Ochem Ch 18 - Aromatic Substitution Reactions
1/97
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
98 Terms
Electrophilic aromatic substitution
A substitution reaction in which an aromatic proton is replaced by an electrophile and the aromatic ring is preserved
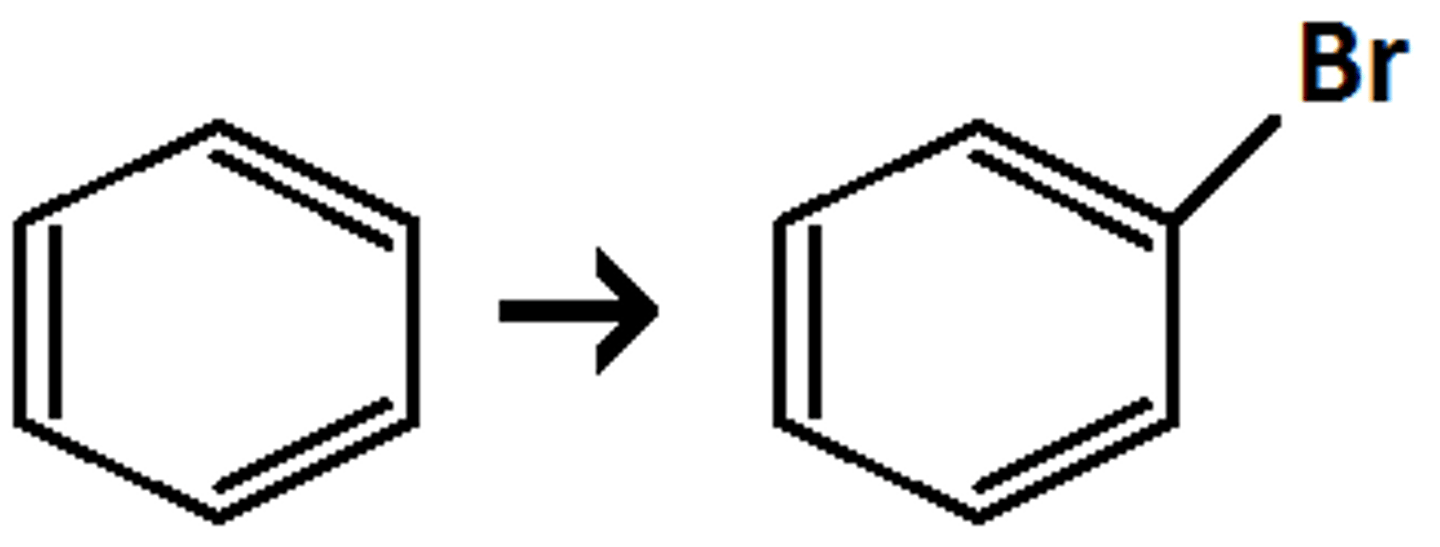
How to add bromine to an aromatic ring?
Reagents: Br2 and FeBr3 (or AlBr3)
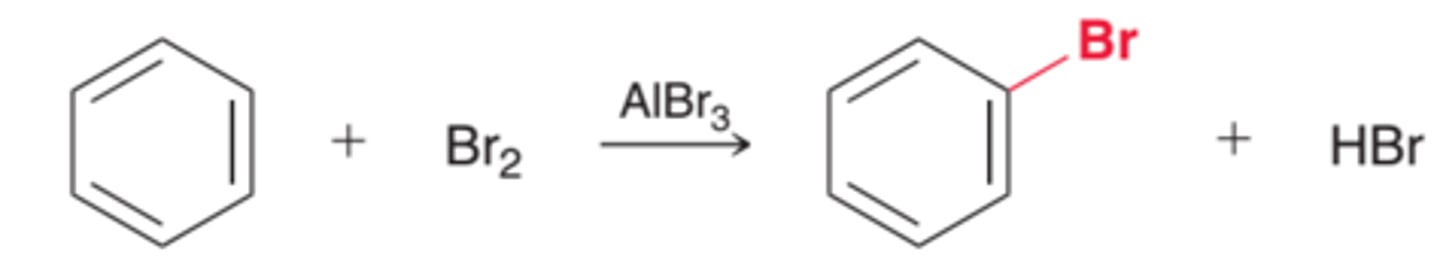
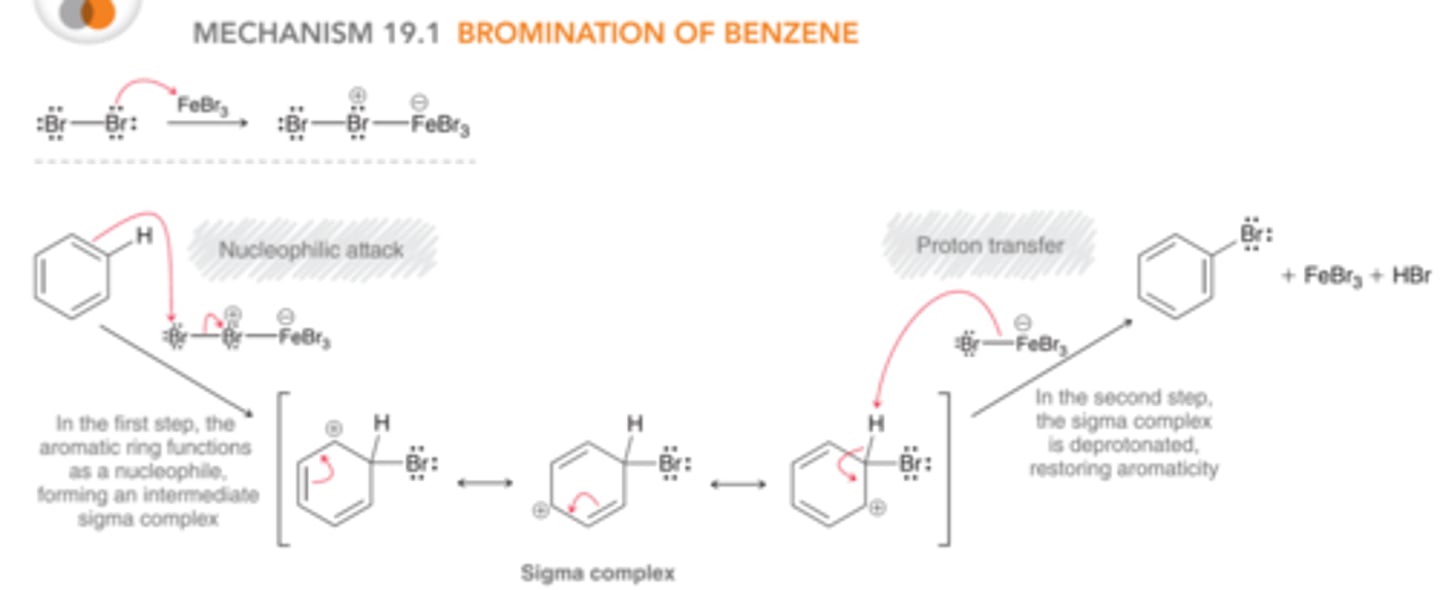
Mechanism: Bromination of Benzene
1st step: The aromatic ring functions as a nucleophile & attacks the electrophilic agent, generating a positively charged intermediate called a sigma complex which is resonance stabilized.
2nd step: The sigma complex is deprotonated, restoring aromaticity & regenerating FeBr3 (the catalyst which is not consumed)
Substitution is an _______________ process while addition is an _______________ process. So, only _________________ is observed.
Substitution is an EXERGONIC process while addition is an ENDERGONIC process. So, only SUBSITUTION is observed.
How to add chlorine to an aromatic ring?
Reagent: Cl2 and AlCl3
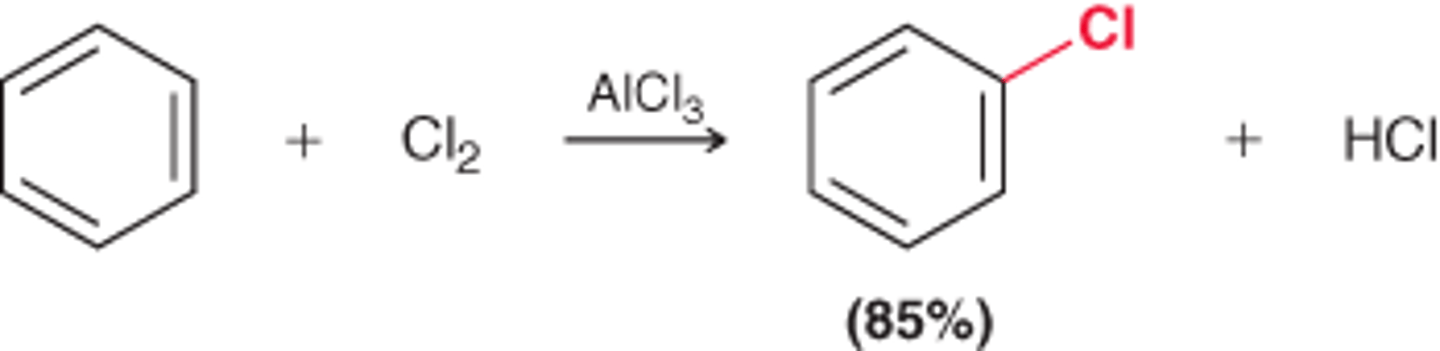
Mechanism: Chlorination of Benzene
1st step: The aromatic ring functions as a nucleophile and attacks the electrophilic agent, generating a sigma complex.
2nd step: The sigma complex is deprotonated, restoring aromaticity & regenerating the Lewis acid (AlCl3)
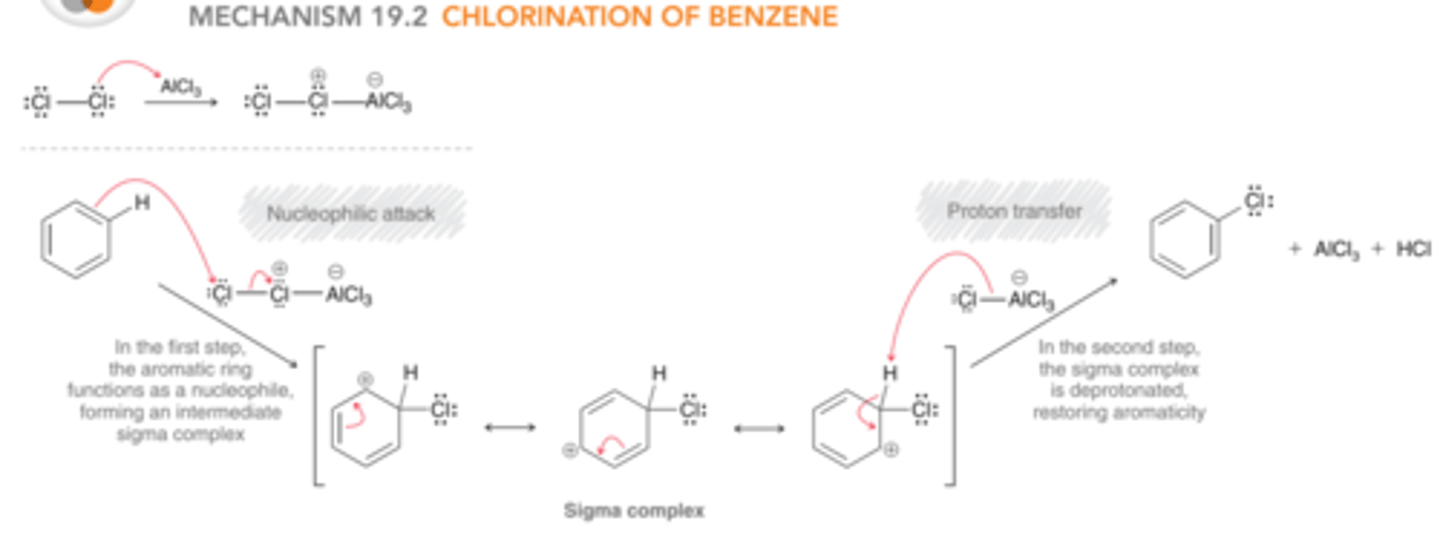
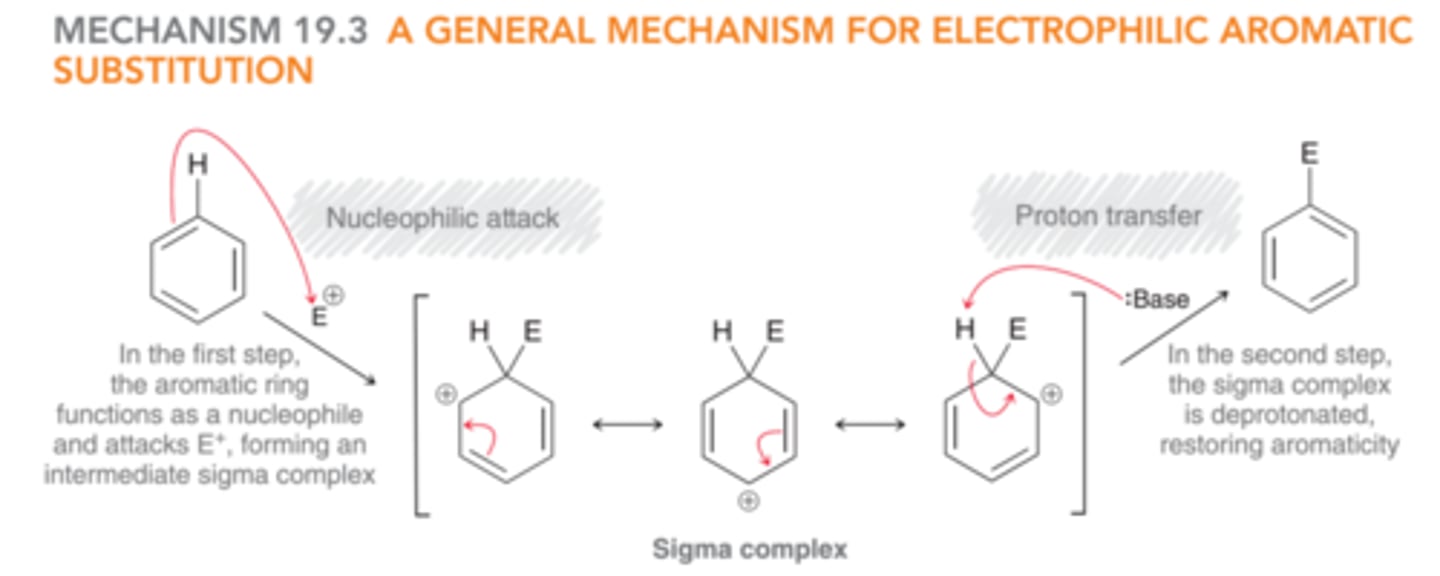
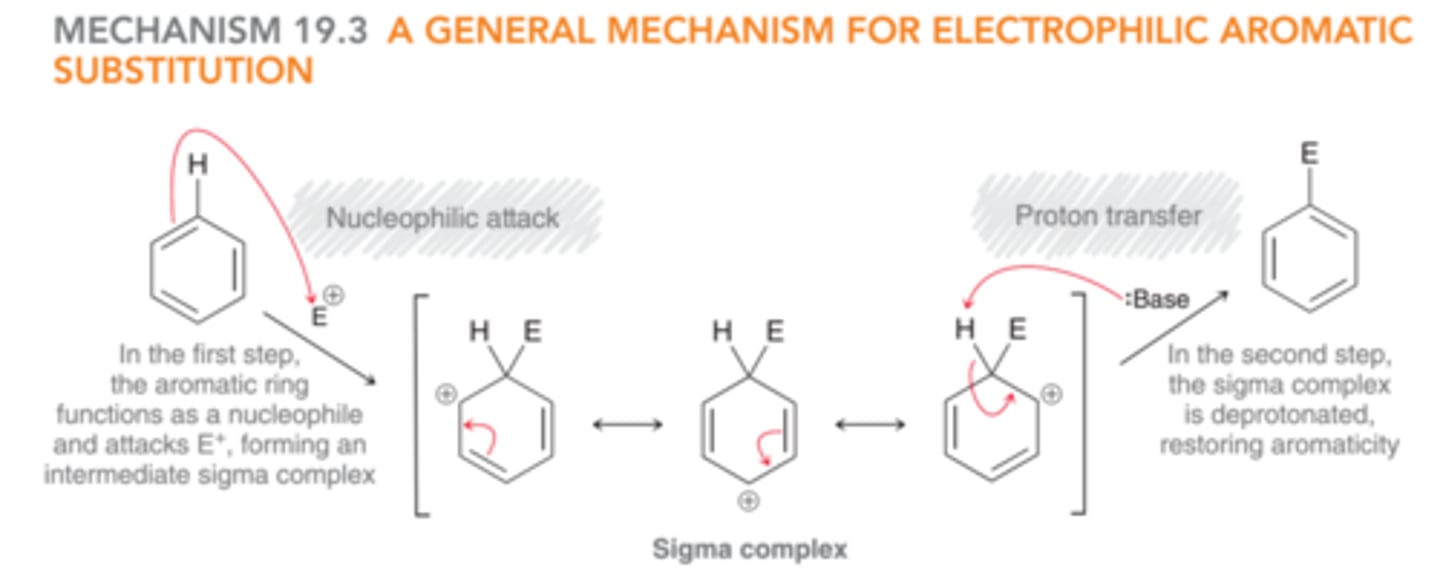
General Mechanism for Electrophilic Aromatic Substitution
1) The aromatic ring functions as the nucleophile & attacks an electrophile to form a sigma complex.
2) The sigma complex is deprotonated to restore aromaticity.
