CHEM canvas quiz + notes (4/2/6)
1/51
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
52 Terms
examples of state function (depends only on initial and final state)?
internal energy (U), enthalpy (H), temp (T), pressure (P), volume (V), Gibbs free energy (G)
examples of path functions (depend on route taken)?
heat (q) and work (w)
1 __ of water = 1 gram of water
mL (NOT liters)
-mcΔT = mcΔT…simpler way of writing left side is basically
-(mcΔT)
The internal energy (U) increases when
energy is added to the system either as heat or work
Under what condition(s) is the enthalpy change, ΔH, of a process equal to the amount of heat (q) transferred into or out of the system?
at constant pressure (ΔH = qp)
how to find q when given equation, ΔHrxn, and mols (or grams)?
q = mols x (ΔHrxn/ coefficient)
formula for KE?
need?
½ (m)(v2)
kilograms for m (not grams)
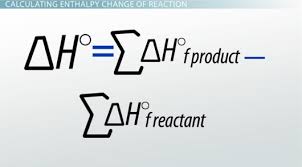
standard enthalpy of reaction is calculated by…
(these problems will have table of values)
which will have greatest temp change when multiple substances of the same mass absorb the same amount of heat?
which will have smallest temp change when multiple substances of the same mass absorb the same amount of heat?
substance with the lowest specific heat capacity
substance with highest specific heat capacity
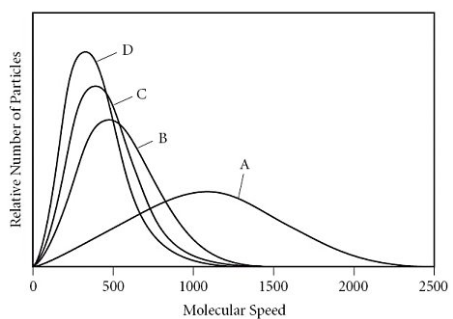
