MCAT chem/phys
1/58
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
59 Terms
Give the definition for enantiomers. What is there R/S configuration?
non-superimposable mirror images that canNOT appear identical with rotation.
>1 chiral center
NO internal plane of symmetry
opposite R/S configuration
give the definition for diastereomers.
Non-super imposable MIRROR images, NOT mirror images
>2 chiral centers
some opposite and some same R/S configuration
what is the difference between constitutional isomers and steroisomers?
Consitutional isomers have DIFFERENT connectivity
stereoisomers have the same connectivity but DIFFERENT spatial arrangements
They both have the same molecular formula.
What are the differences between diastereomers and enantiomers?
Enantiomers are mirror images
enantiomers have all opposite R/S configurations at chiral centers. diastereomers have some same, some different.
For Poiseuille’s Law give the relationship between flow rate and each of the variables (how do changes in each variable effect Q)
Q is proportional to r^4 (double r= increase Q by 16x)
Q=P, more pressure difference = more flow
Q=1/viscosity (double viscosity = ½ flow)
Q= 1/L (double length = ½ flow)
what is the difference between heat capacity and specific heat?
heat capacity: the amount of heat required to raise the temperature of an object by 1 deg C
specific heat: the amount of energy required to raise the temperature of 1 gram of the substance by 1 deg C
What is hyperopia and what causes it? Where is the original image? What lens is used to fix this and how does it work?
hyperopia = farsighted
optical power is not sufficient → image is BEHIND retina
tx w/ converging/convex lens → focal point shifted toward the retina
What is myopia and what causes it? Where is the original image? What lens is used to fix this and how does it work?
myopia = nearsighted
focal point is in FRONT of retina
tx with diverging/concave lens → shift image away from lens
converging/convex lenses create what type of image?
real and inverted
diverging/concave lenses create what type of image?
virtual and upright
what type of wave is light?
a transverse electromagnetic wave
what does it mean for a wave to be “transverse”?
the wave oscillates perpendicular to the direction of travel
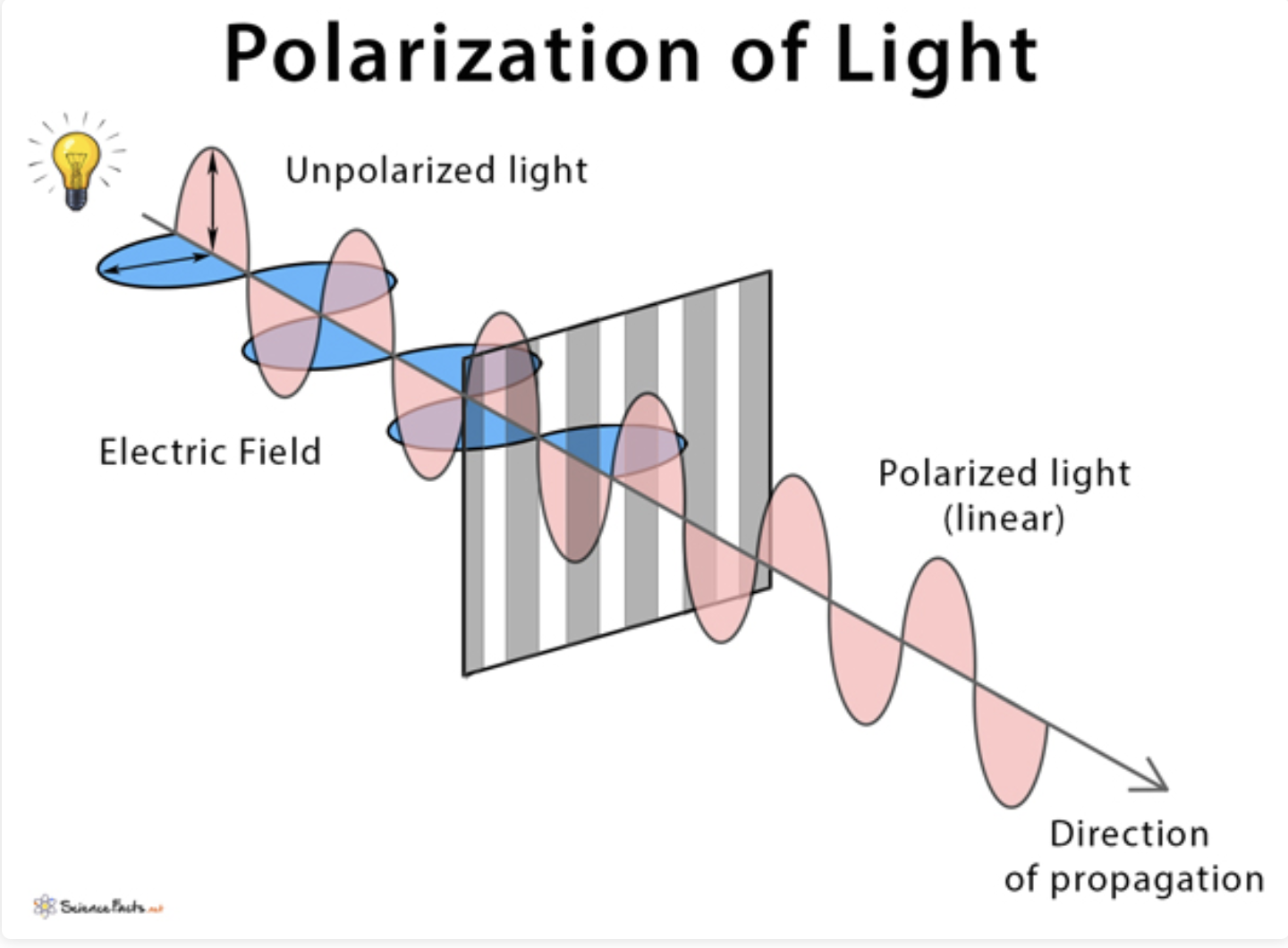
give a one-sentence definition of polarization:
restricting in the electric field of a transverse wave to one direction (unpolarized light usually goes in many directions)
one direction of oscillation
why can light waves be polarized but not sound waves?
