similar shielding
- however, there are more protons in the nucleus as you go across the period, so the nucleus attraction increases
explain why first ionisation energy decrease down a group
- electron shielding increases down the group, so e- further from nucleus
- although proton number increases, force of attraction is not greater than the effect of shielding
- shielding effect > nuclear charge
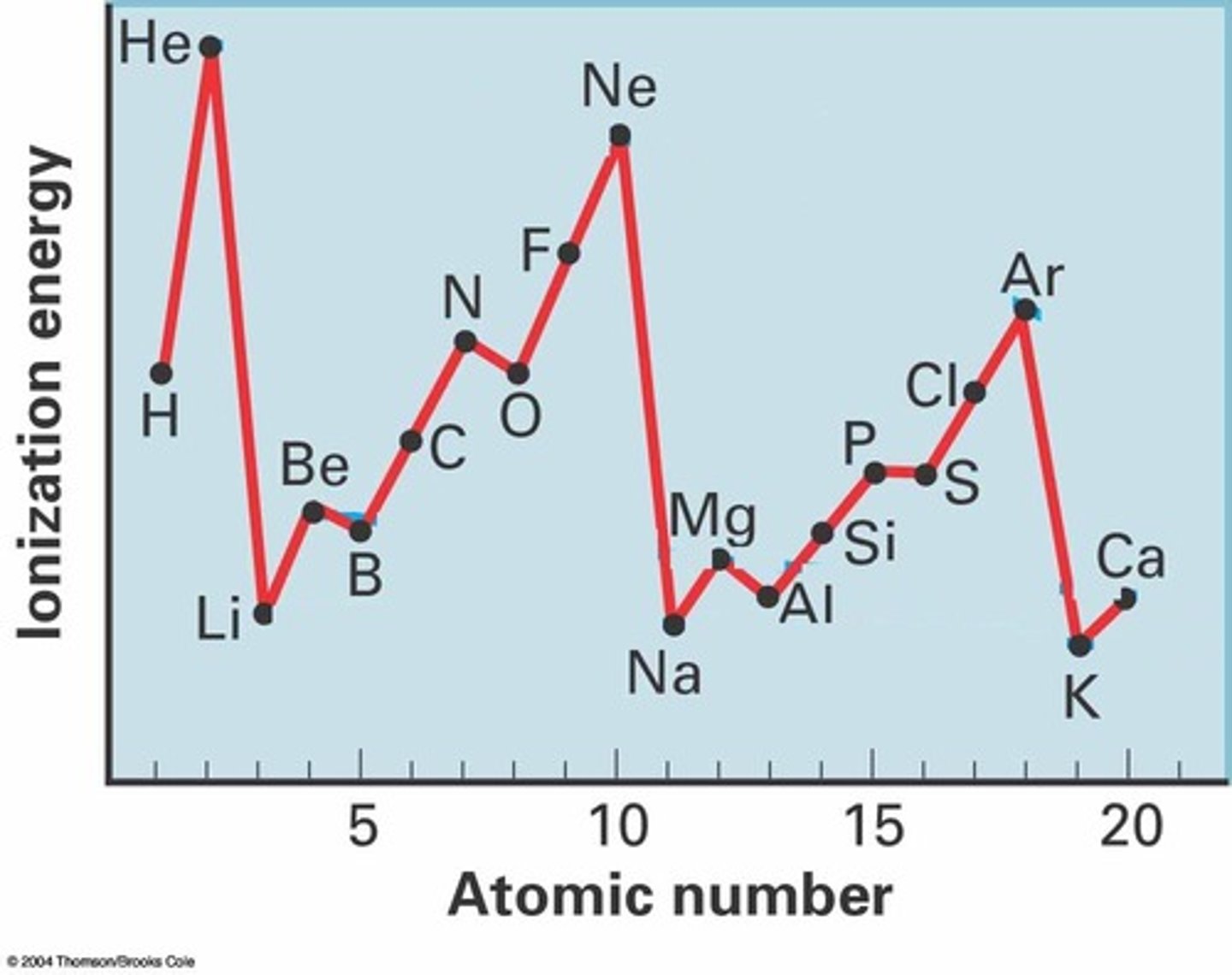
explain why the first ionisation energy of hydrogen is less than that of helium, but greater than that of lithium.
H > He
- He has more protons than H (so greater nuclear charge)
- same shielding effect as He is in the same shell as H
H > Li
- the outer electron for Li is in a higher energy level
- further away from the nucleus
- Li's outermost electron is shielding by inner electrons (1s2)
explain the trend in first ionisation energies for the group containing sulfur. (down a group: in general)
- first ionisation energy decreases down the group because although the number of protons is increasing
- the electrons being removed is further from the nucleus
- giving more shielding from the nucleus
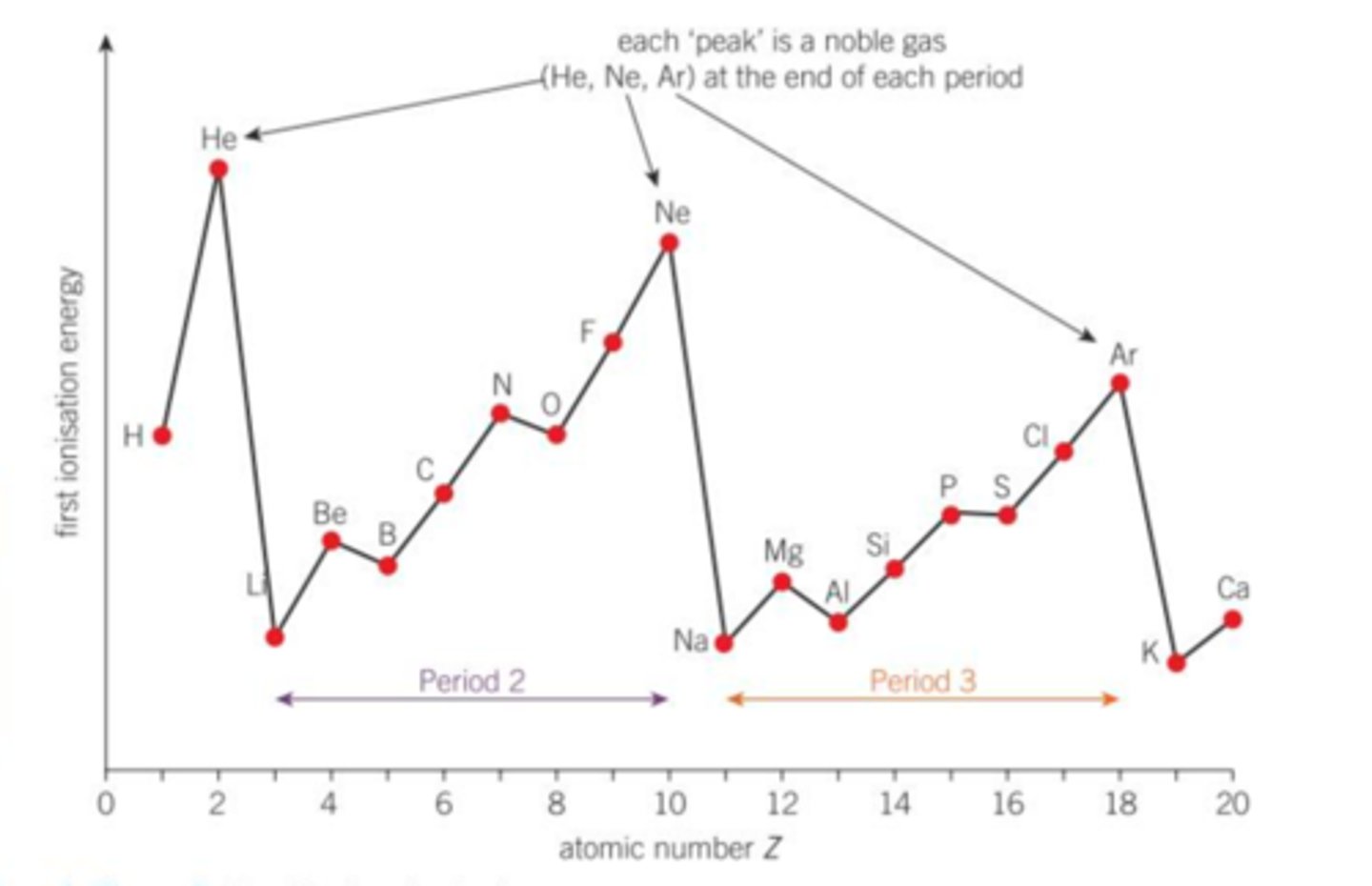