Sulfonation
An electrophilic aromatic substitution reaction in which an SO3H group is installed on an aromatic ring
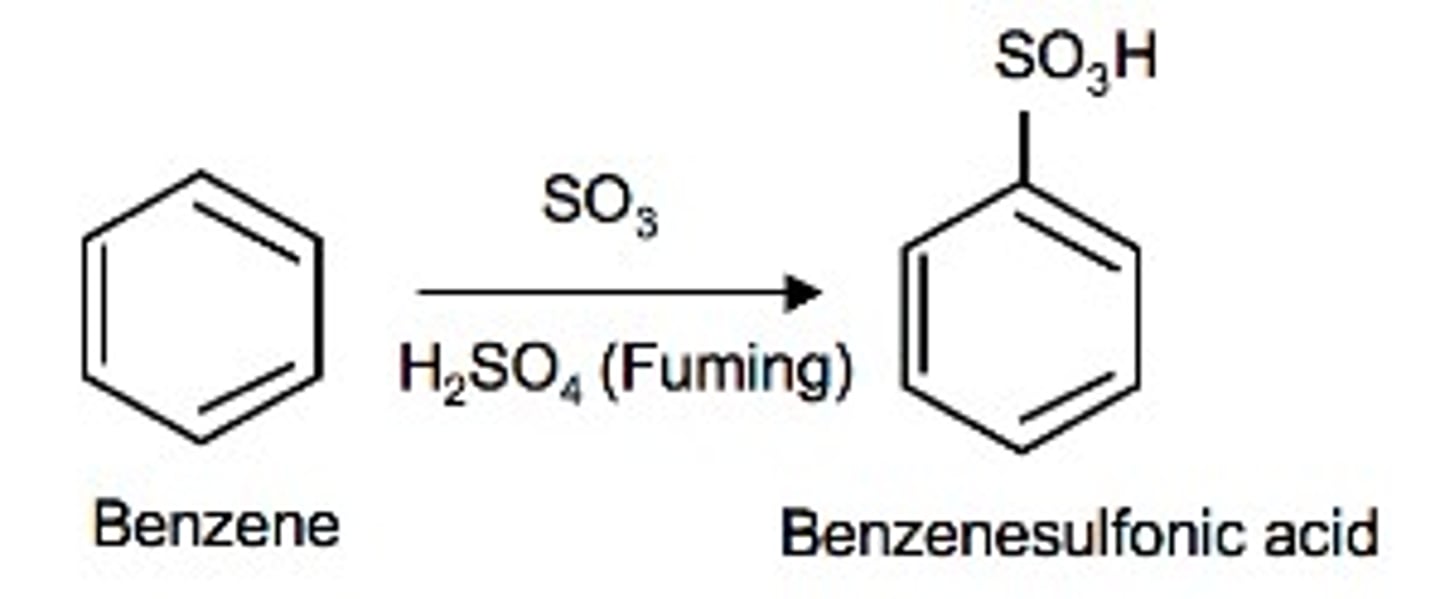
How to add SO3H to a benzene ring?
Reagent: Fuming H2SO4 (a mixture of H2SO4 and SO3 gas)
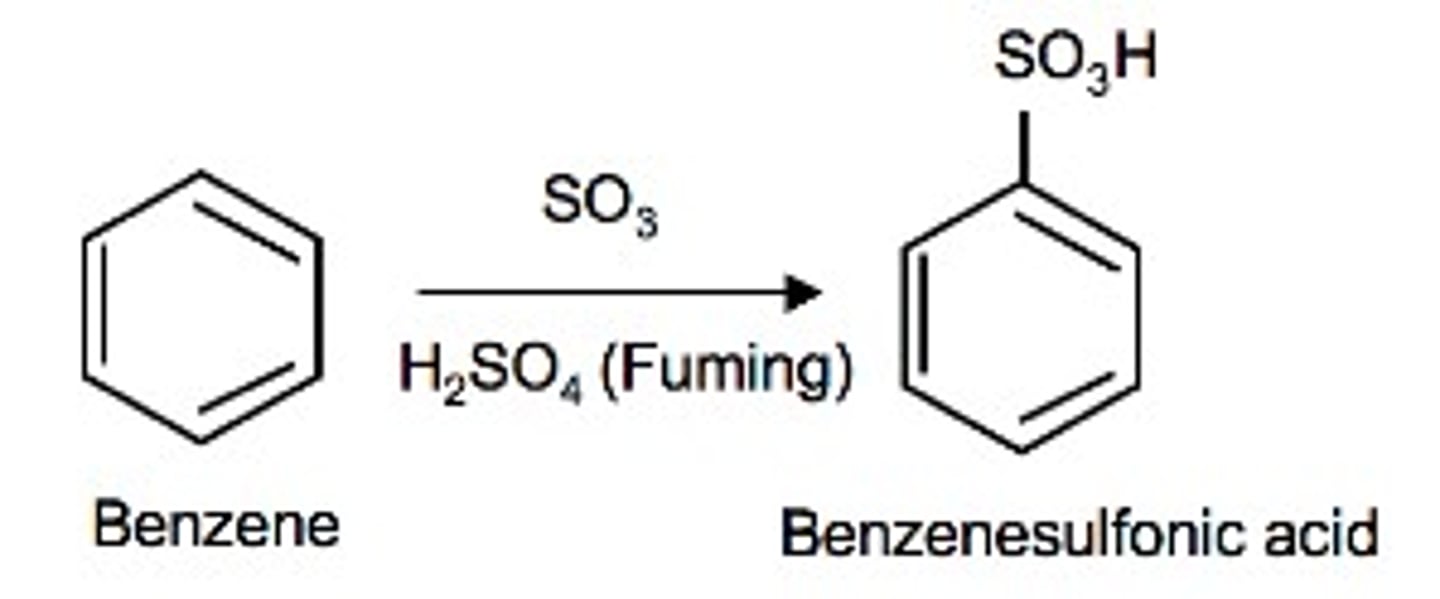
What is the electrophile in sulfonation?
SO3
It is a good electrophile due to the inefficient orbital overlap and the highly polarized bonds.
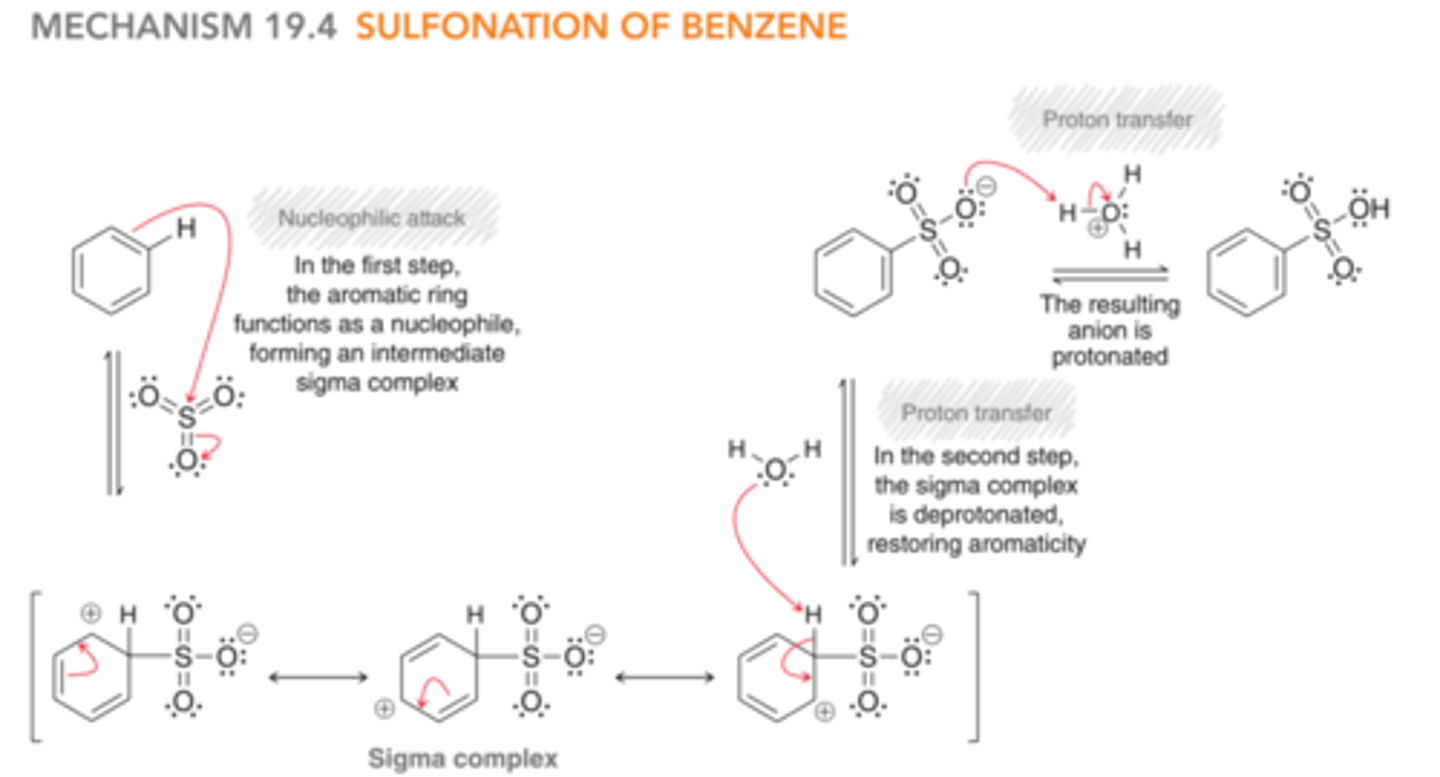
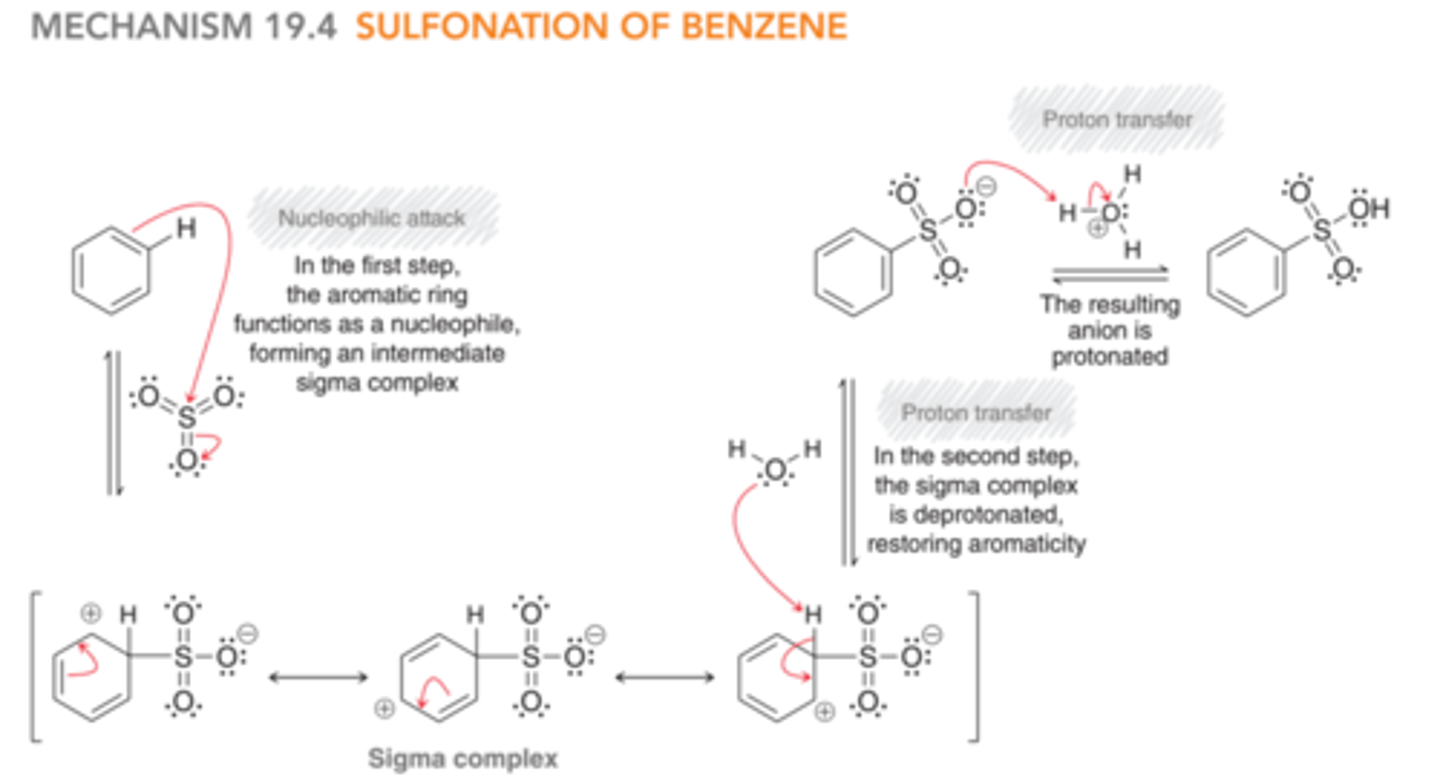
Mechanism: Sulfonation of Benzene
1) The aromatic ring is the nucleophile and attacks sulfur of SO3, forming a sigma complex
2) The sigma complex is deprotonated to restore aromaticity.
3) The SO3 anion is protonated to balance charge
How to reverse from sulfonation?
Use diluted H2SO4 to remove the SO3H group and replace the hydrogen.
Nitration
An electrophilic aromatic substitution reaction that involves the installation of a nitro group (NO2) on an aromatic ring
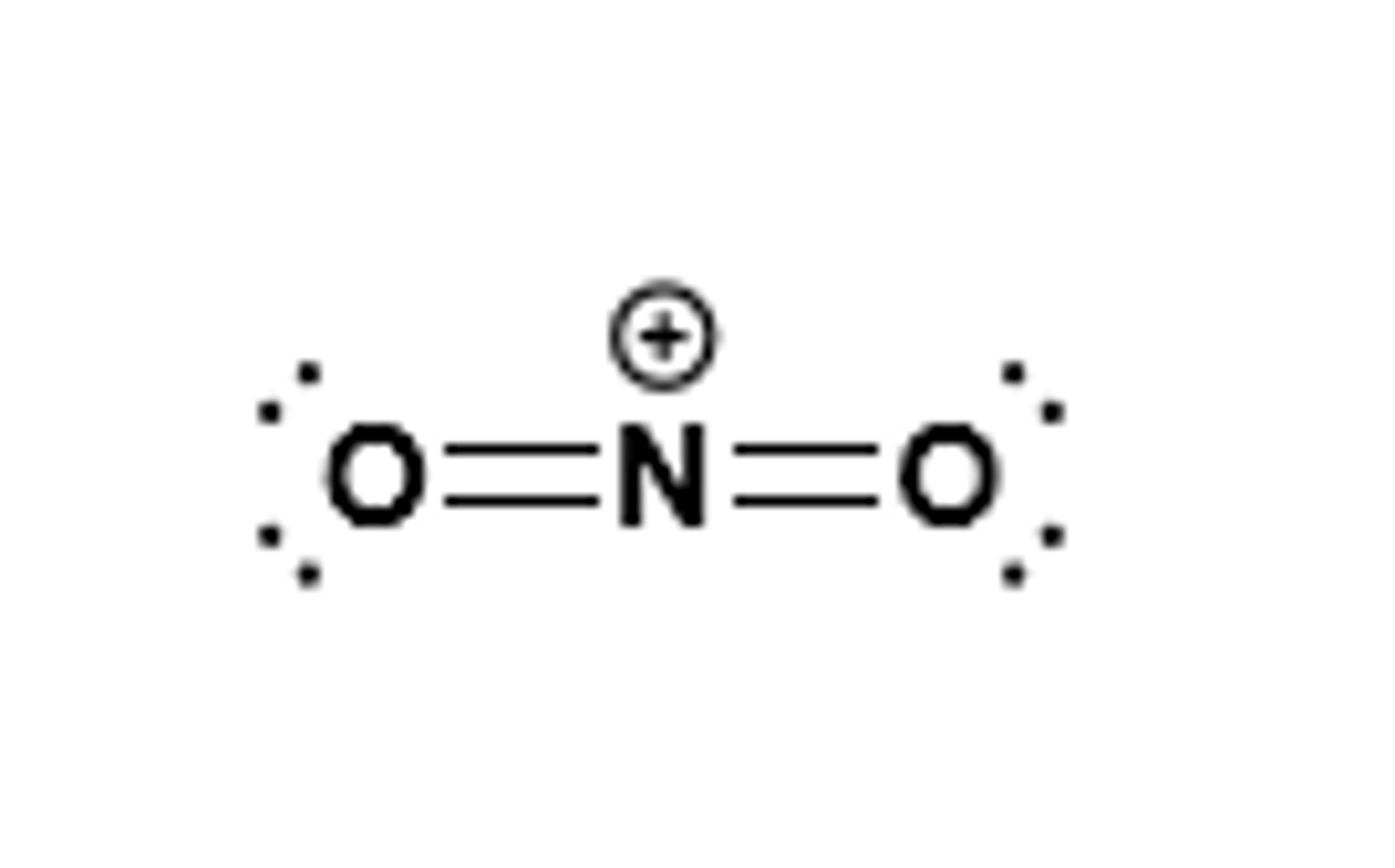
NO2+ is the electrophile
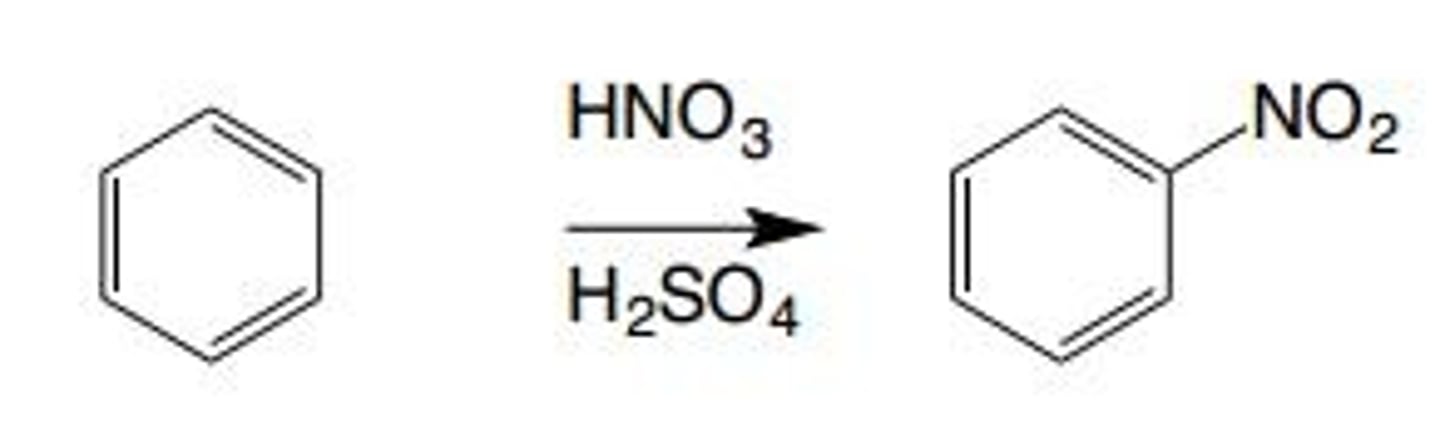
What is the electrophile in nitration?
