MEEN 315 exam 1 conceptual
1/71
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
72 Terms
initial step 1
define system. list assumptions
initial step 2
identify type of system
initial step 3
list properties of a system
System
region we want to study
Surroundings
everything external to the system
Boundary
separates the system from the surroundings.
can be at rest OR in motion
Types of system
isolated, closed, or open
Isolated system
no mass transfer or energy transfer across boundary (ex: perfect insulator)
Closed system
no mass transfer across boundary, but there is energy transfer across the boundary (ex: a closed cold drink cup in a warm room)
Open system
there is mass and energy transfer across the boundary (ex: a classroom door opening)
Macroscopic properties
characterize the gross/total behavior of the entire system
Intensive properties
size independent, not additive
Temperature, pressure, density
Extensive properties
size dependent, additive
Volume, mass, energy (typically converted to intensive)
Specific volume
volume/unit mass
v = V/m = 1/density
Specific energy
energy/unit mass
e = E/V
Equilibrium
state of balance
Process
action causing change in a system from one state to another
Zeroth law of thermo
if 2 bodies are in equilibrium with a 3rd body, they are all in equilibrium with each other, even if not in direct contact
Pressure
normal force exerted by fluid per unit area
Absolute pressure
actual pressure
measured with respect to an absolute vacuum
Gage/Vacuum pressure
used for measuring devices
measured with respect to the absolute pressure of the atmosphere
is everything above sea level
Pressure relations
Pgage + Pother = Pabs
Temperature
degree of hotness and coldness
measured to a definite scale
empirical has limitations, like range
Problem solving steps
known and find
identify what is known
state what needs to be determined
schematic and given data
draw a sketch of the system
label with relevant info
engineering model
list all assumptions and idealizations
analysis
reduce equations to get results
Transient
properties at the same location change with respect to time
Steady state
all properties of a system at the same location do NOT change with time
Forms of energy
macroscopic and microscopic
Macroscopic energy
processed as a whole with respect to an outside source frame
scaled we can directly see
Microscopic energy
processed by molecular structure of a system
thermal, chemical, and nuclear
independent of outside reference frames
sum equals to internal energy (U)
