CHEM EXAM IV
1/37
Earn XP
Description and Tags
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
38 Terms
thermochemistry
study of heat absorbed or released during chemical and physical changes
energy
the capacity to supply heat or do work
work
a change in volume against a pressure
potential energy
energy an object has because of its relative position, composition, or condition
kinetic energy
energy an object has because of its motion
law of conservation of energy
during a chemical/physical change, energy is neither created nor destroyed, but it can change forms
thermal energy
random internal kinetic energy associated with the random motion of atoms and molecules internally in a system
temperature
quantitative measurement of particle speed
hot = high = fast moving particles
heat
the transfer of thermal energy between two bodies at different temperatures
heat flow
increases the thermal energy of one body and decreases the thermal energy of the other until both are the same
exothermic process
heat is released and/or absorbed by solution
ex. combustion reaction that uses a torch
endothermic process
heat is absorbed and/or provided by the solution
ex. cold pack
calorie
amount of energy required to raise one gram of water by 1*C
cal
Calorie / kilocalorie
quantifies food energy content
C
joule
the amount of energy used when a force of 1 newton moves an object 1 meter
J
James Prescott Joule
english physicist
joules are named for him
heat capacity
quantity of heat absorbed or released when a body of matter experiences a change in temperature change
extensive property
does a large pan or small pan have higher heat capacity
large pan
specific heat capacity
quantity of heat required to raise the temperature of 1 gram of a substance by 1 °C
intensive property
calorimetry
measures the amount of heat transferred in or out of a substance
system
substance or substances undergoing the chemical or physical change
surroundings
all other matter that provide or absorb heat from a system
calorimeter
device that measures amount of heat involved in a chemical or physical processf
types of calorimeters
coffee-cup: two “cups” - can be industrial, not actual cups
bomb: used when the reaction releases gas
whole body: can hold a human being, used to compare heat under differing conditions
nutritional calorie
energy derived from metabolism of foods
thermochemistry
deals in the relationships between heat, work, and forms of energy
first law of thermodynamics
—>
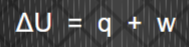
expansion work
pressure-volume work
system pushes back the surroundings against a restraining pressure
surroundings compress a system
-q
-w
what type of reaction (endo/exothermic)
exothermic
state function
values that depend on the state of the substance, and not on how that state was reached
ex. internal energy, density
enthalpy
the sum of a system’s internal energy (U) and the mathematical product of its pressure (P) and volume (V)
most convenient choice for determining heat

enthalpy change
this is pressure-volume work

what does a thermochemical equation represent
changes in both matter and energy

what type of reaction does negative delta H represent? (endo/exothermic)
endothermic

standard state
a commonly accepted set of conditions used as a reference point for the determination of properties under other different conditions
The IUPAC standard state conditions:
Pressure of 1 bar (or 1 atm; 1 bar = 0.987 atm)
Solutions at 1 M concentration

standard enthalpy of combustion
enthalpy change when exactly 1 mole of a substance burns (combines vigorously with oxygen) under standard state conditions
heat of combustion
standard enthalpy of formation
enthalpy change for a reaction in which exactly 1 mole of a pure substance is formed from its constituent free elements in their most stable states under standard state conditions
Hess’s Law
If a process can be written as the sum of several stepwise processes, the enthalpy change of the total process equals the sum of the enthalpy changes of the various steps