mcdb 6 midterm 1 (ch
1/10
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
11 Terms
Energy
the capacity to do work/cause change
Type of transfer and transformation of energy and matter
one way flow
The reactivity of an atom is due to…
the presence of one or more unpaired electrons in the valence shell
An electrons energy level is correlated with…
Its average distance from the nucleus
Electrons can move to higher or lower electron shells by…
Absorbing (higher) or releasing (lower) energy
Valence electrons
those that occupy the outermost (valence) shell
Nonreactive/inert atoms
Atoms with completed valence shells
The chemical behavior of an atom is dependent on the…
number of electrons in its outermost shell, or valence shell
Bonding capacity (valence)
the number of bonds that the atom can form; usually corresponds to the number of electrons required to complete the atom + forming of molecules (electrically neutral and held together by chemical bonds)
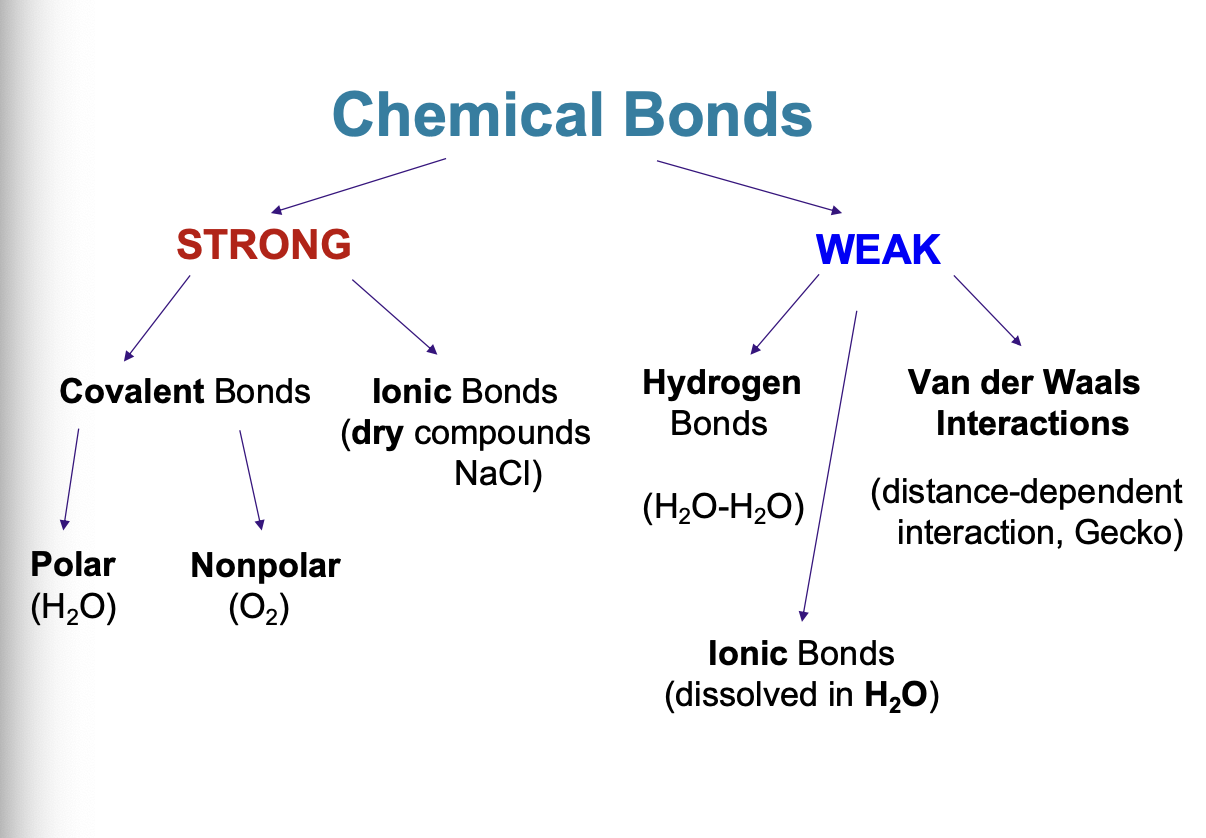
Chemical Bonds