Radioactivity part 2
1/16
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
17 Terms
Ways to detect ionising radiation?
Photographuc film
Geiger-Müller tube
How can photographic film detect radiation?
By becoming darker when they absorb radiation, similar to when it absorbs visible light
The more radiation it absorbs, the darker it is when it is developed
How do people who work with radiation monitor the levels of radiation absorbed?
They wear photographic film badges which are checked regularly
If the film becomes fogged, it shows that the scientist has been exposed to a certain amount of radiation
Goes darker when it absorbs radiation — more radiation = darker
To get an accurate measure of the dose received, the badge contains different materials that the radiation must penetrate to reach the film
These materials may include aluminium, copper, paper, lead and plastic
The Geiger-Müller tube is the most common device to measure and detect radiation.
How does it work? What is an important factor that affects this device?
Each time it absorbs radiation, it transmits an electrical pulse to a counting machine
This makes a clicking sound or displays the count rate
The greater the frequency of clicks, or the higher the count rate, the more radiation the Geiger-Müller tube is absorbing
Because it shows the frequency it matters how close the tube is to the radiation source.
The further away from the source, the lower the count rate detected
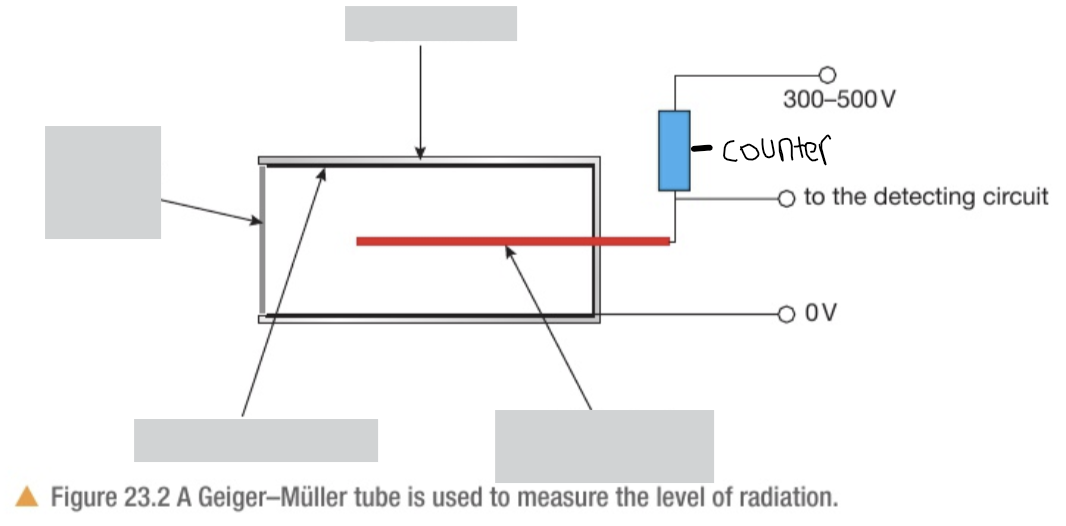
Label the GM tube. Describe the contents inside and the structure
Glass tube with an electrically conducting surface on the inside at 0V
Inside tube has a mixture of inert gases at low pressures
The anode is connected via a high value resistor to a high voltage supply, typically 300-500V
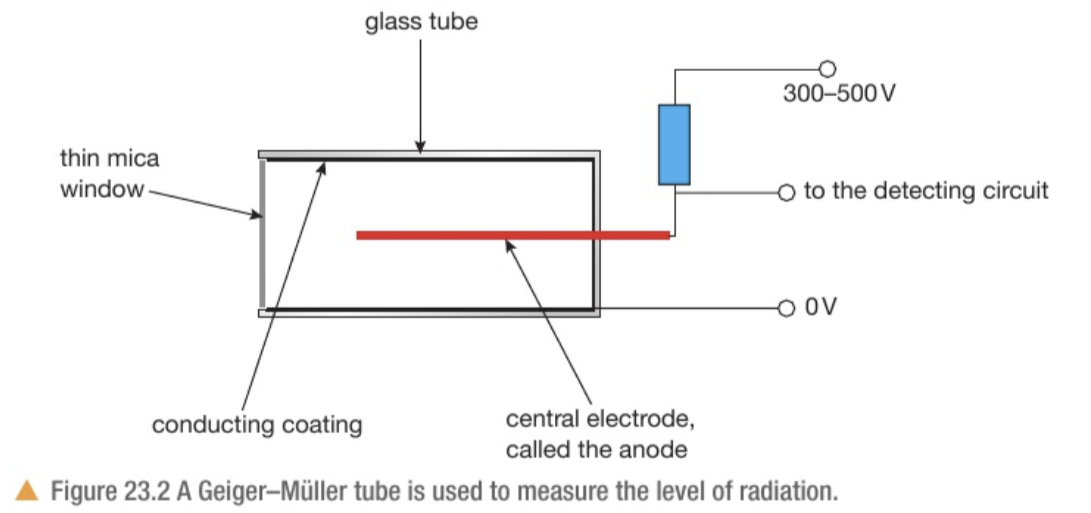
Explain how a GM tube works to measure radiation
The tube contains a mixture of inert gases at very low pressure
