1. Atomic Structure and the Periodic Table
1/51
Earn XP
Description and Tags
Module 1; Objective 1.1 - 1.13
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
52 Terms
1.1 What are the postulates of Dalton’s atomic theory?
Matter is made up of small, indivisible atoms.
Atoms of the same element are identical in mass and properties.
Atoms of different elements have different masses and properties.
Atoms cannot be created, destroyed, or subdivided in chemical reactions.
Compounds are formed when atoms combine in simple whole number ratios.
Chemical reactions involve the rearrangement of atoms.
Why is Dalton’s atomic theory no longer considered completely accurate?
Atoms are divisible (contain electrons, protons, neutrons).
Atoms of the same element are not always identical → isotopes exist.
Atoms can be changed in nuclear reactions.
Atoms have internal structure, not solid spheres.
What is meant by theoretical change in science?
The process by which scientific theories are modified or replaced based on new experimental evidence
What criteria are used to determine whether a scientific theory is accepted?
Agreement with evidence (fits experimental results)
Reliability (consistent results)
Accuracy (close to true value)
Replicability (can be repeated with same results)
Consensus (accepted by scientific community)
Societal factors (technology, ethics, needs)
How does Dalton’s atomic theory illustrate theoretical change?
Dalton’s theory was accepted based on available evidence but was later modified when new discoveries (subatomic particles and isotopes) showed that atoms are divisible and not identical, demonstrating how scientific theories evolve.
1.2 What is the structure of the atom?
An atom consists of a small, dense nucleus containing protons and neutrons, surrounded by electrons moving in regions of space called energy levels (shells).
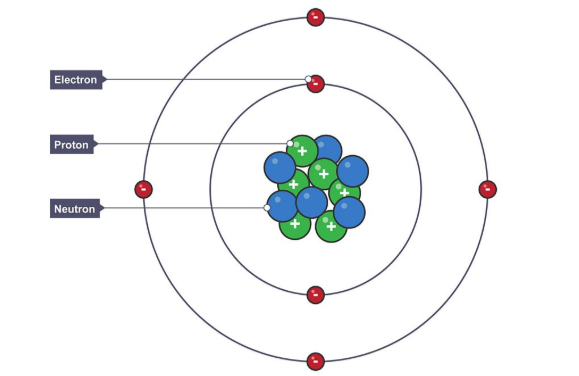
What are the properties of a proton?
Charge: +1 (positive)
Relative mass: 1
Location: nucleus
Behaviour: Deflected in electric and magnetic fields toward the negative plate
What are the properties of a neutron?
Charge: 0 (neutral)
Relative mass: 1
Location: nucleus
Behaviour: Not deflected in electric or magnetic fields
What are the properties of an electron?
Charge: –1 (negative)
Relative mass: 1/1836 (very small)
Location: outside nucleus (energy levels/shells)
Behaviour:
Deflected in electric and magnetic fields toward the positive plate
Deflected more strongly than protons due to smaller mass
How do subatomic particles behave in electric fields?
Protons → deflected toward negative plate
Electrons → deflected toward positive plate
Neutrons → no deflection
How do subatomic particles behave in magnetic fields?
Protons and electrons are deflected (in opposite directions due to opposite charges)
Electrons are deflected more strongly (smaller mass)
Neutrons are not deflected
Why are electrons deflected more than protons in fields?
Because electrons have a much smaller mass (1/1836), so they are more easily accelerated by electric and magnetic fields.
What is mass number?
The total number of protons and neutrons in the nucleus of an atom.
Example:
Carbon-12 → mass number = 12
What are isotopes?
Atoms of the same element that have the same number of protons but different numbers of neutrons, and therefore different mass numbers.
Example:
Carbon-12 and Carbon-14
What is relative isotopic mass (based on the carbon-12 scale)?
The mass of an atom of an isotope relative to 1/12 of the mass of one atom of carbon-12.
Key point:
Carbon-12 is assigned a mass of exactly 12
All other isotopic masses are compared to it
What is relative atomic mass (Ar) based on the carbon-12 scale?
The weighted average mass of the atoms of an element relative to 1/12 of the mass of one atom of carbon-12.
Chlorine Ar ≈ 35.5 (due to mixture of isotopes)
What is radioactivity?
The spontaneous and random disintegration of unstable atomic nuclei, accompanied by the emission of radiation (α, β or γ).
What is a radioactive isotope?
A radioactove isotope is an atom of the same element that has an unstable nucleus and undergoes spontaneous decay, emitting radiation (alpha,beta and gamma) to achieve stability.
What is meant by the neutron-to-proton (n/p) ratio?
The ratio of the number of neutrons to the number of protons in a nucleus, which determines its stability.
Write a nuclear equation for alpha (α) emission.
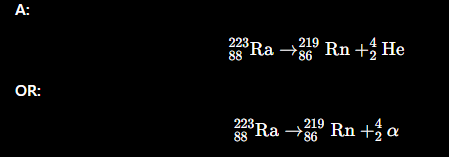
Write a nuclear equation for beta (β) emission.