NO2+ ion, which is present in a mixture of HNO3 and H2SO4 (nitronium ion)
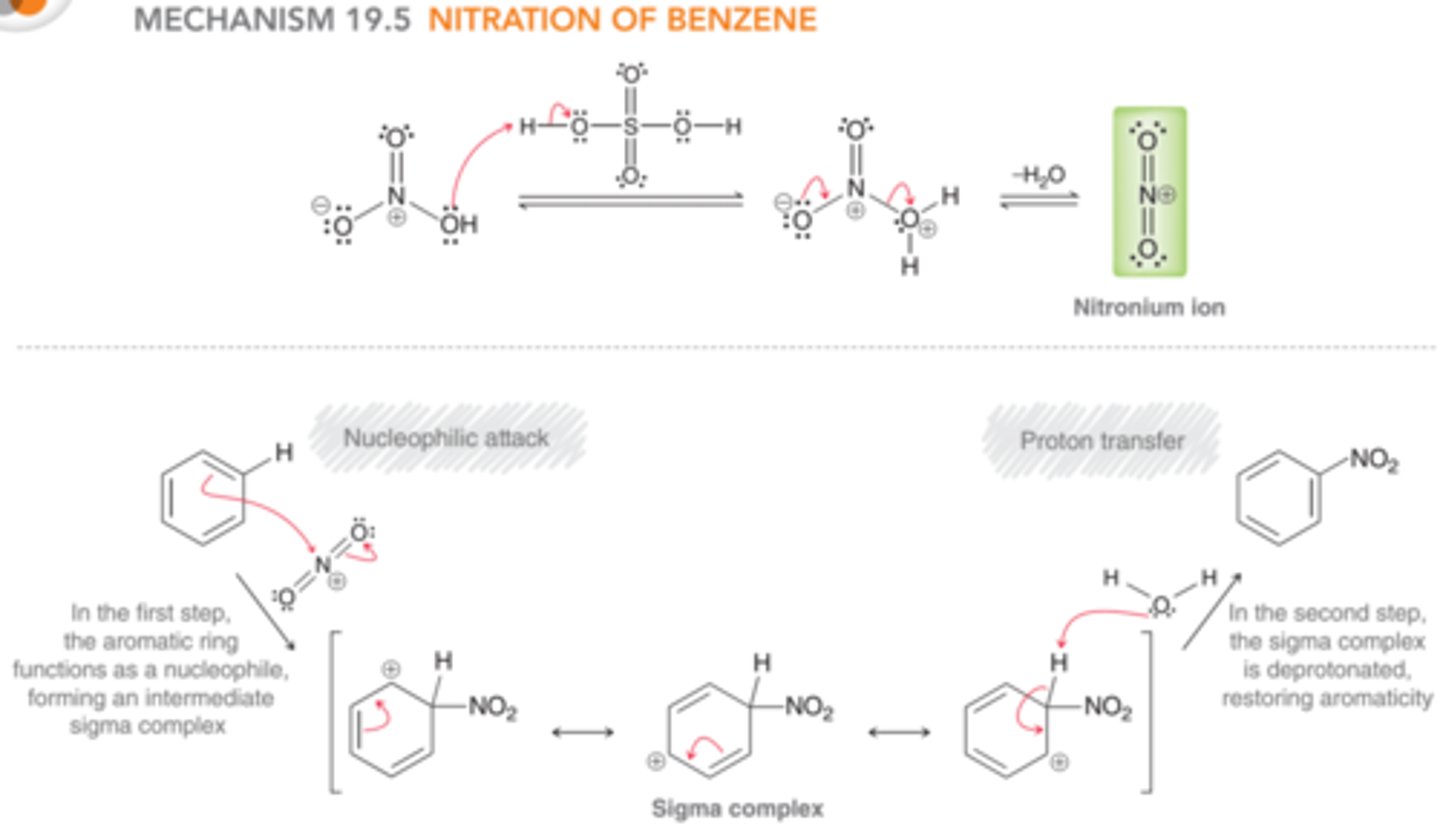
Mechanism: Nitration of Benzene
Nitric acid functions as a base to accept a proton from sulfuric acid, followed by loss of water to produce NO2+
Nucleophilic attack generating a sigma complex, then deprotonation to restore aromaticity
In nitration, what is the base and what is the acid?
Base - nitric acid/HNO3
Acid - sulfuric acid/H2SO4
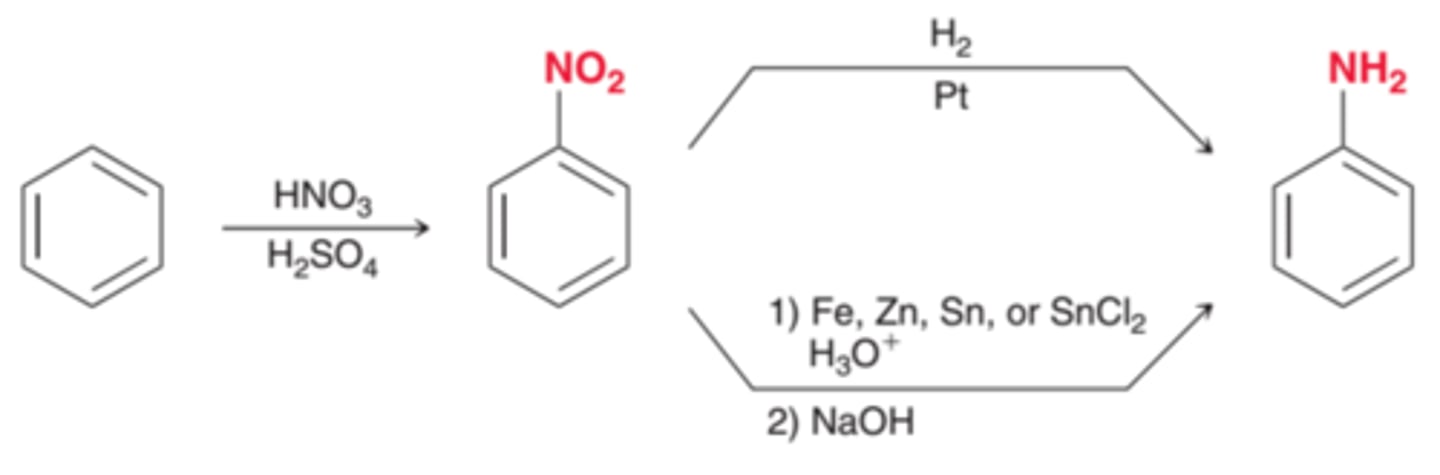
A nitro/NO2 group can be reduced to give an ____________ group by...
NO2 can be reduced to give a NH2 group
1) Fe or Zn, HCl
2) NaOH

How to install an amino group onto an aromatic ring?
1) HNO3/H2SO4
2) Fe or Zn, HCl
3) NaOH
Friedel-Crafts Alkylation
An electrophilic aromatic substitution reaction that installs an alkyl group on an aromatic ring
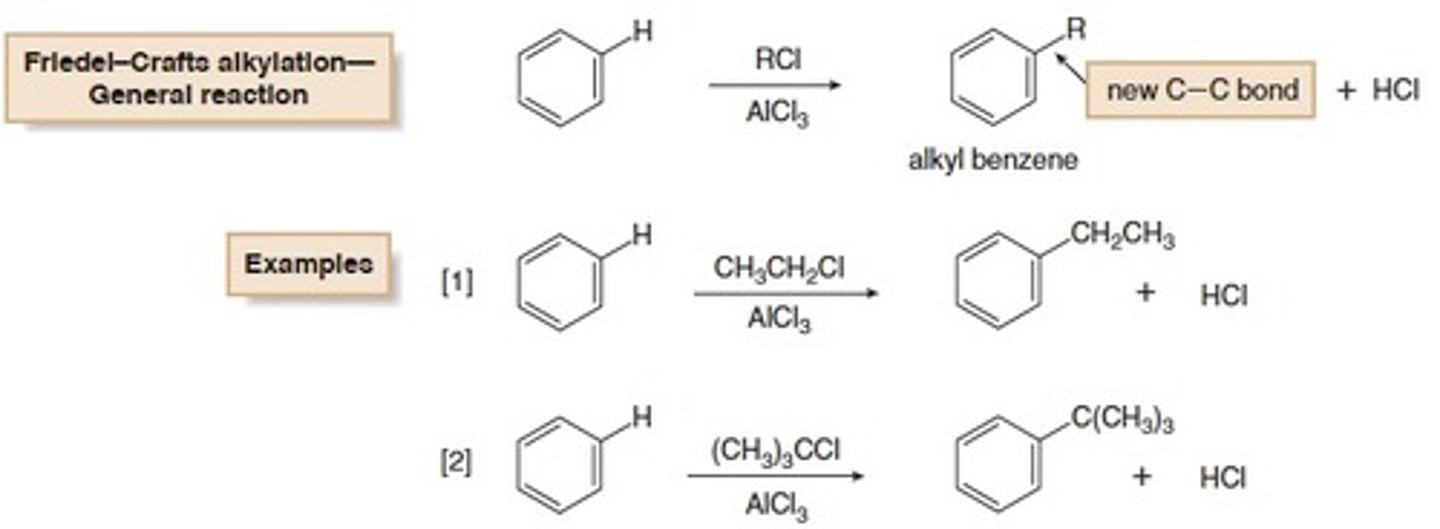
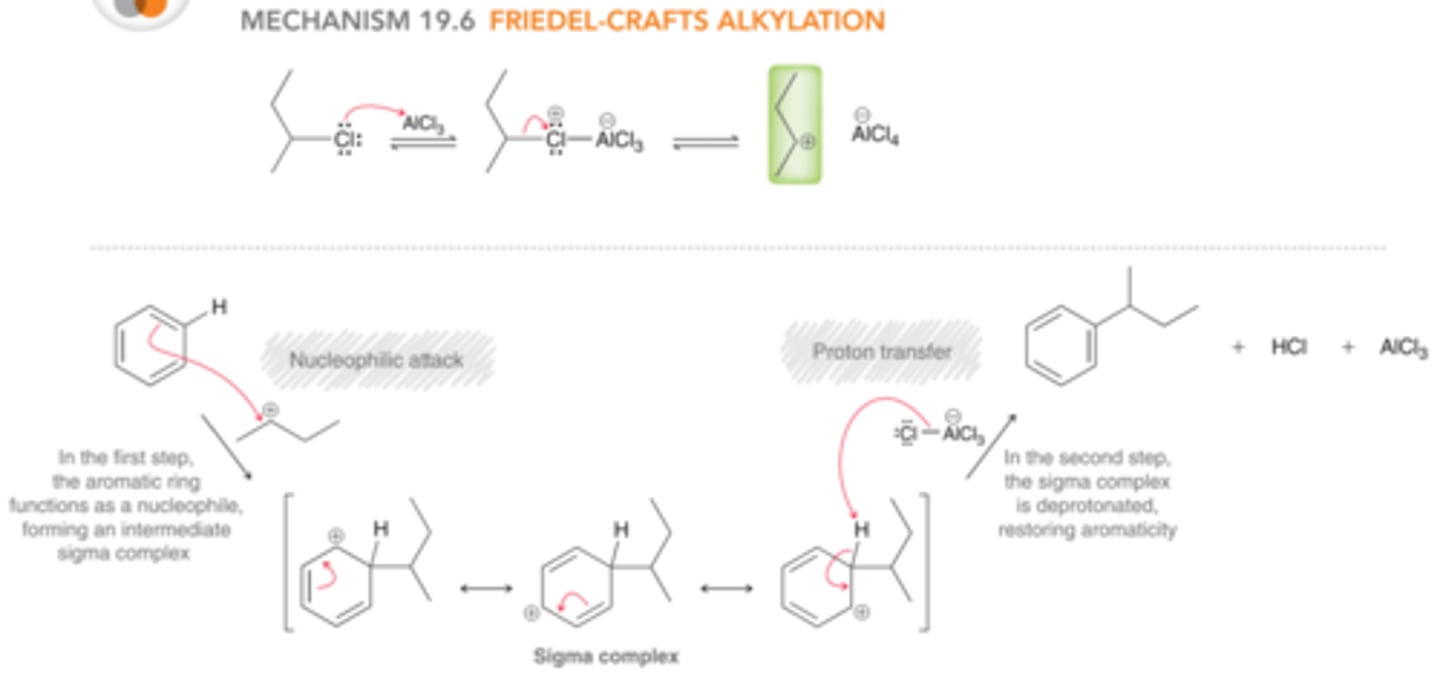
Mechanism: Friedel-Crafts Alkylation
1st step: The aromatic ring functions as a nucleophile and attacks the carbocation, forming a sigma complex
2nd step: The sigma complex is then deprotonated to restore aromaticity
What kind go alkyl halides are converted into carbocation in the presence of AlCl3?
2° and 3°
Most 1° alkyl halides (besides ethyl chloride) cannot be sued because their complexes with AlCl3 undergo rearrangement to form 2° or 3° carbocations.
When alkyl halides in Friedel-Crafts alkylation undergo rearrangement, what kind of product do you get?
a mixture of products
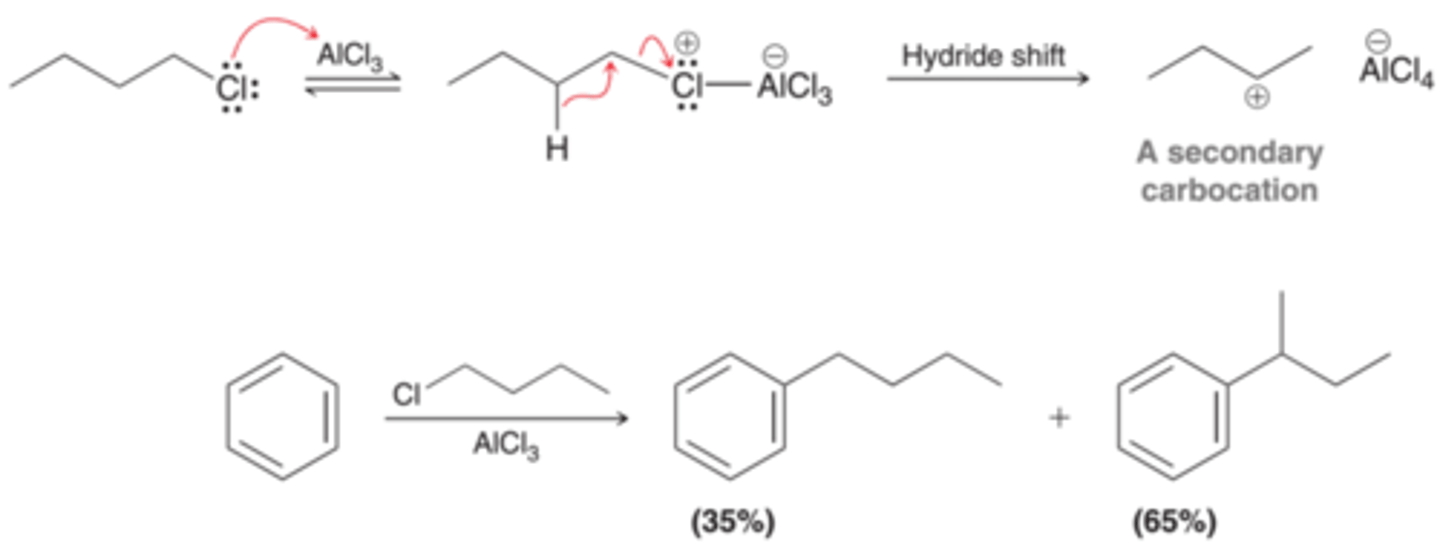
Limitations of Friedel-Crafts Alkylation
1) When choosing an alkyl halide, the C atom connected to the halogen must be sp3 hybridized
2) Installation of an alkyl group activates the ring toward further alkylation. Therefore, polyalkylations often occur (can be avoided by choosing conditions that favor monoalkylation)
3) There are certain groups, such as a nitro group, that are incompatible with a Friedel-Crafts reaction
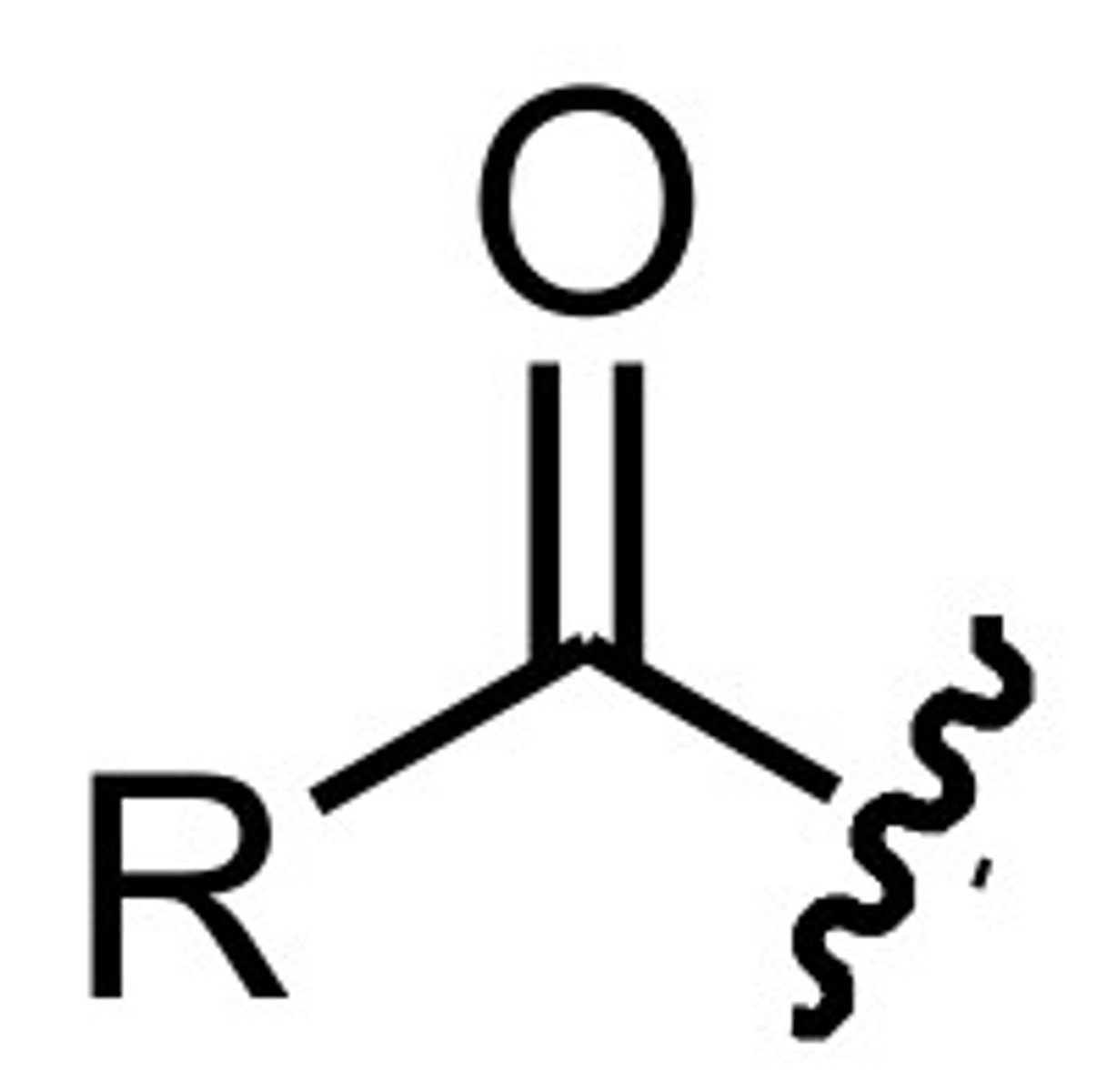
Acyl group
The term describing a carbonyl group (C=O bond) connected to an alkyl group or aryl group
Friedel-Crafts acylation
An electrophilic aromatic substitution reaction that installs an acyl group on an aromatic ring
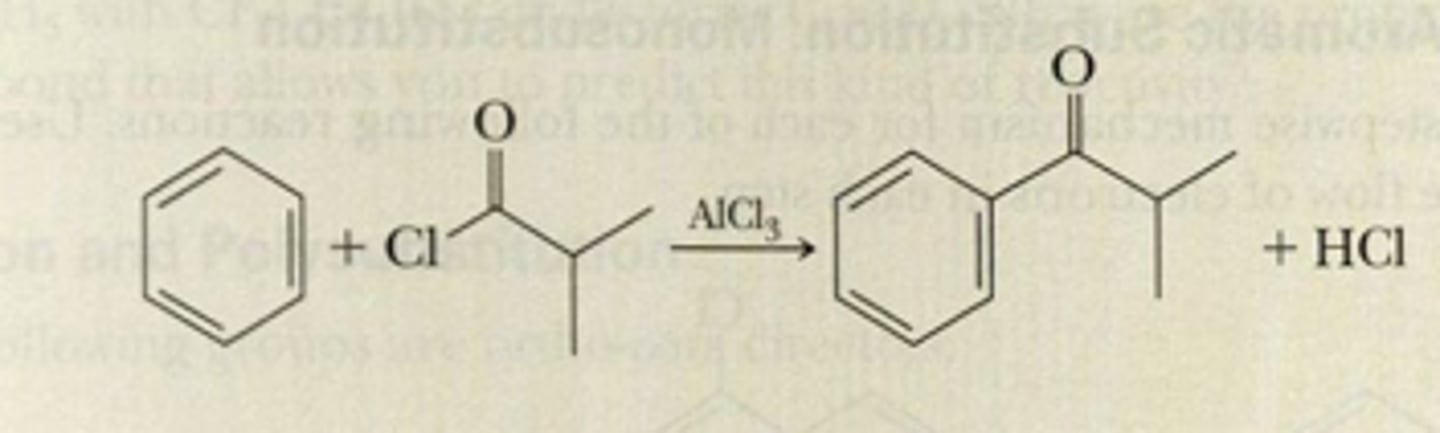
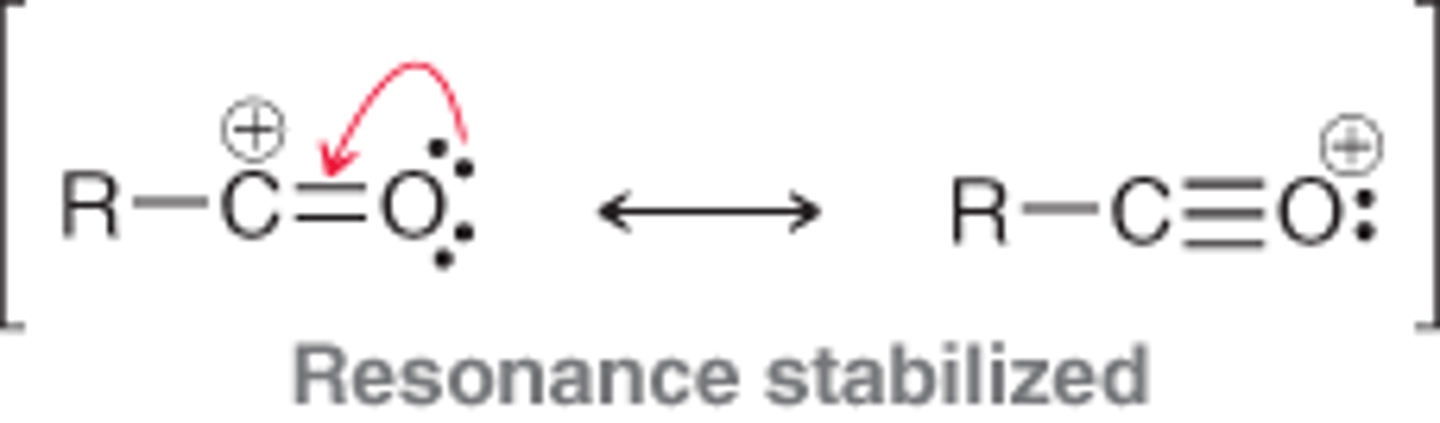
The resonance-stabilized, cationic intermediate of a Friedel-Crafts acylation, formed by treating an acyl halide with aluminum trichloride. Do not rearrange.