sounds waves are longitudinal, light waves are transverse
what 2 fields make up light?
magnetic field (B) and electric field (E)
which field of light determine polarization?
E (electric) field
what does a linear polarization filter do to light?
allows for transmission of electromagnetic radiation whose E field is parallel to the axis of polarization.
inhibits passage of radation whose E field is perpendicular to this axis
what happens to the intensity (I) of unpolarized light if it passes through ONE polarizer?
Intensity becomes ½ of its original
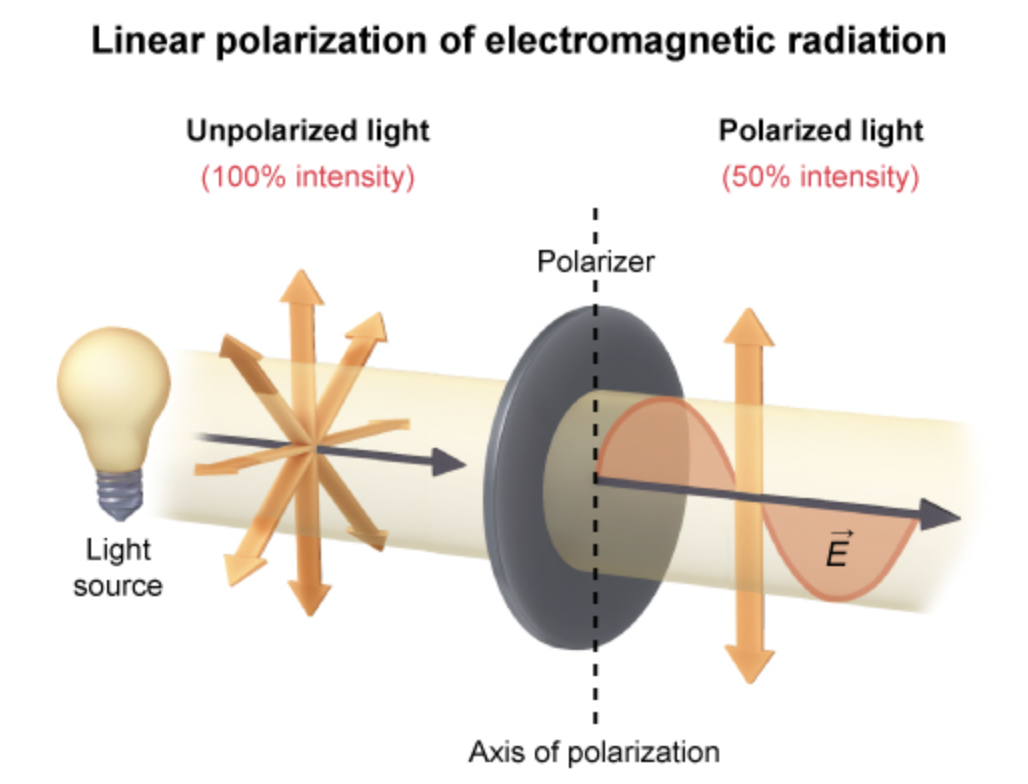
what happens if light hits 2 polarizers at 90 degrees?
