Chemistry - the rate and extent of chemical change
1/55
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
56 Terms
What is the rate of reaction?
How fast the reactants are changed into products
What is the formula for the rate of reaction
Rate of reaction = Amount of reactant used / Time
or
Rate of reaction = amount of product formed / Time
How can you find the rate of reaction on a graph?
draw a tangent at a given time and then find the gradient
How can you find the average rate of reaction on a graph?
total change in product/reactant ÷ total time
Why does the rate of reaction slow down?
There are fewer successful collisions as there are less reactants
What is the collision theory?
The theory that chemical reactions only occur when particles collide with sufficient energy
What is the activation energy?
the minimum amount of energy needed to start a chemical reaction
What can make reactions more likely?
- increasing the frequency of collisions
- increasing the energy they have when they collide
What factors affect the rate of reaction?
- temperature
- concentration
- pressure
- surface area
- catalysts
How does surface area affect the rate of reaction?
- increasing the surface area increases the rate of reaction because more particles are exposed for collisions
How does temperature affect the rate of reaction?
- increasing the temperature also increases the rate of reaction because particles have more kinetic energy.
- meaning particles move faster and collide more frequently
- also, the more energy the particles have, the more particles collide with enough energy to overcome the activation energy
How does concentration affect the rate of reaction?
- Increasing concentration increases the rate of reaction because there are more reactants per volume
- meaning that the particles become more crowded, which will result in a higher frequency of collisions
How does pressure affect the rate of reaction?
Increasing the pressure of gases increases the rate of reaction because the reactant particles are squeezed closer together so collisions occur more often.
How does a catalyst affect the rate of reaction?
A catalyst increases the rate of a chemical reaction by providing an alternative reaction pathway with a lower activation energy, allowing more reactant particles to collide successfully.
What is a catalyst?
A substance which increases the speed of a reaction, without being changed or used up in the reaction
What is a reversible reaction?
A chemical reaction in which the product reform the original reactants
If a reaction is exothermic in one direction, what kind of reaction would it be in the other?
endothermic
What is dynamic equilibrium?
in reversible reactions, occurring in a closed system, where the forward and reverse reaction rates are equal.
What is a closed system?
A system where neither matter nor energy can enter or leave
What are the requirements for equilibrium?
- a reversible reaction
- closed systems
What is the position of equilibrium?
Relative amounts of reactants and products at equilibrium.
What does it mean when equilibrium is towards the right?
there are more products than reactants
What does it mean when equilibrium is towards the left?
There are more reactants than products
What does it mean when equilibrium shifts towards the right?
The number of products increases, and the reactants decrease
What does it mean when equilibrium shifts towards the left?
The number of reactants increases, and the products decrease
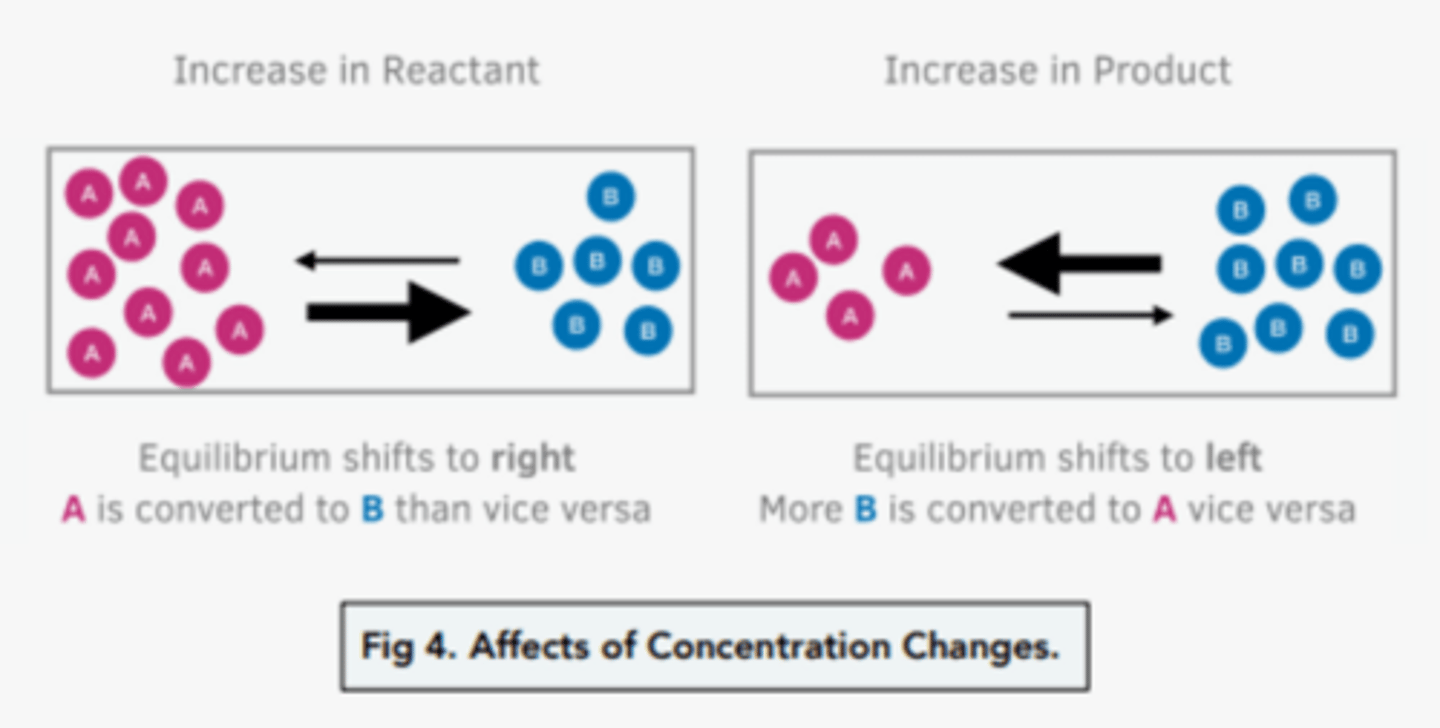
What is Le Chatelier's principle?