Potential energy
position of a system in earth’s gravitational field
Kinetic energy
motion of a system as a whole
Total energy of a system
Esys = KE + PE + U
kJ, Btu
Specific energy of a system
esys = ke + pe + u
kJ/kg, Btu/lbm
Modes of energy transfer
Heat, mass, work
only recognized at the system boundary
NOT property
depend on process paths
Heat
transferred by a temperature difference
moves from high T to low T
Q
Adiabatic process
NO heat transfer
Tsystem = Tsurroundings
only energy transfer is considered at the boundary
Work
energy transfer associated with force exerted on a system in the direction of a distance
W = F*S
Wb = integral( P dV ) ,(expansion and compression)
Quasi-equilibrium
moves very slowly
basically stays at equilibrium state at all times
Isochoric
Volume stays constant
makes work always = 0
Polytropic
pressure and volume are related
n = polytropic index between 0 and infinity
p*V^n = constant
Modes of work
boundary work
shaft work: Ws = T(2 pi n)
electrical work: We = V*I
Total energy
static
property
KE, PE, U
belongs to a system
point function (independent from process path)
exact differential
Transferred energy
dynamic
mode of energy transfer
Q, Emass, W (Wb like isobaric, isochoric, etc)
recognized at boundary
NOT properties
path function (depends on process path)
inexact differential
Process paths
area under curves show changes in work (P-V graph)
First law of thermo
Energy can be transferred between systems, but not created or destroyed
delta Esys = delta Ein - delta Eout
Sign conventions of energy transfer
positive is in/to/on
negative is out/by/from
State principle
state of a simple compressible system is completely specified by 2 independent, INTENSIVE (size indep., not additive) properties
Thermodynamic cycle
system that returns to the initial state at the end of the process
Power cycle
delivers net work transfer to surroundings
Refrigeration/Heat pump cycle
transfers heat from a cold body to a hot body by an addition of work input
Pure substance
constant chemical composition throughout
cannot be separated by physical separation
homogenous
may exist in more than one phase (ex: h2o can be solid, liquid, gas, or mix)
Latent heat
amount of heat energy absorbed or released during a phase change process
Latent heat of fusion (hif)
energy absorbed during melting
equals energy released during freezing
Latent heat of vaporization (hfg)
energy absorbed during vaporization
equal to energy released during condensation
Phase changes
oscillation changes phases
processes are reversable
