bio-orthogonal reactions
1/18
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
19 Terms
what is click chemistry and what are the 3 main principles
the easy connection of two complex molecules
ideally near 100% yield
ideally no by-products
ideally no additional reagents or catalysts
what is a bio-orthogonal reaction (definition + 3 principles)
one that meets “click” criteria but additionally can be defined as a reaction between functional groups
that are not themselves present commonly in nature
that don’t react with any functional groups that are commonly found in biological molecules (e.g. amines, alcohols)
that will react together under conditions compatible with living organisms
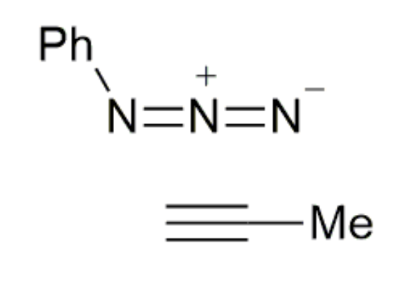
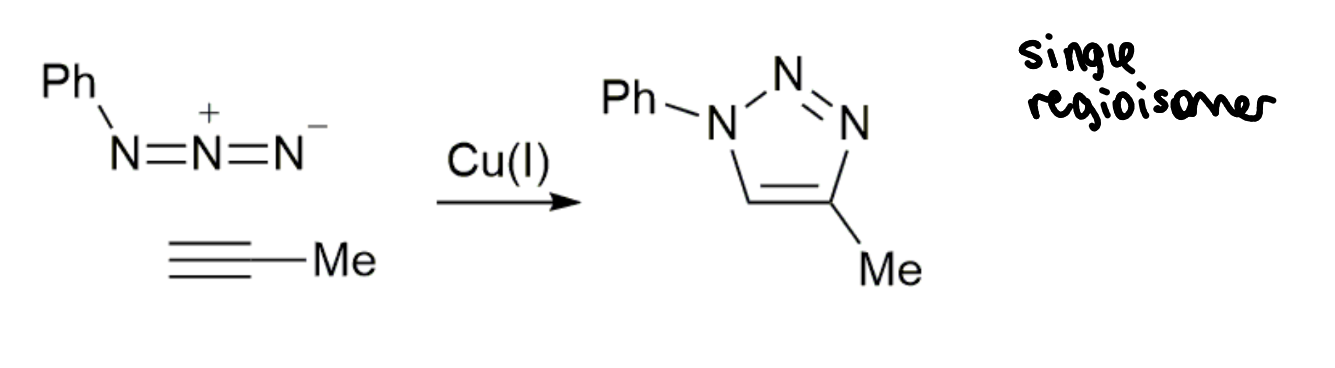
CuAAC reaction
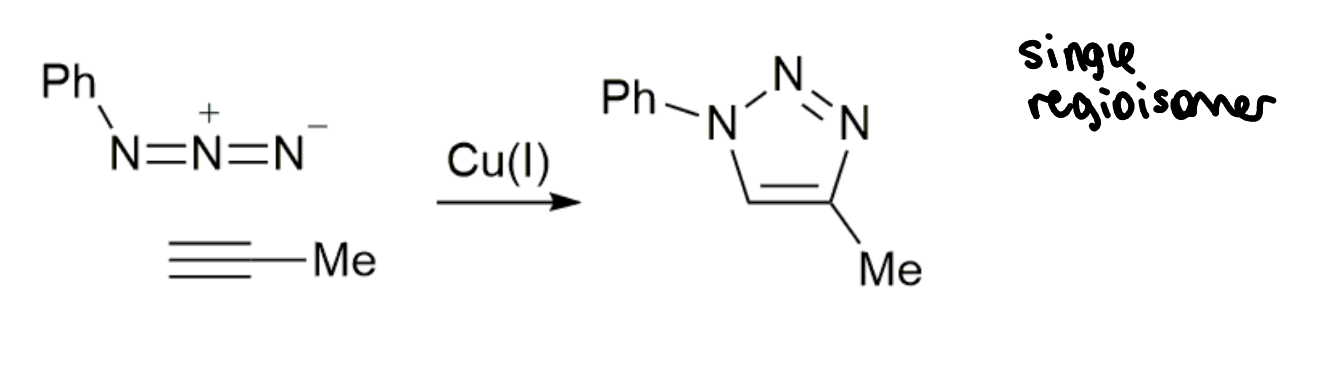
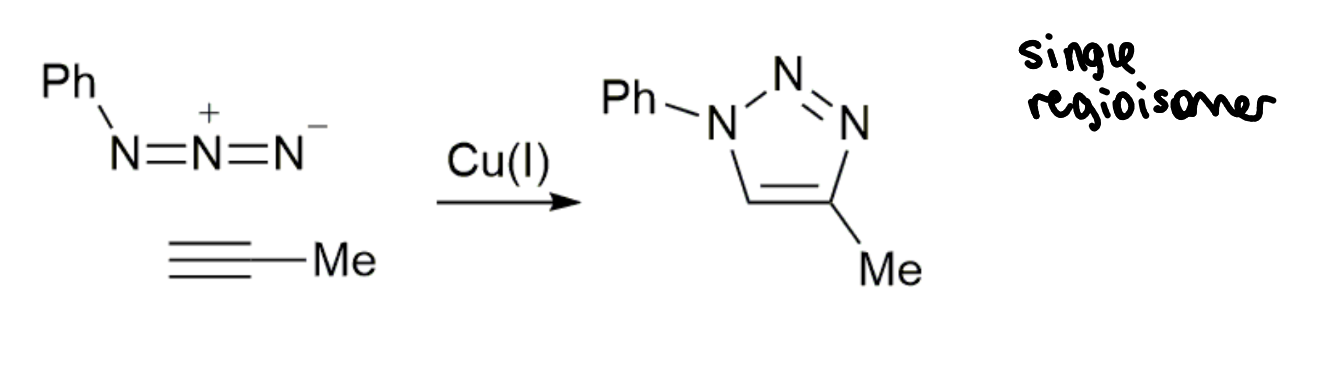
how well does this reaction meet the click criteria?
high yield
no by-products
no additional reagents but does require a catalyst
nearly click but not quite
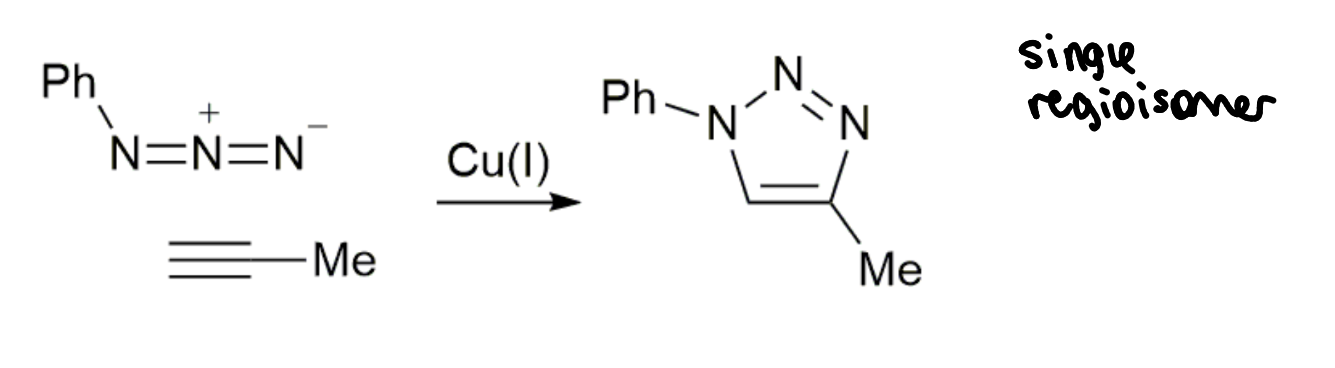
how well does this reaction meet the bioorthogonal criteria?
can it be done in living systems?
azides hardly found in nature
alkynes hardly found in nature
don’t react readily with alcohols, amines etc
CuAAC is compatible with many living systems - can do in cells as long as the presence of Cu is OK
show how this could be used for imaging?