The inside of the tube is lined with a conducting coating at 0V
There is an anode in the middle of the tube connected, via a high value resistor, to a high voltage supply typically 300-500V
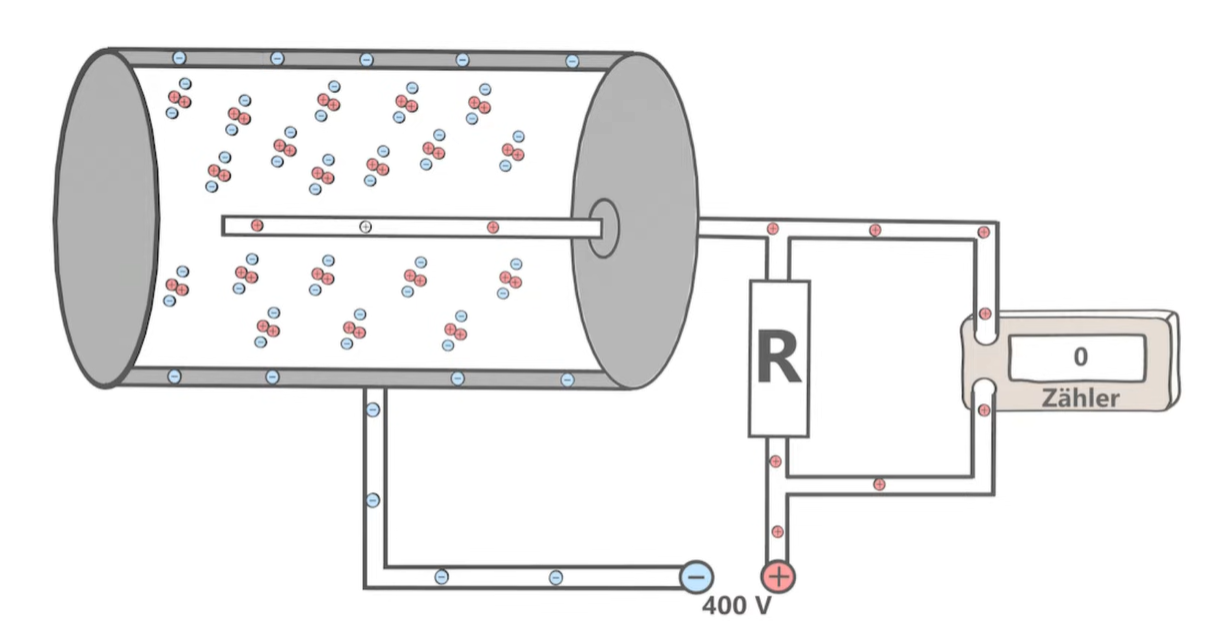
When ionising radiation enters the tube, it ionises the atoms/molecules of the gases, forming ions by kicking out the electron
As the electron is attracted to the positive anode, in its way there it will create an electron avalanche which creates a small current that flows into a counting machine
The counting machine displays a count rate. The higher the count rate the more radiation the tube is absorbing
Meanwhile, the positive ions are attracted to the 0V inner metal coating (it acts as a cathode)
The coating closes the circuit as the current returns to the cathode from the counting machine, neutralising the gas particles
What can we use instead of a counter? How are these different?
Rate meters
These measure the number of ionising events per second, and so give a measure of the radioactivity in becquereles
They ususally have a loudspeaker output so the level of radioactivity is indicated by the rate of clicks produced
Why is a high voltage supply needed? Why is a high value resistor needed? Why do the gases have to be at low pressures?
Voltage connected to anode causes it to be positively charged and a high V causes e- to accelerate fast enough to cause an avalanche to produce a current (the cathode, metal casing is negatively charged as it is connected to other terminal)
Needed so the tube can reset and detect the next current separately
Low pressure means atoms are further apart so e- can travel long enough between collisions to gain energy and produce the avalanche. At high pressure, e- would collide too often
How can the accuracy of results when measuring count rates be improved?
Repeating readings and taking averages
Taking readings over a long period of time
What is background radiation?