NO light passes
what does polarization ALWAYS refer to?
direction of E field
In normal TLC the stationary phase is ______ and the mobile phase is _______
stationary: polar (silical gel, alumina)
mobile phase: nonpolar (hexanes, ethyl acetate)
In normal TLC:
nonpolar compounds will react more strongly with the stationary/mobile phase and procees closer to/farther from the solvent front.
polar compounds will react more strongly with the stationary/mobile phase and proceed closer to/farther from the solvent front.
nonpolar compounds react more w/ the mobile phase because it is nonpolar (like dissolves like) and therefore proceeds closer to the solvent front.
polar compounds react more w/ the stationary phase because it is polar and therefore be farther from the solvent front
which types of compounds (polar/nonpolar) will have a larger Rf in TLC?
nonpolar, because they interact more with the nonpolar mobile phase and move farther down the plate towards the solvent front
What are the 5 steps for balancing a chemical equation? (non redox)
Determine what rxn is occuring (reactants, products, physical states, etc.)
Write unbalanced equation
Balance 1 atom at a time: Start with substance with most elements present
Small whole # coefficients: resolve fractional coefficients by multiplying by a factor
Check each atom to ensure equation is balanced.
What are the steps for balancing a redox rxn chemical equation?
Separate rxns (ox/red): Divide rxn into oxidation and reduction rxns
Balance elements: other than O or H first. Then add H2O or H+ as needed (Double check to see if rxn is occuring under ACIDIC (H+) or BASIC (OH-) conditions)
Balance charge: Add e- to balance charge. Multiply each half-equation by an integer so the number of electrons in each equation is equal. Make sure you multiply every term in the equation by this integer.
Combine half rxns: Add the two half equations together. Cancel and simplify terms that appear on each side
What is the avg molecular wt. of an AA? How can you calculate the molecular wt of a polypeptide?
Avg wt. AA = 110 kDa
Avg wt. polypeptide = # AAs x 110 kDa
What is the oxidation # for Group 17 (halogens)?
-1 unless bonded to a more EN atom (ex: Cl= +1 when HOCl)
What are the oxidation #s for Group 1 metals (alkali metals) and Group 2 metals (alkaline earth metals)?
Group 1 (alkali): +1
Group 2 (alkaline earth): +2
What are the oxidation #’s of Ag, Cd, Zn, Ga, and Al?
Ag = +1
Cd = +2
Zn = +2
Ga = +3
Al = +3
What is the oxidation # for H?
+1 UNLESS bonded to < EN metals from group 1 or 2 (ex: H= -1 when NaH)
What is the oxidation # for O?
2-
what are the 4 steps for IUPAC naming?
Select parent chain
Number the chain based on priority of fxnl groups
Name substituents
Assign numbers to substituents
Complete names
How do you select the parent chain when IUPAC naming? What do you do if there is a tie between options?
longest C chain = parent chain
IF TIE → chain with greatest # of substituents wins
How do you number the substituents on the parent chain when IUPAC naming? (hint: which # should the highest priority fxnl group have)
Highest priority fxnl group = LOWEST possible #
If subs have = priority → all as low # as possible
From highest to lowest priority, list the priority of all fxnl groups when following IUPAC naming.
COOH > anhydrides > esters > amides > aldehydes > ketones > alcohols > alkenes > alkynes > alkanes
How do you name C substituents off of the parent chain? what do you do if there are multiple substitutents of the same type?
Prefix (meth/eth/prop/but/etc.) + -yl
multiple subs of same type → use prefixes (di-, tri-, etc.)
List the prefixes and suffixes for all functional groups from highest to lowest priority.
COOH: carboxy—, —oic acid
anhydride: alkanoyloxycarbonyl—, anhydride
ester: alkoxycarbonyl—, —oate
amide: carbonoyl—/amino—, —amide
aldehyde: oxo—, —al
ketone: oxo—/keto—, —one
alcohol: hydroxy—, —ol
alkene: alkenyl—, —ene
alkyne: alkynyl—, —yne
alkane: alkyl—, —ane
Give the approximate equivalents for the following fractions:
3/2, 9/2, 1/5
3/2 = 1.5
9/2 = 4.5
1/5 = 0.2
Elements with > ionization energy typically have > EN except for which 3 elements?
He, Ne, Ar
when a metal is binding into coordinate covalent bond, which orbital do the non-metal e- bind into?
non-metal e- are binding into free d orbital
If given the known mass of each element in a compound, how can you determine the empirical formula? (2 steps)
determine the # of moles of each element (by dividing the known mass by its molar mass)
Divide the each molar amount by the smallest molar amount to find the relative ratios of each type of atom
How do you find the molecular formula of an element when given the empiricial formula and the molecular wt?
Find the molecular wt of the empirical formula then multiply the each element in the empirical formula until the molecular wt matches that of the problem.
ex: if the MW is double that of the given wt of the empirical formula, you must double the number of each of the elements to find the molecular formula
Provide the resonance frequency and characteristics on IR spectroscopy for carbonyls, nitriles, carboxylic acids, Amines/amides, and alcohols?
