EASC 2502 Module 8: Redox
1/24
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
25 Terms
What are redox reactions?
Reactions driven by electron transfer
What is oxidation?
Loss of electrons
What is reduction?
Gain of electrons
How are redox reactions measured?
Electronical potential (E)
What are the 6 steps of a redox reaction?
1) oxidation numbers
2) half reactions
3) balance elements
4) balance electrons
5) combine reactions
6) add H2O and H+ to balance O and H
What is the electromotive series (EMS)?
Standard electrode potential (SEP, voltage) of half-cell reactions relative to standard hydrogen electrode (SHE).

How do we know the species that will oxidize or reduce?
The more positive species will be reduced, or the species higher on the EMS will get oxidized

What can be said about species that are higher than SHE on the electromotive series?
They have negative SEP, so they give off electricity/electrons -> they are reducing agents, and get oxidized

What can be said about species that are lower than SHE on the electromotive series?
They have positive SEP, so they require electricity to react and accept electrons -> they are oxidizing agents, and get reduced

In a Zn-Cu battery, what is oxidized/reduced?
What is oxidizing/reducing agent?
What is voltage generated?
E Zn = -0.76
E Cu = +0.34
Zn gets oxidized, Cu gets reduced
Zn is the reducing agent, Cu is the oxidizing agent.
V = -0.76 + -0.34 (note sign change of reduced species) = -1.10V
What is y-intercept in the Eh-pH plot?
𝐸ℎ = 1.23 + 0.0148 ∗ log[𝑂2] − 0.0591𝑝𝐻
Y-intercept is 𝐸ℎ = 1.23 + 0.0148 ∗ log[𝑂2]
![<p>Y-intercept is 𝐸ℎ = 1.23 + 0.0148 ∗ log[𝑂2]</p>](https://knowt-user-attachments.s3.amazonaws.com/52d3885d-5245-4b53-ad6e-e20a68bcc131.png)
How do you get the upper and lower stability limits of H2O for the Eh-pH plot?
[O2] of natural waters ranges from 10^-83.1 to 10^0 atm
𝐸ℎ = 1.23 + 0.0148 ∗ log[𝑂2] − 0.0591𝑝𝐻
Substitute the upper and lower ranges of [O2] into the equation.
![<p>Substitute the upper and lower ranges of [O2] into the equation.</p>](https://knowt-user-attachments.s3.amazonaws.com/c782c506-bc3d-44af-9c60-823d5f8d3871.png)
What happens above or below the stability limits?
Water would dissociate
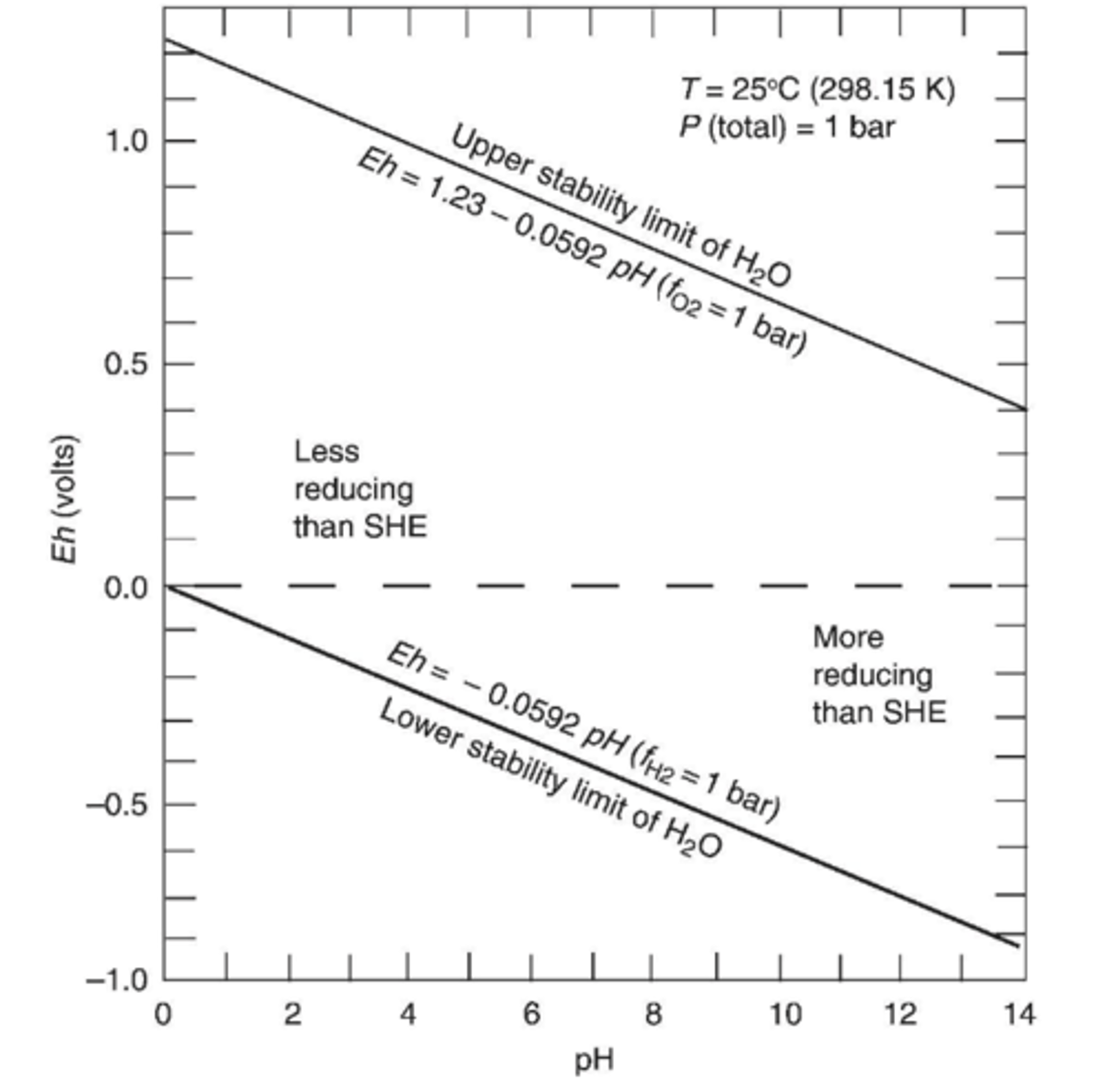
What happens between stability limits?
Water is stable
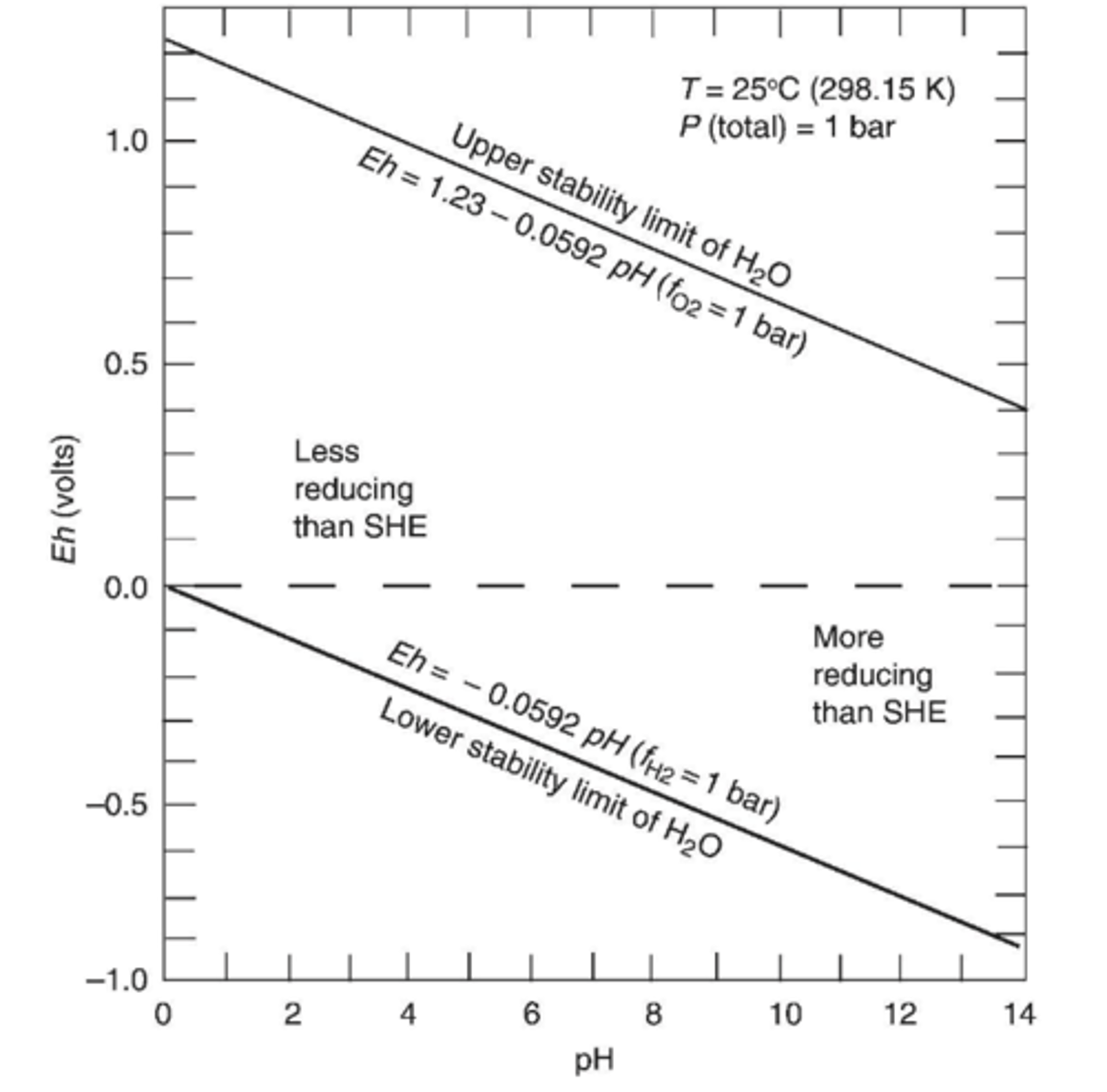
Is the space above Eh = 0 in the stability field more or less reducing than SHE?