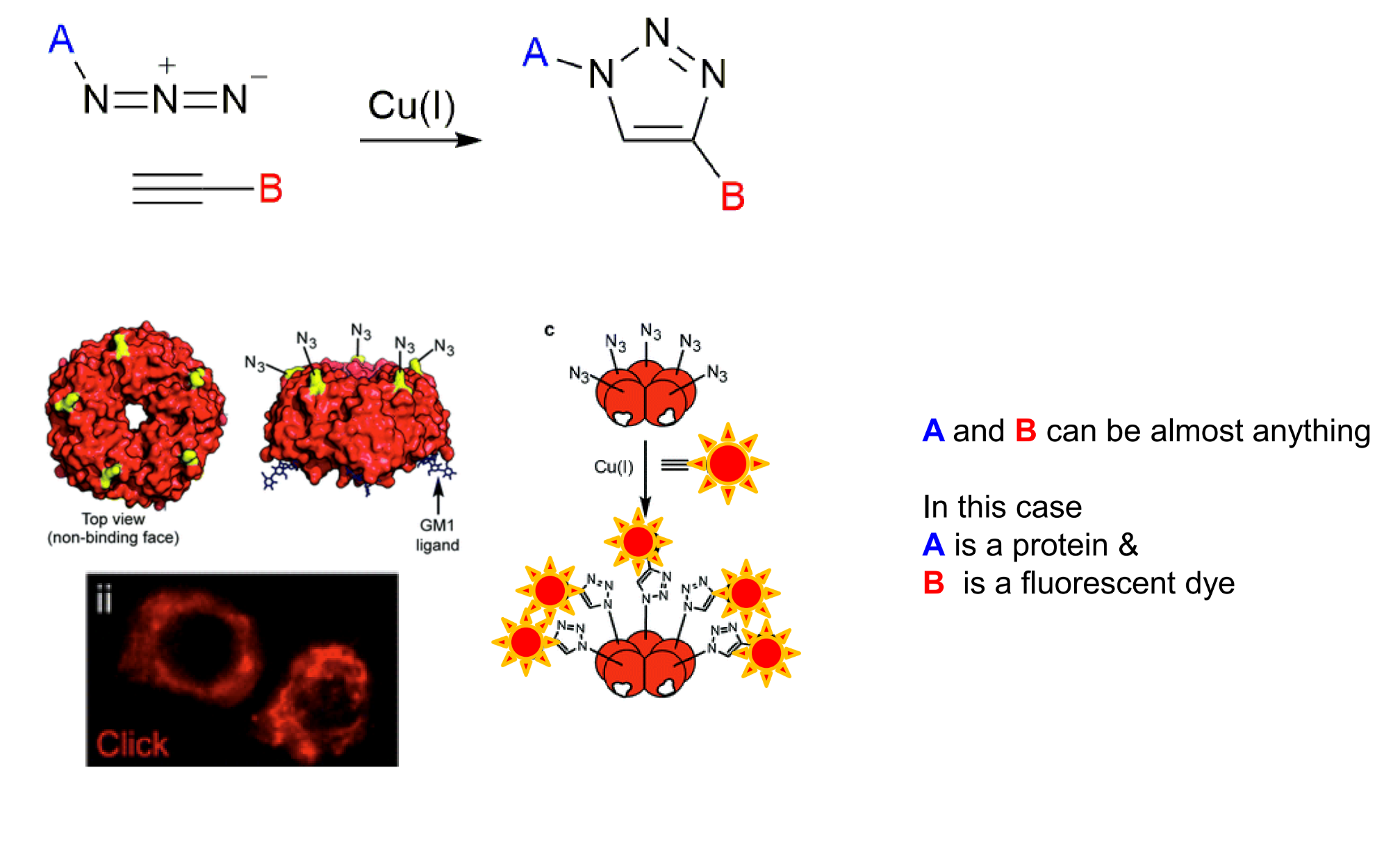
how does SPAAC compare to CuAAC in terms of bioorthogonality?