Maxwell-Boltzmann distribution graph is..?
the one with smallest molar mass is the…?
speed vs number of particles
fastest one (the one farthest to the right)
boyle’s law?
relationship?
constant?
P1V1 = P2V2
inverse (so increasing pressure, decreases volume)
temperature and n (molecules)
Avogadro’s Law?
relationship?
constant?
V1/n1 = V2/n2
Direct relationship (Increasing number of molecules, increases volume)
T and P
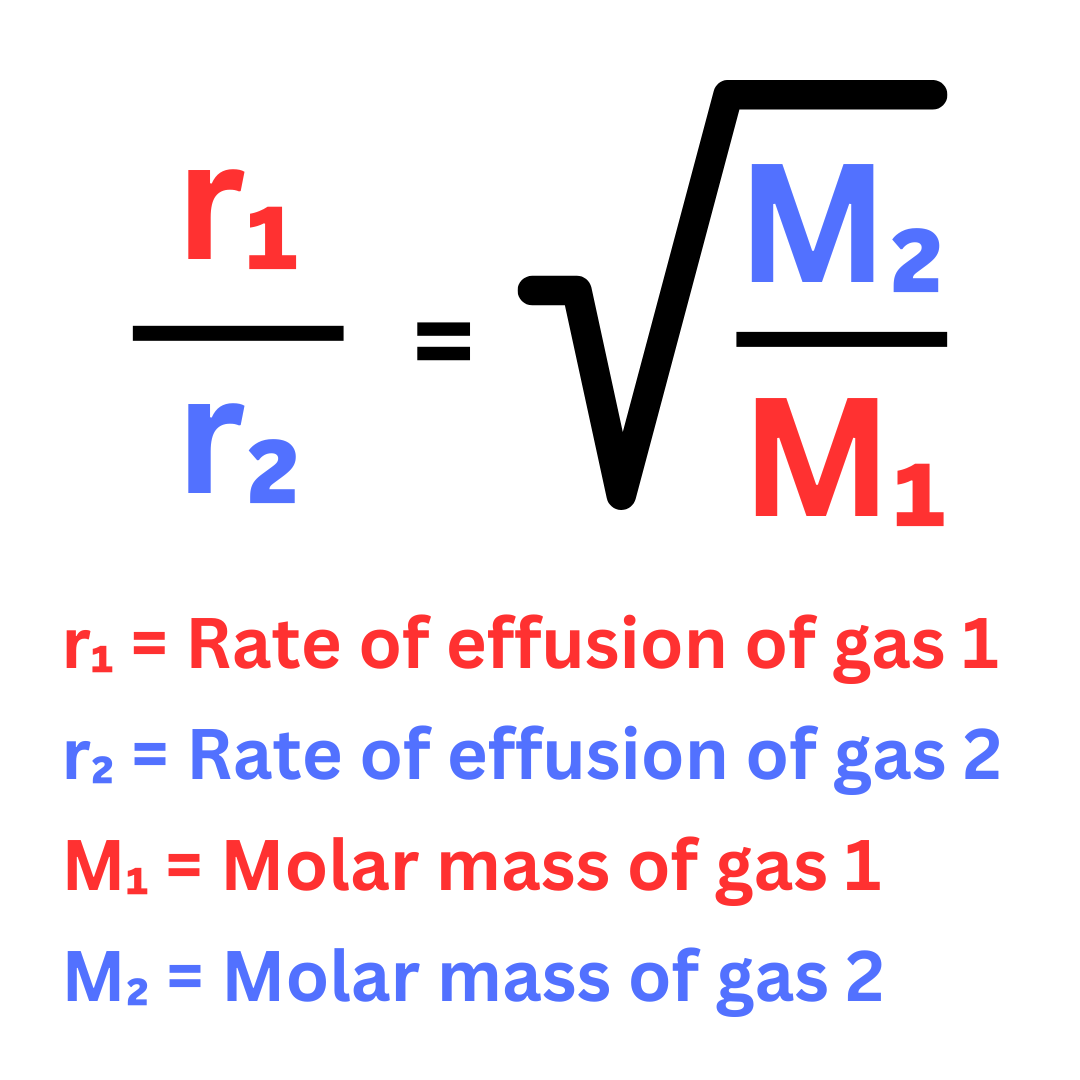
gas with greatest rate of effusion (move the fastest) at given temperature is the one…
according to the graham’s law of effusion, gas is…
with the lowest molar mass
inversely proportional to square root of its molar mass
2 formulas for questions like:
A mixture of 10.0 g of Ne and 10.0 g Ar have a total pressure of 1.6 atm. What is the partial pressure of Ne?
A gas mixture containing 17.0 mol of gas has a total pressure of 760.0 torr. If He gas accounts for 5.30 mol, what is the partial pressure of He?
Mole fraction (X) = moles of component / total moles in mixture
Partial pressure = mole fraction x total pressure
rate effusion formula (Graham’s Law)?
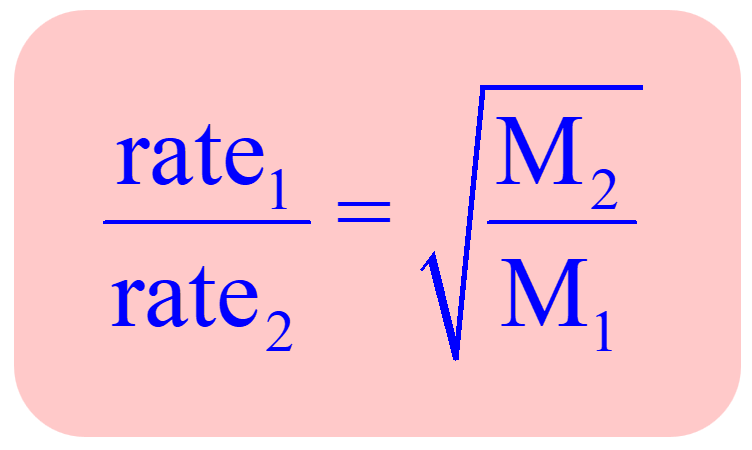
rate effusion formula if times is provided and not rates
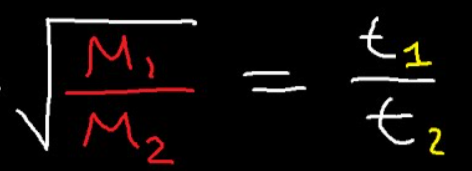
Ideal Gas Law (PV = nRT) when density is given and solving for molar mass?
