Definitions for chemistry, physics and cs (HIGHER)
1/35
Earn XP
Description and Tags
from qs course spec
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
36 Terms
Fetch Execute step 1:
The processor sets up the address bus with the required address
Fetch Execute step 2:
The processor activates the read line on the control bus.
Fetch Execute step 3:
An instruction is fetched from the memory location using the data bus and stored in the instruction register
Fetch Execute step 4:
The instruction in the instruction register is then interpreted by the decoder and carried out.
what is a meson
a particle made of a quark anti-quark pair
what is a baryon
a particle made of 3 quarks
what is a hadron
a particle made of quarks
what is a fermion
a matter or antimatter particle
What type of C to C bonding is saturated
C-C
What type of C to C bonding is unsaturated
C=C
What type of reaction is "hardening" an oil
hydrogenation
What does hardening an oil achieve?
It saturates the molecule, removing kinks and letting it pack together more tightly. This increases LDFS and thus BP making it a solid.
What catalyst is used in esterification
sulphuric acid
why is sulphuric acid used in esterification(2)
to provide H+ ions
to remove H2O
how can you tell an ester has formed(2)
a layer has formed
you can smell the ester
what type of solution is an ester put through after production
carbonate solution
why put an ester through Na2CO3 solution
neutralise excess carboxylic acid
neutralise sulphuric acid catalyst
why are oils good to eat?(2)
they provide E
they provide essential fatty acids
what is the formula for the range of +ve numbers an amount of bits can store?
2^n -1
what is the formula for the range of -ve numbers an amount of bits can store?
2^n
How is the exponent found in floating-point representation?
number of places it has moved from 0.1
logic error
An error in a program that makes it do something other than intended
execution error
An error that occurs while a program is running and causes the program to crash.
how does acidified K2Cr2O7 react with a primary alcohol
the alcohol is oxidised to an aldehyde then to a carboxylic acid
it goes from orange to green
how does acidified K2Cr2O7 react with a secondary alcohol
the alcohol is oxidised to a ketone
it goes from orange to green
how does benedicts/fehlings solution react with an aldehyde
the aldehyde is oxidised to a carboxylic acid
the solution changes from blue to brick red
how does tollens reagent react with an aldehyde
the aldehyde is oxidised to a carboxylic acid
the solution forms a silver mirror on the test tube wall
What is denaturing a protein?
A permanent change in the structure of a protein, causing loss of function due to disruption of its shape.
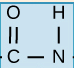
functional group?
peptide/amide link
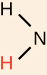
functional group
amine group
when a carboxylic acid reacts with a metal/metal base to form a soap what type of reaction is it
neutralisation
what is the first step in free radical reactions?
initiation
oxidising agents have a ___ electronegativity
high
reducing agents have a ____ electronegativity
low
things that are readily oxidised are at the ___ of the electrochemical series
bottom
things that are readily reduced are at the ___ of the electrochemical series
top