PDA III Local Anesthetics
1/34
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
35 Terms
Local anesthetics
-Drugs applied locally to block transmission of nerve impulses
-Intent is to produce loss of sensation in a limited area
-Actions are reversible and recovery is complete
Local anesthetic formulation
-Topical
-Injection (dental)
-Epidural or intrathecal injection
Peripheral primary afferent nociceptor
-An axon with two nerve endings
-Periphery: on the skin
-Primary: first in the neural pathway
-Afferent: carrying info to a center (spinal cord)
-Nociceptor: neuron that senses pain
-Ion channels open in response to acid, injury, heat, etc.
-Voltage gated Na channels propagate APs from peripheral nerve ending to other axon terminal (dorsal horn of the spinal cord)
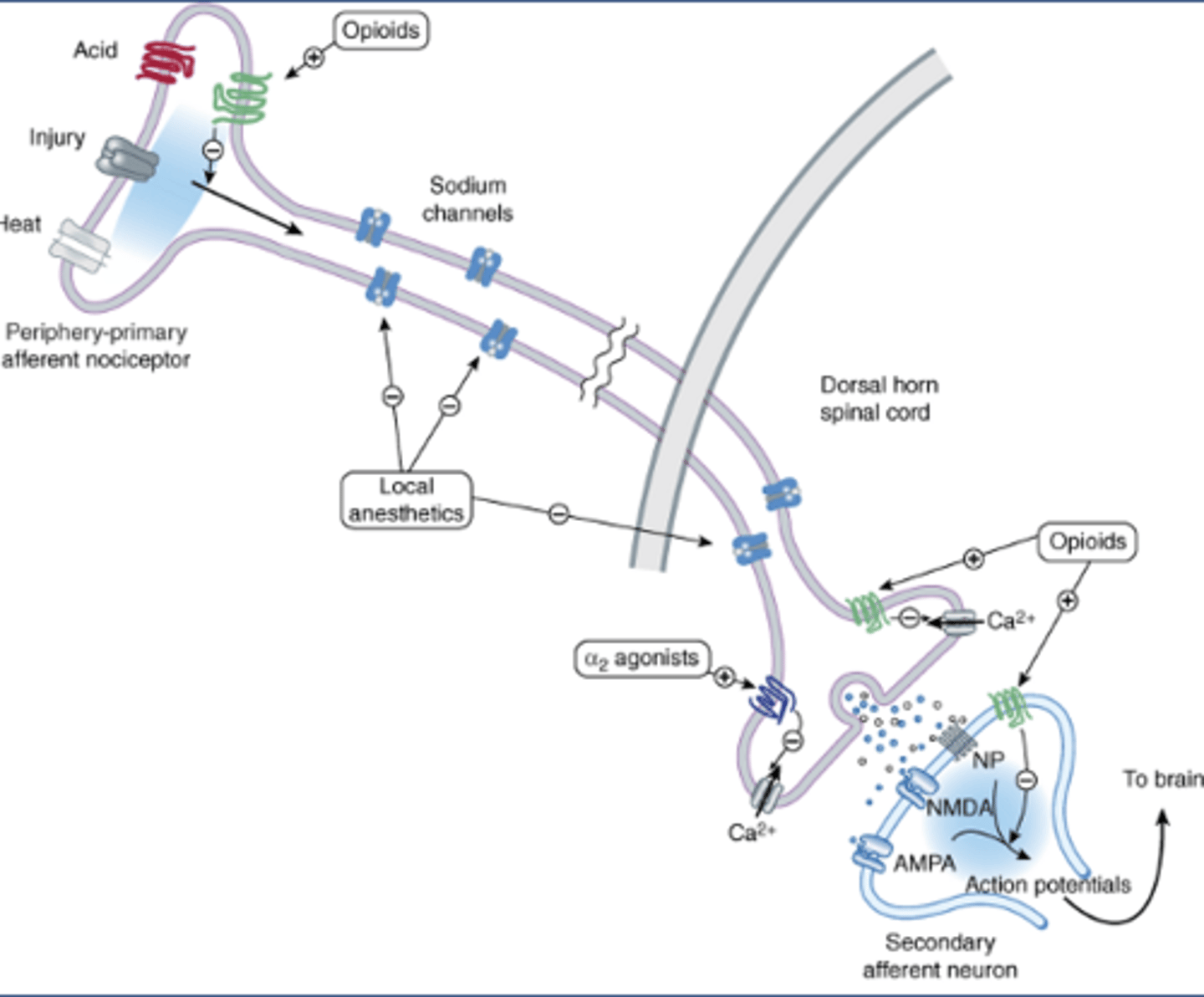
Local anesthetic MOA
-Peripheral-primary afferent nociceptor
-Inhibit voltage gated sodium channels
Na gated sodium channel review
-At rest: Na channels are in resting/closed can be opened conformation (top left)
-If voltage reaches threshold, intermediate closed conformation into open conformation (Na flows in for rising phase of AP)
-At +40 mV, Na channels are at inactivated conformation (refractory conformation/closed can't be opened): absolute refractory period
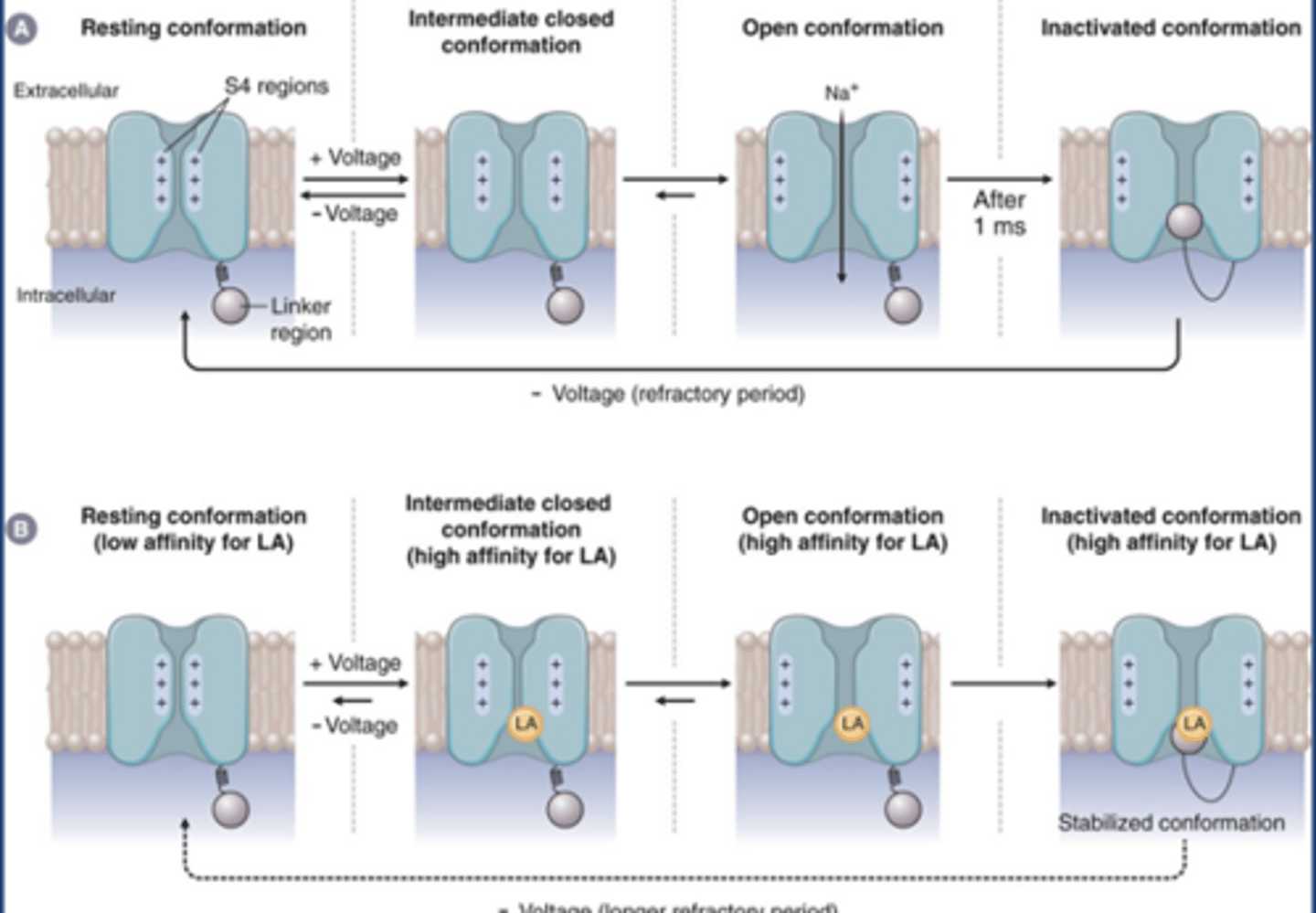
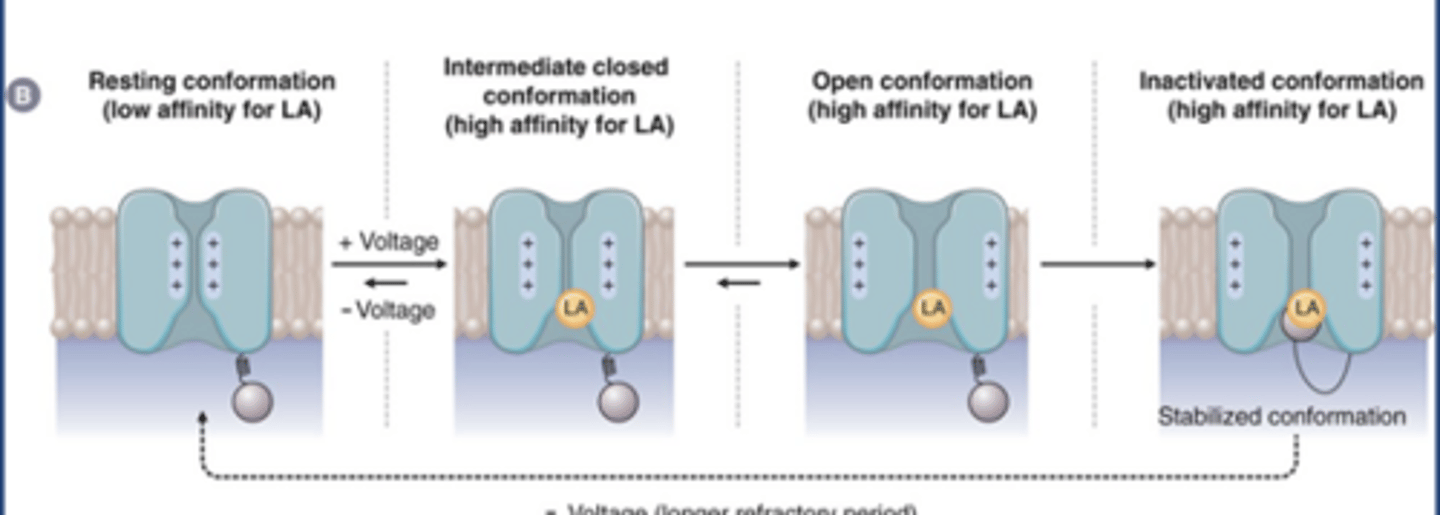
LA Na channel MOA
-LAs don't bind resting conformation (low affinity)
-LAs may bind intermediate closed conformation and open conformation
-LAs have highest affinity at the inactivated/closed can't be opened conformation