Less reducing (more positive than SHE)
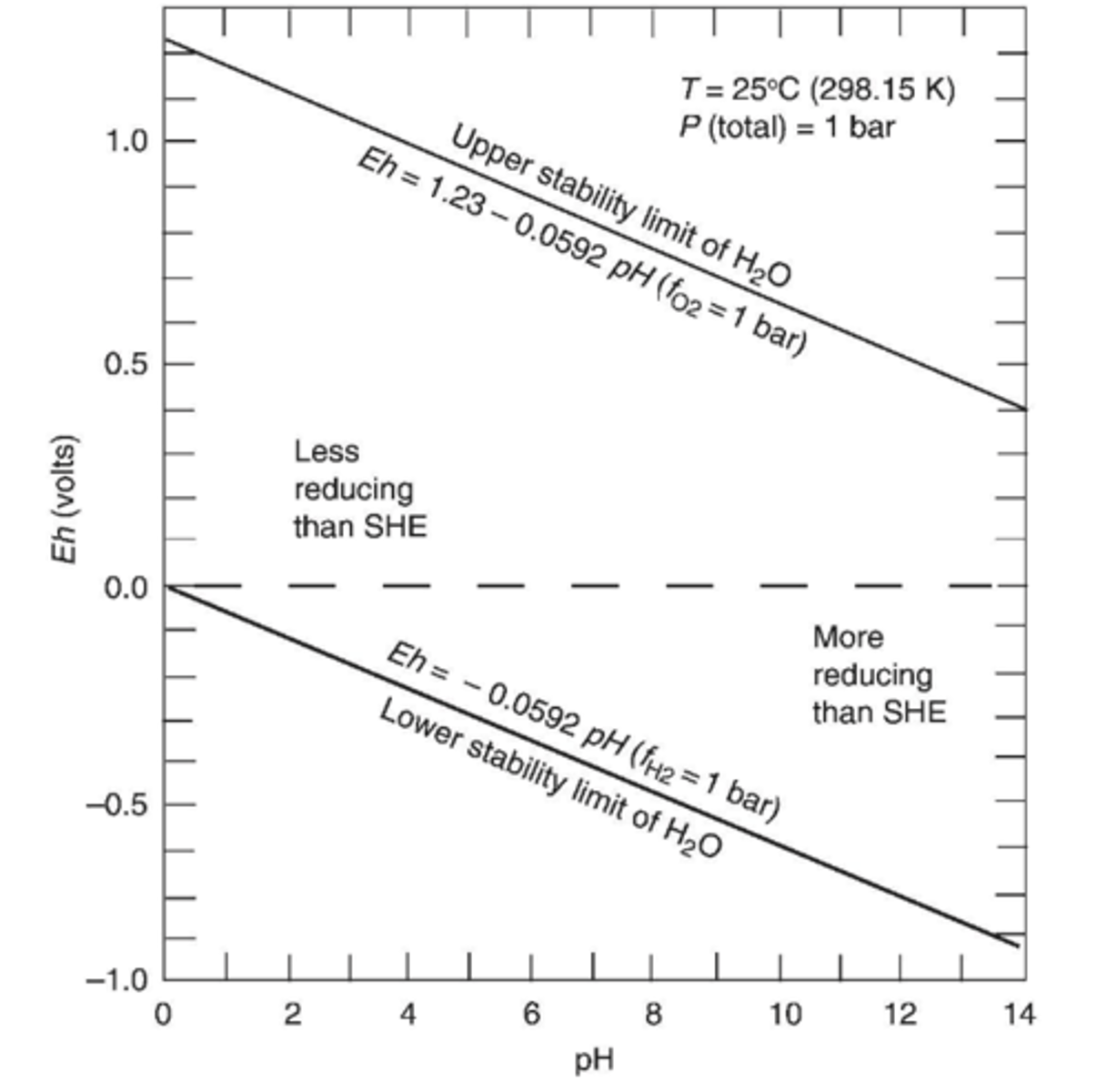
Is the space below Eh = 0 in the stability field more or less reducing than SHE?