acylium ion
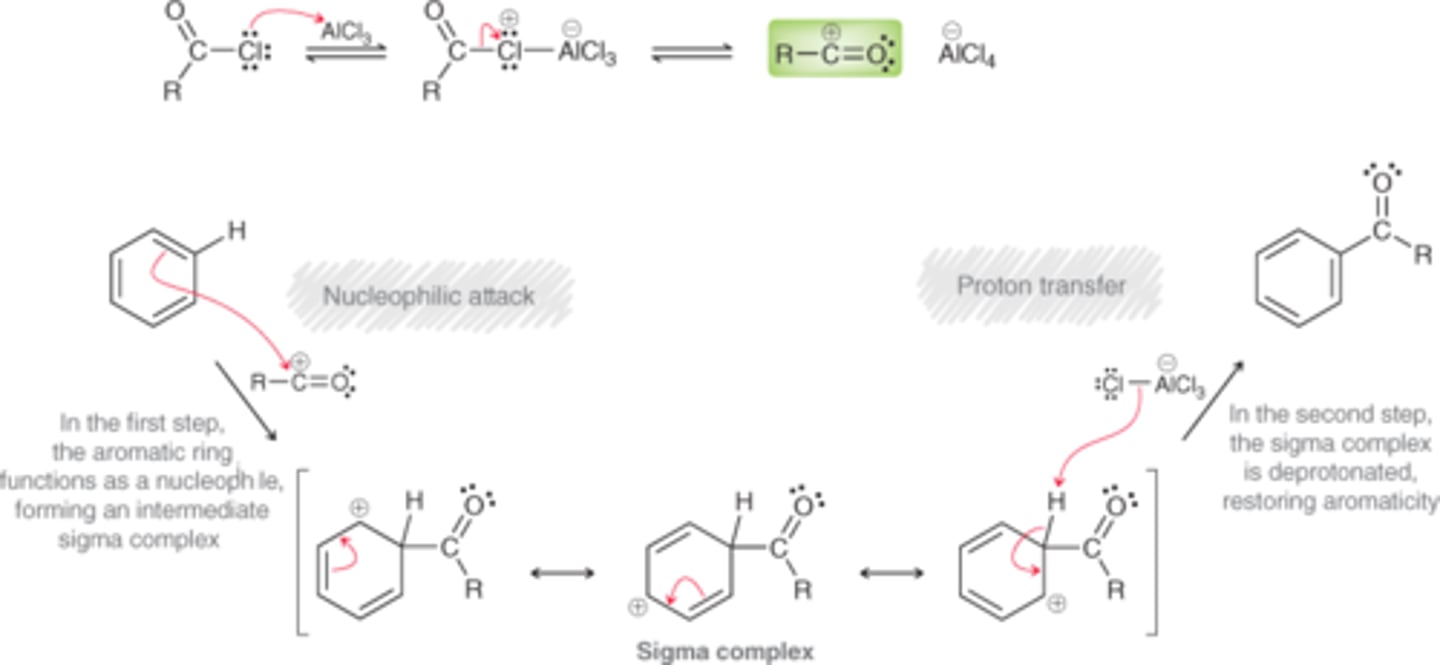
Mechanism: Friedel-Crafts Acylation
The acylium ion is attacked by benzene, which produces an intermediate sigma complex, when is then deprotonated to restore aromaticity.
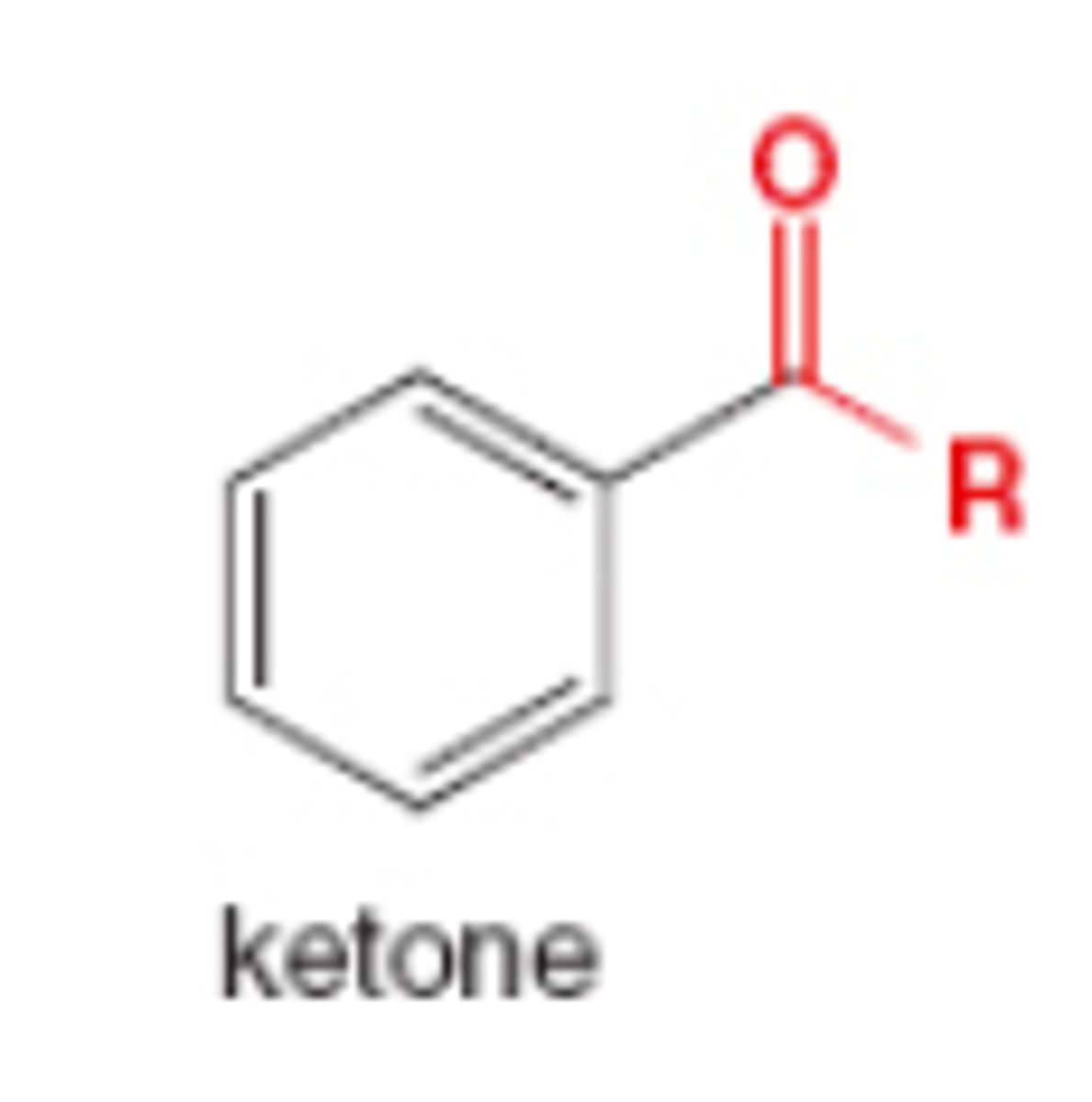
The product of a Friedel-Crafts acylation is a...
aryl ketone
Clemmensen reduction
A reaction in which a carbonyl group is completely reduced and replaced with a methylene (CH2) group
Zn(Hg)
--------->
HCl, heat
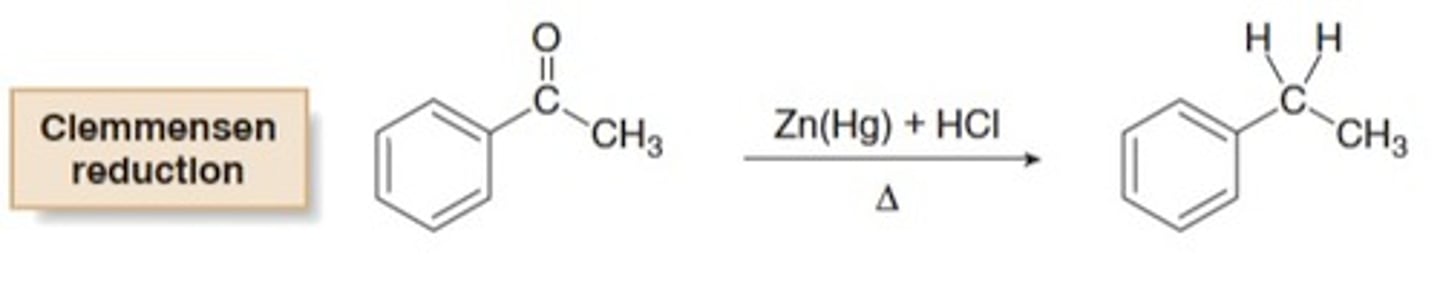
When a Friedel-Crafts acylation is followed by a Clemmensen reduction, the net result is the...
Why is this method useful?
Installation of an alkyl group
Useful for installing alkyl groups that cannot be installed with a direct alkylation process due to carbocation rearrangements that would give a mixture of products
Polyacylation is not observed
For a substituted aromatic ring, the effect of an electron-donating substituent that increases the rate of electrophilic aromatic substitution
Activate
Why are alkyl groups considered activators?
Alkyl groups are electron donating, which stabilizes the positively charged sigma complex & lowers the energy of activation for its formation.
a group that directs the regiochemistry of an electrophilic aromatic substitution reaction such that the incoming electrophile is installed at the ortho or para positions
Ortho-para director
*All ACTIVATORS are ____________ directors.*
*All ACTIVATORS are ORTHO-PARA directors.*
Is a methoxy group an activator or deactivator?
activator
Resonance pictures show that methoxy donates electron density to the ring which stabilizes the positively charged sigma complex & lowers the activation energy.
For a substituted aromatic ring, the effect of an electron-withdrawing substituent that decreases the rate of electrophilic aromatic substitution
Deactivate
Is a nitro group an activator or deactivator?
Nitro is a deactivator. It is electron-withdrawing due to the positive charge on nitrogen.
Electron-donating groups are called _______________ and are _______________ directors.
Electron-donating groups are called ACTIVATORS and are ORTHO-PARA directors.
Donating electron density to the ring would stabilize the positively charged sigma complex and activate the reaction.
Electron-withdrawing groups are called _______________ and are _______________ directors.
Electron-withdrawing groups are called DEACTIVATORS and are META directors.
Withdrawing electron density from the ring would destabilize the positively charged sigma complex & deactivate the reaction.
Halogens are the exception. They are electron-withdrawing groups/deactivators and ortho-para directors.
An electron-withdrawing group that directs the regiochemistry of an electrophilic aromatic substitution reaction such that the incoming electrophile is installed at the meta position
Meta director
Most deactivators are ______________ directors.
Most deactivators are META directors.
Halogens are deactivators that are ortho-para directors.
Why are halogens ortho-para directors even though they are deactivators (electron withdrawing)?
Halogens are electronegative & are inductively electron withdrawing. In resonance pictures, halogens are electron-donating
In this case, induction is the dominant factor for halogens and so halogens withdraw electron density from the ring, destabilizing the positively charged sigma complex and raising Ea.
Halogens are _____________ directors.
Ortho-para (even though they are deactivators)
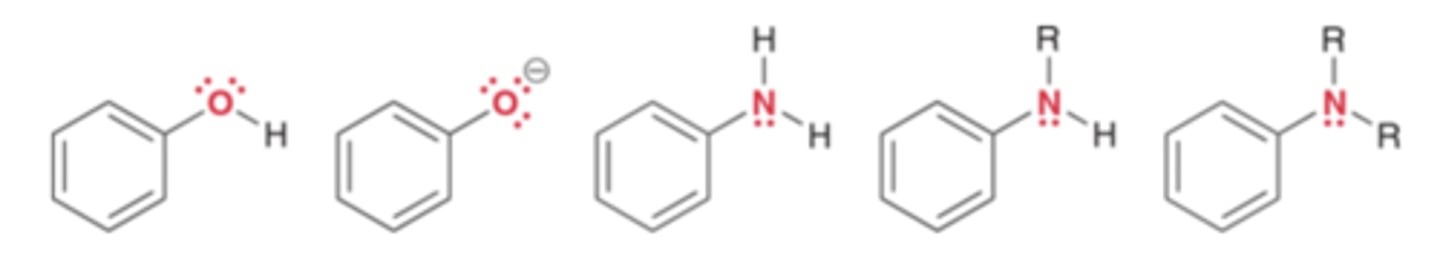
Groups that strongly activate an aromatic ring toward electrophilic aromatic substitution, thereby significantly enhancing the rate of the reaction
Strong activators
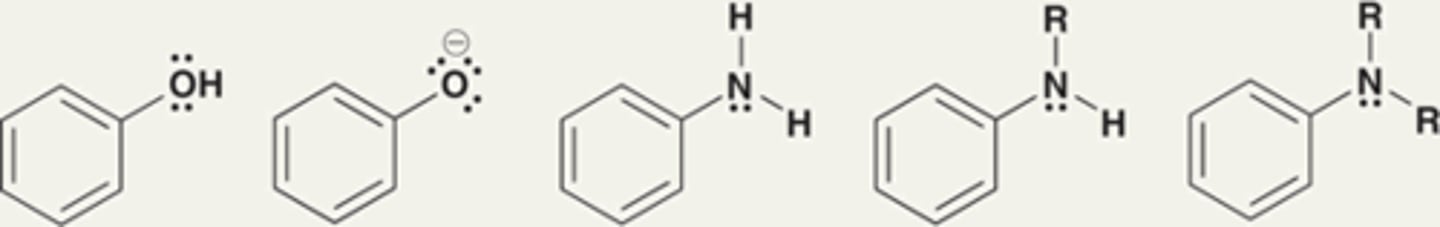
Strong activators are characterized by...
The presence of a lone pair immediately adjacent to the aromatic ring. The lone pair is delocalized into the ring.