-Blocking the channel (gives slight effect)
-Stabilizes the channel in its inactivated form
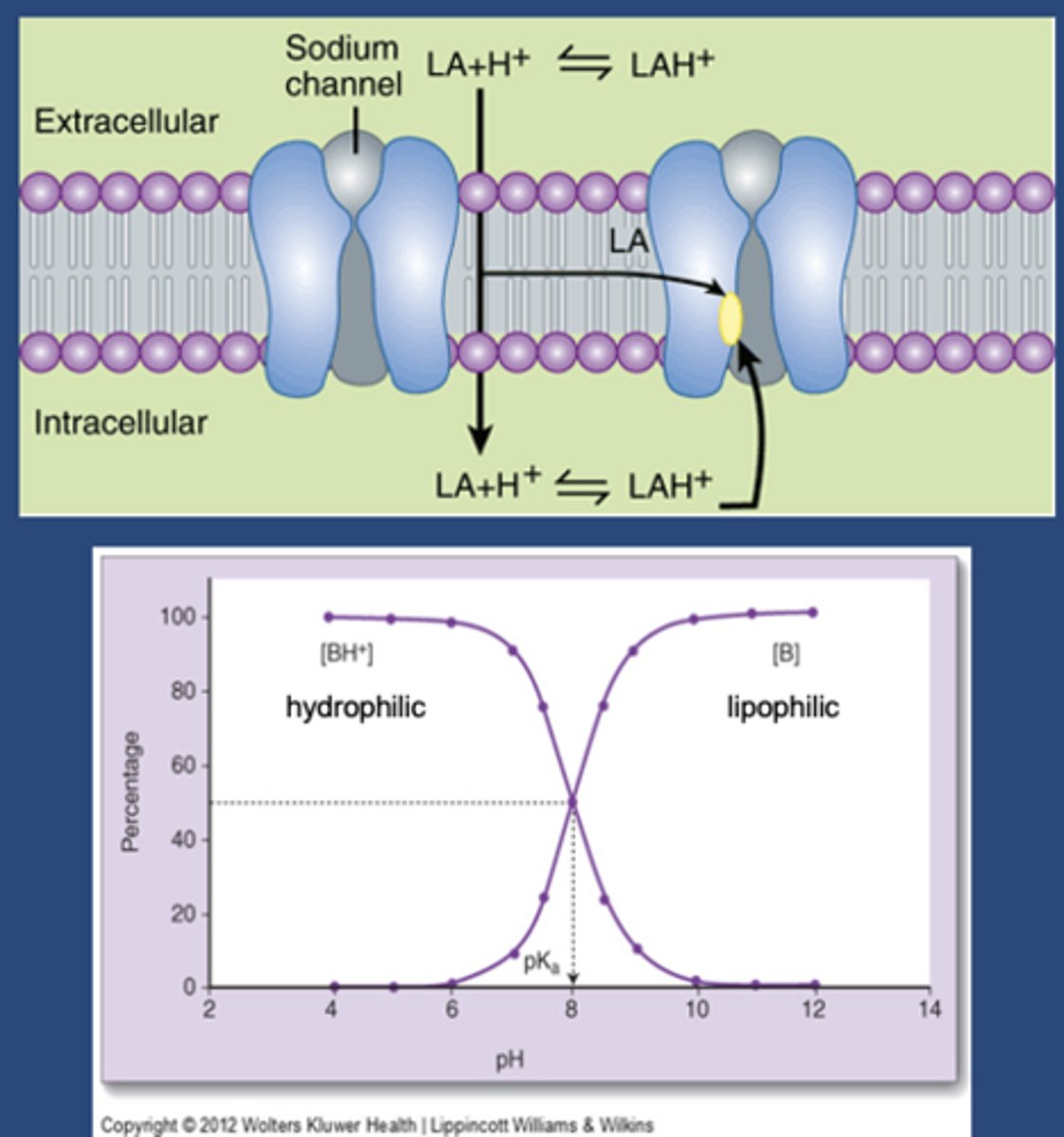
LA chemistry
-May be charged or uncharged (protonated or deprotonated)
-Weak bases
-pKa 8-10
-At 7.4, LA will be more charged than uncharged
-LA has to go through the lipid bilayer to get to its side of action (small minority of drug gets to site of action)
-Uncharged amount that gets in will make an equilibrium toward its charged form
-Charged form is the form that binds at its binding site
Cocaine
-Acts as a LA when rubbed on the skin
-Blocks sodium channels
-Other cocaine molecules are blocking NE reuptake, NE constricts blood vessels (specific to cocaine)
-Constricted blood vessels disallow the drug to be taken to other parts of the body (holds the cocaine to allow for a longer duration of action