M = dRT/P
Ideal Gas Law (PV = nRT) when density is given and solving for density?
D = PM/RT
(basically PM = dRT)
long wavelengths have a ___ frequency and ___ energy
short wavelengths have ___ frequency and ___ energy
low, low
high, high
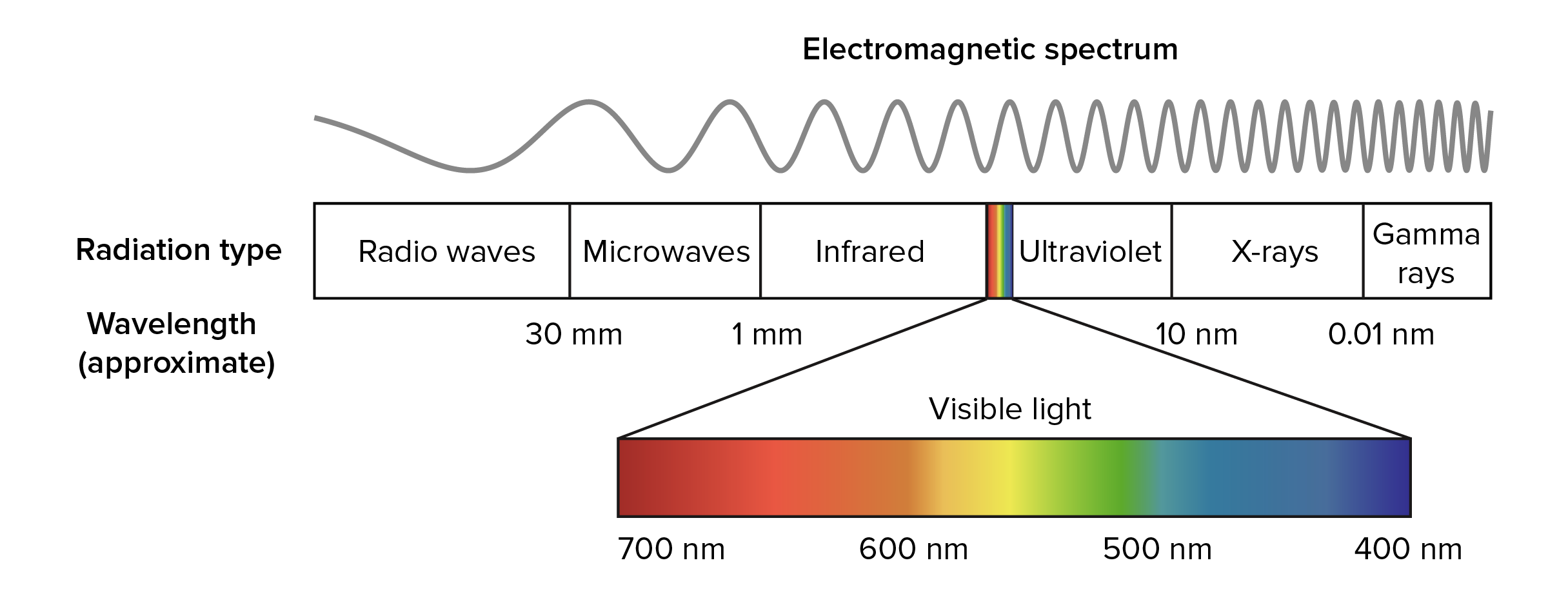
longest wavelength (lowest energy) to shortest wavelength (highest energy)?