Carbonyl (C=O): 1700, sharp and deep
nitriles (CN, triple bond): 2250, sharp and deep
COOH (OH*): 3000, broad
Amine/Amide (NH): 3300, BOTH broad
amine → shallow
amide → deep
alcohol (OH): 3300, broad
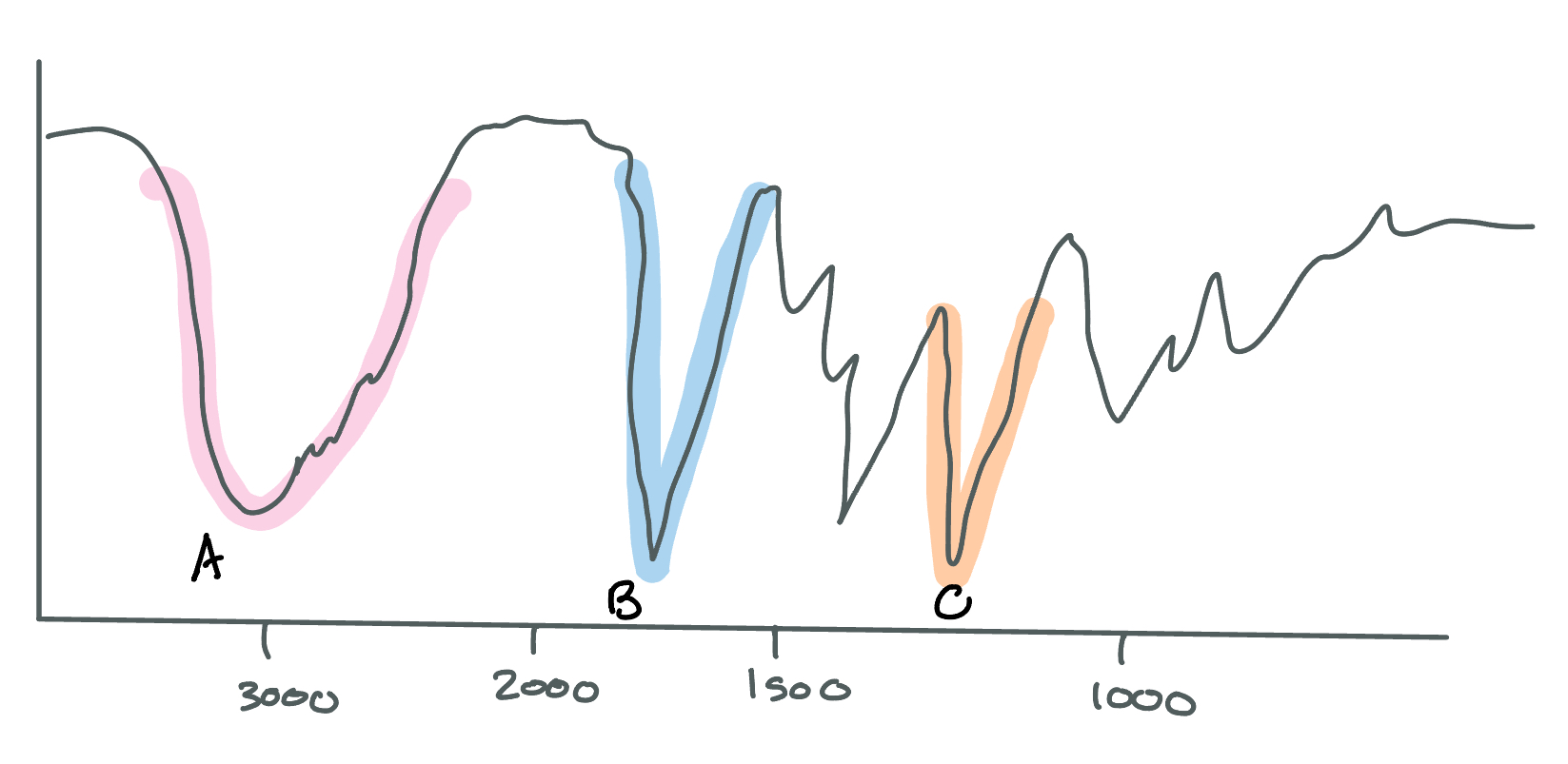
What type of bonds are shown at locations A and B on this IR spectroscopy?