It is low level ionising radiation that is produced all the time. It has a number of sources and some are natural (cosmic rays from space, rocks, foods) while some are artificial
Describe the natural sources of background radiation
From rocks and buildings (the earth) which release radioactive gases of radon and thoron
From space which produces cosmic rays
Radiation in living things
Radon and thoron gas.
How/Where are they formed?
What type of radiation does it emit?
Why is radon gas only detected by a Geiger counter?
Why does the amount of background radiation produced in this way varies from place ot place?
Radon gas from rocks in the Earth’s crust and buildings
An isotope of uranium is a radioactive element that occurs naturally in all rocks and soils and decays very slowly and two of its decay products are gases: radon and thoron
These gases come out of radioactive rocks in the Earth’s crust. They are dense gases so they build up in the basements of buildings
Radon gas is an alpha emitter
Because radon gas is tasteless, colourless and odourless
Becuase some parts of the Earth’s crust have higher amounts of radioactive material
Background radiation from space
How/Where do they release radiation? Type of radiation? How are we protected?
Nuclear reactions (creo q es nuclear fusion) in stars and supernovae produce cosmic rays (very energetic particles) that hit the Earth
Lower energy cosmic rays are given out by the Sun
When they collide with molecules in the air, this leads to gamma radiation
Our atmosphere gives us protection from cosmic rays, but some still reach the Earth’s surface
Background radiation from living things (food and drink)
Atoms that make up our bodies were formed in the nuclear reactions that take place in supernovae. Some of these atoms are radioactive
As we breathe we take in the radioactive isotope of cabron, carbon-14.
Carbon-14 and other radioactive isotopes are eaten by humans (and animals) because they are present in all living things and naturally occurring radioactive elements can get into food and water we eat since they are in contact with rocks and soils containing these elements
Artificial radiation sources
Leaks of radioactive material from nuclear power stations
Testing nuclear weapons in the atmosphere
Radioactive tracers used in industry and medicine
Radiation theraoy and X-rays
Are artificial or natural sources of radiaiton the most significant contributors?
Natural sources:
Radon gas from rocks and buildings
Food and drink
Cosmic rays
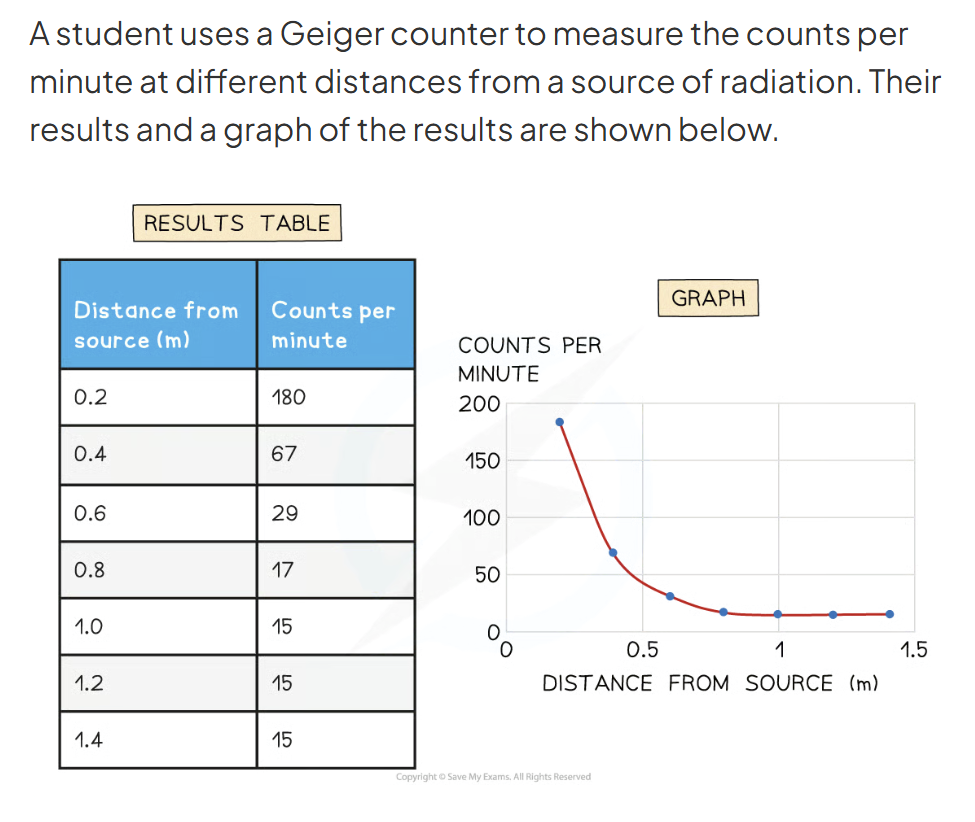
Determine the background radiation count. Explain why