Groups that moderately activate an aromatic ring toward an electrophilic aromatic substitution reaction
Moderate activators
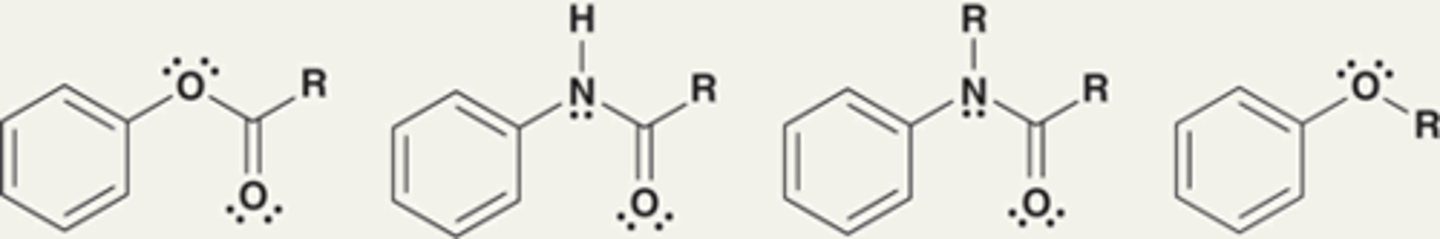
Moderate activators are characterized by...
Exhibit a lone pair that is already delocalized outside of the ring.
Alkoxy groups are moderate activators.
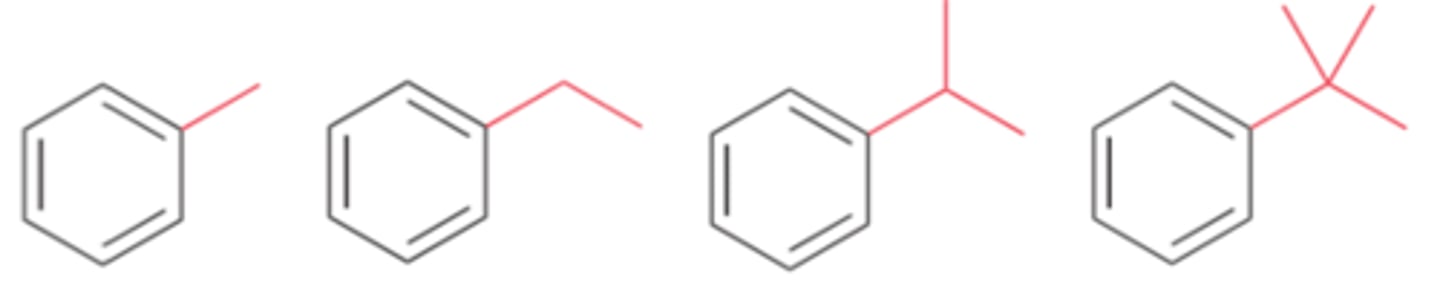
Weak activators include...
alkyl groups
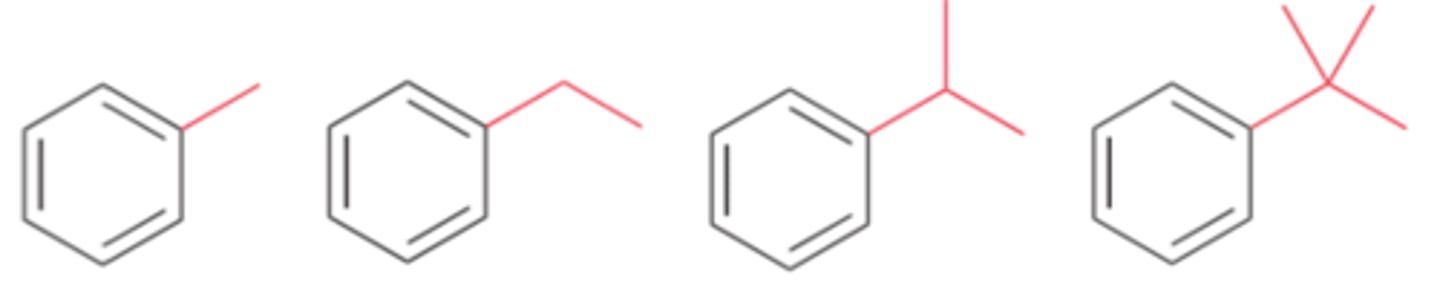
Groups that weakly activate an aromatic ring toward electrophilic aromatic substitution, thereby enhancing the rate of the reaction
Weak activators
Groups that weakly deactivate an aromatic ring toward electrophilic aromatic substitution, thereby decreasing the rate of the reaction
Weak deactivators
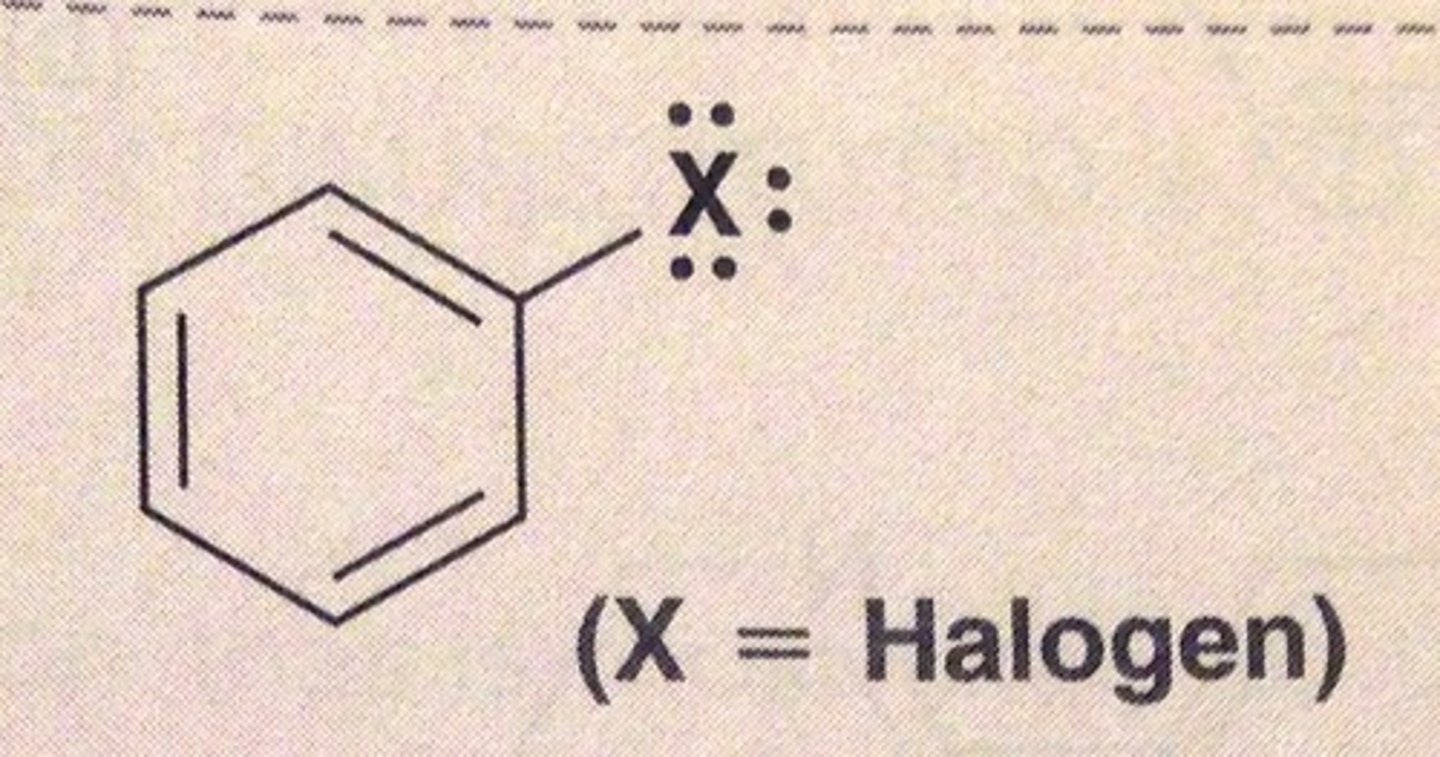
Weak deactivators include...
halogens
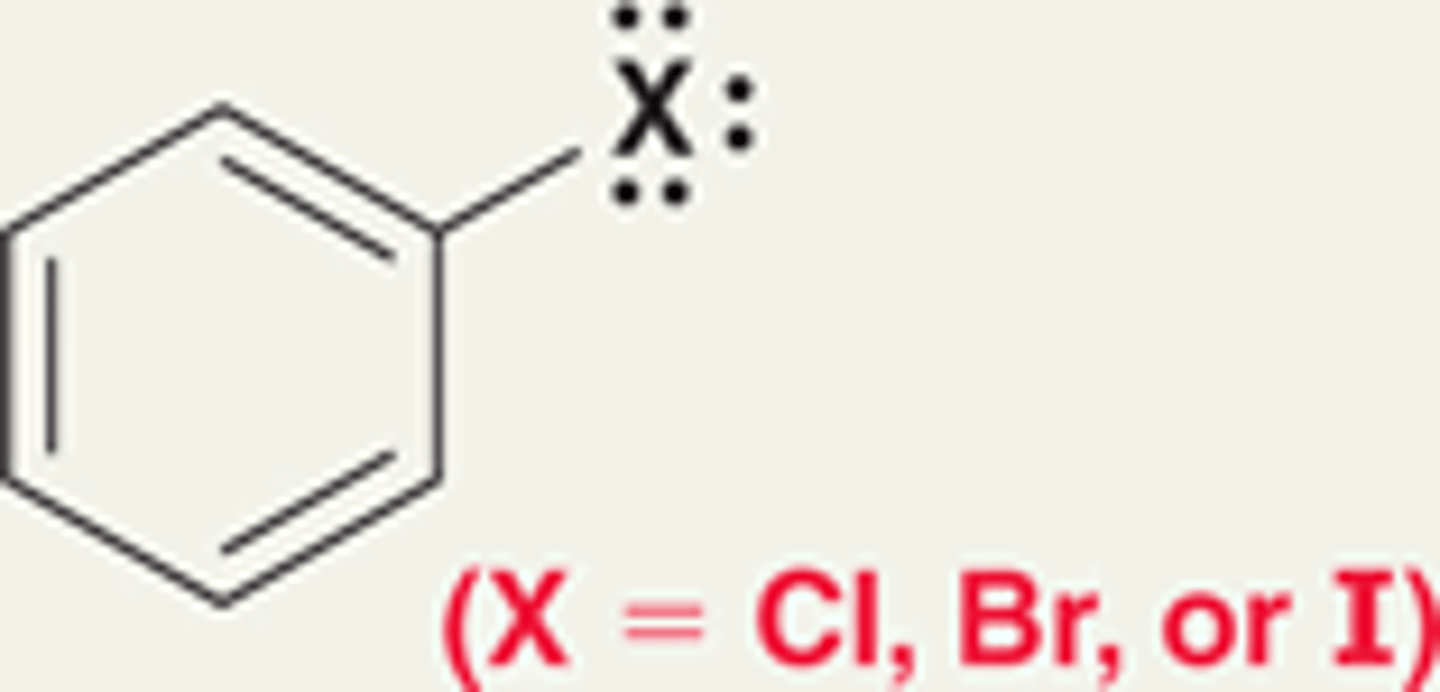
Groups that moderately deactivate an aromatic ring toward an electrophilic aromatic substitution reaction
Moderate deactivators
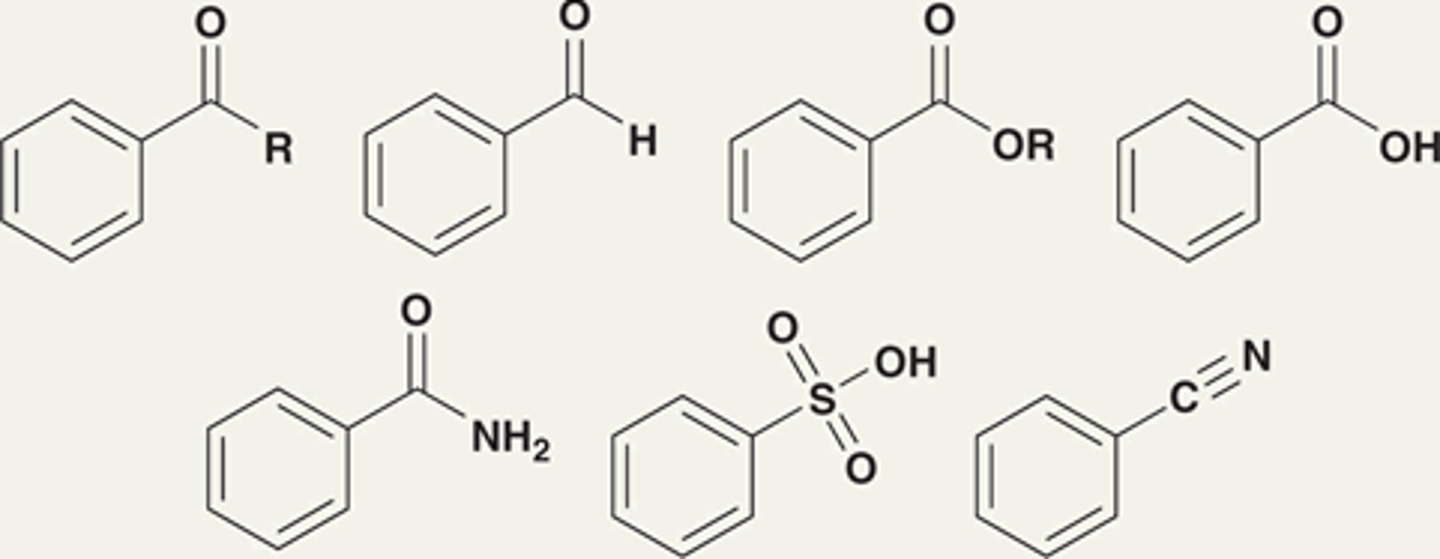
Moderate deactivators are characterized by...
Having a π bond to an electronegative atom, where the π bond is conjugated with the aromatic ring.
Each withdraws electron density by resonance.
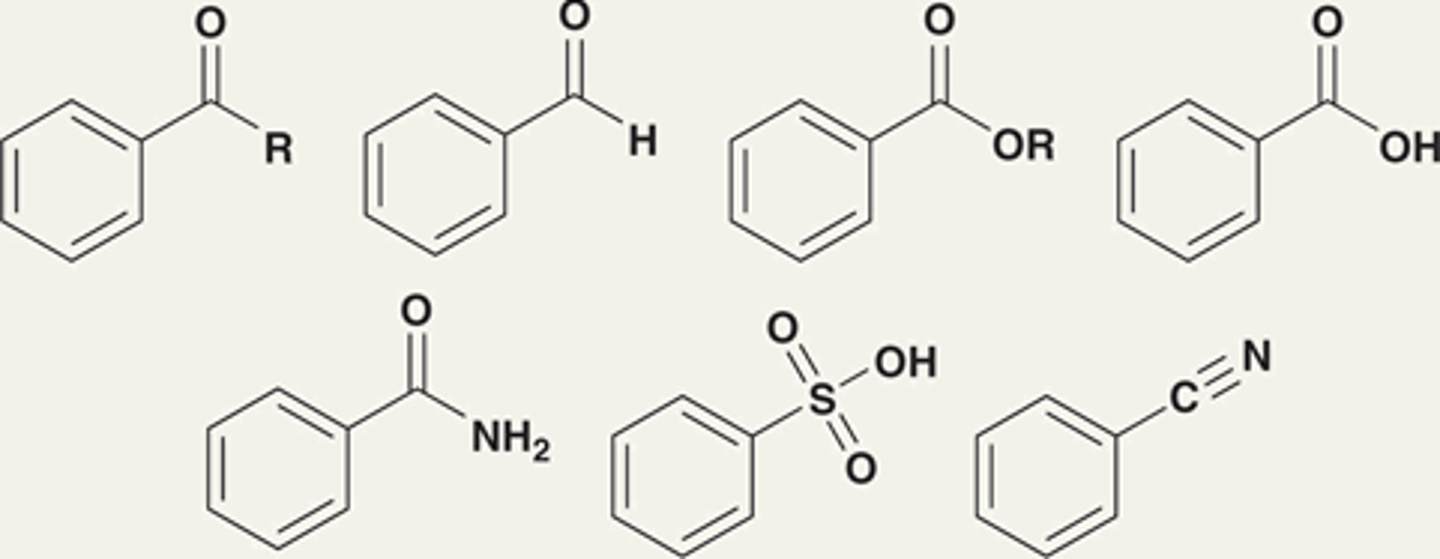
Groups that strongly deactivate an aromatic ring toward electrophilic aromatic substitution, thereby significantly decreasing the rate of the reaction
Strong deactivators
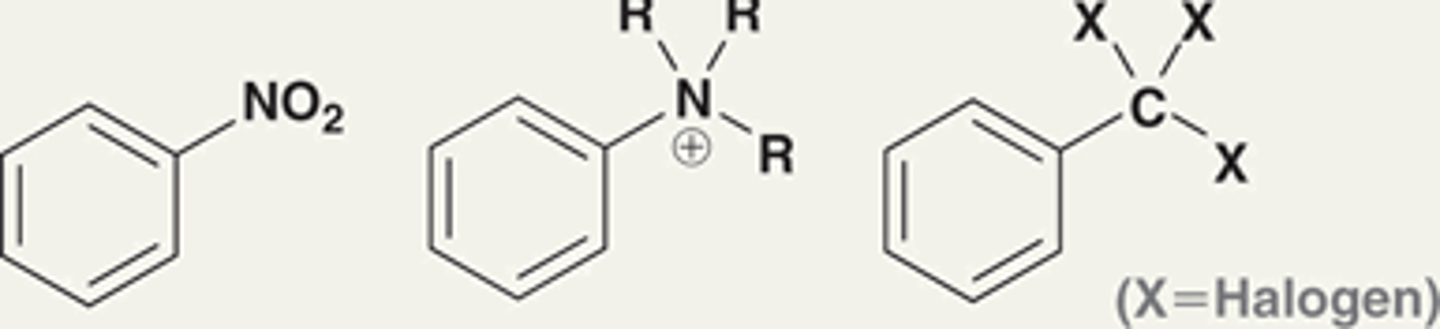
What are the strong deactivators?
Nitro (NO2) group
NR3+ group
CX3 group
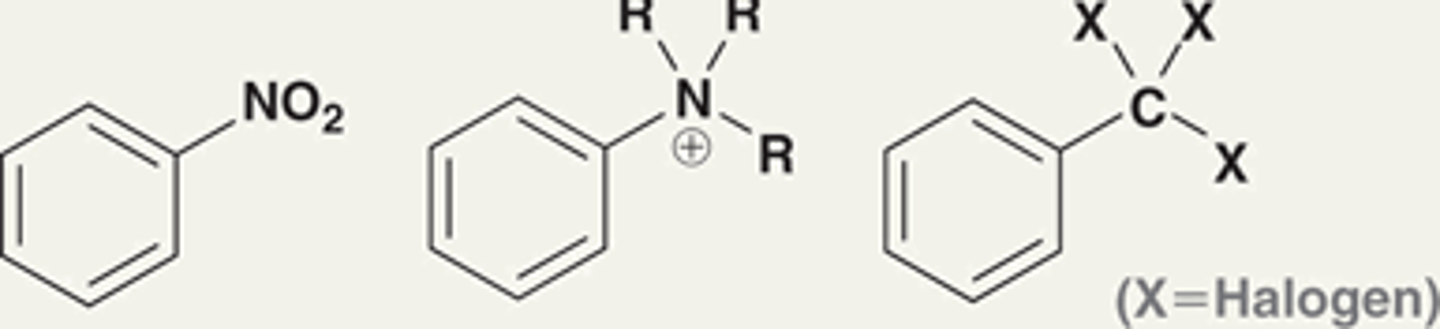
All electron-donating groups are _____________ directors.