A: O-H in a COOH
3000
broad
B: C=O
1700
sharp and deep
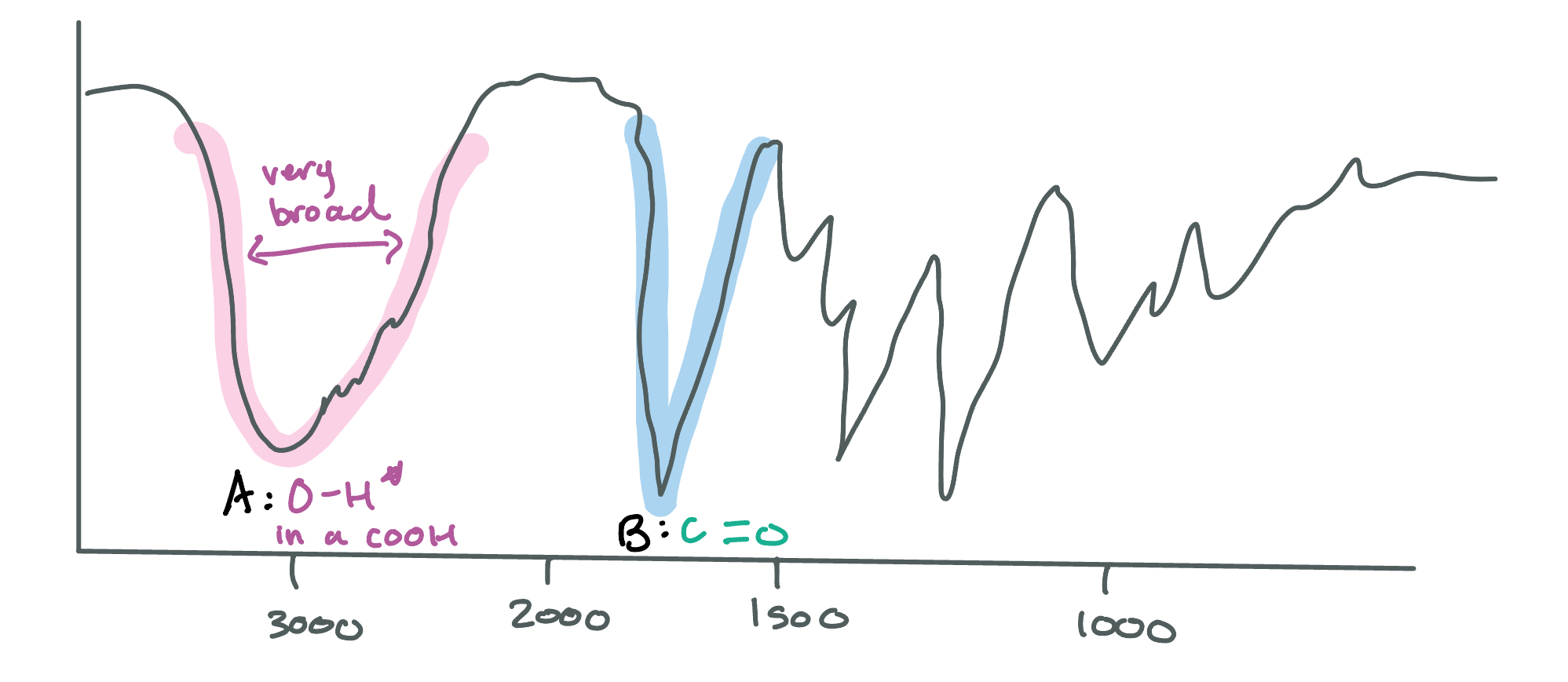
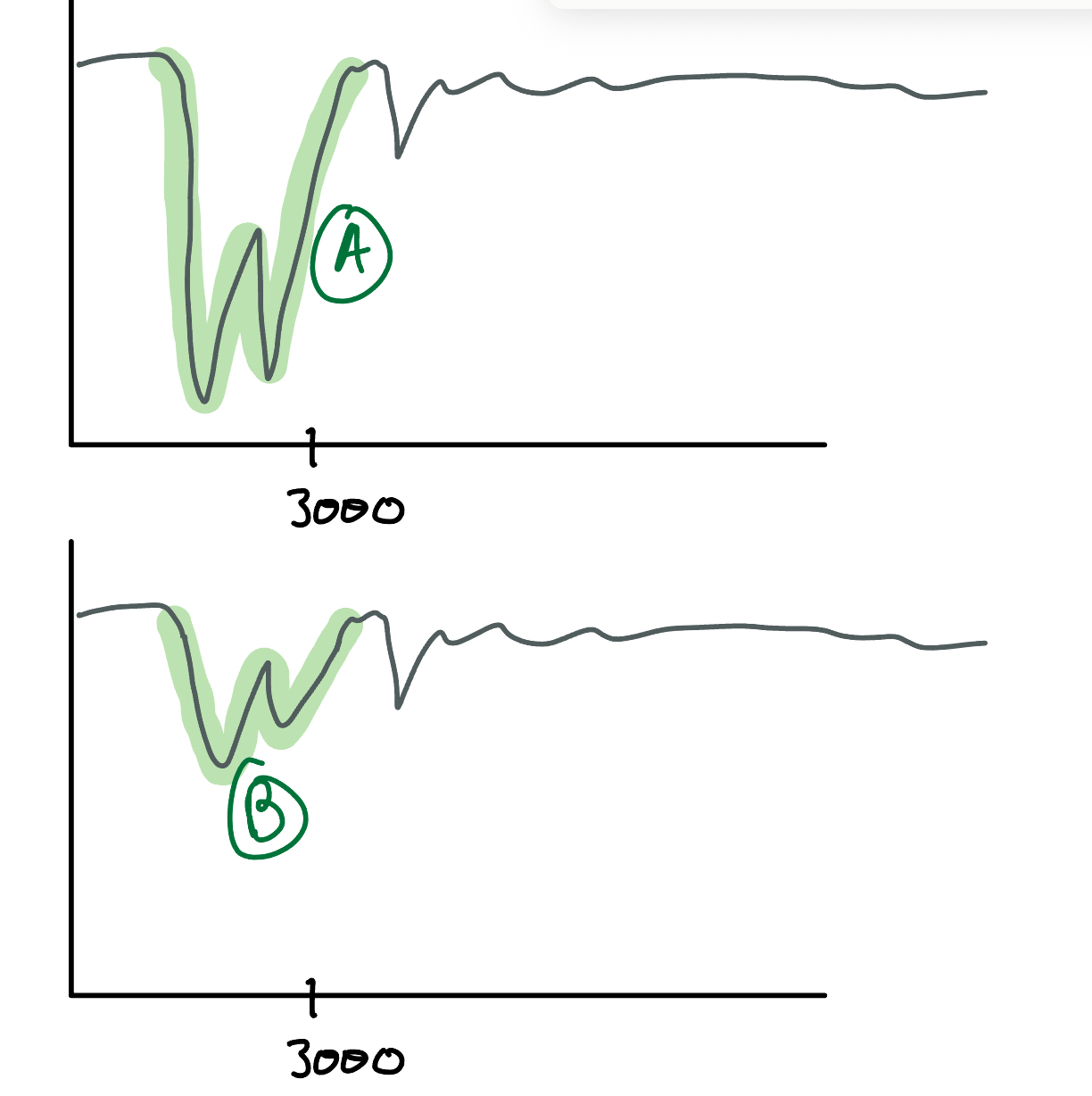