If a dynamic equilibrium is disturbed by changing the conditions (like concentration, pressure, or temperature), the system shifts its equilibrium position to counteract the change and restore balance
What factors affect the position of equilibrium?
temperature, pressure, concentration
How does increasing the temperature affect the position of equilibrium?
- if the temperature is increased, the equilibrium shifts to the endothermic reaction
- endothermic reactions absorb heat, so if the endothermic reaction is favoured, it will counteract the change
How does decreasing the temperature affect the position of equilibrium?
- if the temperature is decreased, the equilibrium shifts towards the exothermic reaction
- Exothermic reactions release heat, so if the exothermic reaction is favoured, it will counteract the change
Do catalysts affect equilibrium?
No, they only speed up reactions and have an equal effect on both the forward and backwards reaction
How does increasing the pressure affect the position of equilibrium?
- increasing the pressure shifts the equilibrium to the side with fewer gas particles
- This reduces the total number of particles by reacting more particles together to form a smaller number of product molecules
- This reduces the number of particles that collide with the container walls, undoing the initial change
How does decreasing pressure affect the position of equilibrium?
- decreasing the pressure shifts the equilibrium to the side with the most gas particles
- This increases the total number of particles, leading to more collisions with the container walls, undoing the initial change
How does changing the concentration affect the position of equilibrium?
- if the reactants increase, equalibrium shifts to the right to make more product to counteract this change
- if the product increases, equalibrium shifts to the left to make more reactants to counteract this change
What is the Haber process?
a method used to produce ammonia
Formula for the Haber process
N₂(g) + 3H₂(g) ⇌ 2NH₃(g)
Is the forward reaction of the Haber process exothermic or endothermic?
exothhermic
Where do the reactants of the Haber process come from?
- nitrogen comes from the air
- hydrogen is made from natural gas or from cracking hydrocarbons
If the pressure is decreased, which side will be favoured?
the side with the greater total number of moles of gas
If the pressure is increased, which side will be favoured?
the side with fewer moles of gas
If the temperature is decreased, which side will be favoured?
the side with the exothermic reaction
If the temperature is increased, which side will be favoured?
