Identifying an Unknown Alcohol - Lab 13
1/50
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
51 Terms
Purpose
Identify an unknown liquid alcohol by qualitative chemical tests and by comparison of a derivative melting point to literature values.
Nuclear Magnetic Resonance spectroscopy and Infrared spectroscopy
______________________ are the major spectroscopic techniques used by organic chemists.
Simple Qualitative Tests
Much insight can be gained by using ___________ to determine the identity of unknowns
Structures of unknown compounds
_______________ of unknown compounds can be determined by
- comparing physical properties
- Performing functional group tests
- Checking the melting points of the derivatives against those of known compounds reported in literature
Solubility properites
______________ and chemical reactivity become apparent during these qualitative tests
Classification Tests
Qualitative tests are called ______________________ because they support or refute the presence of certain structural features of a molecule.
For example: Certain classification tests indicate the presence of a particular functional group - such as an aldehyde or an alcohol. Other tests indicate a functional group that easily undergoes oxidation. Other tests show the presence of an acidic hydrogen or other feature.
Inviolable rules
There are two _________________ when performing classification tests.
- First, perform the test exactly as described
- Second, always perform tests on a known compound that will result in a known positive and on known negative. Then perform the test on the unknown reagent to have a direct visual comparison and to confirm that the reagents are good and you are performing the test properly.