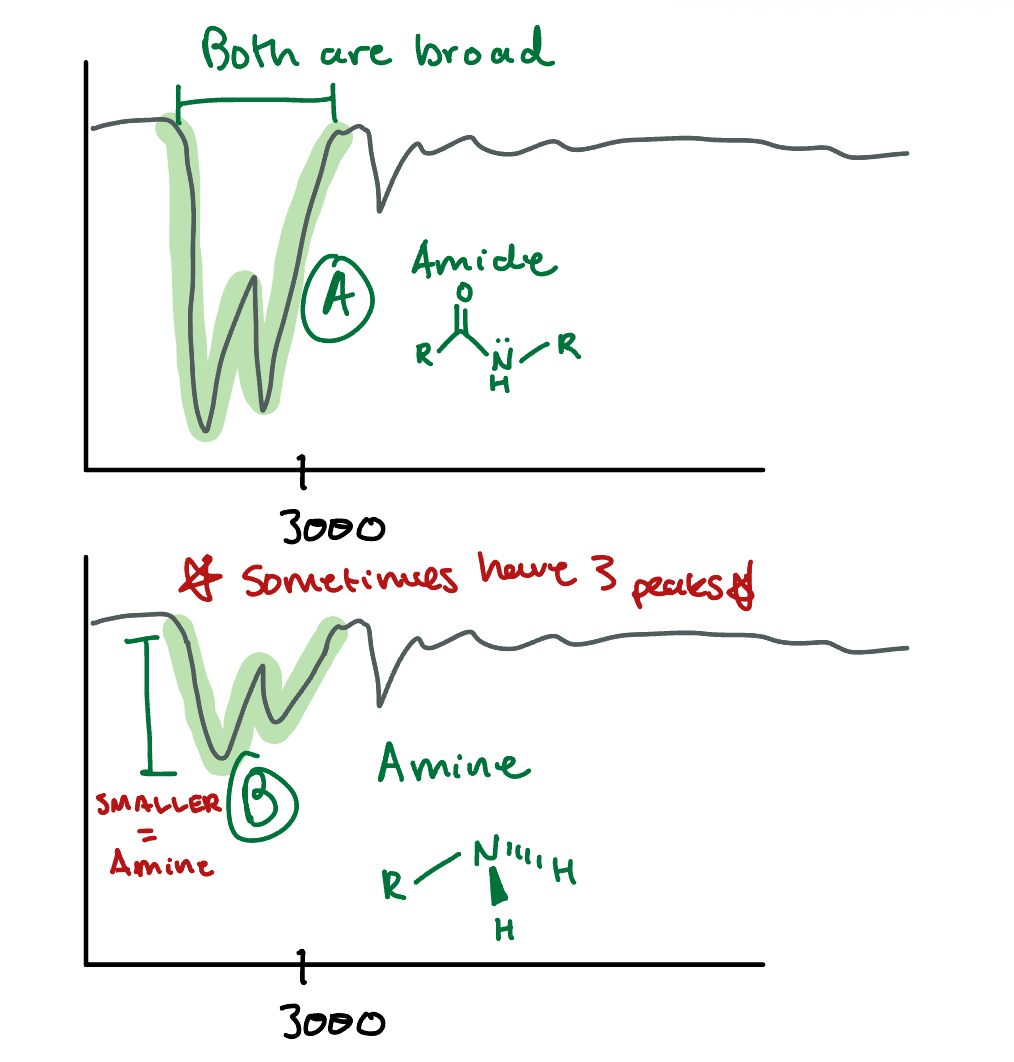
Identify which peak (A or B) in this IR spectroscopy is an amide and which one is an amine. How do you know?
