elements, compounds and mixtures
1/12
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
13 Terms
what are elements?
substances that contain only one type of atom
in a diagram, they are show as atoms of a single colour or size
what are compounds?
substances that are formed when two or more atoms chemically combine
in a diagram, they are show as different coloured atoms joined together in fixed groups
what are mixtures?
a physical combination of two or more substances that are mixed together and no chemical reaction occurs
can be made from elements and/or compounds
what are the melting and boiling points of a pure substance like?
pure substances, like elements and pure compounds, melt and boil at fixed temperatures
what are the melting and boiling points of a mixture like?
mixtures usually don’t have a fixed melting or boiling point and melt or boil over a range of temperatures. This is because the presence of impurities lowers the melting point and raises the boiling point.
Substances in a mixture also aren’t chemically combined, so the components have different melting and boiling points
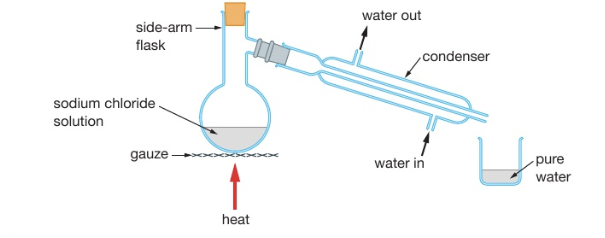
describe simple distillation
used to: separate the solvent from a solution
it works because the dissolved solute has a much higher boiling point than the solvent
method: when the solution is heated, the solvent evaporates, rises, and passes down the condenser, where it is cooled and condensed. The pure liquid is then collected in the beaker.
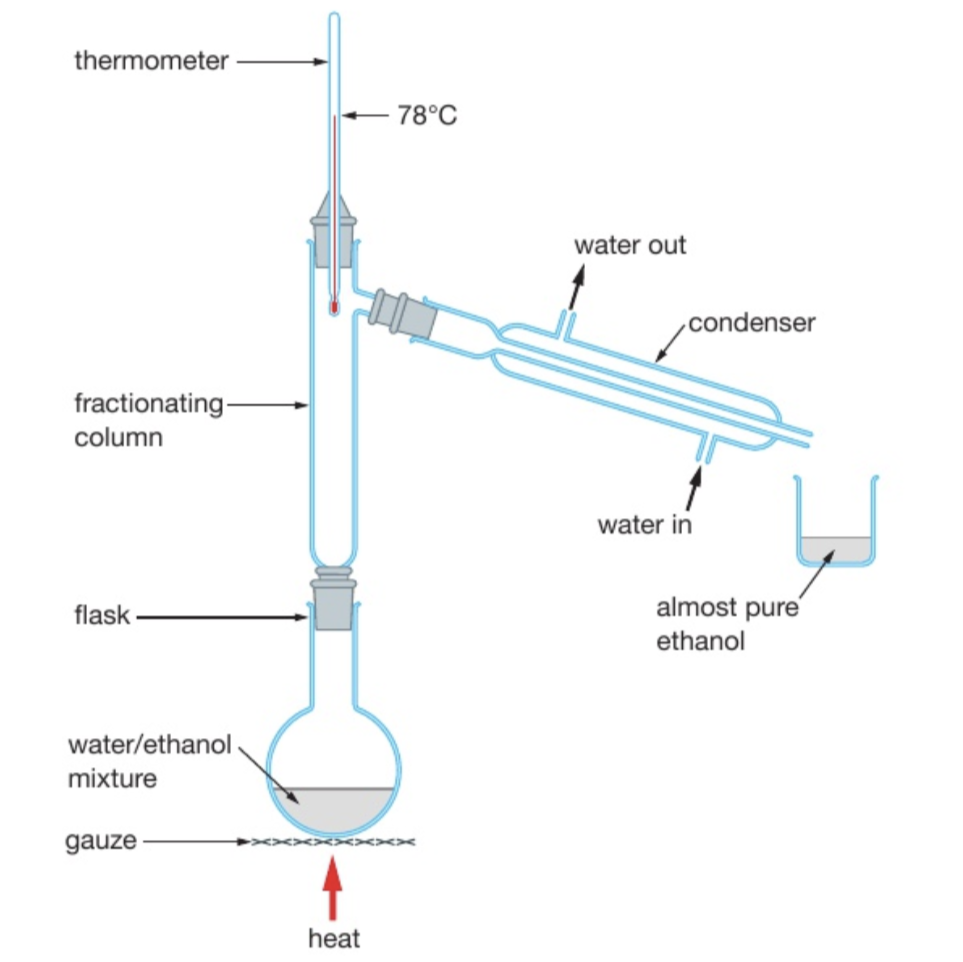
describe fractional distillation
used to: separate a mixture of liquids
it works because different liquids have different boiling points
method: the mixture is heated and evaporates. the vapours rise through the fractionating column, which is hot near the bottom and cooler near the top, and condenses when it reaches a point in the column that has a temperature lower than its boiling point. Therefore, the column separates liquids that condense at different temperatures. The liquid with the higher boiling point will then trickle back into the flask, and the liquid with the lower boiling point is collected in the beaker.