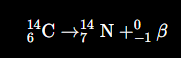
Write a nuclear equation for gamma (γ) emission.
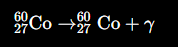
How does the neutron-to-proton ratio relate to radioactive decay?
Nuclei with an unstable n/p ratio undergo radioactive decay to become more stable
α and β emissions help adjust the n/p ratio toward stability
What are radioisotopes used for? (Give at least three uses)
Medicine (diagnosis and treatment):
Used as tracers to detect diseases in organs
Used in radiotherapy to destroy cancer cells
Example: Iodine-131 for thyroid disorders
2. Industry (leak detection and thickness control):
Used to detect leaks in pipelines
Used to measure thickness of materials (e.g. paper, metal)
Example: Beta emitters used in thickness gauges
3. Archaeology (carbon dating):
Used to determine the age of fossils and artefacts
Example: Carbon-14 dating
How do you calculate relative atomic mass (Ar) given isotopic masses and abundances?
Where:
mmm = isotopic mass
aaa = percentage abundance
Divide by 100 because abundances are in %
Method (how to calculate):
Multiply each isotopic mass × its abundance (%)
Add all the results together
Divide the total by 100
Final answer = relative atomic mass (Ar)
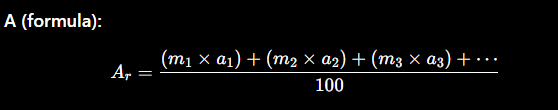
How do emission spectra provide evidence for discrete energy levels in atoms?
Emission spectra show separate lines of specific wavelengths, not a continuous spectrum. This indicates that electrons can only occupy fixed (discrete) energy levels and emit energy only when they move between these levels.
What is the Bohr model of the atom?
A model proposed by Niels Bohr which states that electrons occupy fixed energy levels (shells) and can only move between these levels by absorbing or emitting quantised energy.

What is meant by the emission spectrum of hydrogen?
The emission spectrum of hydrogen is a line spectrum produced when electrons in excited hydrogen atoms fall from higher energy levels to lower energy levels, emitting photons of specific wavelengths.

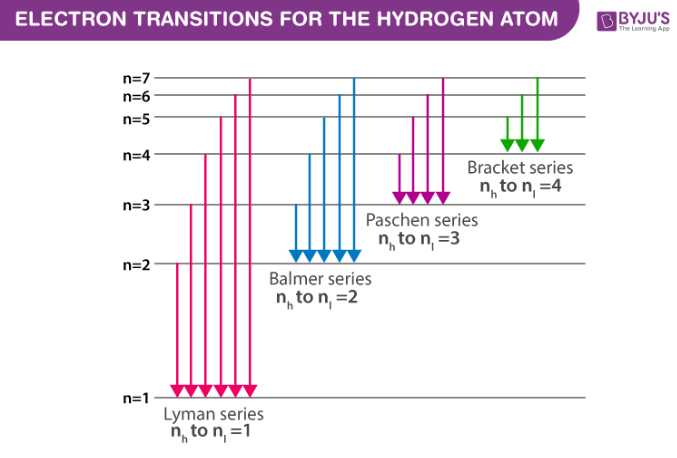
What are the Lyman and Balmer series?
Lyman series: Transitions where electrons fall to the n = 1 level (ultraviolet region)
Balmer series: Transitions where electrons fall to the n = 2 level (visible region)
What is the energy change equation for electron transitions?
ΔE=hν
Where:
ΔE= energy difference between levels
h = Planck’s constant
ν = frequency of emitted radiation
How does emission spectra support the idea of quantised energy levels?
Because only specific wavelengths (lines) are observed, it shows that electrons can only lose or gain fixed amounts of energy, meaning energy levels in the atom are discrete, not continuous.
What is a principal quantum number?
The principal quantum number (n) indicates the main energy level (shell) occupied by an electron.
Key idea:
n = 1, 2, 3, 4…
Higher n = higher energy level and larger distance from nucleus
What are atomic orbitals?
Regions of space around the nucleus where there is a high probability of finding an electron.
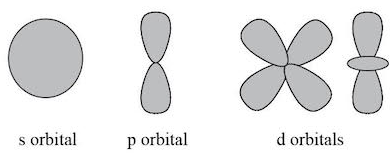
What are s, p and d orbitals?
s orbitals: spherical shape, hold 2 electrons
p orbitals: dumbbell shape, three orientations (px, py, pz), hold 6 electrons
d orbitals: more complex shapes, five orientations, hold 10 electrons
How do the energies of 4s and 3d orbitals compare?
The 4s orbital is lower in energy than 3d when empty, so it fills first. However, once electrons are added, 3d becomes lower in energy than 4s, which is why 4s electrons are lost first in ion formation.
How do you write electronic configurations using s, p and d orbitals?
1s → 2s → 2p → 3s → 3p → 4s → 3d → 4p
Aufbau principle: fill lowest energy first
Pauli exclusion principle: max 2 electrons per orbital (opposite spins)
Hund’s rule: electrons fill orbitals singly first before pairing
What is the electronic configuration of elements up to atomic number 30 (Zn)?