NH Bond: Amide/Amines, 3300, Broad and looks like a W
A: AMIDE because the peaks are DEEP
B: AMINE, same thing but shallow
**DO not confuse these with CH bonds, CH bonds are at a lower frequency than 3300**
What are the 2 types of gel electrophoresis? How do they differ in separating particles?
Native Gel: separates by charge AND size
SDS Page: separates by size ONLY; molecules are coated in NEG charge so everything is the same charge **NOTE: everything is denatured in SDS page**
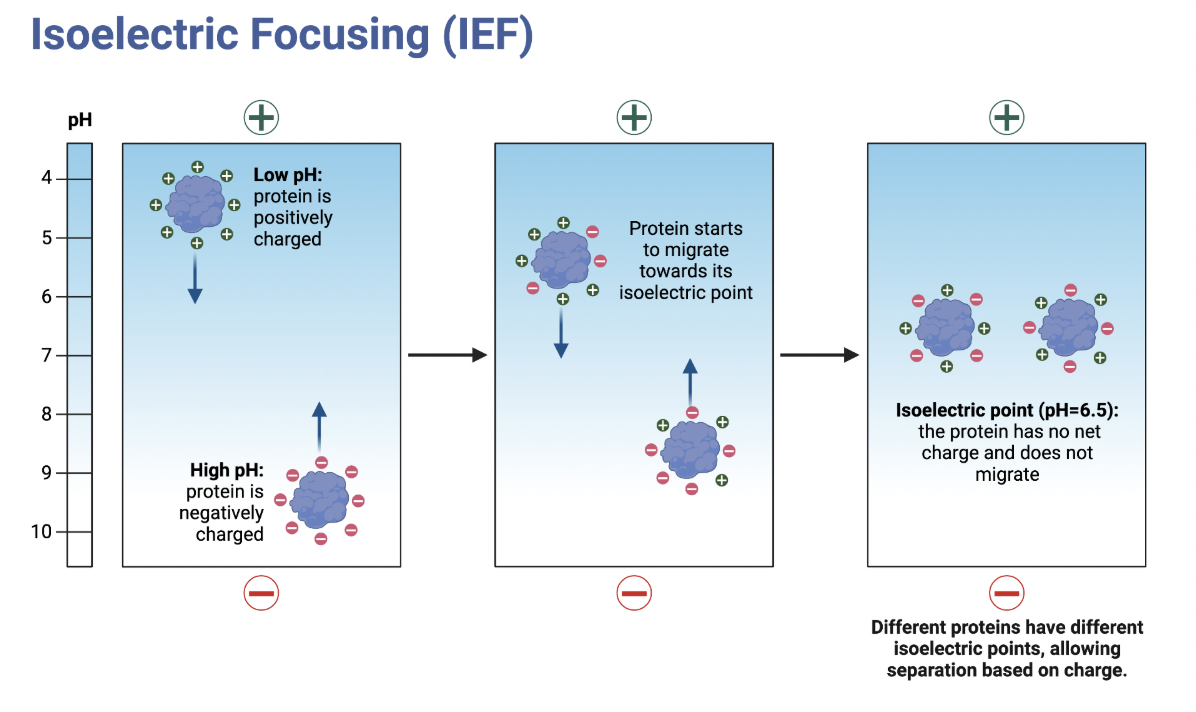
What is isoelectric focusing electrophoresis and how does it work?
separates proteins by pH using their charge
The gel has a gradient of different pHs.
Proteins initially move up/down gel based on charge, but stop when they reach their Ip because their charge is 0.