ROYGBIV (red, orange, yellow, green, blue, indigo, violet)
moving toward red (or radio waves) = wavelength ____
moving toward violet (gamma rays) = wavelength ____
increases, decreases
speed of light © formula is…
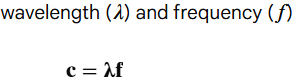
wavelength measured in…
frequency measured in…
meters (m)
hertz (Hz or S-1)
speed of light equation relationship?
wavelength and frequency are inversely proportional (wavelength increases, frequency decreases)
Gases behave in an ideal manner at ___ temperature and ___ pressure
high, low
The pressure of gases are the result of the ____ of collisions and the ___ energy of the collisions
frequency, kinetic
(heat increases both)
rate of effusion means
molecular speed means
how many molecules pass through a tiny hole over time (mol/s)
how fast a molecule moves (m/s)
All gases at the same absolute temperature and pressure will have the same average____, but not the same
kinetic energy
molecular speed or rate of effusion
system absorbing J (heat) =
system releasing/producing J (heat) =
+,-
STP means
also is when T and P are
standard temperature and pressure
0 C and 1 atm
common molecular weight units…
atomic mass unit (amu or u), Dalton (Da), grams per mole (g/mol)
formula for u need for question below?
steps?
A 31.5 mL aliquot (sample) of H2SO4 (aq) of unknown concentration was titrated with 0.0134 M NaOH (aq). It took 23.9 mL of the base to reach the endpoint of the titration. The concentration (M) of the acid was ________.
mols/L = M (molarity)
convert mL to L, balance equation, find mols with mols/L = M, find mols of H2SO4 using coefficients, find M with mols/L = M
endothermic or exothermic: ammonium thiocyanate and barium hydroxide are mixed at 25 C: temp dropped?
formula?
25 C is the surroundings (water in this case) and dropped because system absorbed energy (ΔTsys = -ΔTsurr)
electron shell capacities for 1st shell, 2nd shell, 3rd shell, 4th shell, 5th shell?
2, 8, 18, 32, 50
quantum numbers question is almost always about the
last electron in electron configuration

angular momentum quantum shapes and letters (fancy l) for 0, 1, 2
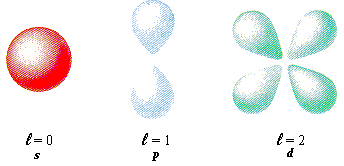
how to solve for energy (E) when given wavelength, planck’s constant (h), speed of light ©
how to solve for energy (E) when given plank’s constant (h) and frequency (v)

1 meter to nanometer (nm)
1 nanometer to meters (m)
1 meter to micrometers (µm)
1 micrometer (µm) to m
109
10-9
106
10-6
Mn is [Ar] 4s2 3d5
Mn2+ is ?
[Ar] 3d5 (remove from biggest number first, does NOT apply for adding)
the valence shell number when looking at electron configuration is…
the highest number
ex: valence shell is 6 in [Xe] 6s24f145d8
in quantum number, n =
formula to figure max number of electrons in an atom when only given n?
subshells
2n2
steps (including formula) for orbitals with given only n = 5?
(hint: orbitals = possible mfancy l values)
identify fancy l which is 0, 1, 2, 3, 4 then use 2(fancy l) + 1 for each one and add all
in quantum numbers: Formula for max numbers of electrons in an atom when given only n and fancy l?
(hint you only use the fancy l to figure answer)
0 = s = 2
1 = p = 6
2 = d = 10
3 = f = 14
in quantum numbers: Max numbers of electrons in an atom when given n, fancy l, and mfancy l
always 2
ΔHrxn stands for?
enthalpy change of the entire reaction
1 cm = __ millimeters (mm)
only condition when mm is same as mmHg is…
10
liquid is mercury (mmHg) and measuring height difference in manometer
formula for manometer, trying to solve for pressure of gas?
ΔH in this situation means…
Pgas = Patm+ ΔH
height difference in mmHg
temp increase and molecules of gas increases and volume is CONSTANT (closed container) then pressure…
increases (more collisions)
temp increase and molecules of gas increases and volume increase (like balloon) then pressure…
stays the same (gas spread out)
steps in solving molar mass when density is NOT given?
solve for n (PV = nRT) and use grams/MM = n
Based on the kinetic-molecular theory, at a constant temperature, all gas molecules have the same…
average kinetic energy