Lucas Test
Hydrochloric Acid (HCl) Can be used to replace the -OH group of alcohol with Cl-.
SN1
The rate of this _____ rxn (Lucas test with HCl removing OH to Cl0) depends on the stability of the carbocation formed at the carbon bonded to the -OH group.
Tertiary
LUCAS TEST: In general, ________ alcohol react fastest
Primary
LUCAS TEST: _________ Alcohol react the slowest.
resonace
Lucas Test: Alcohol such as allyl alcohol and benzyl alcohol, which can produce ____________ resonance stabilized carbocations, will react just as fast as tertiary alcohols.
Lucas reagent
is a solution of zinc chloride in concentrated HCl. It is a lewis acid that increases the acidity of the solution.
Lucas Test Positve
__________ occurs when the non-polar alkyl halide formed in the reaction begins to separate from the aqueous solution to form a second layer. The solution will appear cloudy.
ZnCl2
For the lucas test to work, the alcohol must be soluble in the aqueous ZnCl2 solution. In practice, only alchols containing no more than seven or eight carbon atoms will dissolve in the lucas reagent.
Lucas Tertiary
react within 30 seconds with resonance stabilized carbocations
Lucas Secondary
React in one to three minutes
Lucas Primary
No reaction in 60 minutes
Lucas Test Reaction
Look on Paper
TCICA
1,3,5 - Trichloroisocyanuric Acid
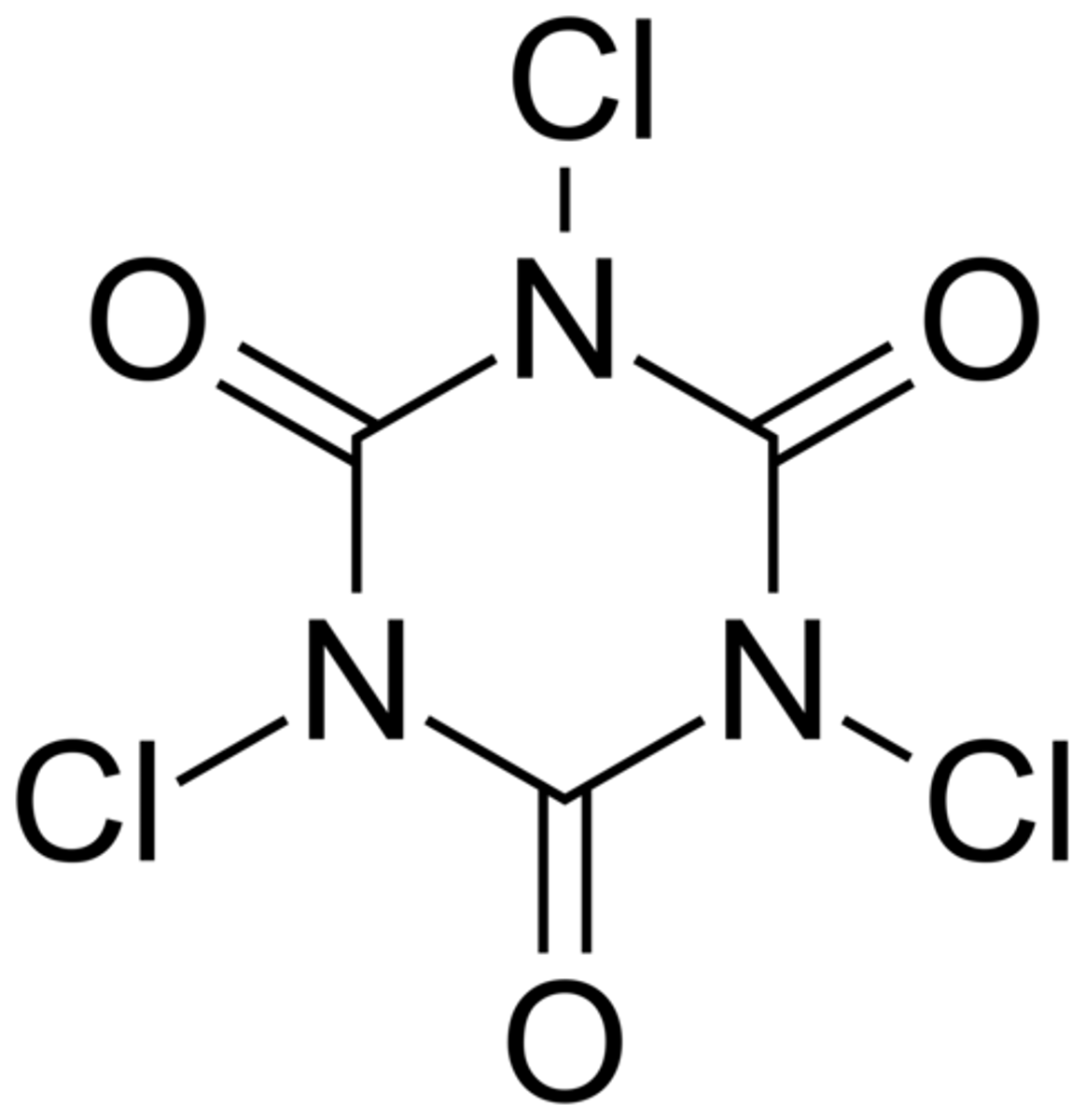
TCICA
is a common source of chlorine used to disinfect swimming pools.
TCICA
In acid solutions, ______ slowly releases chlorine, which is an oxidizing agent.
oxidizable
TCICA test is rapid in the presence of an ___________ compound like a primary or secondary alcohol.