allows spontaneous reaction with azides with no need for copper catalyst - ticks all the criteria for bio-orthogonal reaction
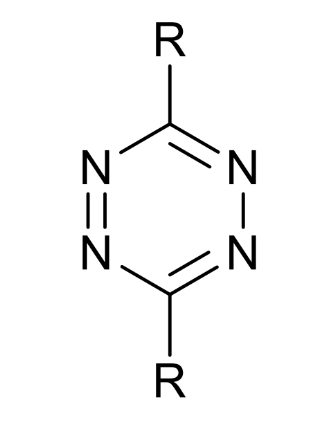
what type of molecule is this
tetrazine
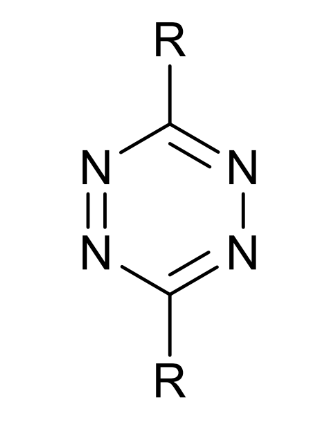
how can this react and why
highly electron-poor diene
LUMO is reactive
can undergo inverse electron demand cycloadditions
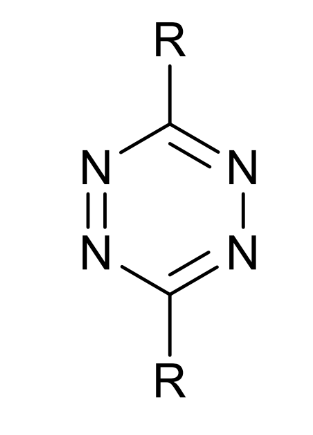
tetrazines in nature?
scarcely found, do not react with alcohols, amines etc
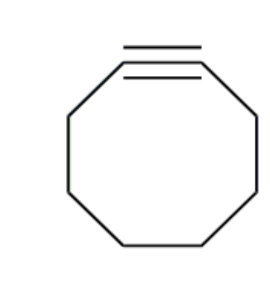
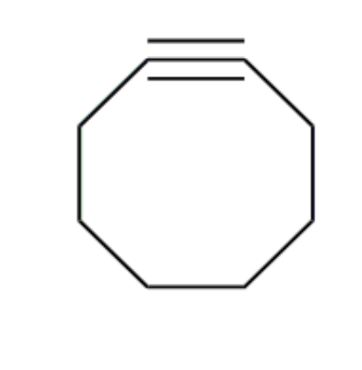
energy + orbitals of cyclooctyne
high energy HOMO
electron rich, strained
