AP BIO UNIT 1
1/13
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
14 Terms
hydrogen bonds
bond between negative atom and a hydrogen
polar covalent bonds between O and H
IN the water molecule
hydrogen bonds between O and H
BETWEEN other molecules
cohesion
water molecules attracted to other WATER molecules
surface tension
adhesion
water molecules attracted to other POLAR molecules
capulary action (water moving up root —> leaf)
high specific heat
water must absorb large amount of energy to change temperature
maintains our body temp
ice on top of lakes
high heat of vaporization
large amount of energy needed to make liquid evaporate
sweat released to absorb body heat
hydrolysis
ADD water to BREAK bond
dehydration synthesis
REMOVE water to FORM bond
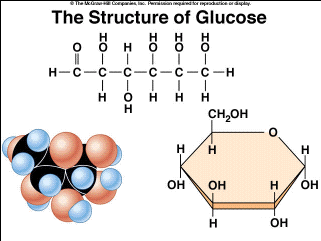
carbohydrates
elements: C H O (1:2:1)
bonds: covalent
linear and branched chains
monosaccharide: glucose, fructose, galactose
polysaccharide: cellulose, chitin
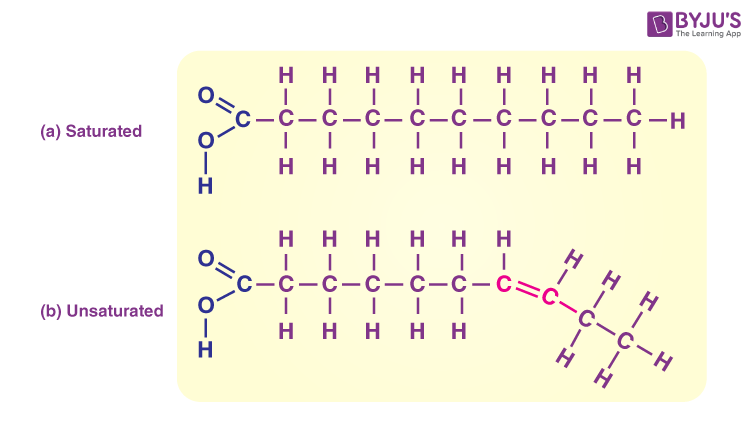
lipids
elements: C H O P
fats: saturated and unsaturated, energy storage
phospholipids: head and tail, form lipid bilayers
steroid: support growth, metabolism
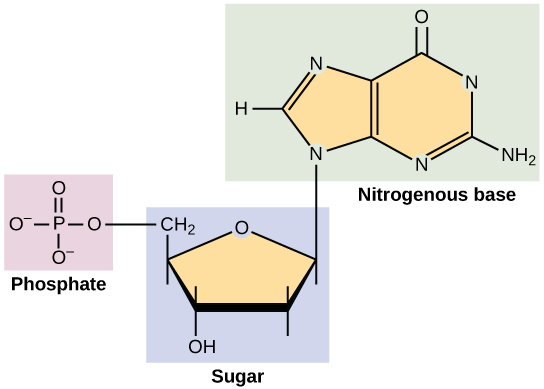
nucleic acids
elements: C H O N P
DNA and RNA
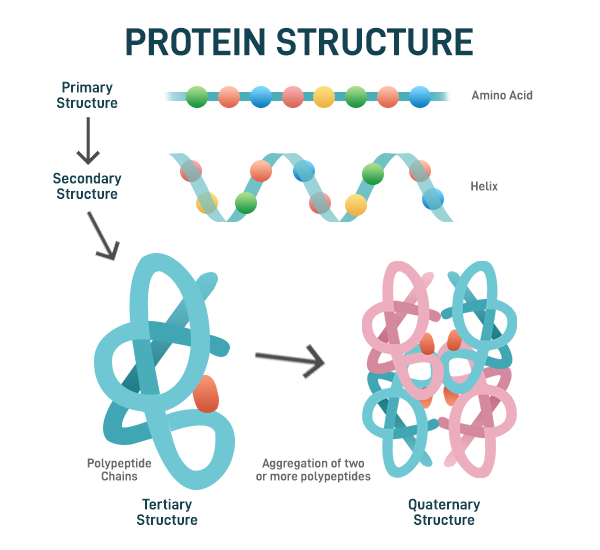
proteins
elements: C H O N S
amino acids
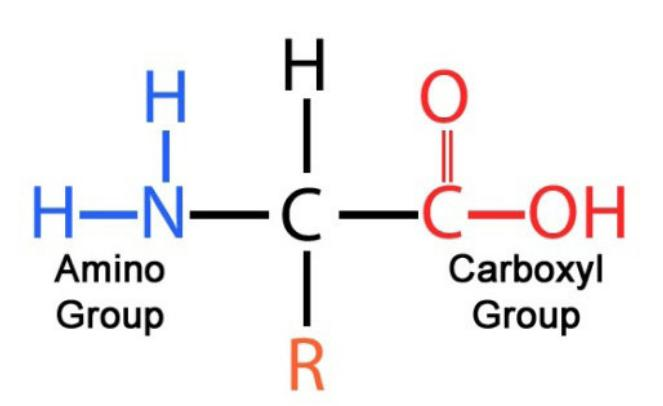
levels of protein structure
primary: chain of amino acids
secondary: bonds between backbone of alpha helix
tertiary: any bonds between R groups, 3D structure
quaternary: any bonds between R groups of polypeptides