TCICA Product
Isocyanuric Acid (which is very soluble in water, but precipitates to form the solvent acetonitrile)
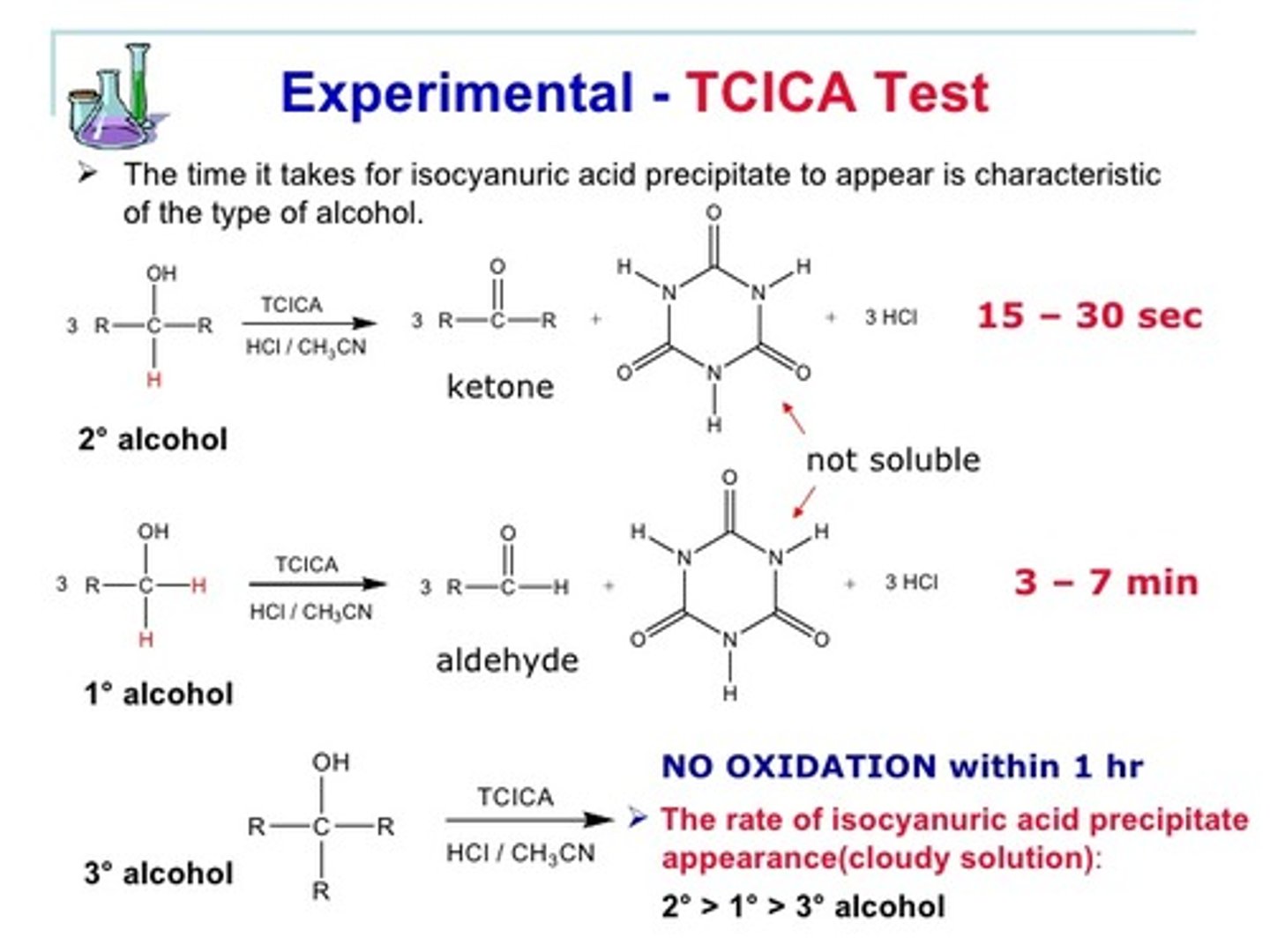
time
TCICA Test: The ______ it takes for isocyanuric acid to precipitate is characteristic of the type of alchol.
TCICA Primary Alchols
Produce precipitate within 3-7 minutes, although some can take up to 20 minutes
TCICA Secondary Alchols
React fastest, within 15-30 seconds
TCICA Tertiary Alcohol
Are not oxidizable at room temperature and can produce no precipitate within an hour
TCICA Rxn
Look at paper.
Derivative
Unknown compounds can be reacted with various reagents to give new compounds called
Stable
________, crystalline derivatives can be used to help identify unknowns by comparing the derivative melting point with literature value.
Purity
For any derivative, _______ is more important than yield.
Small
Only a _________ amount of derivative is needed for melting point, but it must be pure to give accurate results
Several
______- derivatives can be made to confirm unknown alcohol.
nucleophic
Alcohol are only mildly _______, however, so a reactive electrophile is needed to form a solid derivative in a relatively short time period.