show cycloaddition of tetrazines with cyclooctyne
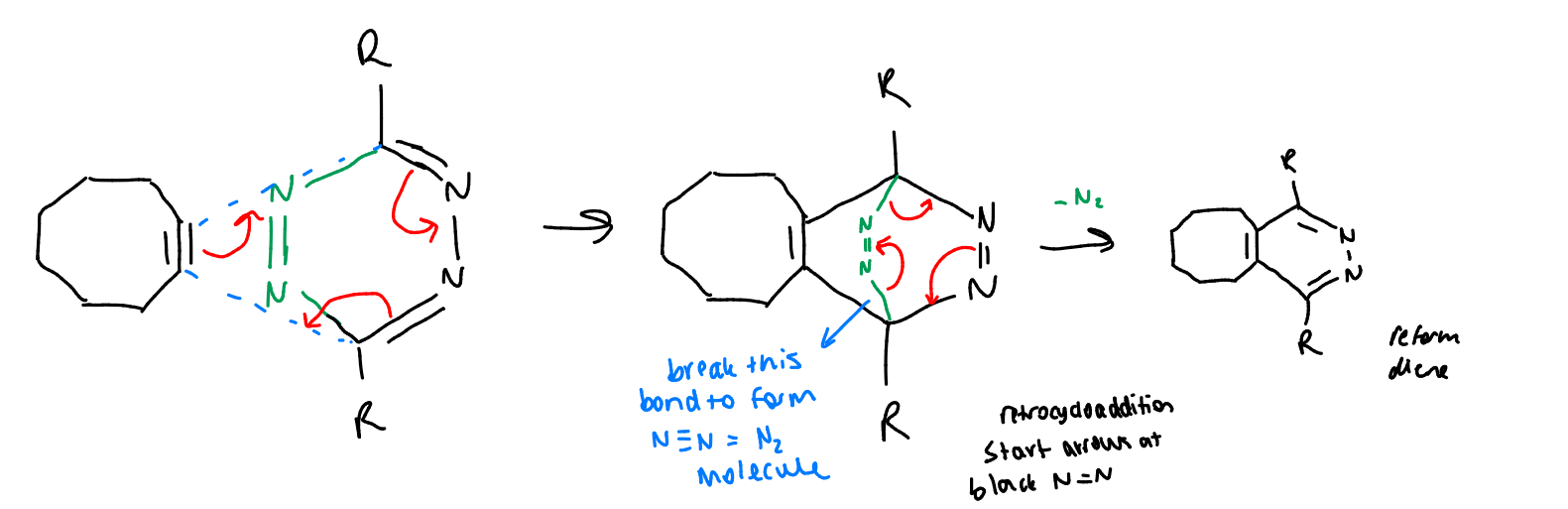
bioorthogonality?
there is an N2 byproduct but this is likely to be harmless to living systems
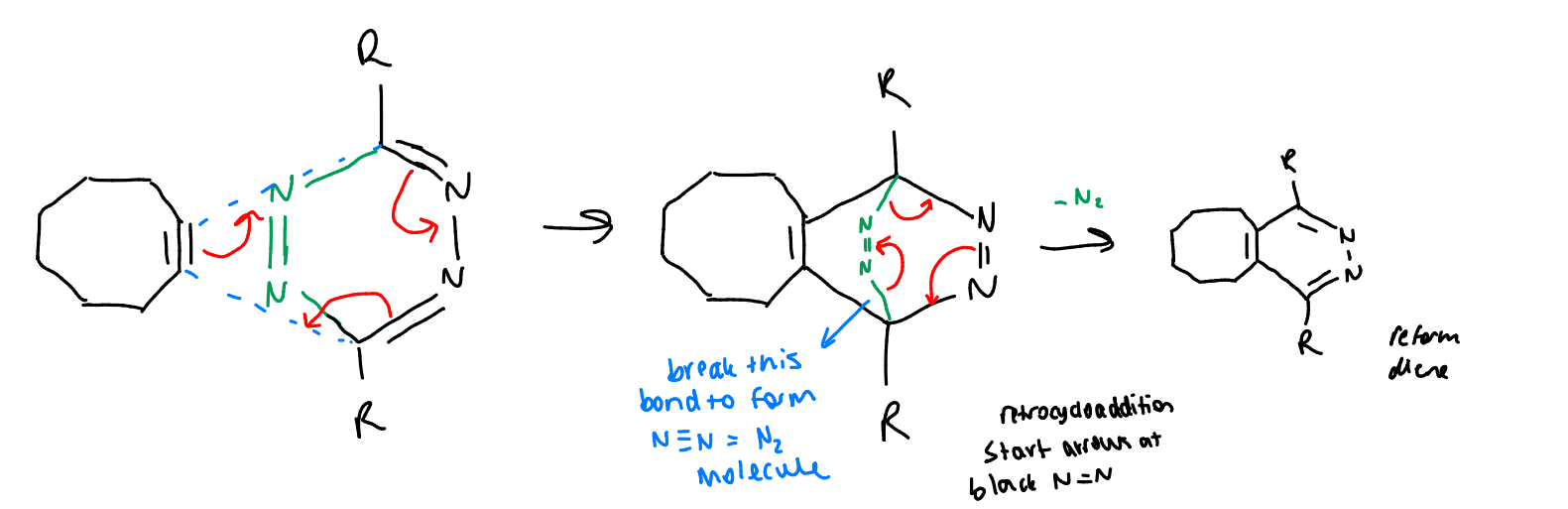
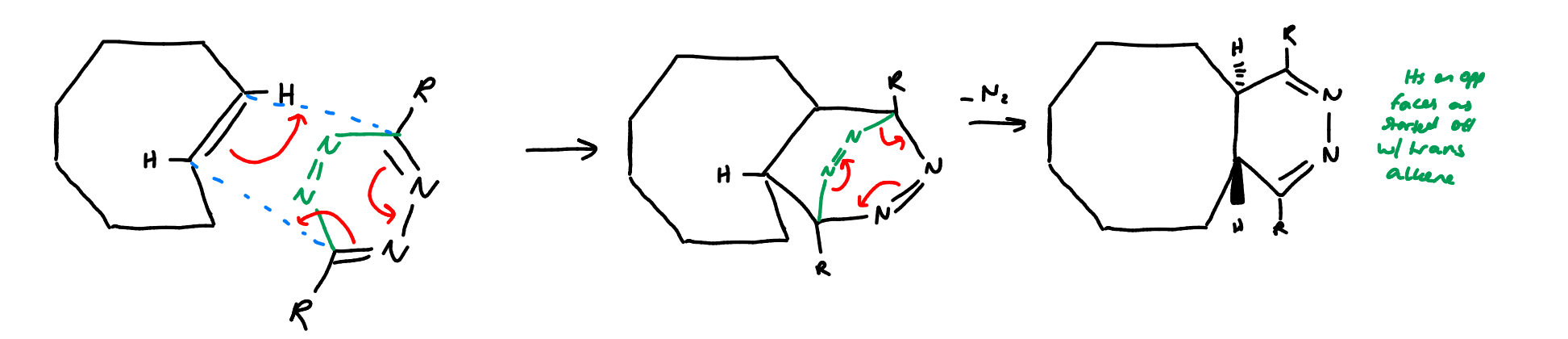
show the reaction of tetrazines with trans-cyclooctene
comment on bioorthogonality?