explain the trend in first ionisation energies for period 3 (across a period: in general)
- Cl has a higher nuclear charge than S because it has more protons
- as a result atomic radius is smaller
- however similar shielding effect due to same period
explain why the first ionisation energy of oxygen is lower than that of nitrogen (✧ special case: 6 < 5)
- oxygen's electron configuration (1s² 2s² 2p⁴) has an added electron in the 2p sublevel compared to nitrogen (1s² 2s² 2p³).
- oxygen ha 4 electrons in its 2p subshell
- nitrogen's stable, half-filled 2p sublevel requires more energy to remove an electron compared to oxygen's configuration.
- spin pairing has occurred for the first time in oxygen
- resulting in an increase in electron-electron repulsion
- so less energy is required to remove first electron from oxygen
✧ the same occurs with sulfur in group 3 (3p subshell)
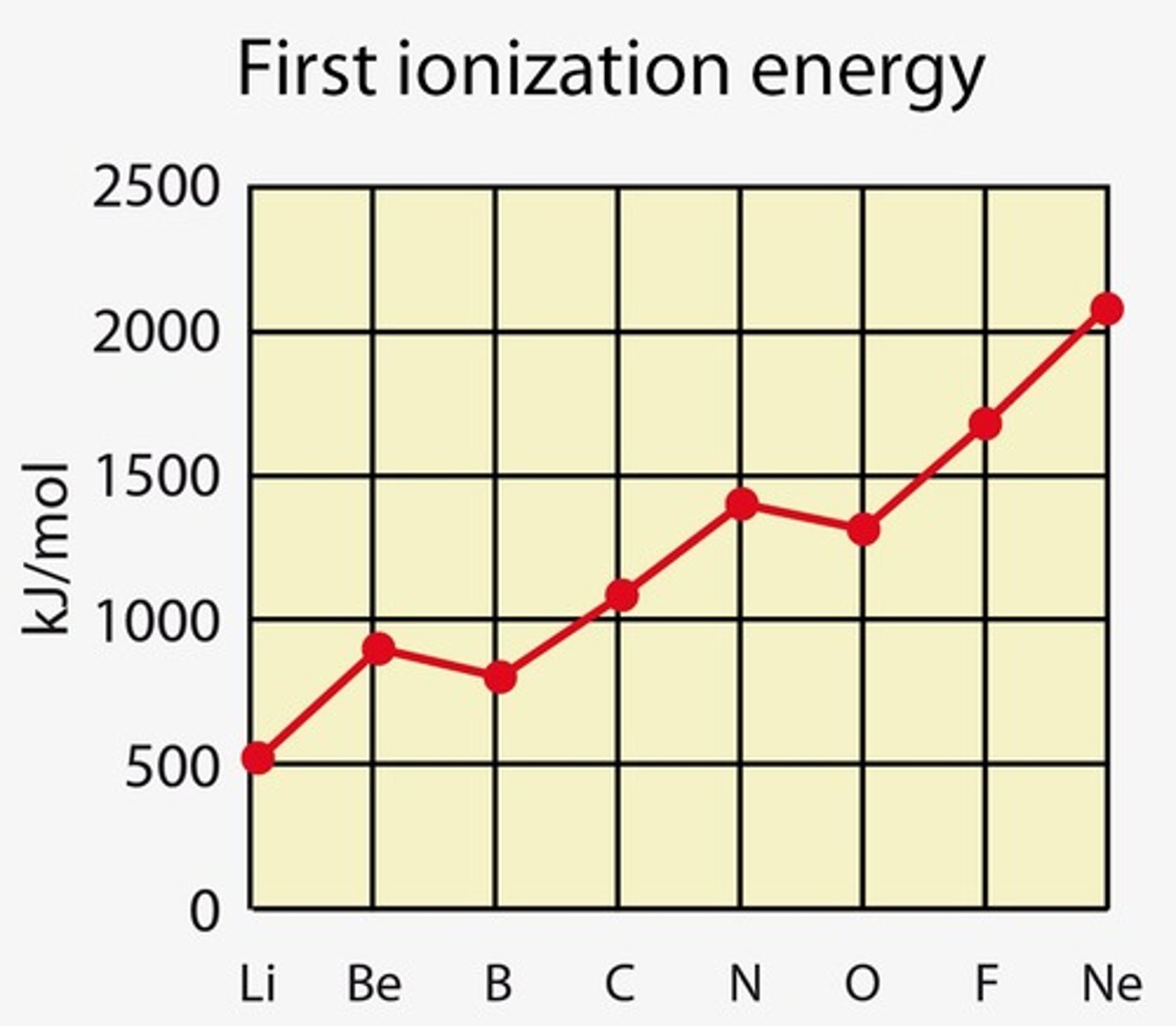
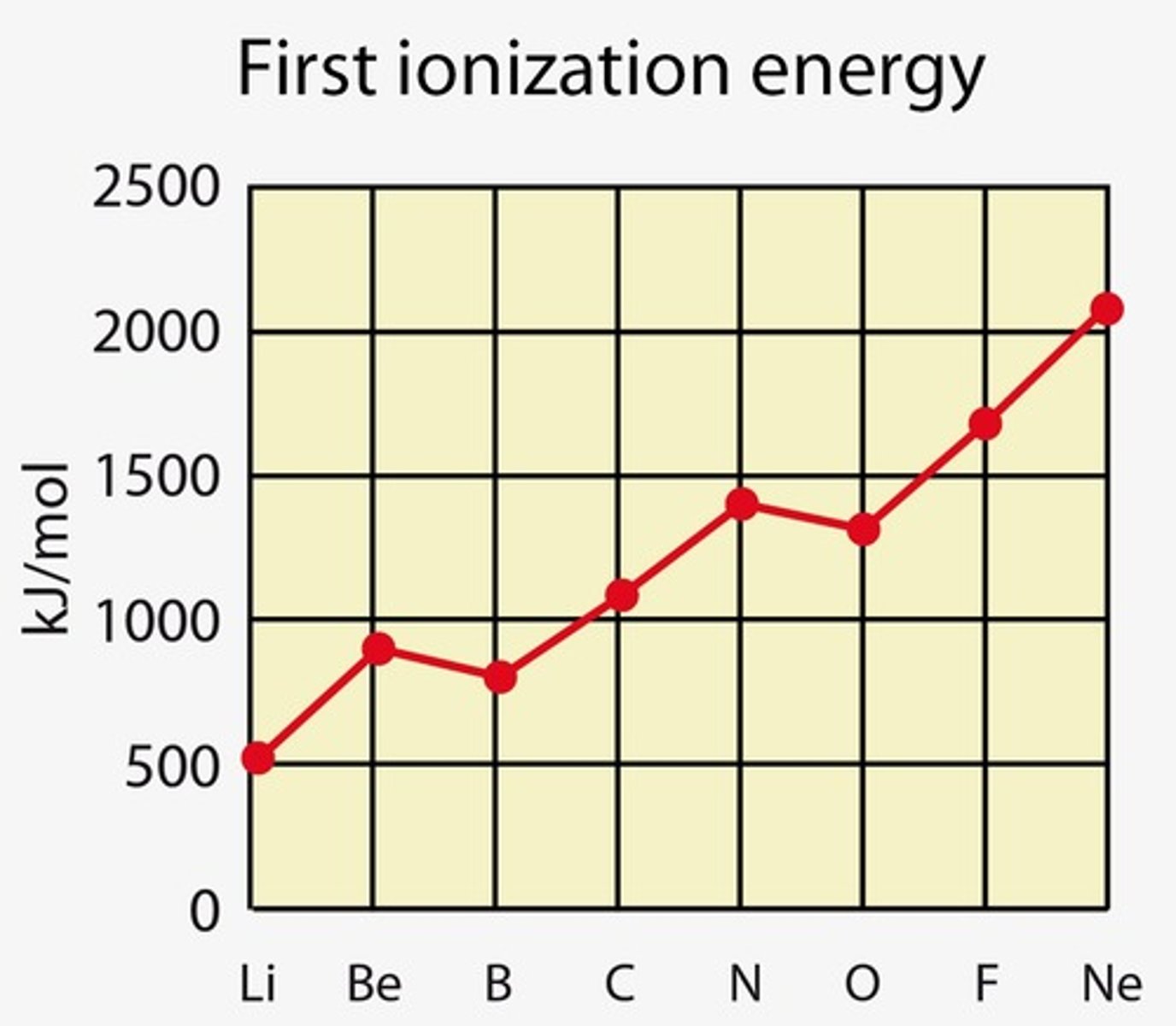
explain why the first ionisation energy of boron is lower than that of beryllium (✧ special case: 3 < 2)
- boron's addition of an electron to the 2p subshell, unlike beryllium's stable 2s level, makes electron removal easier due to higher energy levels
- electrons in boron's 2p subshell are further from the nucleus hence requiring less energy to remove compared to beryllium's 2s electrons
- so less energy required to remove first electron from boron
✧ the same occurs with aluminium in group 3 (3p subshell)
the first ionisation energy of sodium is 496kJ mol-1
explain why the first ionisation energy of magnesium is higher than that of sodium
- magnesium atom has more protons than sodium
- so it has a greater nuclear charge
- outer electron in magnesium atom is in the same sub-shell as sodium atom
- so shielding effect is the same for both
- so the force of attraction is greater in magnesium atom than sodium atom
explain the large difference between the first and second ionisation energies of lithium
- electron configuration of lithium is 2, 1
- hence second electron removed is from the inner shell
- second electron is closer to the nucleus
a student suggested that the difference in the rates of reaction of strontium and barium with water is due to the difference in the sum of their first and second ionisation energies.
discuss this suggestion.