Tertiary Alchols
_____________ have significant steric hindrance that slows their rate of reaction.
Acidic
Alcohol can dehydrate under strongly __________ conditions.
3,5-dinitrobenzoate ester
The alcohol derivative that best balances these above considerations is the ___________.
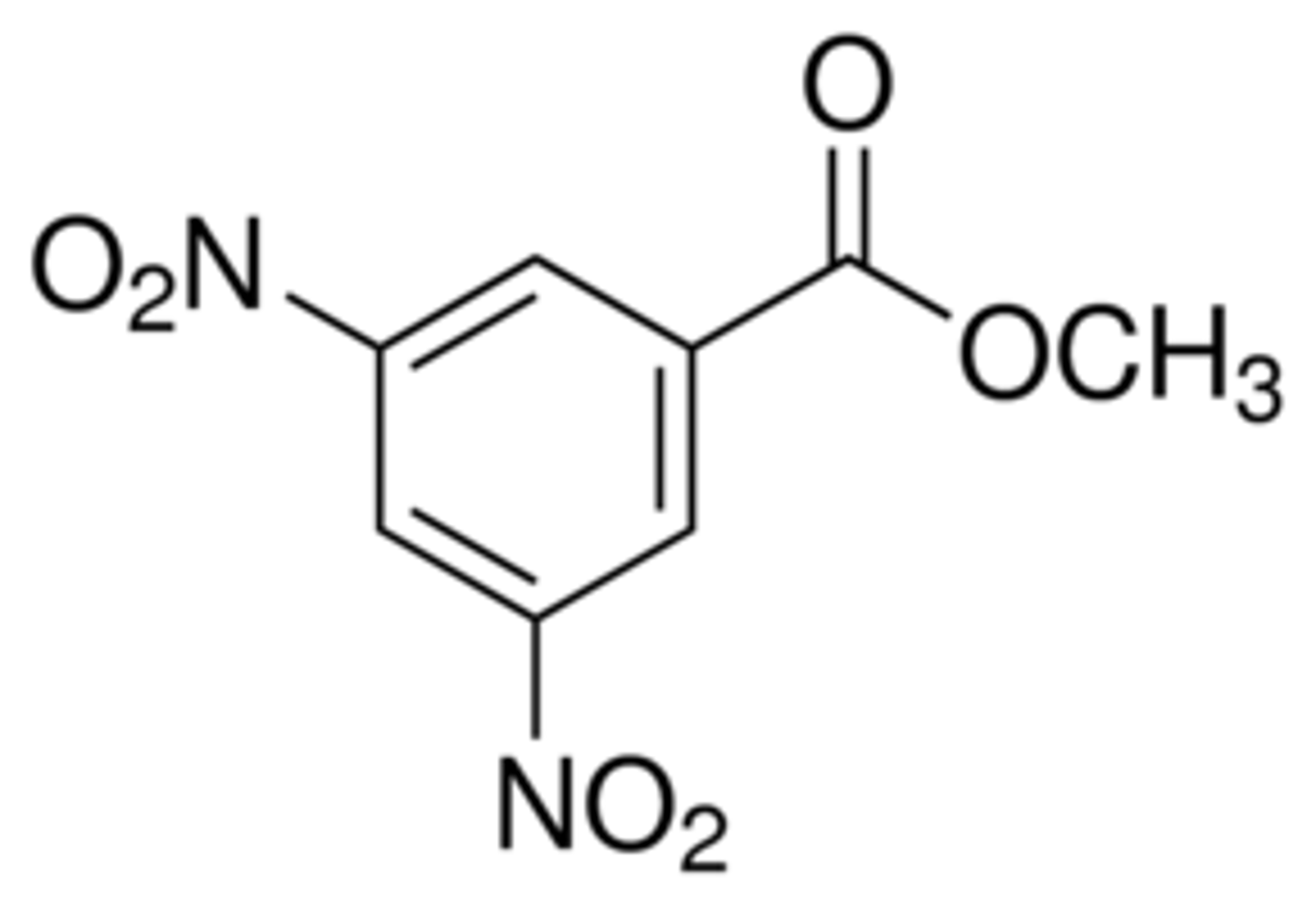
3,5-dinitrobenzoyl chloride
