A level Chem 1.10 The Equilibrium Constant
1/12
Earn XP
Description and Tags
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
13 Terms
Methanol can be manufactured in a reversible reaction as shown. CO(g) + 2 H2(g) ⇌ CH3OH(g) ΔHө = –91 kJ mol–1
Give an expression for the equilibrium constant (Kp) for this reaction (1)
(1)
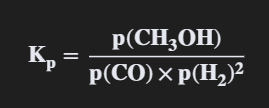
Methanol can be manufactured in a reversible reaction as shown. CO(g) + 2 H2(g) ⇌ CH3OH(g) ΔHө = –91 kJ mol–1
A 0.230 mol sample of carbon monoxide is mixed with hydrogen in a 1:2 mol ratio and allowed to reach equilibrium in a sealed flask at temperature T. At equilibrium the mixture contains 0.120 mol of carbon monoxide. The total pressure of this mixture is 1.04 × 104 kPa
Some more carbon monoxide is added to the mixture in part (b). The new mixture is allowed to reach equilibrium at temperature T.
State the effect, if any, on the partial pressure of methanol and on the value of Kp (2)
Partial pressure of methanol increases (1)
Kp stays the same/no effect (1)
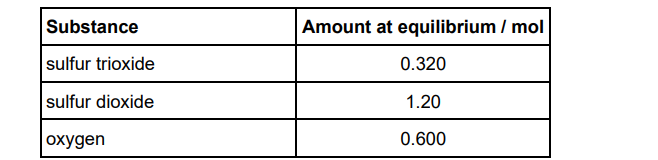
A different mass of sulfur trioxide was heated and allowed to reach
equilibrium at 1050 K
2SO3(g) ⇌ 2 SO2(g) + O2(g)
The amounts of each substance in the equilibrium mixture are shown in the table.
For this reaction at 1050 K the equilibrium constant, Kp = 7.62 x 105 Pa
Calculate the mole fraction of each substance at equilibrium.
Give the expression for the equilibrium constant, Kp
Calculate the total pressure, in Pa, of this equilibrium mixture. (4)
Mole fraction SO3=0.15
Mole fraction SO2=0.57
Mole fraction O2 =0.28 (1)
Kp = (pSO2)x(pO2)/(pSO3)2 (1)
Kp x (0.15)2 /(0.57)2x(0.28)
P=1.91×105 (Pa) (1)
For this reaction at 1050 K the equilibrium constant, Kp = 7.62 x 105 Pa
For this reaction at 500 K the equilibrium constant, Kp = 3.94 x 104 Pa
Explain how this information can be used to deduce that the forward reaction is endothermic. (2)
Kp is higher at higher temperature or converse (1)
At higher temperature more dissociation occurs / more products are formed / equilibrium shifts to the right/forward direction (1)
Nitrogen reacts with hydrogen in this exothermic reaction
N2(g) + 3 H2(g) ⇌ 2 NH3(g)
Which change increases the equilibrium yield of ammonia but has no effect on
the value of the equilibrium constant Kp?
A Add a catalyst
B Increase the partial pressure of
nitrogen
C Decrease the temperature
D Decrease the total pressure (1)
B (1)
This shifts the equilibrium to the right (towards ammonia) because the system tries to use up the extra N₂.
Nitrogen and hydrogen were mixed in a 1:3 mole ratio and left to reach equilibrium in a flask at a temperature of 550 K.
The equation for the reaction between nitrogen and hydrogen is shown.
N2(g) + 3H2(g) ⇌ 2NH3(g)
When equilibrium was reached, the total pressure in the flask was 150 kPa and the mole fraction of NH3(g) in the mixture was 0.80 Calculate the partial pressure of each gas in this equilibrium mixture.(3)
p(NH3) = 0.80×150=120kPa (1)
Remaining pressure for N2 and H2 : 150-120=30kPa
Initial mole ratio N₂ : H₂ = 1 : 3
p(N₂)=1/4×30=7.5 kPa (1)
p(H₂)=3/4×30=22.5kPa (1)
An equation for this formation of methanol is given below.
CO2(g) + 3H2(g) ⇌ CH3OH(g) + H2O(g)
Suggest one environmental advantage of manufacturing methanol fuel by this reaction (1)
The carbon dioxide (produced on burning methanol) is used up in this reaction (1)
An equation for this formation of methanol is given below.
CO2(g) + 3H2(g) ⇌ CH3OH(g) + H2O(g)
Suggest why the catalyst used in this process may become less efficient if the carbon dioxide and hydrogen contain impurities (1)
Impurities (or sulfur compounds) block the active sites (1)
An equation for this formation of methanol is given below.
CO2(g) + 3H2(g) ⇌ CH3OH(g) + H2O(g)
Use Le Chatelier's principle to justify why the reaction is carried out at a high pressure rather than at atmospheric pressure. (3)
4 mol of gas form 2 mol (1)
At high pressure the position of equilibrium moves to the right to lower the pressure / oppose the high pressure (1)
This increases the yield of methanol (1)
The equation for the complete combustion of butane is C4H10 + 6.5 O2 → 4 CO2 + 5 H2O
What is the mole fraction of butane in a mixture of butane and oxygen with the minimum amount of oxygen needed for complete combustion?
A 0.133
B 0.153
C 0.167
D 0.200 (1)
A
“the mole fraction of butane in a mixture of butane and oxygen”
This question is about some gas mixtures at equilibrium.
This reaction can be used to make hydrogen.
H2O(g)+CO(g) ⇌ H2(g)+CO2 (g) ΔH = –41 kJ mol–1
State why the equilibrium constant (Kp) for this reaction has no units.(1)
(The amount in) moles of products is the same as reactants (so the units (of partial pressure) cancel out) (1)
Give two features of a reaction in dynamic equilibrium. (2)
forward and reverse reactions proceeding at equal rates (1)
concentrations (of reactants and products) remain constant (1)
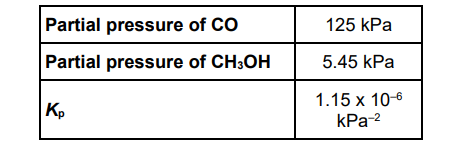
A different mixture of carbon monoxide and hydrogen is left to reach equilibrium at a temperature T. Some data for this equilibrium are shown in the table below.
CO(g) + 2H2(g) ⇌ CH3OH(g)
Use the Kp value from the table above to calculate a value for Kp for the following reaction at temperature T.
CH3OH(g) ⇌ CO(g) + 2H2(g) (2)
(1/1.15×10-6) =8.7(0)x10-5 (1)
kPa2 (1)