the side with the endothermic reaction
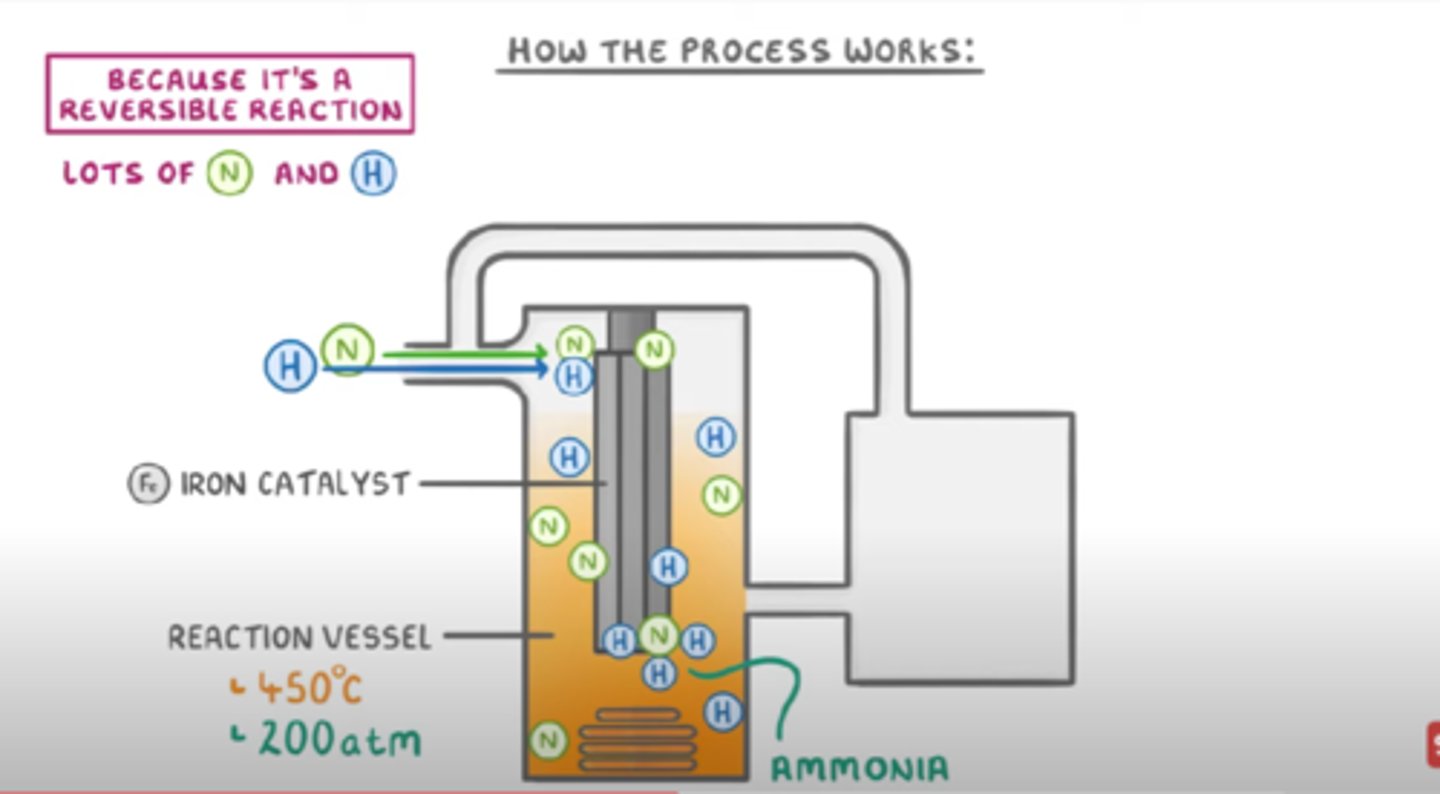
How does the Haber process work?
1. A gas stream containing hydrogen and nitrogen enters the system
2. The nitrogen and hydrogen mixture is compressed to a pressure of 200 atm and heated to 450 degrees
3. The hydrogen and nitrogen enter a reaction vessel containing iron catalyst
4. The mixture of gases emerges from the reactor and enters the cooling chamber. The ammonia liquifies and is separated
5. The unreacted nitrogen and hydrogen are returned to the reaction vessel via the compressor
What is ammonia used for?
- Used to make Nitric Acid HNO₃
- Used to make fertilisers - Ammonium Nitrate
What are the conditions for the Haber process?
- 450°C
- pressure of 200 ATM
- iron catalyst
Why does the Haber process happen at 450°C?
- to balance reaction speed with production yield
- lower temperatures favour ammonia formation because the forwards reactioon is exothermic, however they make the reaction too slow
- so 450°C is a compromise
Why does the Haber process happen at 200 ATM?
- High pressure gives a higher yield since there are fewer molecules on the right-hand side
- However, very high pressure is dangerous and expensive
- So 200 ATM is used as a compromise
Why is a catalyst used in the Haber process?
to significantly speed up the reaction rate between nitrogen and hydrogen, allowing it to reach equilibrium faster
Why are the nitrogen and hydrogen recycled in the Haber process?
- only about 15% of nitrogen and hydrogen react each time the gases pass through
- The unreacted gases are recycled back into the reactor to improve efficiency
What elements are found in NPK fertilisers?
- nitrogen
- phosphrous
- potassium
What do plants use nitrogen for?
- needed to make proteins and chlorophyll.
- helps plants grow new leaves.
- without it, plants look stunted and yellow
What do plants use phosphorus for?
- needed for healthy root growth
- is also used to make DNA
What do plants use potassium for?
- is needed to make enzymes
- is also involved in respiration and photosynthesis
Why are NPK fertilisers used?
- Plants often don't get enough of these minerals naturally from the soil, so NPK fertilisers replace the missing minerals
Examples of nitrogen sources in NPK fertilisers?
- ammonium nitrate (ammonium + nitric acid)
- Ammonium sulphate (ammonium + sulfuric acid)
- ammonium phosphate (ammonium + phosphoric acid)
Examples of phosphorus sources in NPK fertilisers?
- calcium phosphates (single and triple super phosphates)
Examples of potassium sources in NPK fertilisers?
- potassium chloride
- potassium sulfate