- sum of first two ionisation energy for barium is lower as barium loses its outer electrons more easily
- barium is a bigger atom with a larger atomic radius
- barium has more shielding
- these factors outweigh the fact that barium has more proton (and greater nuclear charge)
- or the fact that barium is more reactive
explain the meaning of the term mass spectrometry
- a graphical display
- the x-axis gives the mass/charge ratio
- while y-axis gives the % abundance
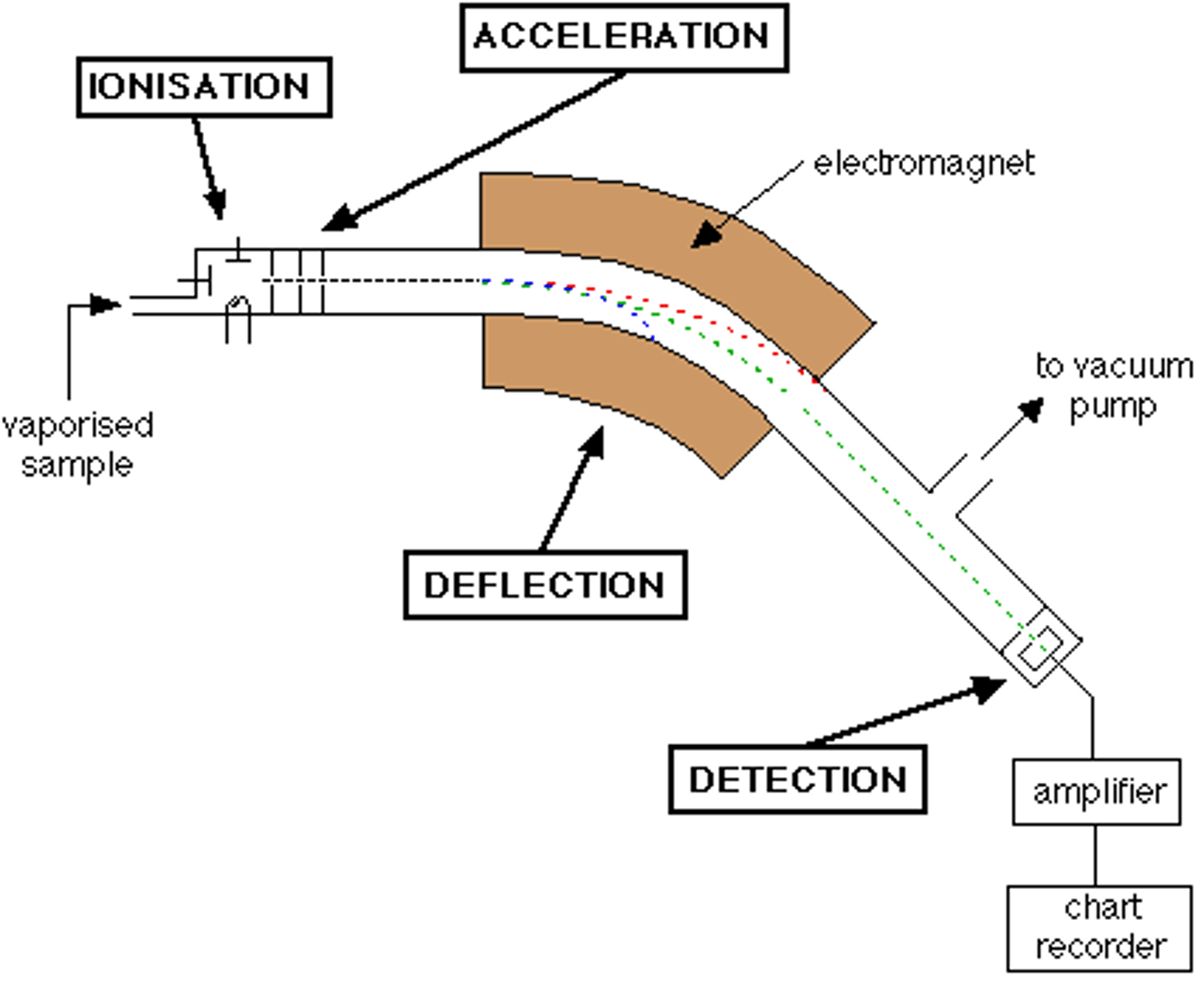
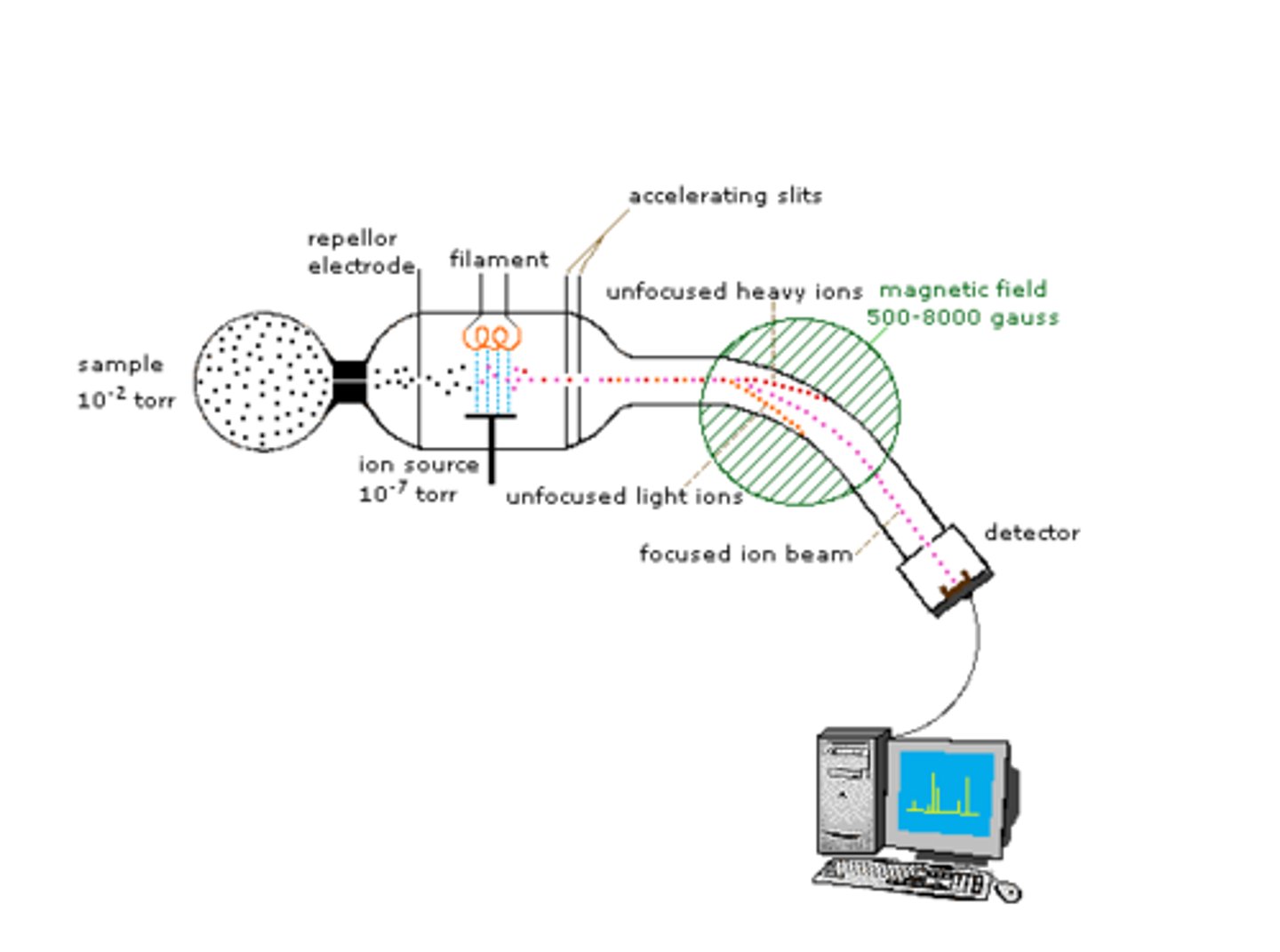
label a mass spectrometer
- only ions on the green pathway are deflected by the magnetic field and detected
- ions on the red pathway have greater mass so there are deflected less
- ions on the blue pathway have smaller mass so they are deflected more
write the equation, including state symbols, to show the third ionisation energy of magnesium
Mg2+ (g) => Mg3+ (g) + e-
what is the main function of a vacuum pump in a mass spectrometer?