dones’t fully meet the criteria due to N2 byproduct, but likely to be OK
alkenes are quite common in nature but it seems that we can get away with cyclooctenes
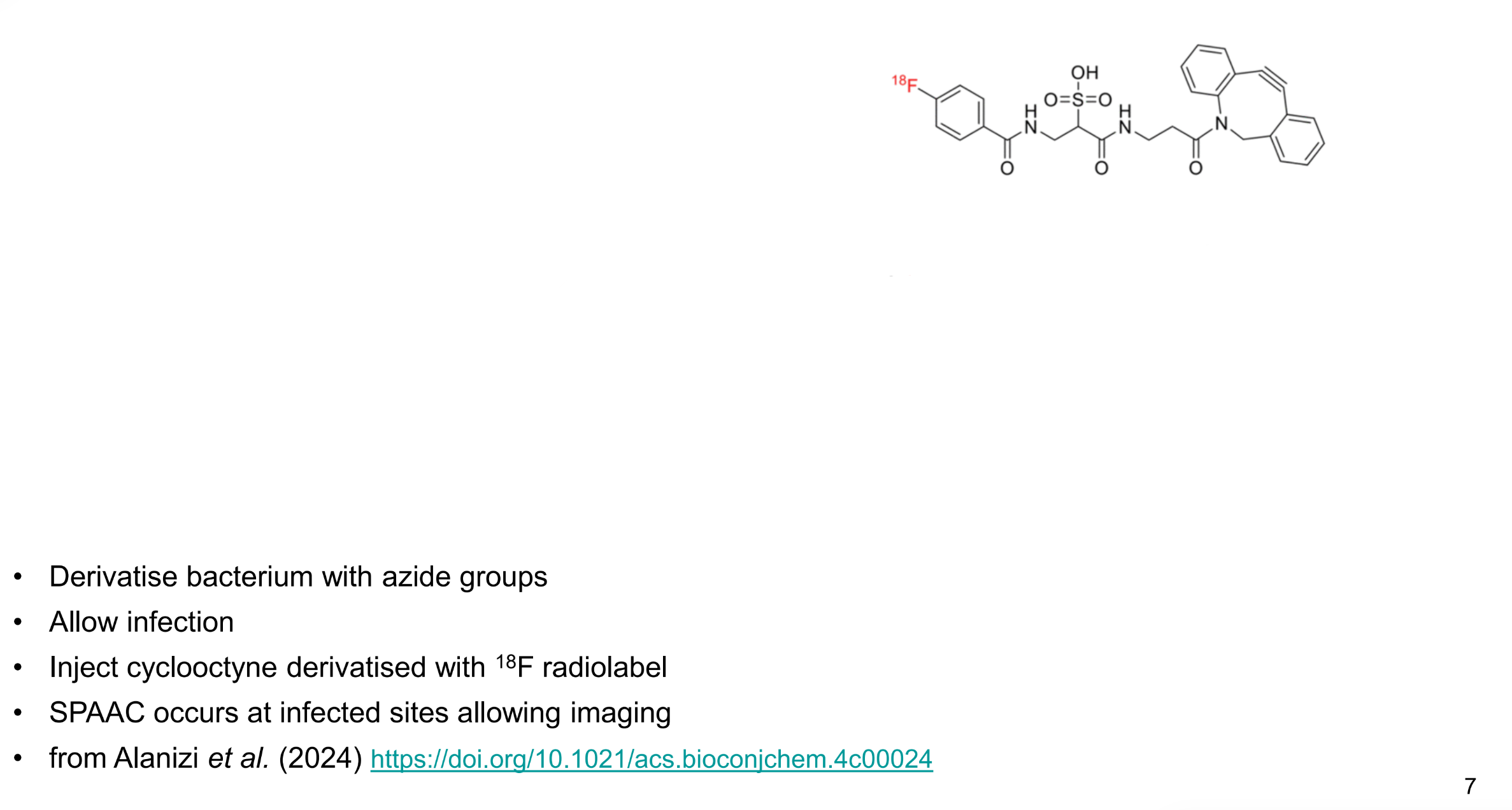
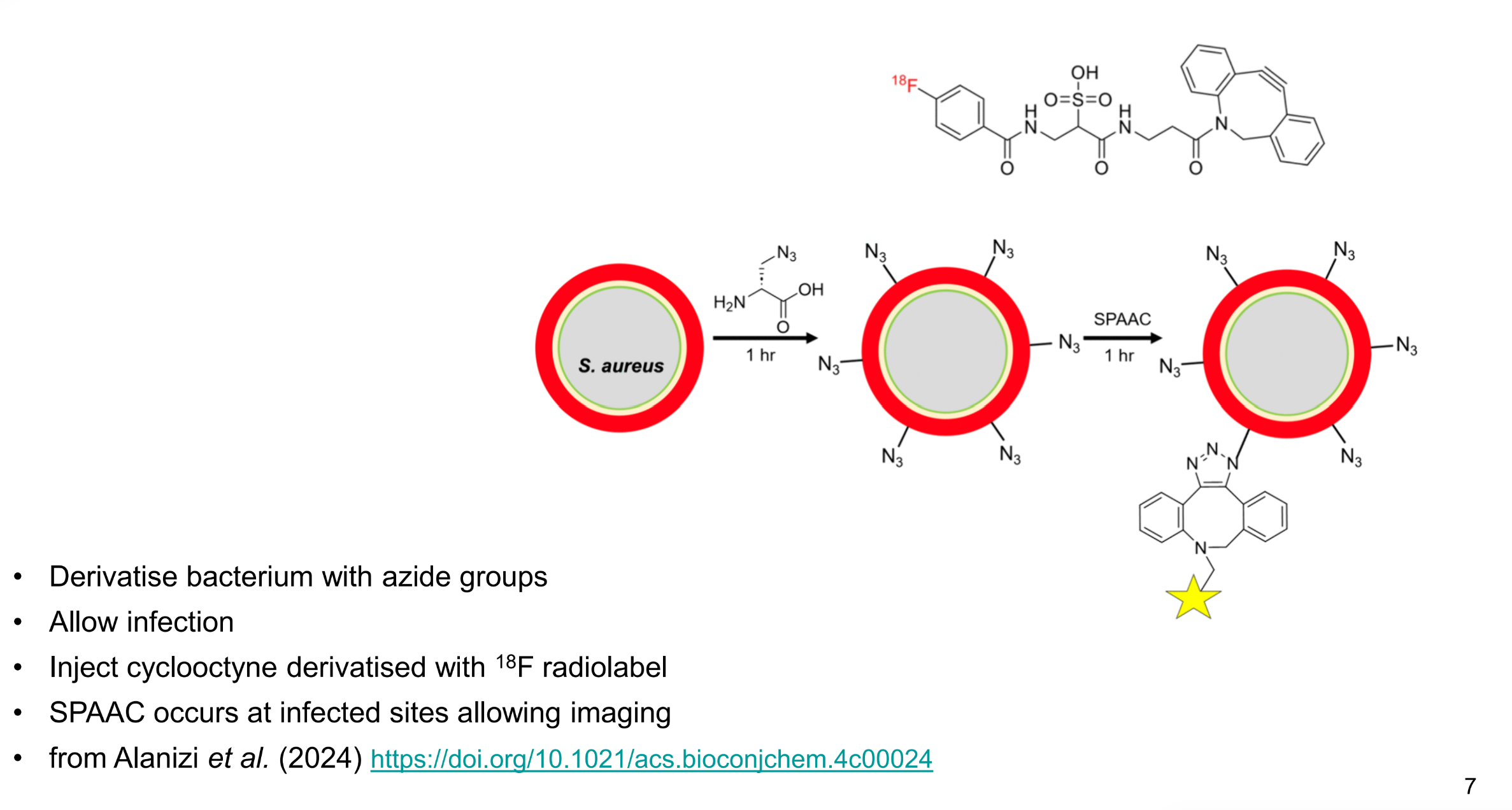
how are bioorthogonal reactions used to tell where bacteria is in the body
derivatise bacterium with azide groups
allow infection
inject cyclooctyne with 18F radiolabel
SPAAC occurs at infected sites allowing imaging
draw a schematic to represent this
what are liposomes
synthetic vesicles that can be used to transport drugs through the body
give an example of liposomes being used with the chemistry seen here
liposomes derivatised with a cyclooctyne
drug incorporated into the liposome
once in the body the liposomes can be reacted with injected antibodies that have azides attached
with appropriate choice of antibody the drug can be targeted to a particular cell and be carried there in the liposome
how can a bioorthogonal reaction be used to treat a tumour
antibody carries attached drug to tumour
after the cycloaddition reaction, tautomerisation and further reactions release CO2 and the drug
the drug can enter the nearby tumour cell
the bioorthogonal reaction is the one used to release the drug from the antibody-drug conjugate