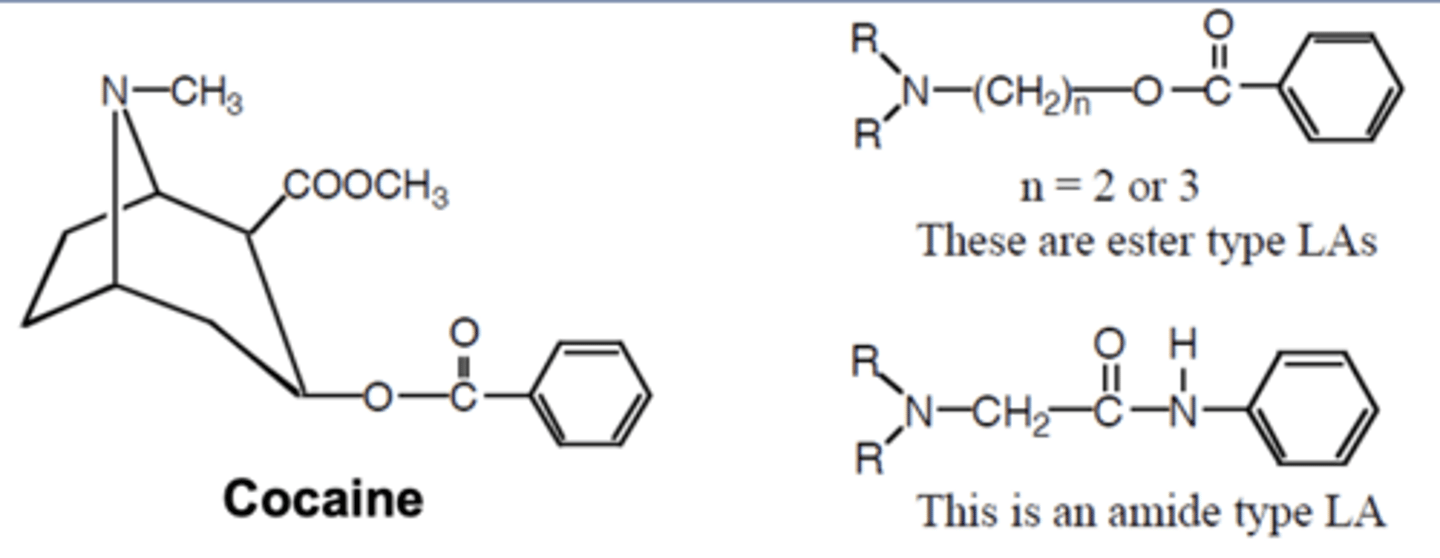
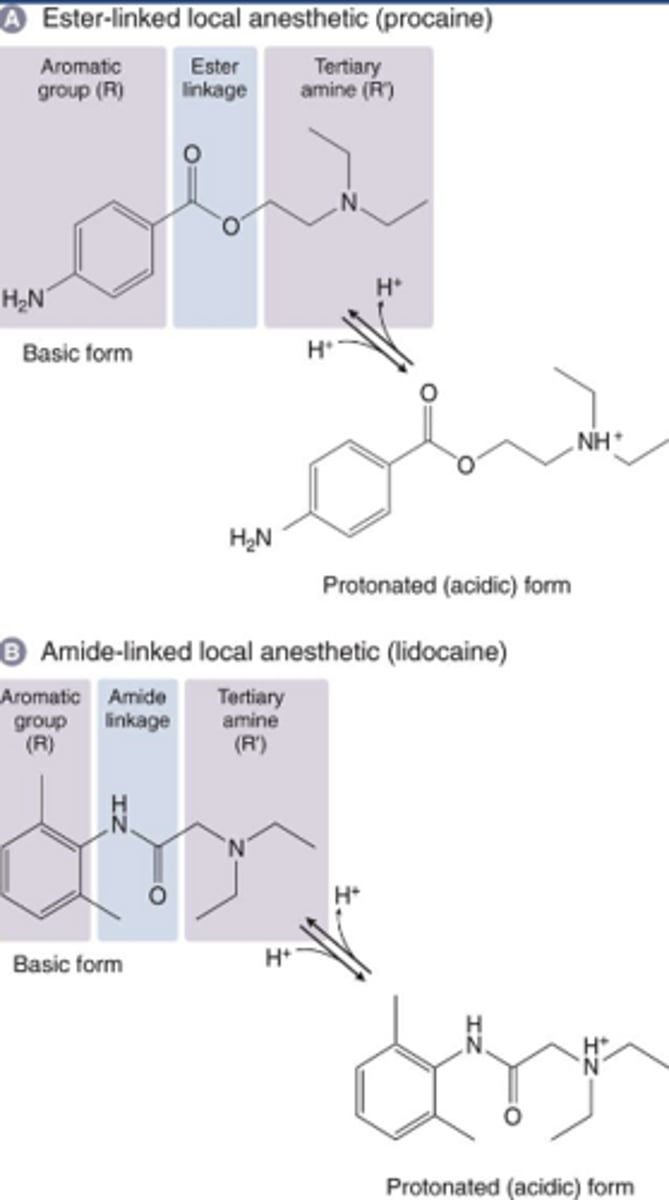
Cocaine chemistry
-Tertiary amine is hydrophilic
-Aromatic ring is homophobic
-Anesthetics may be ester or amide (note differences in structure)
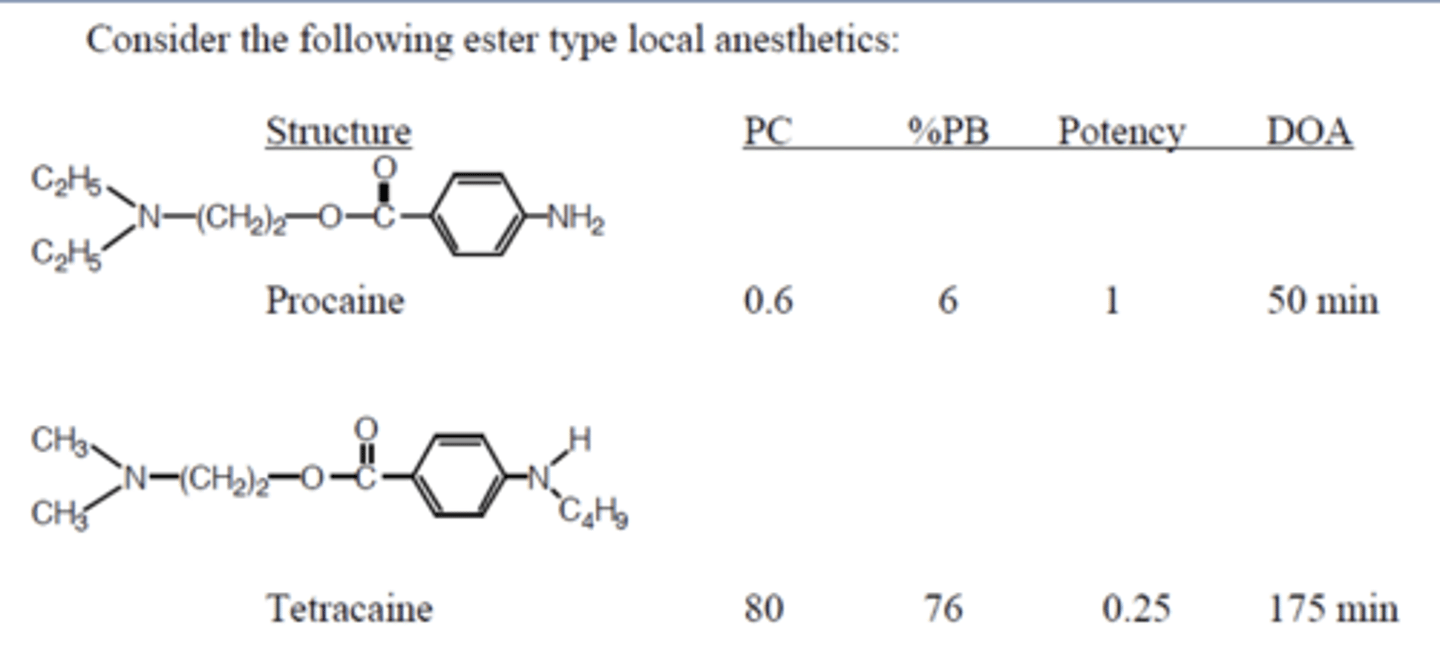
LA S-A-R
-Tetracaine more potent than procaine
-PC: partition coefficient
-Higher PC allows higher potency, protein binding, and duration of action (drugs stick to fat and proteins in the local area and stays there longer)
-True for both ester and amides
LA onset
-Depends on rate of diffusion which depends on ability to penetrate tissue (lipophilicity)
-More un-ionized form = quicker onset
-LAs are amines (weak bases) with a pKa of 8-10, so diffusion would be favored by alkaline conditions