- decrease the pressure so that less heat energy is required to vapourise the sample
- to remove traces of previous samples and air molecules
name the feature of the mass spectrometer responsible for the behaviour of the ions in the focused ion beam
deflection by electromagnetic field
mass spectrometry: ionisation
- a stream of high speed electrons bombards the gaseous sample
- knocks off one or more electron to give a positively charged (1+) cation
mass spectrometry: acceleration
- beam of gaseous cations is forced between 2 negatively charged plates with potential difference around them
- causes the ions to accelerate
mass spectrometry: ion drift
- ions are deflected by a magnetic field into a curved path
- the radius of the path is dependent of the m/z ratio
mass spectrometry: fragmentation
- refers to the rupturing of bonds of diatomic molecules (eg Cl2)
- Cl2+ => Cl + Cl+
- this process creates smaller cations and neutral atoms known as radicals

mass spectrometry: detection
- cations with a particular m/z ratio hit the detector (only the middle stream)
- the detector then counts the number of cation strikes
- the more ions, the higher the current, hence the higher the abundance on the spectra
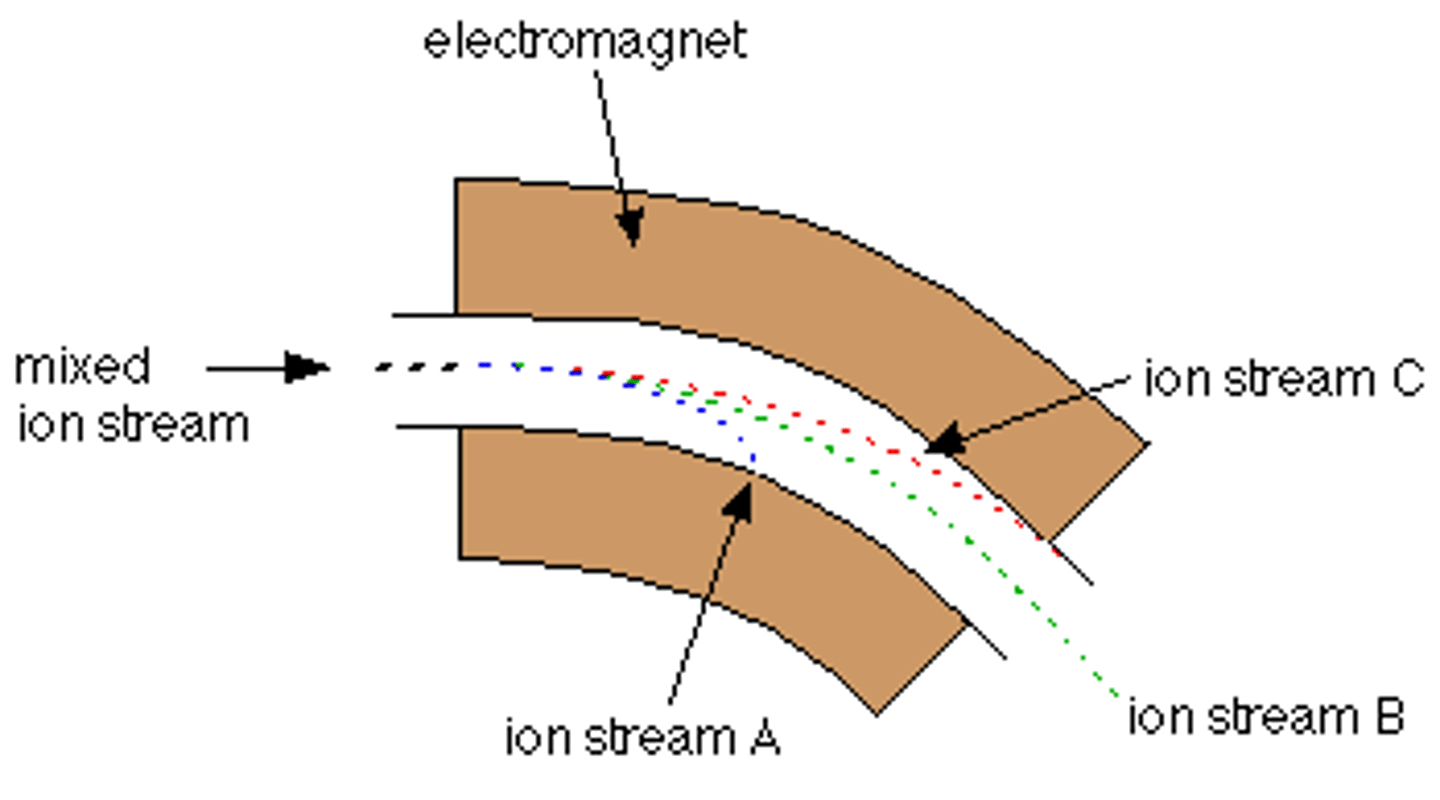
what determines the path of an ion in a mass spectrometer?
- the radius of the path depends on the value of their m/z
- ions that are heavier with larger m/z follow a larger radius curve (C)
- if an ion acquires a 2+ charge, it will be deflected more like stream A (m/z is halved)
state how the relative abundance of the two isotopes can be found.
- compare the intensity of signal / number of particles in each isotope detected
- in a mass spectrometer
a sample of the element, Cl₂, was analysed in a mass spectrometer.
chlorine has 2 isotopes.
what is the total number of peaks, due to ions with a single positive charge, which could be seen in the mass spectrum?
