Introduction part 2: combustion, substitution and addition
1/7
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
8 Terms
All organic compounds at IGCSE undergo combustion reactions
What is combustion?'
Chemical reaction in which a substance reacts with oxygen (burns) to form products and heat
What products are formed during the combustion of hydrocarbons in:
Excess oxygen
Limited supply of oxygen
Excess: complete combustion produces CO2 and H2O + large amount of heat energy
Limited: incomplete combustion produces CO or carbon (soot) and H2O + large amount of heat energy
Explain why incomplete combustion of hydrocarbons might be dangerous for humans
Carbon monoxide is a colourless, odourless and very poisonous gas
It is poisonous because it reduces the ability of the blood to carry oxygen around the body
This will make you ill or you may even die, because not enough oxygen gets to the cells in your body for respiration to provide energy
What is propane frequently used for when burned?
Used in camping stoves outdoors
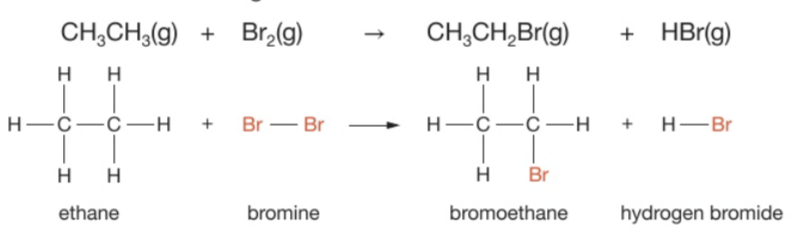
What is a substitution reaction?
A chemical reaction in which an atom or group of atoms in a molecule is replaced by a different atom or group of atoms
Give an example of a substitution reaction + give the equation (displayed or structural)
Methane reacts with bromine under ultraviolet light
What is an addition reaction?
A chemical reaction in which one molecule adds to another without taking anything away, to form a single larger molecule (no other products are formed)
Only alkenes undergo addition reactions
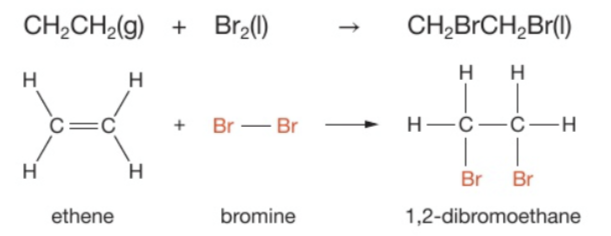
Give an example of an addition reaction + the equation (displayed or structural)
For example, ethene reacts with bromine to produce 1,2-dibromoethane