-Changes with infected tissue
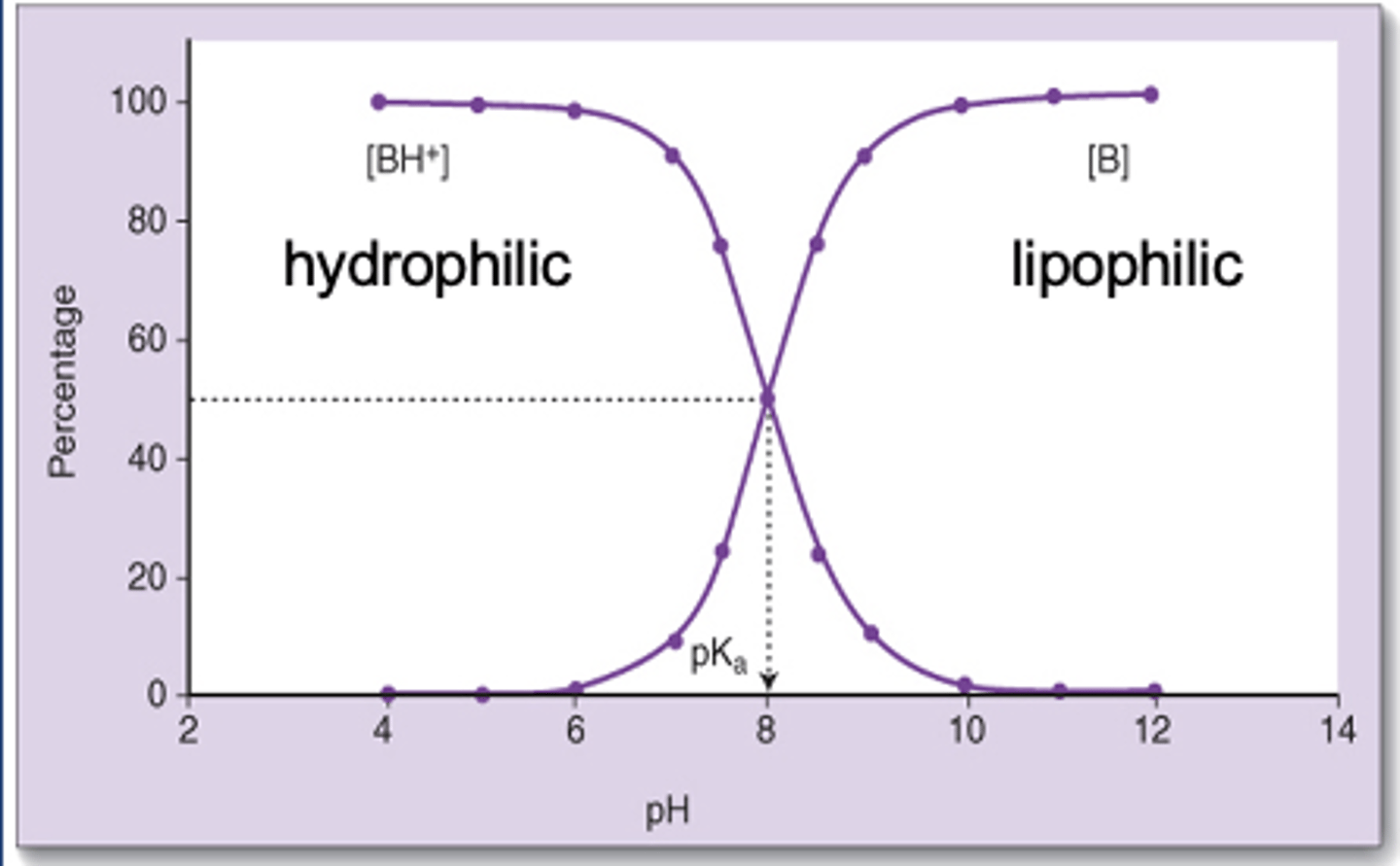
Infected tissue
-Lower pH, favoring LA's charged form
-LAs are less effective in infected tissue
-Bicarbonate (HCO3) can be added to the preparation and will raise the concentration of the unionized form and shorten onset block
Bicarbonate
-HCO3 can be added to the preparation and will rais
LA loss of effect
-Due to diffusion away from site of action
-Due to distribution away from site of action by the blood
LA duration of action
-Epi or phenylephrine: alpha 1 agonists increase duration of action by constricting blood vessels
-LAs tend to either vasodilate or have no effect on blood vessels (except cocaine and prilocaine)
-LAs with high PC are highly tissue bound: longer DOA
LA metabolism
-Doesn't play a role in DOA
