SAAT Physics 4 Section 4-6 (Quantum Theory, The Atom, Solid-State Electronics) - تحصيلي
1/77
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
78 Terms
Objects glow when
They reach a high temperature
Electromagnetic radiation
Depends on the temperature and other properties of an object, typically consists of a continuous distribution of wavelengths from the infraref, visible, and ultraviolet portions of the spectrum
The distribution of the intensity of the different wavelengths
Varies with temperature
Blackbody radiation
The radiation emitted by a blackbody, which is a perfect radiator and absorber and emits radiation based only on its temperature
The intensity of blackbody radiation at three different temperatures
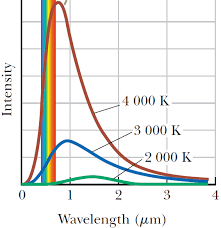
Classical theory’s prediction for blackbody radiation (blue curve) did not correspond to the experimental data (red data points) at all wavelengths
Planck’s theory is the only one that corresponds with the data
Total energy of a resonator with frequency formula
En = nhf
The photoelectric effect
When light strikes a metal surface, the surface may emit electrons
Photoelectrons
Refer to the electrons that are emitted in the photoelectric effect
Photosensitive
Refer to those surfaces that exhibit the photoelectric effect
According to classical physics,
Light waves of any frequency should have sufficient energy to eject electrons from the metal if the intensity of the light is high enough
At lower intensities,
Electrons should be ejected if light shines on the metal for a sufficient time period
Electrons would take time to
Absorb the incoming energy before acquiring enough kinetic energy to escape from the metal
Increasing the intensity of the light waves should
Increase the kinetic energy of the photoelectrons, and the maximum kinetic energy of any electron should be determined by the light’s intensity
Threshold frequency
The minimum frequency of electromagnetic radiation (such as light) required to eject electrons from a metal surface (differs from metal to metal)
If the light frequency exceeds the threshold frequency,
The photoelectric effect is observed
The number of photoelectrons emitted is proportional to
The light intensity; but the maximum kinetic energy of the photoelectrons is independent of the light intensity
Planck’s formula
E = hf
The work function is equal to hft
ft is the threshold frequency for metal
Photons with energy greater than hft eject
Electrons from the surface of and from within the kinetic energy
Maximum kinetic energy of a photoelectron
KE = hf - hf_1
Arthur Compton
Found that, if light behaves like a particle, then a collision between an electron and a photon should be similar to a collision between two billiard balls
The amount that the wavelength shifts depends on
The angle through which the proton is scattered
Compton shift is difficult to detect using visible light, but
It can be observed using electromagnetic waves with much shorter wavelengths (like X-rays)
Matter waves
The wave-like behaviors exhibited by particles of matter (such as electrons, protons, and atoms) as part of wave–particle duality in quantum mechanics
Wavelength of matter waves
λ = h/p
The uncertainty principle
It is fundamentally impossible to measure both the precise position and momentum (velocity) of a quantum particle (like an electron) simultaneously
An electron's location is described by a probability distribution
Simultaneous measurements of position and momentum cannot be completely certain.
Rutherford's model of the atom
Describes the atom as a miniature solar system with a tiny, dense, positively charged nucleus at the center containing most of the mass, surrounded by negatively charged electrons orbiting it
Region of the atom as a nucleus
The positive charge and most of the mass of an atom is concentrated in an extremely small volume
Orbits
Electrons surrounding the nucleus revolve around it with very high speed in circular paths.
Electrons being negatively charged and nucleus being a densely concentrated mass of positively charged particles are held together by
A strong electrostatic force of attraction
Bohr model of an atom
A hydrogen atom (Z = 1) or a hydrogen-like ion (Z > 1), where the negatively charged electron confined to an atomic shell encircles a small, positively charged atomic nucleus and where an electron jumps between orbits, is accompanied by an emitted or absorbed amount of electromagnetic energy (hν)
Hydrogen emission spectrum
When electrons in an atom or a molecule absorb energy and get excited, they jump from a lower energy level to a higher energy level, and they emit radiation when they retur to their onginal states
Importance of the hydrogen spectrum