All electron-donating groups are ORTHO-PARA directors.
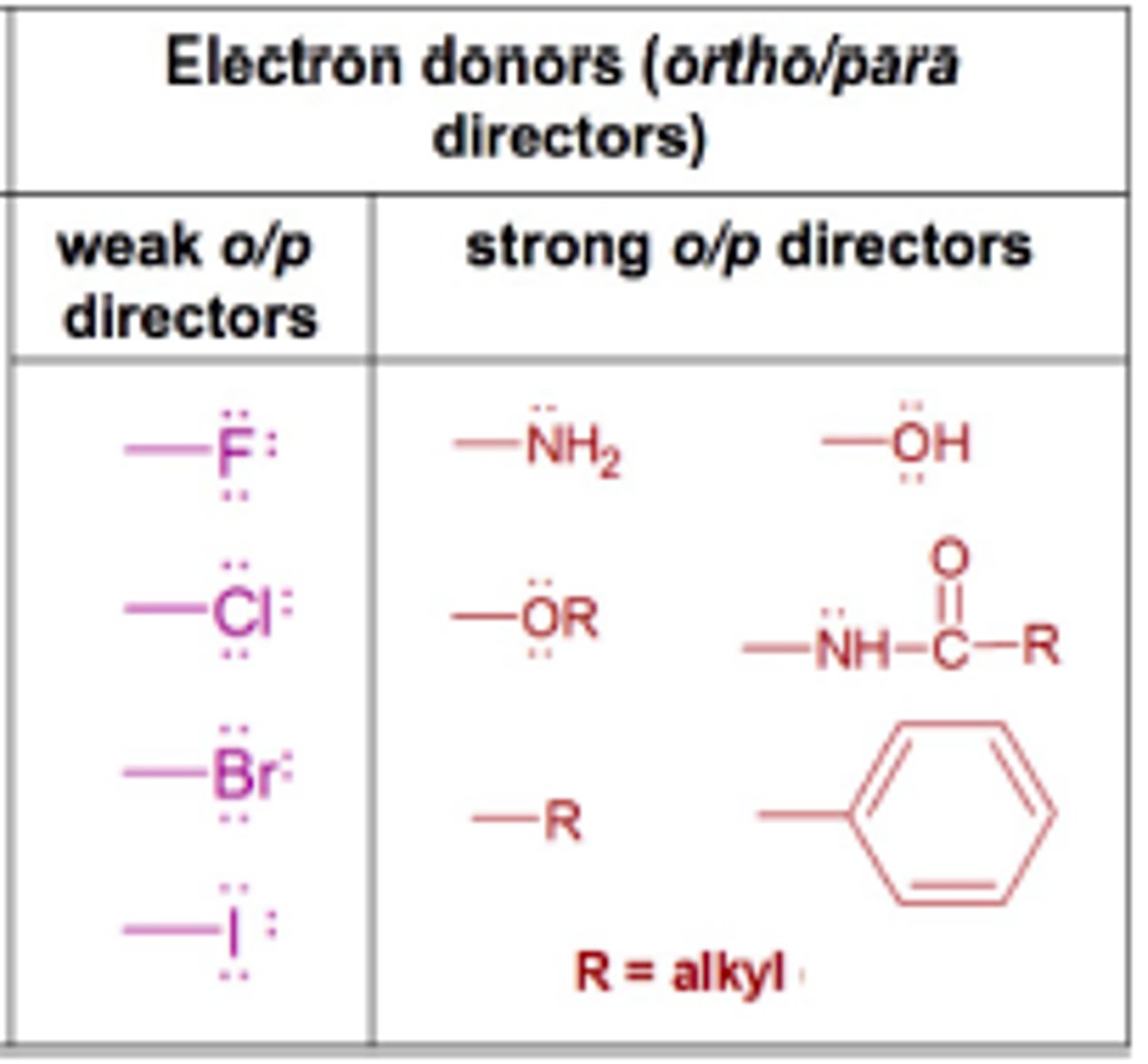
All electron-withdrawing groups are _____________ directors, except the _____________.
All electron-withdrawing groups are META directors, except the HALOGENS.
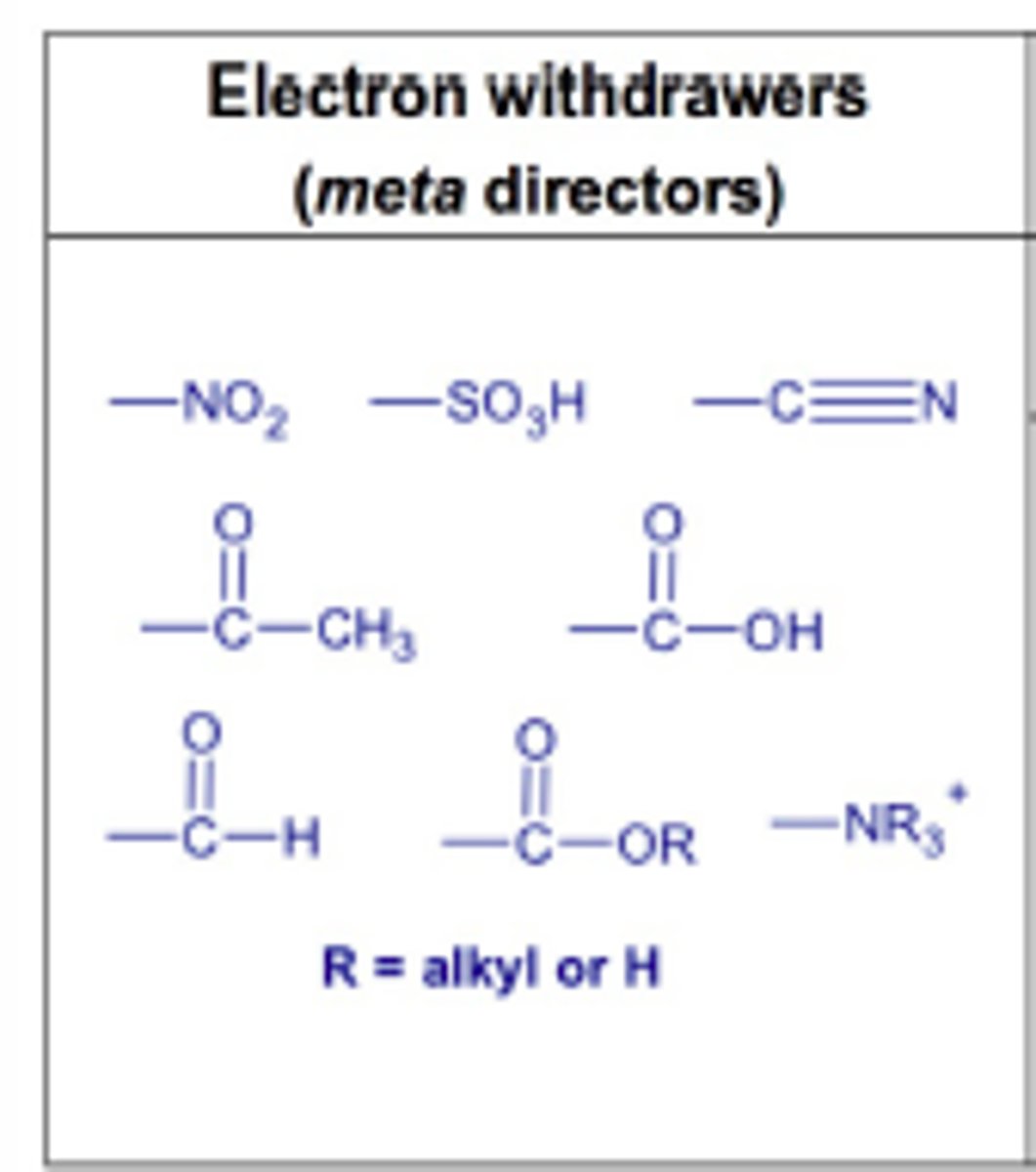
Halogens are (activators OR deactivators) and _____________ directors.
Halogens are DEACTIVATORS and ORTHO-PARA directors.
Deactivators because of withdrawing electrons by induction.
Ortho-para directors due to those positions have one additional resonance picture (stability).
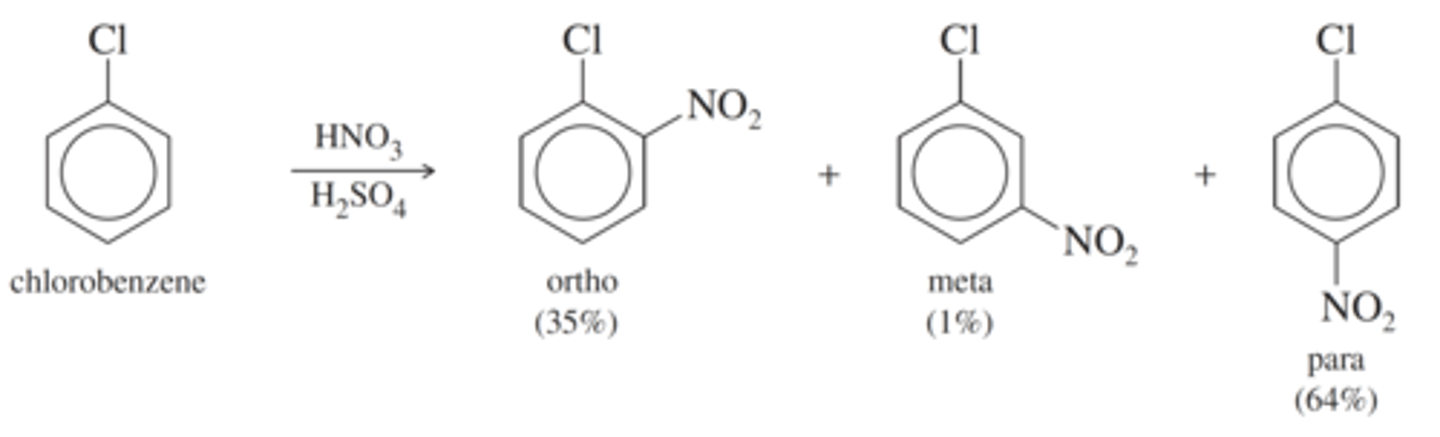
In cases where the directing effects of the substituents compete, what dominates?
The more powerful activating group dominates
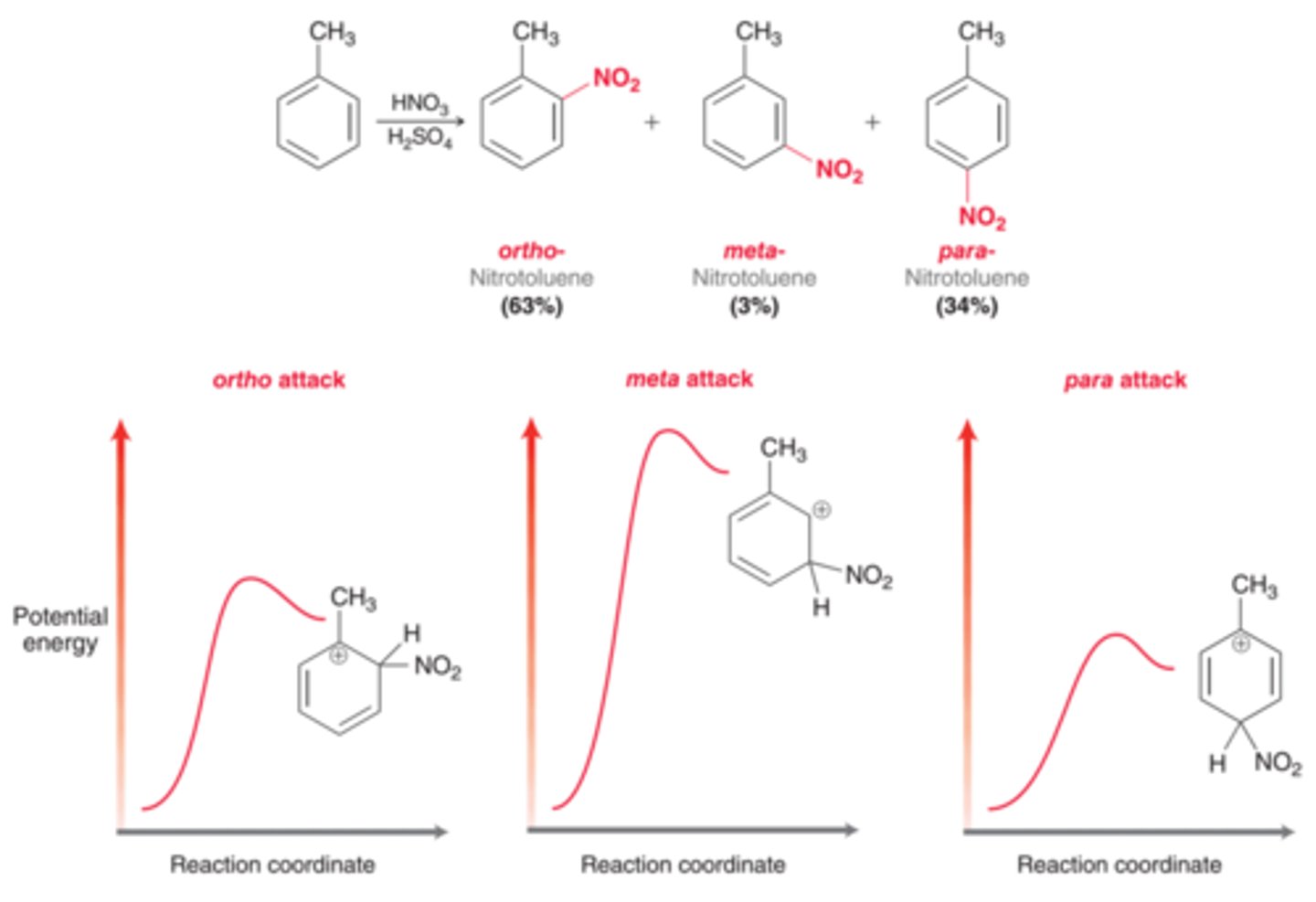
For most monosubstituted aromatic rings, what product dominates?
The para product dominates over the ortho product as a result of steric considerations
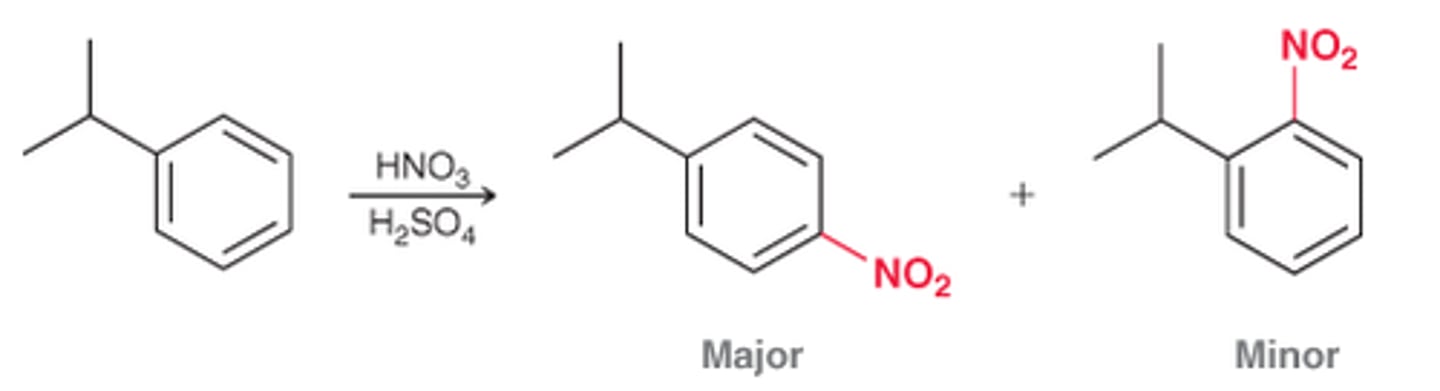
For toluene, the ratio of ortho & para products is...
sensitive to the conditions used, such as the solvent
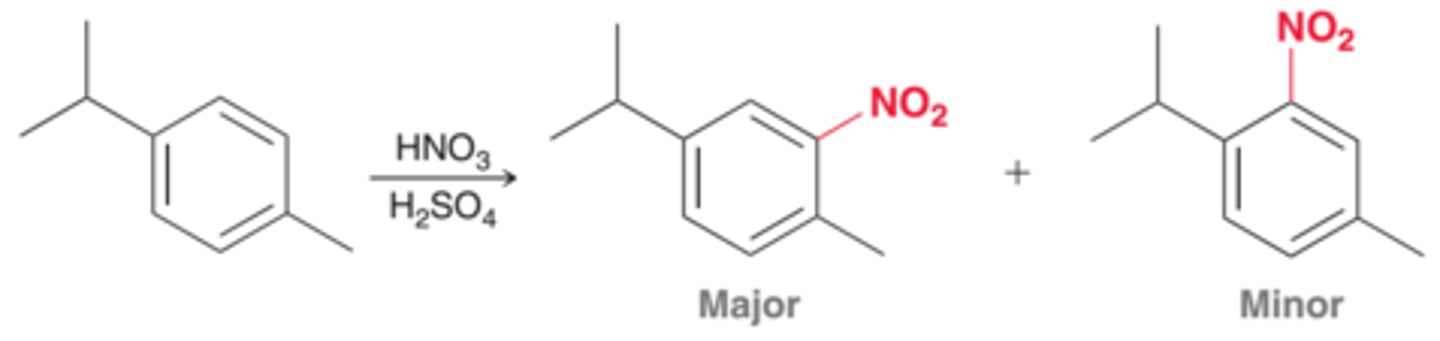
For 1,4-disubstituted aromatic rings, what product is most likely to occur?