5
- (³⁵Cl)⁺
- (³⁷Cl)⁺
- (³⁵Cl)⁺(³⁵Cl)⁺
- (³⁵Cl)⁺(³⁷Cl)⁺
- (³⁷Cl)⁺(³⁷Cl)⁺
a phosphorus atom has mass number 31.
phosphorus has one naturally occurring isotope with mass number 31.
chlorine exists as two isotopes with mass numbers 35 and 37.
give the formulae and m/z ratio of the ions responsible for the molecular ion peaks in the mass spectrum of phosphorus(III) chloride, PCl3.
136 - P(³⁵Cl)₃+
138 - P(³⁵Cl)₂³⁷Cl+
140 - P³⁵Cl(³⁷Cl)₂+
142 - P(³⁷Cl)₃+
the mass spectrum of a diatomic molecule, X₂, has peaks at the following m/z values for the X₂+ ion:
32, 33, 34, 35, 36
deduce the formulae of all the species responsible for each of the peaks in the mass spectrum of X₂, identifying element X and showing clearly the isotopes present
32 - ¹⁶O=¹⁶O+
33 - ¹⁶O=¹⁷O+
34 - ¹⁶O=¹⁸O+ / ¹⁷O=¹⁷O+
35 - ¹⁷O=¹⁸O+
36 - ¹⁸O=¹⁸O+
in a sample of iron, the ions 54Fe2+ and 56Fe3+ are detected.
calculate their m/z ratio and determine which ion is deflected more inside the spectrometer.
54Fe2+: 54/2 = 27
52Fe3+ 56/3 = 18.7
- 56Fe3+ has a smaller m/z ratio so it will be deflected more