The 3,5-dinitrobenzoate ester is formed by the reaction of the alcohol and _________
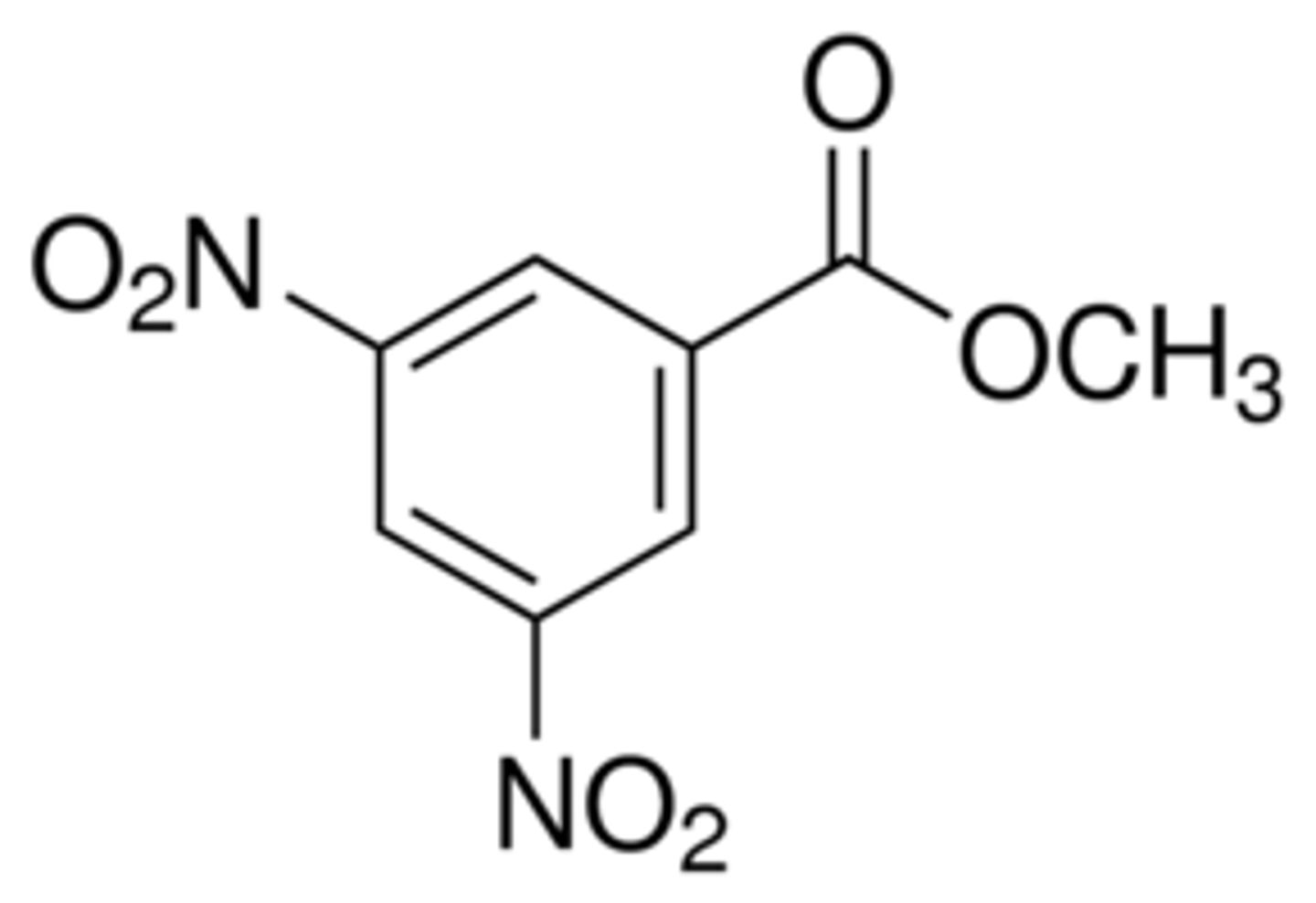
Derivative Reaction
Look on paper
4-Dimethylaminopyridine (DMAP)
For alcohol that are slow to react to create the derivative, particularly tertiary alcohol, a catalyst can be added. _______________ is an excellent acylation catalyst.