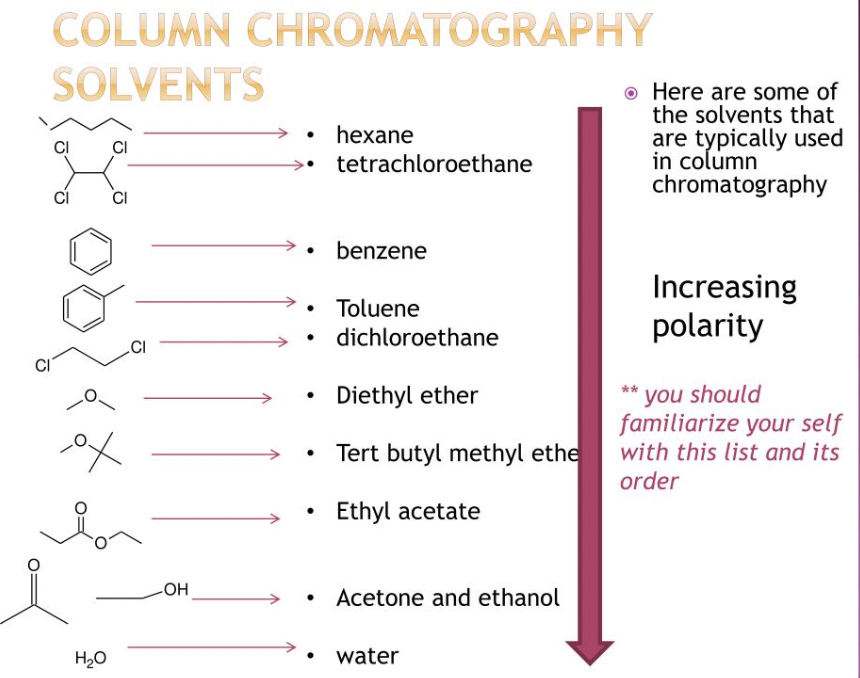
List 4 common polar solvents and 4 non polar solvents used in chromatography?
Polar:
water
DMSO
DMF
acetone
Nonpolar
benzenes
diethyl ethers
alkanes
toluene
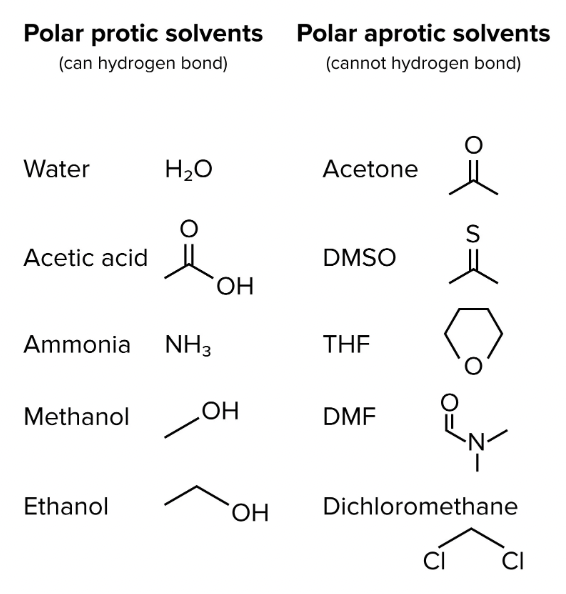
What is the difference between polar protic solvents and polar aprotic solvents? Give examples of each.
Polar protic solvents: CAN form H bonds and have a hydrogen atom attached to an electronegative atom (O or N, NOT HF)
water
alcohols (methanol, ethanol)
acetic (ethanoic) acid
ammonia (NH3)
tBuOH
Polar aprotic solvents: canNOT form hydrogen bonds and do not have an acidic hydrogen
acetone
DMSO (dimethyl sulfoxide)
DMF (N,N-Dimethyl Formamide)
what is the difference between Ksp and Kf in solutions?
Ksp: eq constant for solubility of OG compound in soln.
Kf: eq constant for formation of complex ions in soln
measures stability of a complex ion formed b/w metal ions and ligands
What are the 3 types of aqueous solns? describe and give examples of each.
Strong electrolyte: ionize completely in H20, allow high electrical conductivity. Ex: Strong acids/bases, salts
Weak electrolyte: partially dissociate into ions, weaker electric conductivity. ex: weak acids (acetic), weak bases
Non-electrolyte: do NOT dissolve into ions at all, do NOT conduct electricity. ex: sugars, alcohols
Higher ionization energy usually corresponds with higher EN, except for which 3 elements?
He, N, Ar
What type of orbitals of the central atom are involved in bonding in tetrahedral compounds? (think of how many bonds)
sp3, 4 bonds
What type of orbitals of the central atom are involved in bonding in trigonal bipyramidal compounds? (think of how many bonds)
sp3d, 5 bonds
What type of orbitals of the central atom are involved in bonding in octahedral compounds? (think of how many bonds)
sp3d2, 6 bonds
How many orbitals in the s subshell?
1 orbital
how many orbitals in a p subshell?
3 (x,y,z directions)
How many orbitals in a d orbital?
5
how many orbitals in an f shell?
7