-May be important upon systemic absorption
-Different for esters and amides
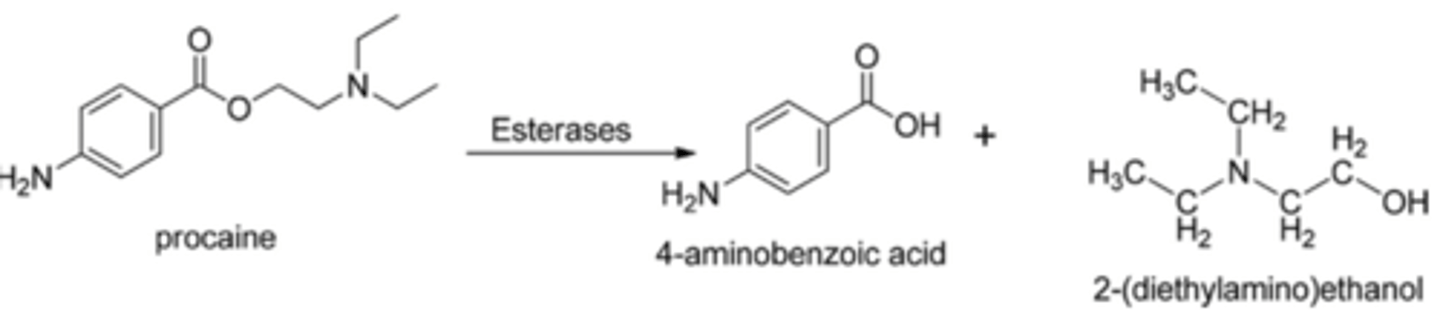
Ester metabolism
-Hydrolyzed by plasma esterases
-Faster hydrolysis means less systemic toxicity
-Metabolite of procaine is PABA and can cause allergic reactions
Amide metabolism
-Metabolized by liver enzymes
-Extremely rare to be allergic to these drugs or their metabolites
-If there is a reaction, check the preservatives first
LA systemic toxicity
-CNS effects
-CV effects
-Absorption plays a role
LA role of absorption in systemic toxicity
-Absorption of the drug into systemic circulation leads to toxicities
-We can limit absorption with a vasoconstrictor
-Esters are less toxic than amides because they're metabolized faster; amides require passage through liver