DMAP
has the added advantage of being a base, thereby removing the HCl as it is formed, preventing the acid-catalyzed dehydration of the alcohol.
DMAP Reaction
Look on Paper
As the lucas test involves ZnCL2 in concentrated HCl - Which is an extremely strong acid - there is a possibility of irritation. For this reason, lab coats, safety goggles, and gloves must be worn when handling this substance.
What precautions should be used when performing the lucas test?
Tertiary alcohol contain a carbon adjacent to a hydroxide carrying no hydrogens (rather there are only R groups). To oxidize an alcohol to carbonyl compounds, forming the C =O double bond, the removal of alpha H is required. In its absence, tertiary alcohol resist oxidation. Primary and secondary alcohol have these alpha H's readily available, so they are easily oxidizable.
Why are primary and secondary alcohol relatively easy to oxidize, but tertiary alcohol resist oxidation
Without qualitative tests, it is more difficult to know if alcohol is a mixture of primary, secondary, and tertiary or not. You don't take into account if the reagents aren't working properly as there is nothing to compare an unknown to.
What are the disadvantages of not performing the qualitative tests on known alcohols?
1. DMAP is a base required to abstract a proton from alcohol (R-OH) to form the product.
2. During the reaction, HCl formation takes place, hence DMAP is used for HCl Neutralization.
What are the two functions of DMAP?
This matches what secondary alcohol would react like. The Lucas test reacted in 1-3 minutes and TCICA test was immediate. This along with the fact that the melting point was around 114 makes me thing the unknown is cyclopentanol which is a secondary and melts at 115.
One of the Unknowns from table 1 gave the following results. Identify the unknown:
Lucas test: Cloudy in 2.5 minutes
TCICA Test: Precipitate in 20 seconds
3,5-dinitrobenzoate melting point: 113-114 C
Refractive index: 1.4535
Experimental Procedure:
1. Use 4 clean test tubes and add 2 ml of lucas reagent to each with 4 drops of respective alcohol
- Primary = 1- pentanol
- secondary = 2-pentanol
- Tertiary = 2-methyl-proponal
2. Watch RXN (Primary = No Rxn, Secondary = 2 min/ Tertiary =15 seconds)
3. Obtain 4 clean test tubes and add .5 mL of TCICA solution to each with one drop of HCl. Mix before adding one drop of the respective alcohol to each tube.
4. Watch Rxn (Primary = 6 minutes, secondary = 20 seconds, tertiary = no reaction)
5. Take 0.23 grams of 3,5-dinitrobenzol chloride into a test tube and add 8 drops of unknown. Place tube in hot water bath and observe. Let this cool, and break up derivative.
6. Add 5ml of sodium carbonate and mix for 30 seconds and filter and rinse with water. Transfer dried erivitive into a test tube and add 2 ml of 95% ethanol and a boiling chip. HEat.
7. Remove from hot water bath and let cool. Vaccum filter crystals and wash with cold ethanol.
8. Check melting point.
NMR
IR
Physical Properties
Functional Group Tests
Melting Points of Derivatives
Variety of techniques can be used to determine unknown alchols including
DMAP Advantages
1. Steric Hindrance slows the rate in tertiary alcohol so a Catalyst speeds up the reaction
2. DMAP is a better nucleophile than the alcohol which helps the rate of rxn.
3. since tertiary alcohol readily dehydrate - DMAP prevents the reaction from becoming acidic enough to dehydrate.