It’s evidence showing that the atom’s electronic structure is quantized
Absorption and emission
The electron absorbs the energy and jumps to a higher energy level; in the reverse process, emission, the electron returns in the ground sale by releasing the extra energy it absorbed
When the light from an atomic gas is passed through a prism or a diffraction grating,
The dispersed light appears as a series of distinct, bright spectral lines
The Balmer series
The part of the hydrogen emission spectrum which is responsible for the excitation of an electron to the second shell from any other higher shell
Laymen series - ultra violet light
The transition to the first shell from any other higher shell
Balmer series - visible light
The transition to the second shell from any other higher shell
Paschen series - infrared rays
The transition to the third shell from any other higher shell
Bracket series
The transition to the fourth shell from any other higher shell
Pfund series
The transition to the fifth shell from any other higher shell
Laser
A device that stimulates atoms or molecules to emit light at particular wavelengths and amplifies the light (typically producing a very narrow beam of radiation)
Acronym of laser
Light amplification by the stimulated emission of radiation
Lasers in medicine
Eye surgeries
Lasers in industries
Cutting and wielding metals, checking metal bars, checking and measuring tunnels and tubes
Lasers in science and technology
Measuring movement of the tectonic plates, and in scientific experiments
Band theory of solids
A theoretical model explaining the states of electrons, in solid materials, that can have values of energy only within certain specific ranges
In atoms, electrons are filled with respective energy orbits following Pauli’s exclusion principle
Two atomic orbitals combine to form a molecular orbit with two distinct energy levels
Pauli’s exlusion principle
No two identical fermions (such as electrons) in an atom or molecule can occupy the same quantum state simultaneously
Energy band in solids
Having many energy bands including valence band, conduction band, forbidden band
Valence band
Energy band that consists of valence electrons energy levels (presemt below the conduction band and the electrons of this band are loosely bound to the nucleus of the atom)
Conduction band
Energy band that consists of free electrons energy levels
(for electrons to be free, external energy must be applied such that the valence electrons get pushed to the conduction band and become free)
Forbidden band (also known as the forbidden gap)
The energy gap between the valence band and the conduction band
The electrical conductivity of a solid is determined by the
Forbidden gap and the classification of the materials such as conductors, semiconductors, and insulators
The difference between the valence band and the conduction band
The band gap or energy gap
In semiconductors, current conduction by holes is as important as
Electron conduction; important to become familiar in thinking of the holes as mobile particles carrying positive charges and negative charges
Diodes
Used to protect circuits by limiting the voltage and to also transform AC into DC
Semiconductors
Like silicon and germanium are used to make the most of the diodes
Even though most semiconductors transmit currents in a single direction,
The way which they transmit differs
Detailed definiton of a diode
A two-way terminal electronic component that conducts electricity primarily in one direction
Diodes have high resistance on one end and
Low resistance on the other end
P-type semiconductors
An intrinic semiconductor doped with boron or indium
The majority of carries in p-type semiconductors
Are holes
Electrons are minority carriers
In p-type semiconductors
In a p-type semiconductor, the hole density is
Much greater than the electron density
The acceptor energy level of the p-type is close to the
Valency bond and away from the conduction brand
N-type semiconductors
An instrinsic semiconductor doped with phosphorus or antimony as impurity
The majority of charge carriers in the n-type electrons
Are electrons
Forward-biased diode
When there’s a small drop of voltage across the diode and the current is conducting
Reverse-biased diode
When the battery’s voltage is dropped completely
Zero-based diode
The voltage potential across the diode is zero
Transistor
A type of a semiconductor device that can be used to both conduct and insulate electric current or voltage
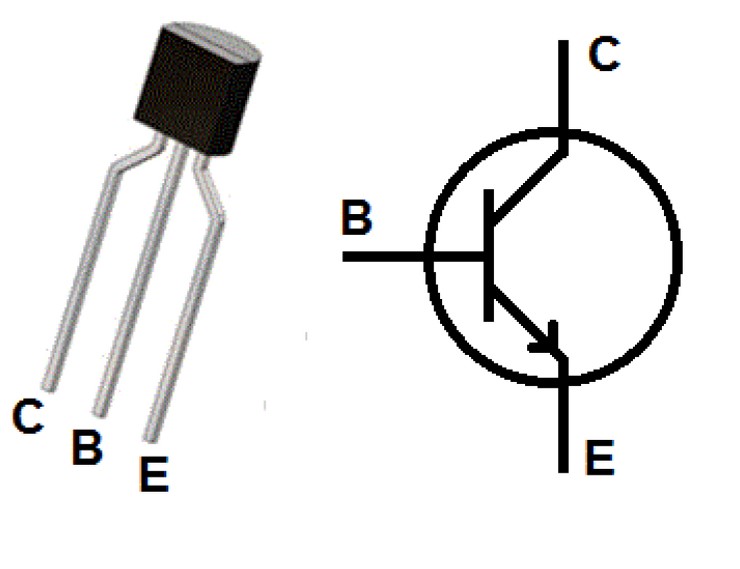
P-N-P transistor
Where one n-type material is introduced or placed between two p-type materials
N-P-N transistor
When one p-type material is present between two n-type materials
PNP transistor image
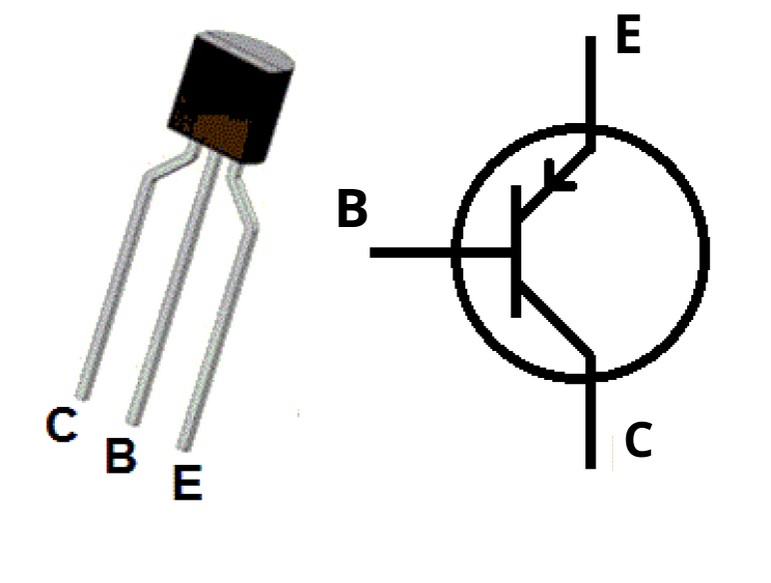
NPN transistor image