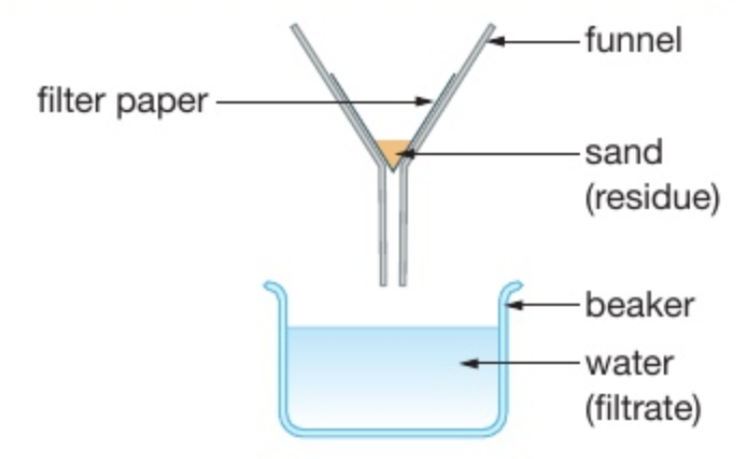
describe filtration
used to: separate an insoluble solid from a liquid
method: pour a mixture containing an insoluble solid and a liquid into a funnel lined with filter paper. The solid will get caught in the filter paper whilst the liquid will be collected in the beaker.
describe crystallisation
used to: separate a soluble substance from a solvent
method: heat the solution in an evaporating basin to evaporate some of the solvent until it become a saturated solution and crystals start to form. Turn off the bunsen burner and allow more crystals to form as more water evaporates and the solution cools. You can then separate the crystals from the mixture by filtration.
what is a chromatogram?
a representation of the separated components of a mixture. It shows the soluble pigments that were present in the mixture
how does a chromatogram provide information about the composition of a mixture?
A pure substance will leave 1 spot on the chromatogram
a mixture will leave two or more spots in a vertical column
the higher the spot, the more soluble the substance
by matching the colour and height of the spots, you can identify the pigments which were present in the mixture.
how can the calculation of Rf values identify the components of a mixture?
the Rf value helps to identify components of a mixture as substances with the same Rf value are likely to be the same substance
Rf = distance moved by a spot/distance moved by the solvent front
Rf values don’t have a unit and are always between 0 and 1
how can you investigate paper chromatography using inks/food colourings?
draw a pencil line across the chromatography paper, about 1cm away from the bottom
put a spot of the mixture of dyes on the pencil line and let it dry
suspend the chromatography paper in a beaker containing a small amount of solvent so that the solvent is just below the pencil line
put a lid on the beaker so the solvent doesn’t evaporate
when the solvent has moved up the paper to about 1cm from the top, remove the paper and draw a pencil line to show where the solvent got to. This is the solvent front.
leave the paper to dry so that all the solvent evaporates