- it also has a larger positive charge so it will be more attracted to the negative pole of the magnet within the mass spectrometer
what does a small m/z ratio indicate?
- fragments that are small
- fragments with a high positive charge
- a combination of both
what can the mass spectrometer detect?
- m/z value
- relative abundance of each ion
- position of peaks give atomic mass
- peak intensity gives relative abundance
explain why a chemical species must be positively charged to be detected by a mass spectrometer
- only positively charged species can be accelerated by the potential difference
- the charged ions are deflected by the magnetic field and counted by the detector
give a reason why the logarithm of ionisation energy is used in order to plot a graph
- the range of numbers is too large
- logarithms make it easier to plot
how can you calculate the Ar of an element from its spectra
Ar = (m/z x abundance) / total abundance
what does the tallest peak on a mass spectrum symbolise?
- relative molecular mass of the molecule
- this peak is known as the molecular ion peak (M+)
why are the peak height corresponding to m/z ratios of similar height?
- peak height is directly proportional to abundance
- suggests a similarity in abundance of both isotopes
a sample of gallium contains 2 peaks at 69 and 71.
the heights are 145mm and 95mm respectively. calculate the relative abundance of each isotope of gallium to one d.p.
Ga-69: 145x100 / (145+95) x100 = 60.4%
Ga-71: 95x100 / (145+95) x 100 = 39.6%
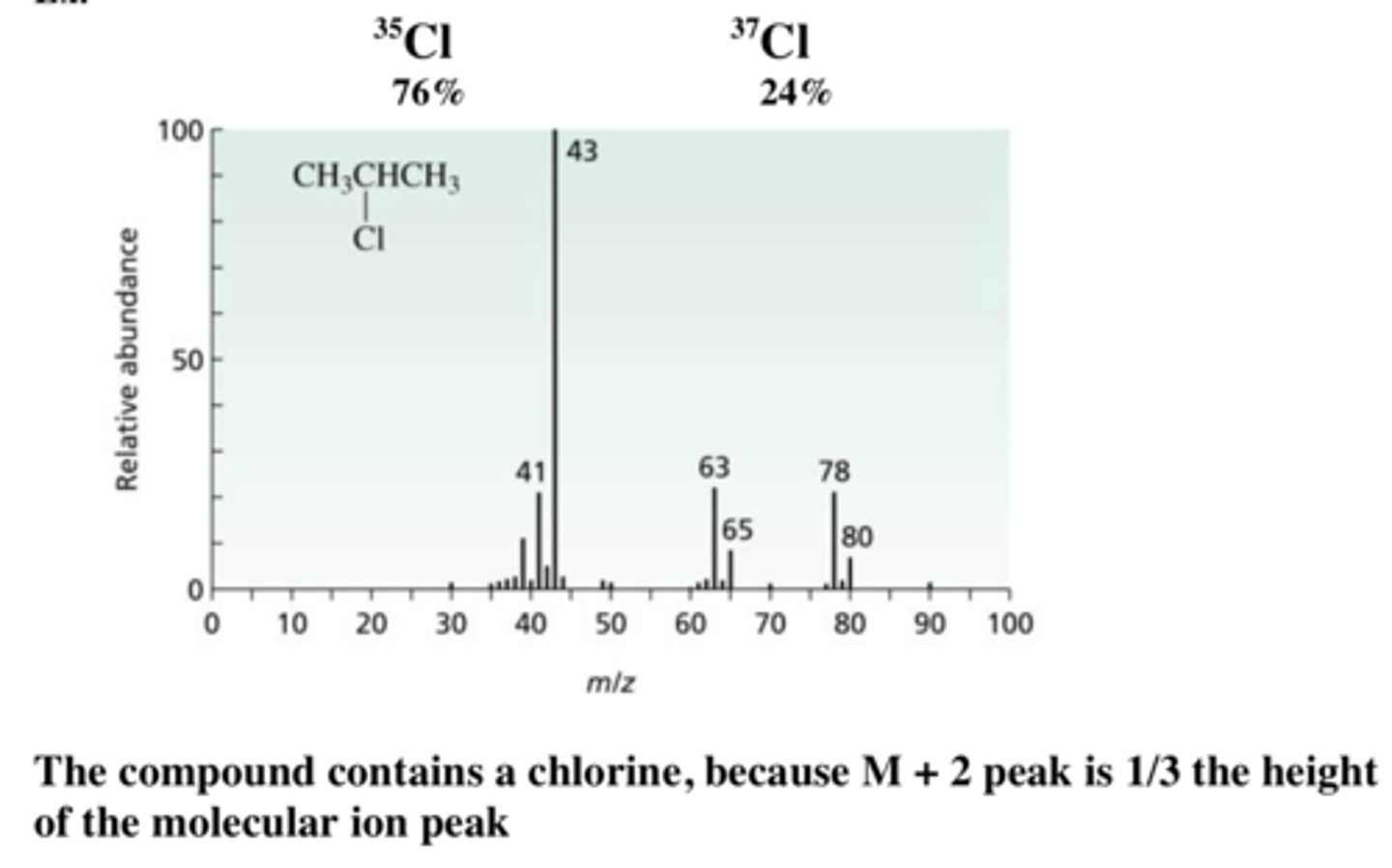
describe the characteristic pattern of a chlorine spectra
- 3:1 ratio for Cl+ ions
- 3:6:9 ratio for Cl2+ ions
- this is because some isotopes are more common than others
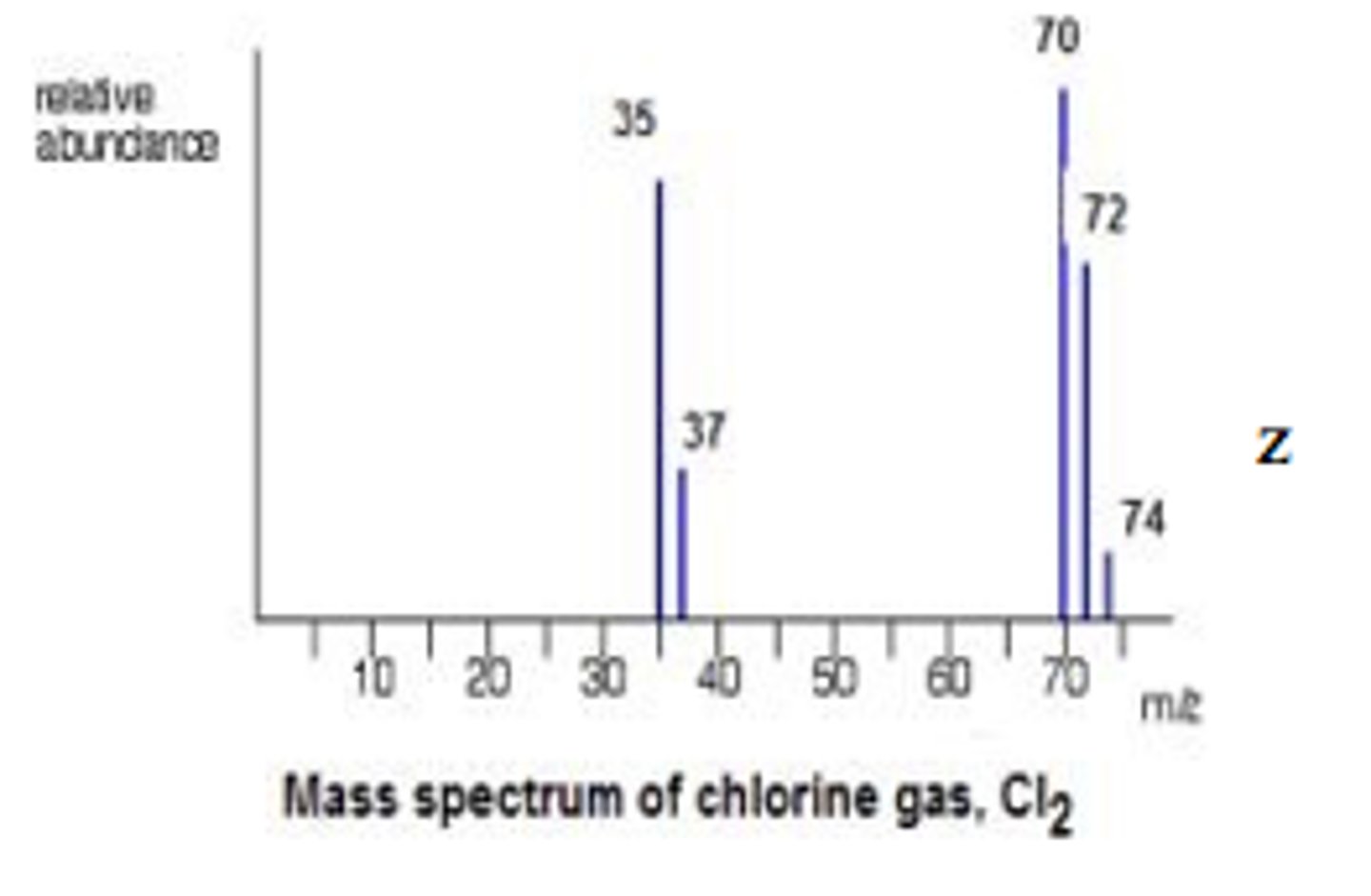
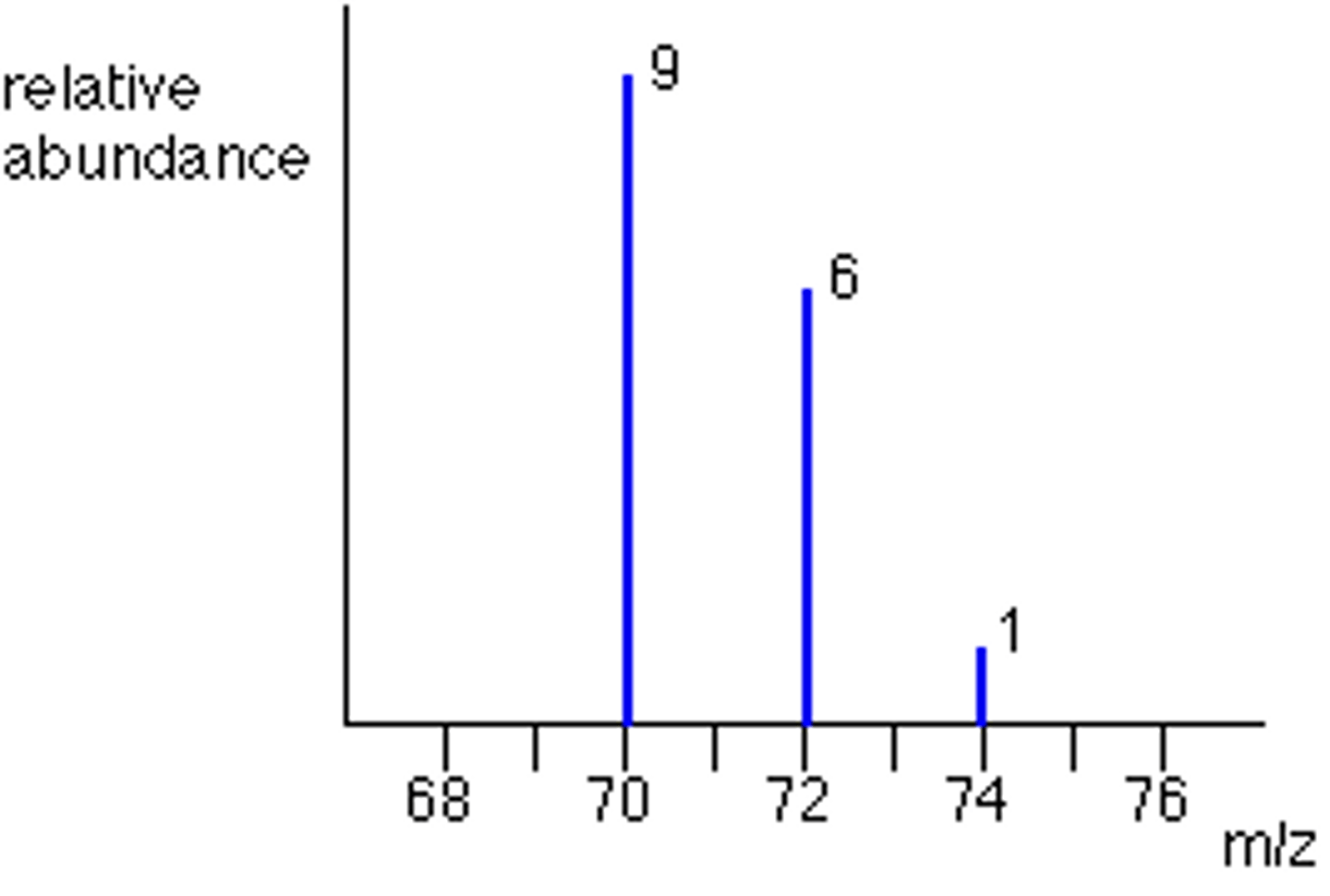
a sample of chlorine contained 75 % of ³⁵Cl and 25 % of ³⁷Cl.
complete the mass spectrum to show the peaks you would expect for the molecular ion from this sample of chlorine gas.
calculations:
70: 3/4 x 3/4 = 9/16
72: 3/4 x 1/4 = 3/6
72: 3/4 x 1/4 = 3/6
74: 1/4 x 1/4 = 1/16
hence the ratio is 9:6:1
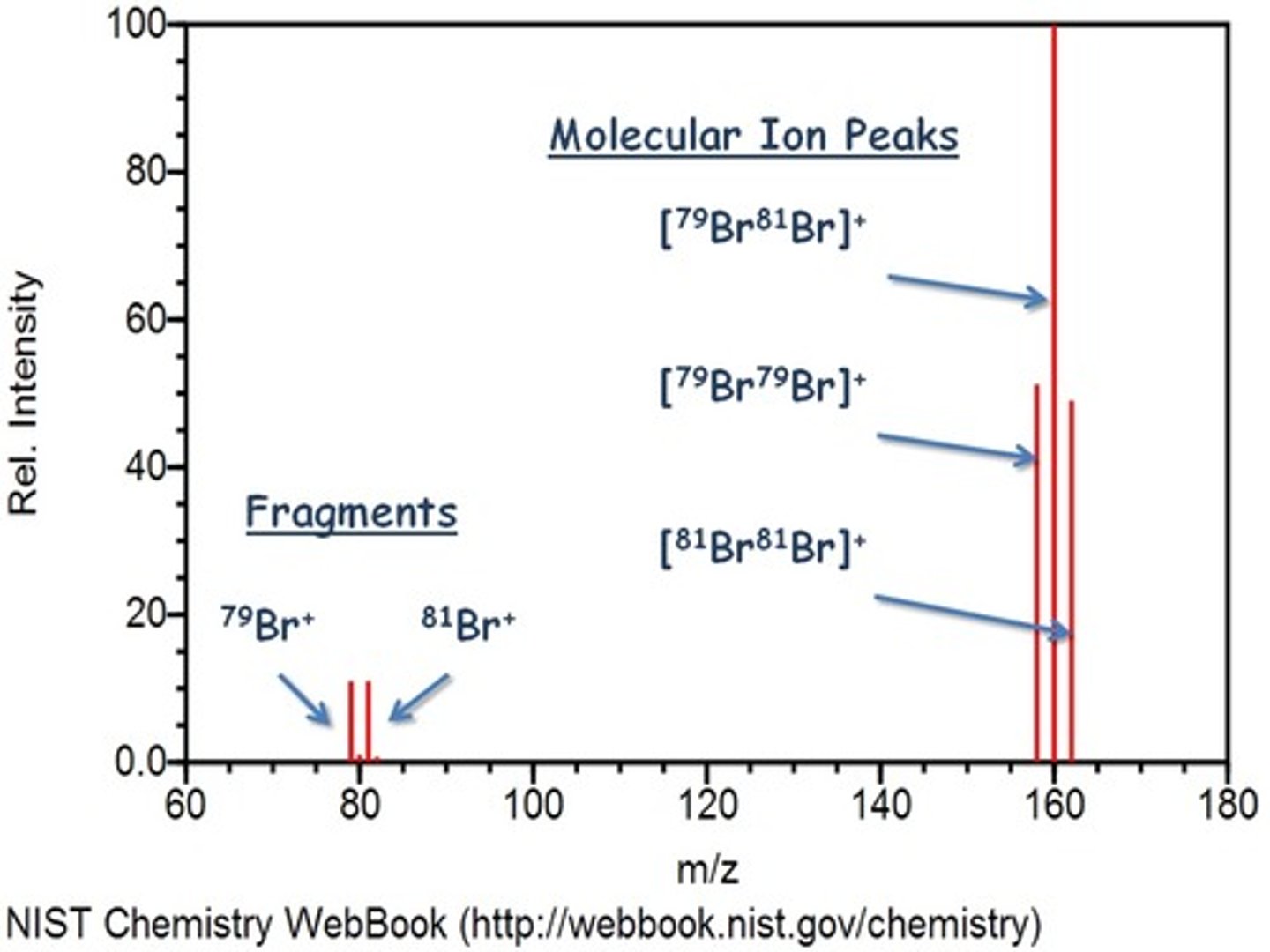
describe the characteristic pattern of a bromine spectra
- 1:1 ratio for Br+ ions
- 1:2:1 ratio for Br2+ ions