-If the LA is highly protein bound, this can limit free conc in the plasma
CNS systemic toxicities
-Tremor
-Nervousness
-Seizures followed by coma and respiratory depression
CV systemic toxicities
-Decreased excitability and force of contraction
-Vasodilation
Systemic toxicity treatment
-No direct pharmacological antidote
-Supportive measures: maintain airway and ventilation and BP
-Antiseizure meds
LA clinical uses
-Infiltration anesthesia
-IV regional anesthesia
-Peripheral nerve blockade
-Central neural blockade
-Topical anesthesia
Infiltration anesthesia
-Intradermal and subq injections
-Topical anesthetics are a subtype of infiltration anesthesia
IV regional anesthesia
-Rarely used due to toxicity risk
-Into a vein in a limb
-Used with tourniquet-occluded limb
Peripheral nerve blockade
-Single or multiple nerves
-Injected near nerve bundle
Central neural blockade
-Epidural or spinal (intrathecal)
Ester LAs and their durations of action
-Cocaine
-Procaine
-Chloro-procaine
-All 3 are short acting
Amide LAs and their durations of action
-Lidocaine: intermediate acting
-Mepivicaine: intermediate
-Prilocaine: intermediate
-Bupivacaine: long acting
-Etidocaine: long
-Tetracaine: long
Short acting
<60 mins
Intermediate acting
90-120 minute
Long acting
>180 min
Benzocaine
-Lacks aliphatic amino group
-Water insoluble, hydrophobic, and lipophilic: very low systemic absorption
-Used topically on open sores and wounds
EMLA
-Eutectic mixture of LAs (lidocaine and prilocaine)
-Applied to skin and covered with a dressing
-Produces anesthesia in 30-60 mins and lasts 1-2 hours