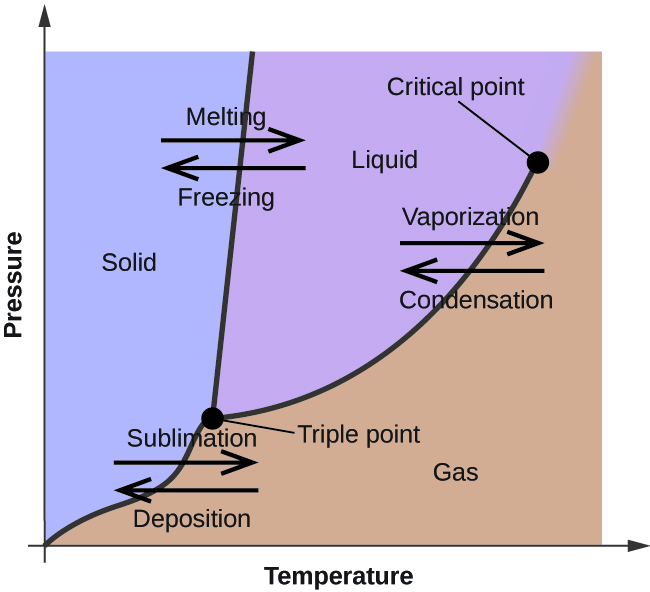
Phase diagram (P-T)
supercritical: liquid and vapor do not differentiate past critical point
triple point: all 3 phases coexist in equilibrium
melting line shows whether a substance expands or contracts with freezing (moves left = expands, moves right = contracts)
Liquid-vapor phase change process states
compressed liquid: substance NOT about to vaporize
saturated liquid: liquid about to vaporize
saturated mixture: liquid and vapor states coexist in equilibrium
saturated vapor: vapor about to condense
superheated vapor: vapor NOT about to condense
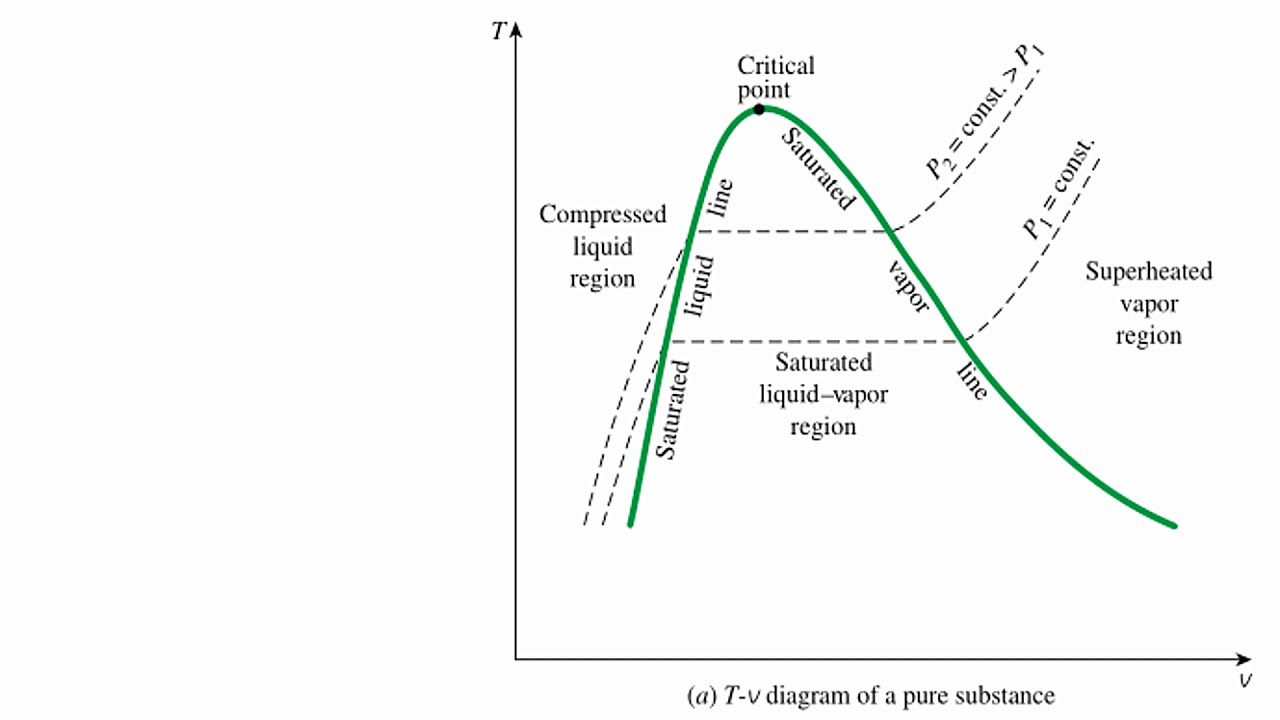
T-V diagram
saturation temp (Tsat): temp when a pure substance changes phase at a given pressure
saturation pressure (Psat): pressure when a pure substance changes phase at a given temp
Tsat = f(Psat): there are multiple Tsats that depend on the pressure!
Tsat2 >Tsat1 and P2 > P1
saturated liquid and saturated vapor curves meet at the critical point
the linear sections go from low to high from L to R
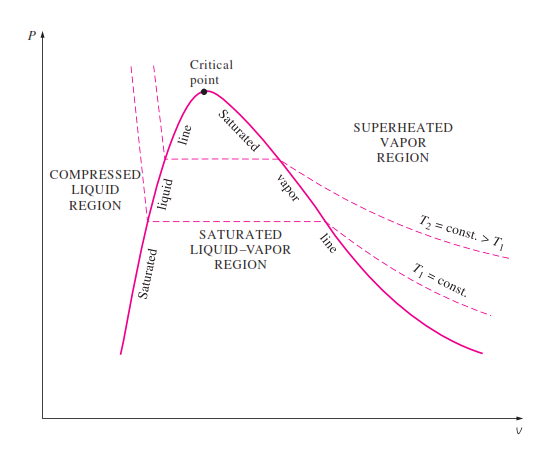
P-V diagram
Psat is the line in the sat mix region
T1 is isotherm (constant temperature)
relies on the state principle
the linear sections go from high to low from L to R
Quality (x)
mg/mt
x = 0: saturated liquid
x = 1: saturated vapor
x between 0 and 1: saturated mixture
Linear interpolation
finds a value between 2 known values
can only do this between 2 points, do NOT extrapolate
v < vf
compressed liquid
vf < v < vg
saturated mixture
v > vg
superheated vapor
T < Tsat at a given P OR P > Psat at a given T
compressed liquid
T > Tsat at a given P OR P < Psat at a given T
superheated vapor
Ideal gas equation of state
relates pressure, temp, and specific volume
pv = RT
pV = mRT
pV - nRT
For ideal gas, R = Rbar/MW
Real gases
hypothetical gas with molecules that take up negligible space and have no interactions
low density, high T, low P
Compressibility factor (Z)
accounts for deviation of real gases from ideal gas behavior at a given T and P
Z = vactual / videal
Principle of corresponding states
Z is the same for all gases at the same values of the reduced temperature and reduced pressure
Tr = T/Tc
Pr = P/Pc
Specific heat capacity
heat energy required to raise the temperature of the unit mass of a substance by 1 degree
constant volume specific heat (cv)
constant pressure specific heat (cp)
Cp is ALWAYS greater than Cv for all substances
Incompressible substance
substance whose specific volume or density is constant
solids and liquids