The site that is less sterically hindered
For 1,3-disubstituted rings, it is very unlikely that substitution will occur at...
the position between the 2 substituents due to it being too sterically hindered
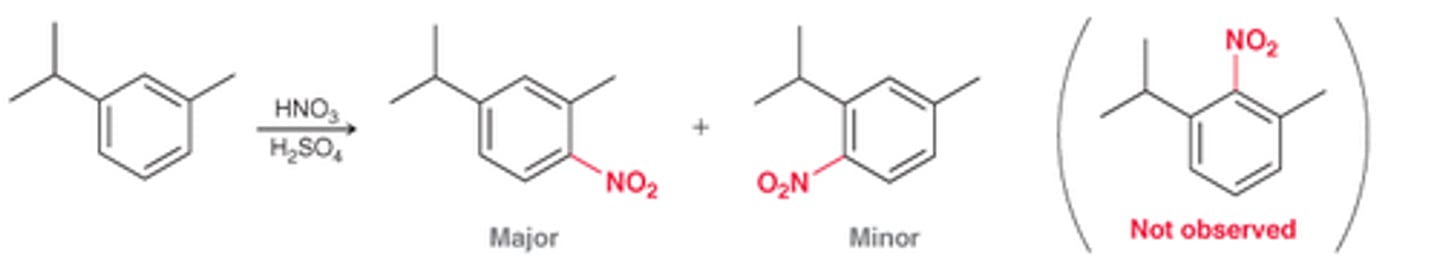
Blocking group
A group that can be readily installed and uninstalled. Used for regiochemical control during synthesis.
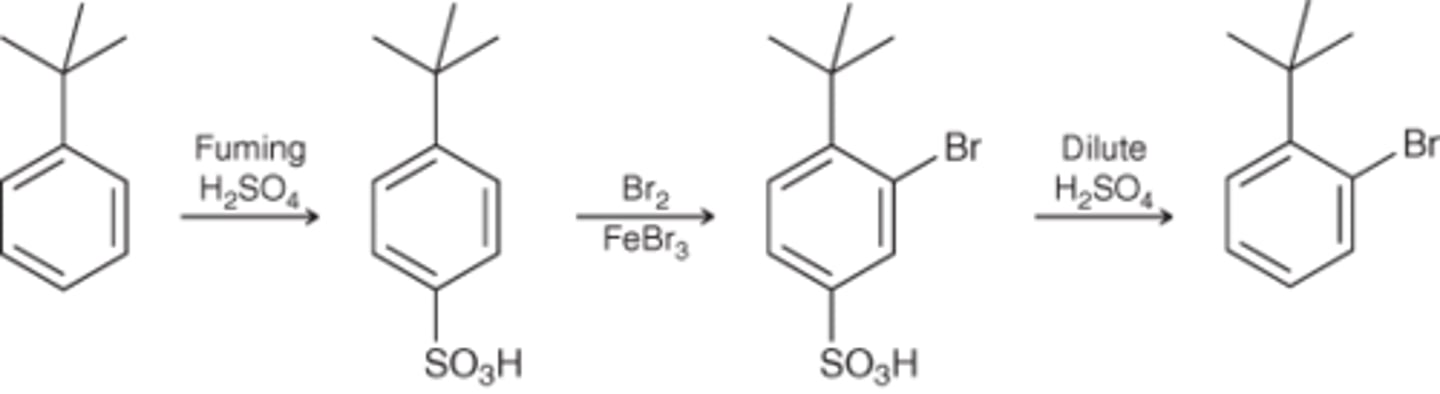
What is the blocking group used?
SO3H
Sulfonation
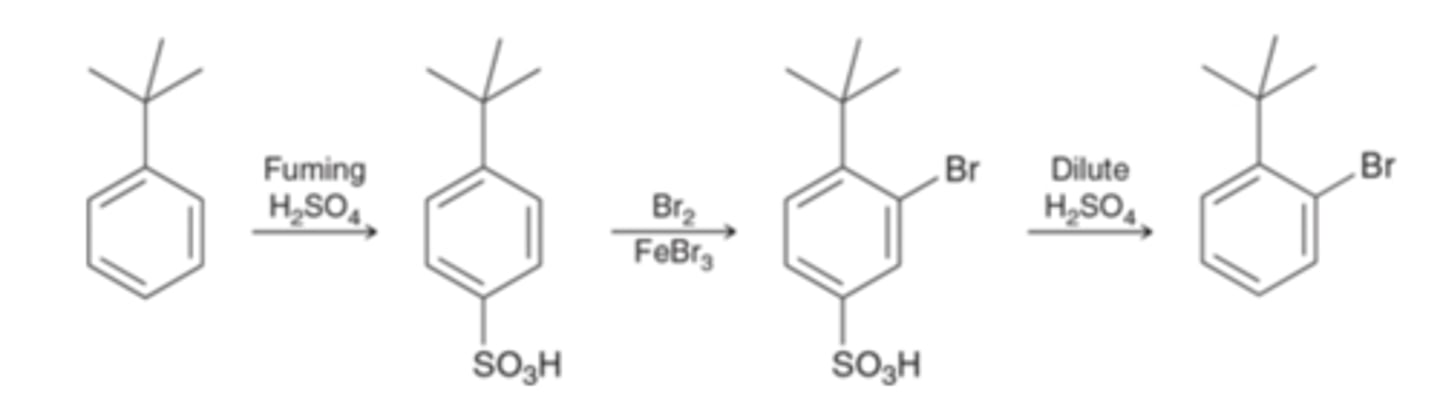
What reagents?
Benzene → Bromobenzene
Br2, FeBr3
What reagents?
Benzene → Chlorobenzene
Cl2, AlCl3
What reagents?
Benzene → Nitrobenzene
HNO3, H2SO4
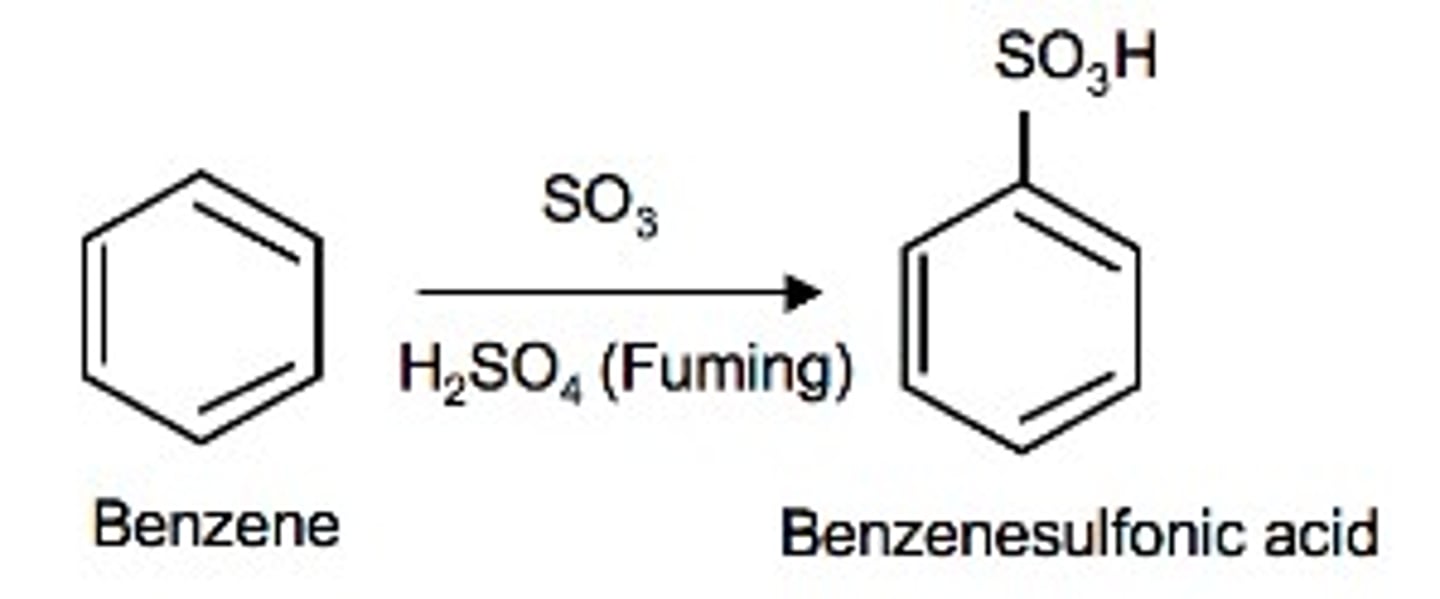
What reagents?
Benzene → benzenesulfonic acid
fuming H2SO4
What reagents?
Benzene → toluene
CH3Cl, AlCl3
What reagents?
Benzene → acetophenone
ClCOCH3, AlCl3
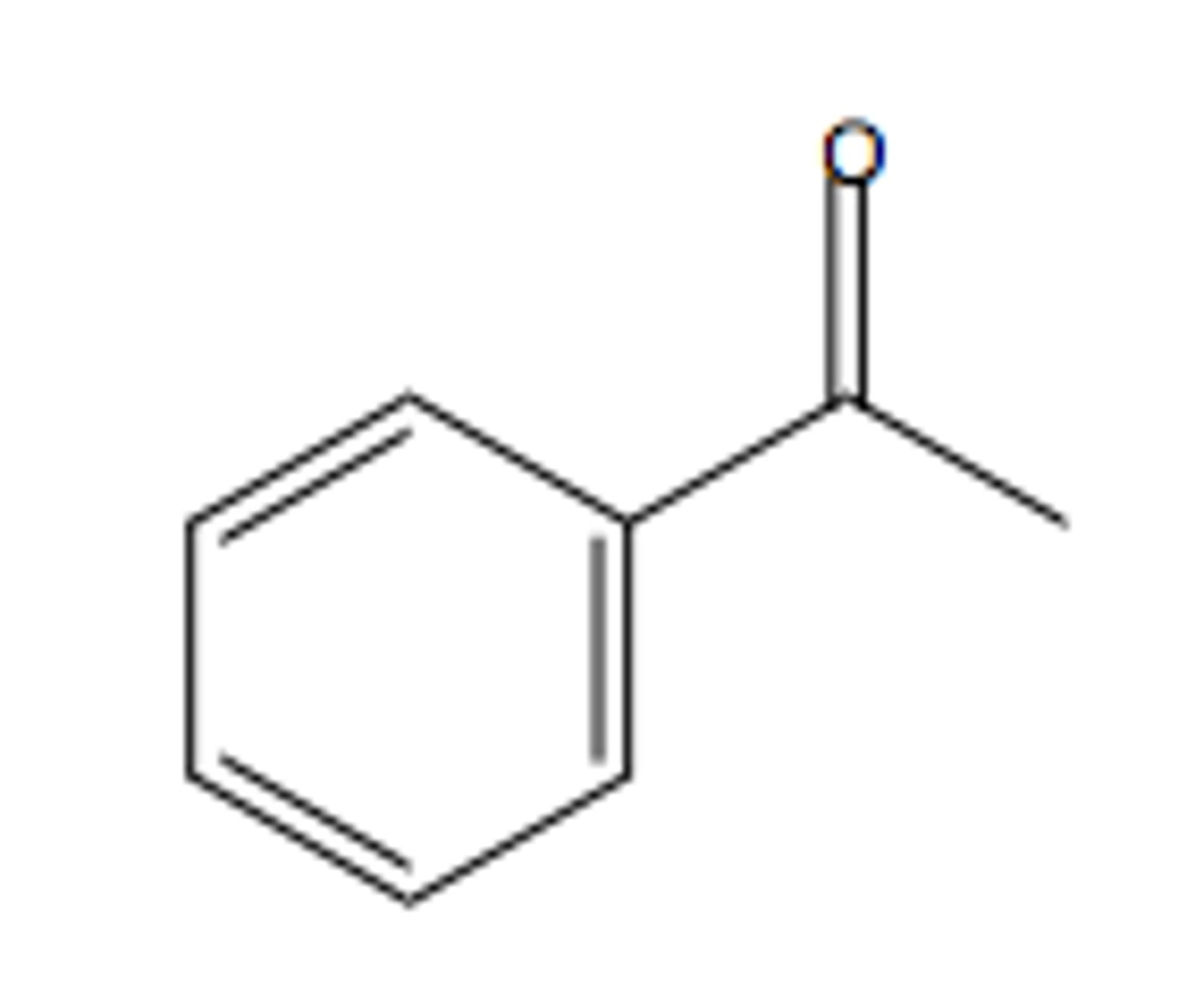
What reagents?
Benzene → aniline
1) HNO3, H2SO4
2) Fe, HCl
3) NaOH
What reagents?
Benzene → Benzene with CBr3 group
1) CH3Cl, AlCl3
2) excess NBS
What reagents?
Benzene → benzoic acid
1) CH3Cl, AlCl3
2) Na2Cr2O7, H2SO4, H2O
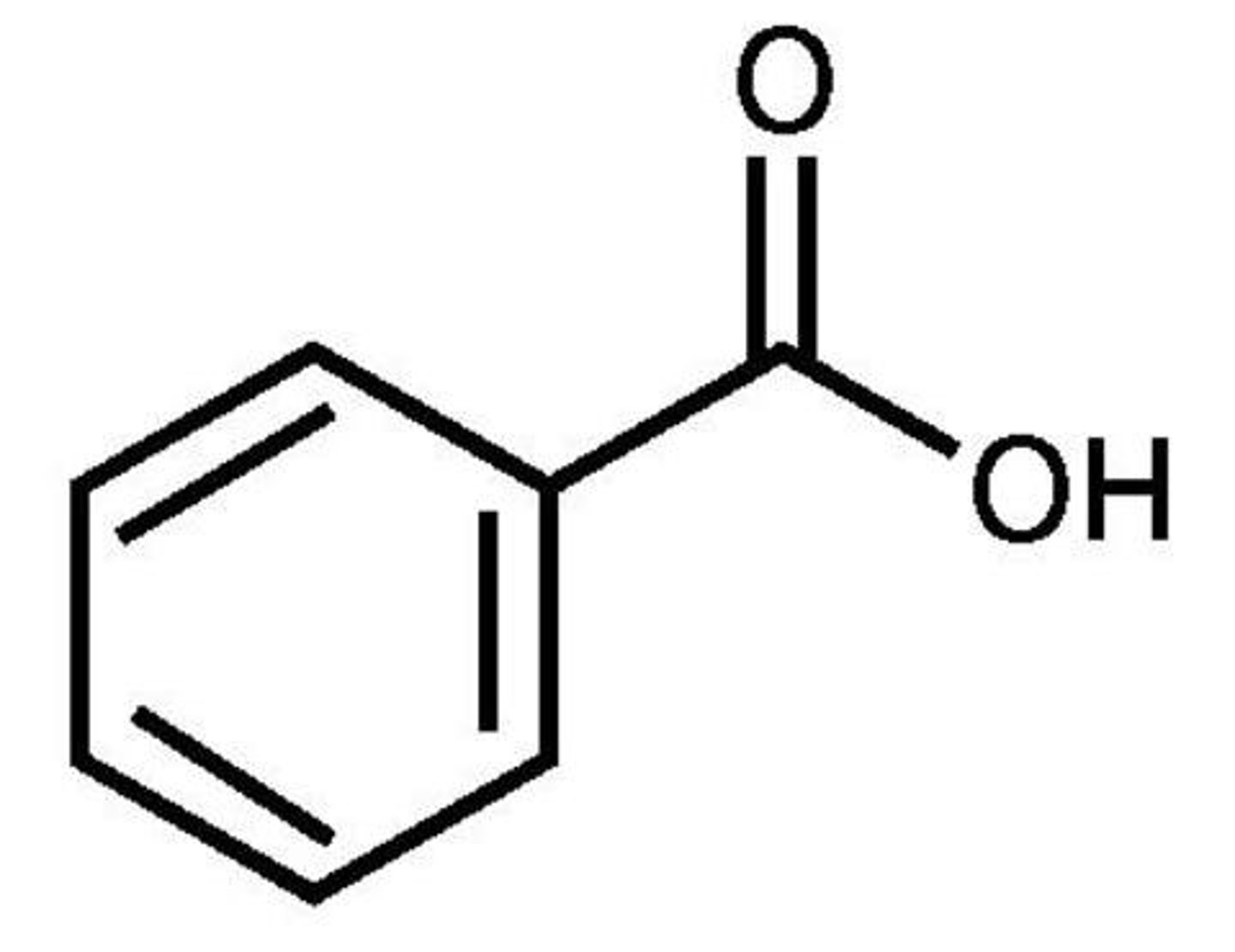
What reagents?
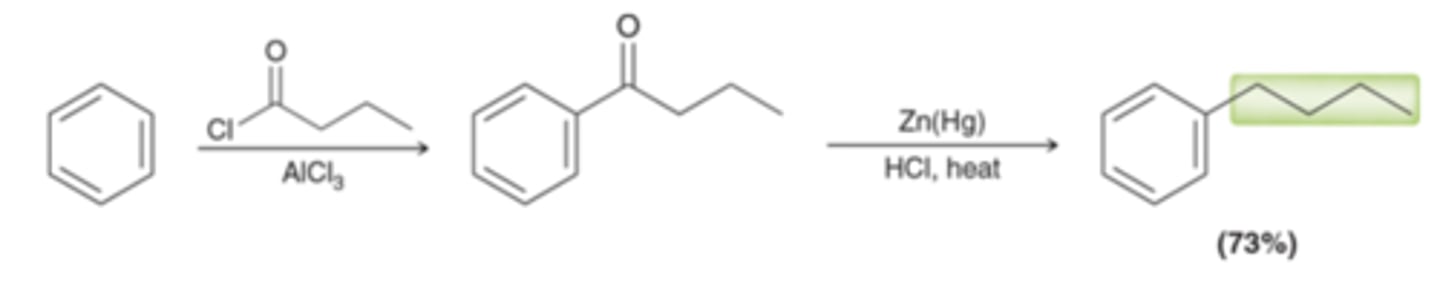
Benzene → propyl benzene
Friedel-Crafts acylation followed by Clemmensen reduction
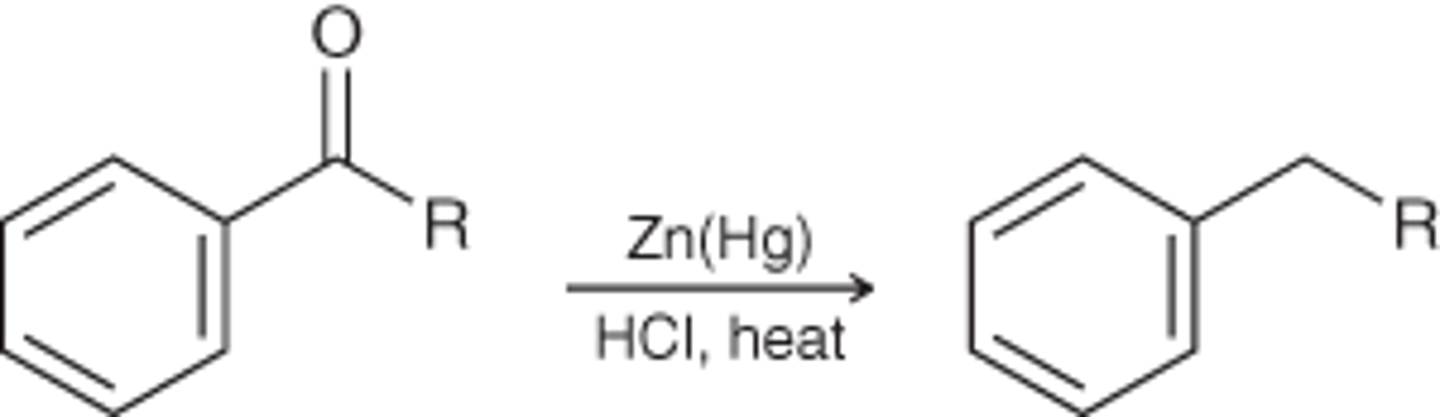
Nitration cannot be performed on a ring that...
has an amino group
A Friedel-Crafts alkylation or acylation cannot be performed on rings that...
are moderately or strongly deactivated
The ring must be _______________________ for Friedel-Crafts to occur.
activated or weakly deactivated
Nucleophilic aromatic substitution
A substitution reaction in which an aromatic ring is attacked by a nucleophile, which replaces a leaving group.