H (1): 1s¹
He (2): 1s²
C (6): 1s² 2s² 2p²
Ne (10): 1s² 2s² 2p⁶
Na (11): 1s² 2s² 2p⁶ 3s¹
Mg (12): 1s² 2s² 2p⁶ 3s²
Al (13): 1s² 2s² 2p⁶ 3s² 3p¹
Ar (18): 1s² 2s² 2p⁶ 3s² 3p⁶
K (19): [Ar] 4s¹
Ca (20): [Ar] 4s²
Sc (21): [Ar] 4s² 3d¹
Fe (26): [Ar] 4s² 3d⁶
Zn (30): [Ar] 4s² 3d¹⁰
Why is 4s filled before 3d?
Because the 4s orbital is lower in energy than 3d in the neutral atom, so it fills first. However, in ions, 4s electrons are lost before 3d electrons.
What is first ionisation energy?
First ionization energy is the energy required to remove the most loosely held electron from one mole of gaseous atoms to produce one mole of gaseous ions with a charge. It is measured in and represents the strength of attraction between the nucleus and valence electrons.
How does atomic radius affect first ionisation energy?
As atomic radius increases, first ionisation energy decreases.
Reason:
Outer electrons are further from the nucleus, so they experience less attraction and are easier to remove.
How does nuclear charge affect first ionisation energy?
As nuclear charge increases, first ionisation energy increases.
Reason:
A stronger positive nucleus attracts electrons more strongly, making them harder to remove.
How does shielding affect first ionisation energy?
As shielding increases, first ionisation energy decreases.
Reason:
Inner electrons repel outer electrons, reducing the effective attraction from the nucleus.
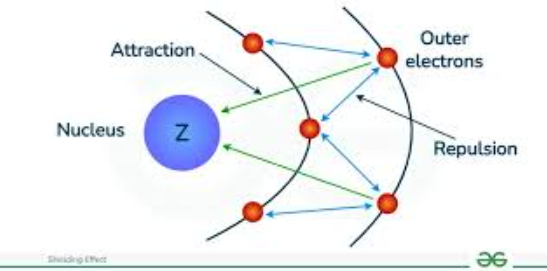
What is shielding effect?
The shielding effect (or screening effect) is the phenomenon where inner-shell electrons reduce the attraction of the nucleus on valence (outer-shell) electrons, due to inner electrons repelling outer ones. This reduces the effective nuclear charge felt by valence electrons, making them easier to remove and increasing atomic radius
What is the trend in first ionisation energy across Period 3?
First ionisation energy generally increases from Na → Ar due to:
Increasing nuclear charge
Electrons being added to the same shell (similar shielding)
Decreasing atomic radius
Why are there small drops in ionisation energy across Period 3?
There are two key drops:
1. Between Mg and Al
Mg: 3s²
Al: 3s² 3p¹
Reason:
The electron removed from Al is in a 3p sub-shell, which is:
Higher energy than 3s
Further from the nucleus
Easier to remove
👉 So ionisation energy drops from Mg to Al
2. Between P and S
P: 3p³ (half-filled, stable)
S: 3p⁴ (one paired electron)
Reason:
In sulfur:
One 3p orbital has a paired electron
Repulsion between paired electrons makes removal easier
👉 So ionisation energy drops from P to S
How does ionisation energy data provide evidence for sub-shells?
The non-uniform pattern of ionisation energies in Period 3 (especially the drops from Mg → Al and P → S) shows that electrons are removed from different sub-shells (3s and 3p) with different energies.
Conclusion:
This proves that electrons occupy sub-shells with different energy levels, not just a single shell.
What conclusion about atomic structure is supported by ionisation energy trends?
Ionisation energy data supports the idea that:
Electrons exist in shells and sub-shells
Sub-shells (s, p, d) have different energies
Electron removal depends on sub-shell type and electron pairing
How do successive ionisation energies help determine electronic configuration?
Successive ionisation energies increase gradually, but a very large jump indicates that all electrons in a particular shell have been removed, and the next electron comes from a closer (inner) shell.
What does a large jump in successive ionisation energy mean?
A large jump shows that:
The next electron is from a lower energy level (inner shell)
It is closer to the nucleus
It experiences less shielding and stronger attraction
How do you use ionisation energy data to determine electronic configuration?
Look at the values of successive ionisation energies
Identify where the big jump occurs
Count how many electrons were removed before the jump → this gives the number of electrons in the outer shell
Use this to write the electronic configuration
Example: If a large jump occurs after the 3rd ionisation energy, what does this mean?
The atom has 3 outer shell electrons
After removing 3 electrons, the next one is from an inner shell
👉 So configuration ends in: …3s² 3p¹ (or similar outer 3 electrons) depending on element
What is the key principle behind this method?
Electrons are removed in order of increasing energy, and a large jump in ionisation energy shows a transition from valence electrons to core electrons.