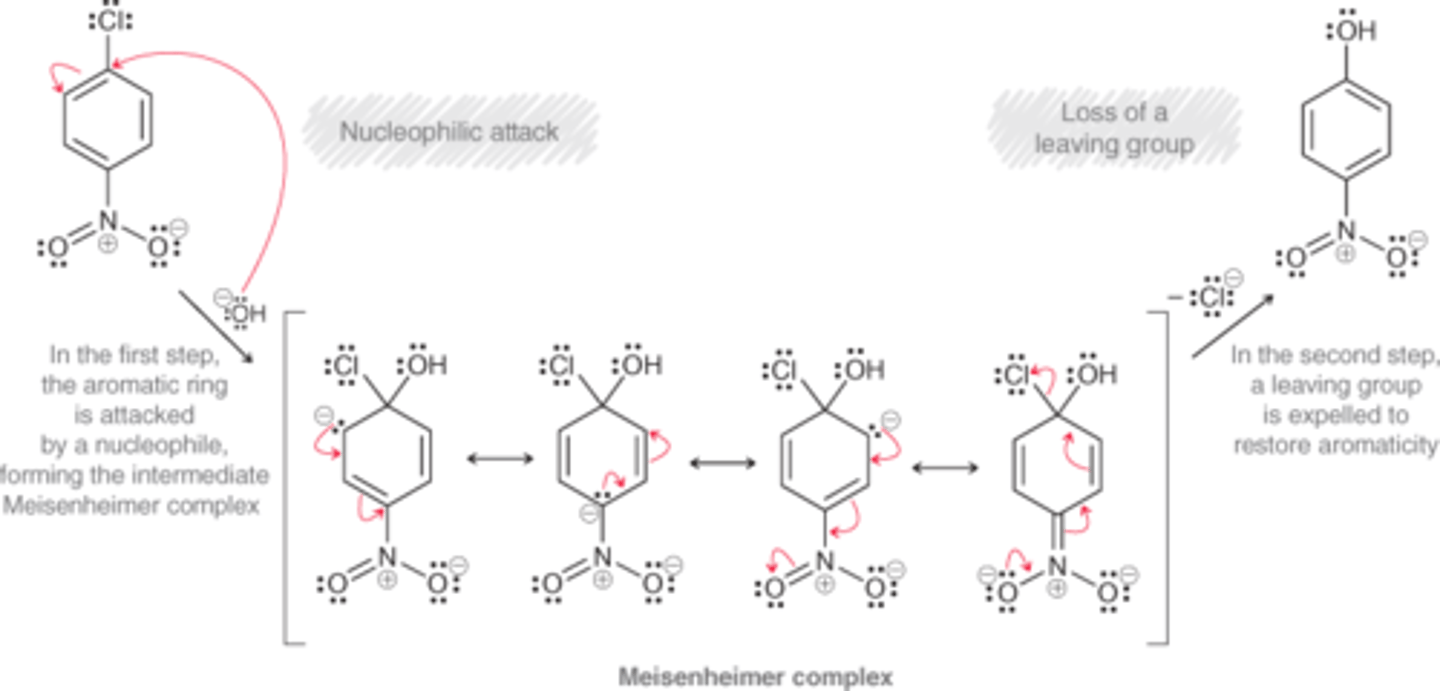
What are the 3 criteria for nucleophilic aromatic substition?
1) The ring must contain a powerful electron-withdrawing group (typically a nitro group)
2) The ring must contain a leaving group (usually a halide)
3) The leaving group must be either ortho or para to the electron-withdrawing group. If the leaving group is meta to the nitro group, the reaction is not observed.
Mechanism for Nucleophilic Aromatic Substitution (SnAr)
Nucleophilic Attack
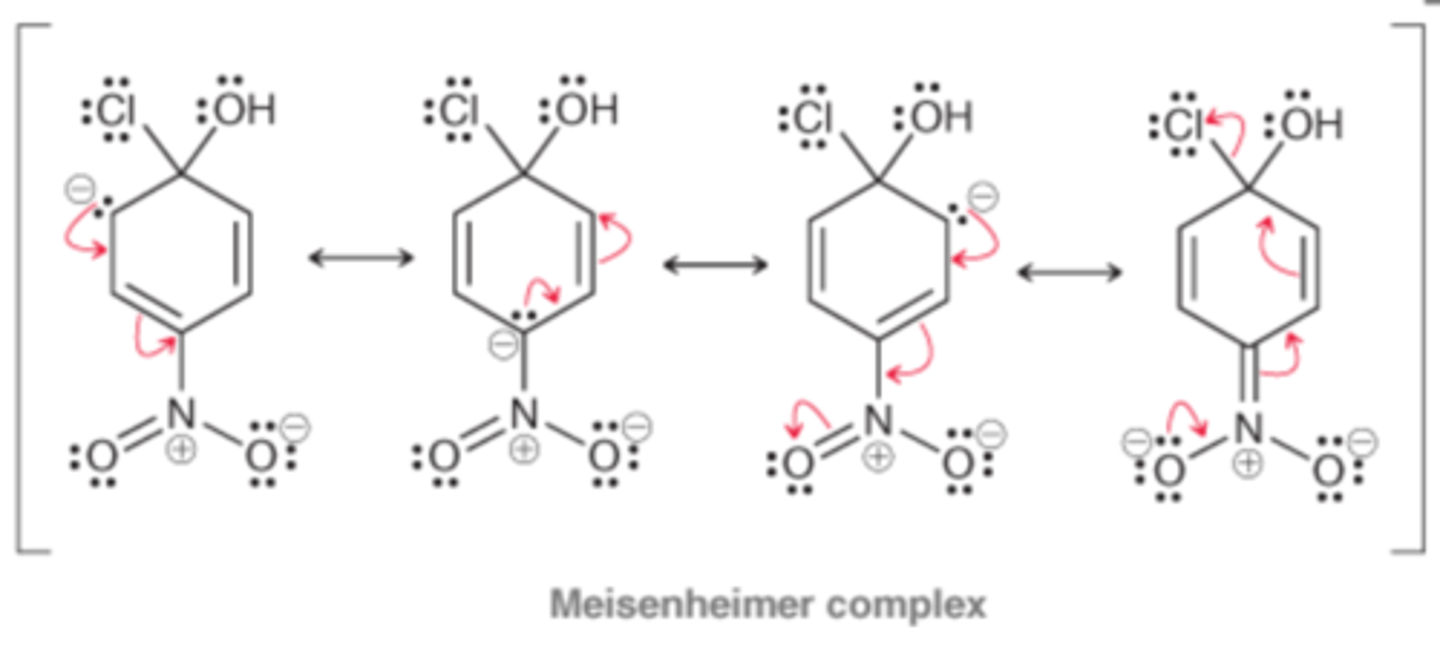
Meisenheimer complex
Loss of leaving group
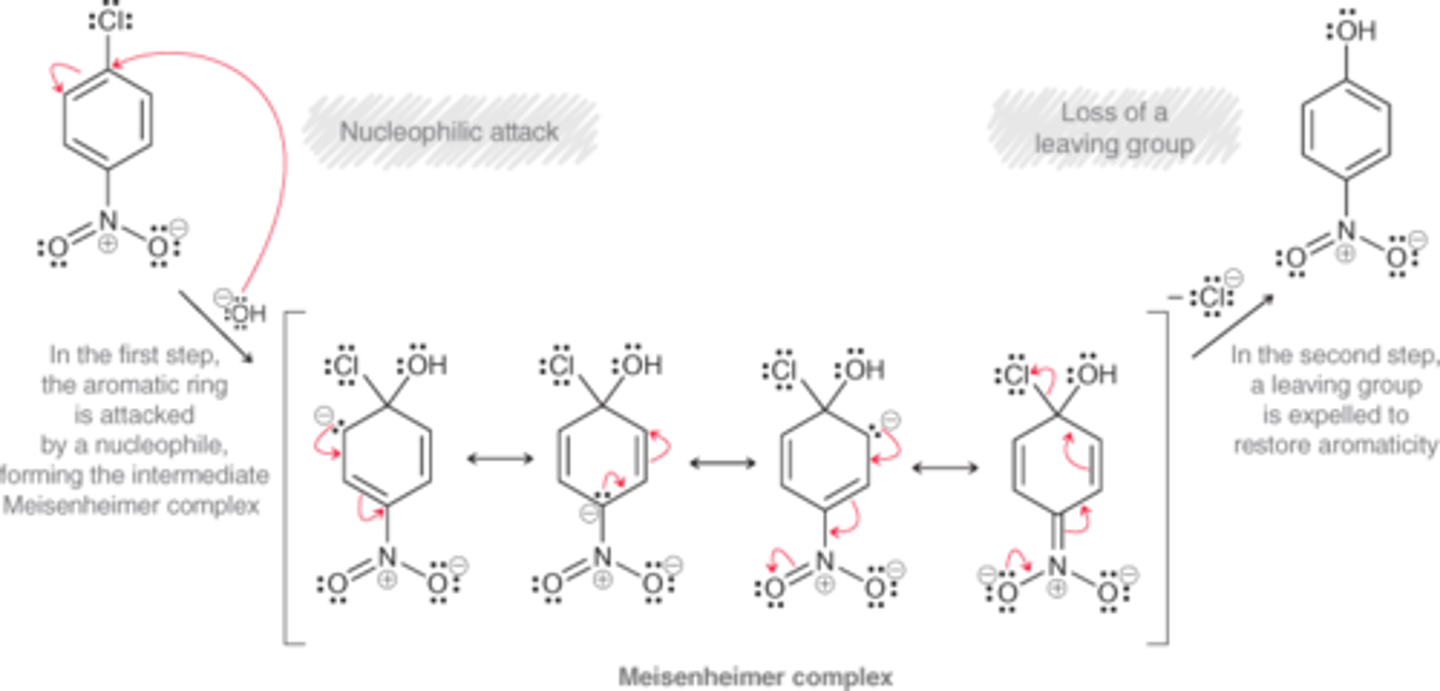
The resonance stabilized intermediate of a nucleophilic aromatic substitution reaction
meisenheimer complex
Meisenheimer Complex vs Sigma Complex
Meisenheimer Complex has a negative charge that is resonance stabilized
Sigma Complex has a positive charge that is resonance stabilized
Why is a nitro group needed for nucleophilic aromatic substitution?
The nitro group is a temporary reservoir for electron density. The nucleophile attacks the ring, dumping its electron density into the ring, where it is temporarily stored on the nitro group. Then, the nitro group releases the electron density to expel a leaving group
In nucleophilic aromatic substitution, when hydroxide is used as the attacking nucleophile the product is...
a phenolate ion
Need an acid to protonate the product
1) Excess NaOH, heat
2) H3O+
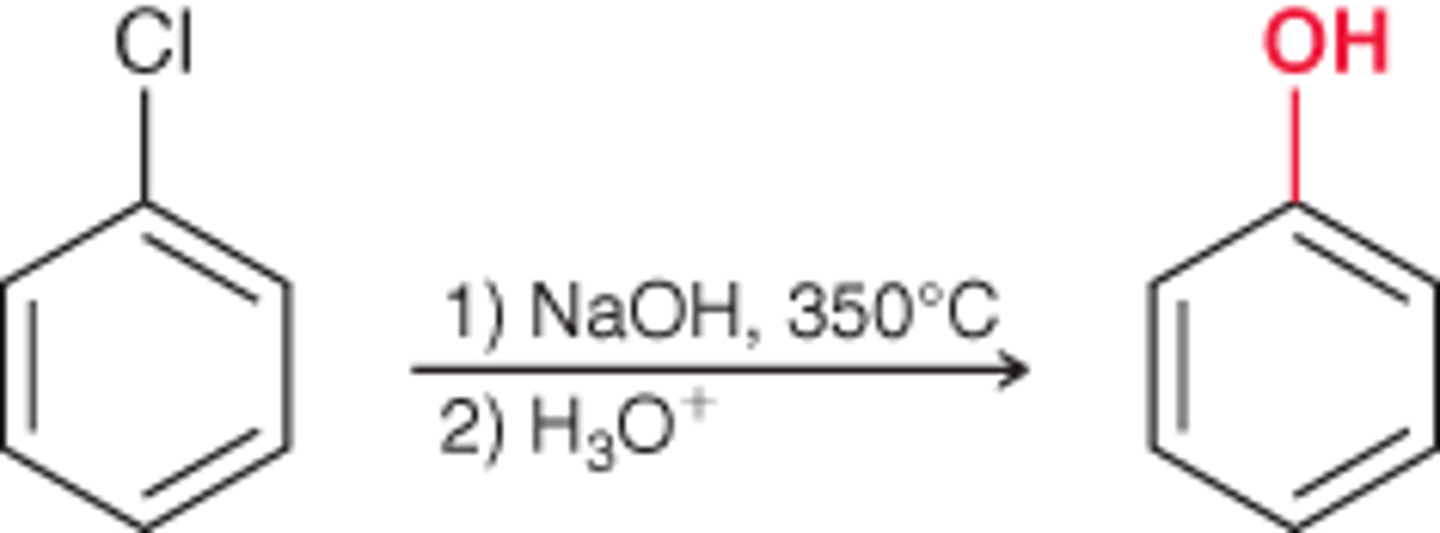
A reaction that occurs between chlorobenzene and either NaOH (at high temperature) or NaNH2.
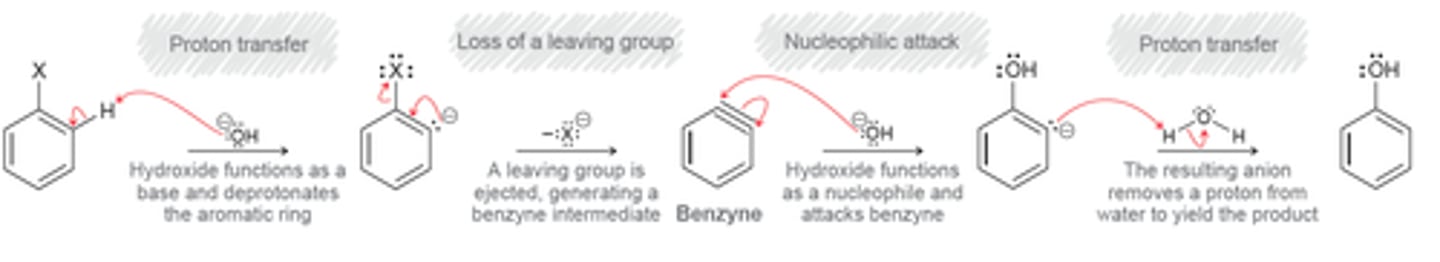
Elimination addition
Elimination addition mechanism
proton transfer
loss of a leaving group
nucleophilic attack
proton transfer
Reagents for elimination addition to add a OH
1) NaOH, heat
2) H3O+
Reagents for elimination addition to add an NH2
1) NaNH2, NH3(l)
2) H3O+
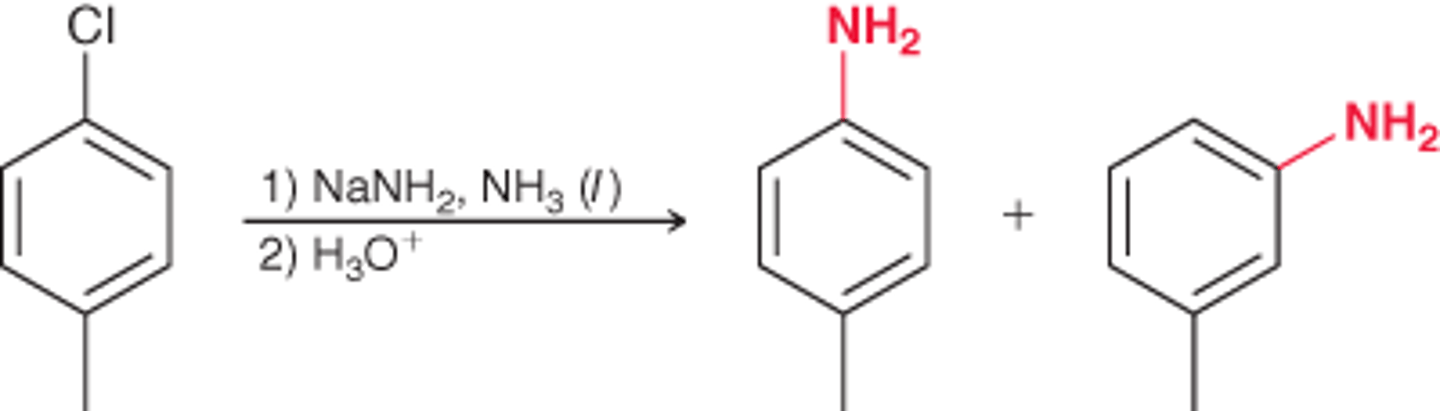
Benzyne
A high-energy intermediate formed during the elimination-addition reaction that occurs between chlorobenzene and either NaOH (at high temperature) or NaNH2.
In elimination-addition, benzyne can be attacked at...
either end of the triple bond
Benzyne + Furan
Diels-Alder cycloaddition
The triple bond in benzyne results from the...
overlap of sp2 orbitals
Electrophilic aromatic substitution proceeds via a...
Nucleophilic aromatic substitution proceeds via a...
Elimination-addition proceeds via a...
Electrophilic aromatic substitution proceeds via a sigma complex
Nucleophilic aromatic substitution proceeds via a Meisenheimer complex
Elimination-addition proceeds via a benzyne intermediate
In electrophilic aromatic substitution, the incoming substituent....
In nucleophilic aromatic substitution and elimination-addition, a negatively charged leaving group....
replaces a proton
is expelled
In electrophilic aromatic substitution, _____________________________ groups deactivate the ring toward attack
In nucleophilic aromatic substitution, an ______________________________ group is required in order for the reaction to proceed
In electrophilic aromatic substitution, electron-withdrawing groups deactivate the ring toward attack
In nucleophilic aromatic substitution, an electron-withdrawing group is required in order for the reaction to proceed
If the reagents are nucleophilic, the reaction is a...
nucleophilic aromatic substitution if it meets the 3 criteria
OR
elimination-addition
If the reagents are electrophilic, the reaction is a...
electrophilic aromatic substitution