More reducing (less positive than SHE)
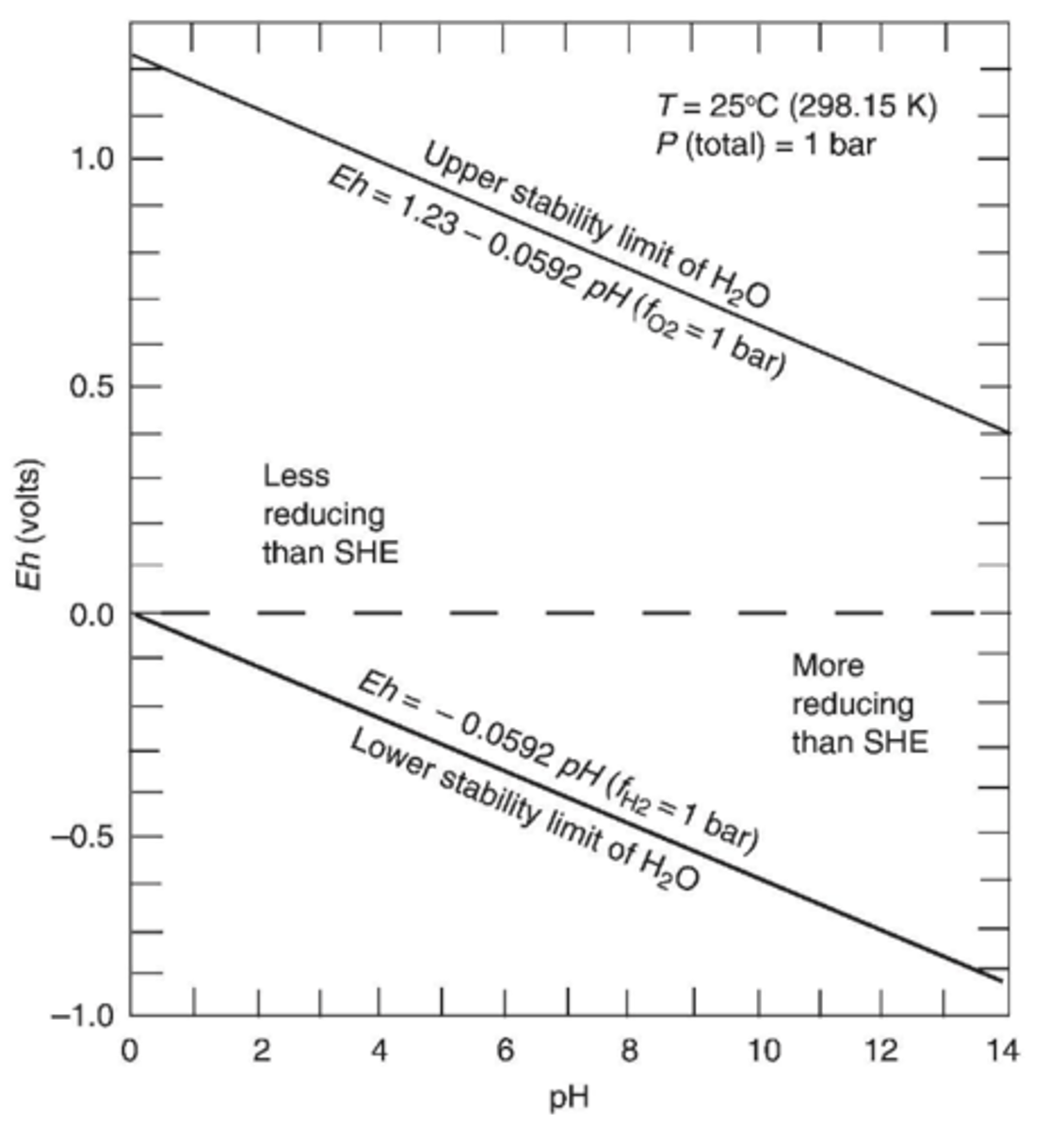
Are environments in contact with the atmosphere oxidizing or reducing?
Are environments isolated from the atmosphere oxidizing or reducing?
In contact: oxidizing ex. mine waters
Isolated: reducing ex. marine waters
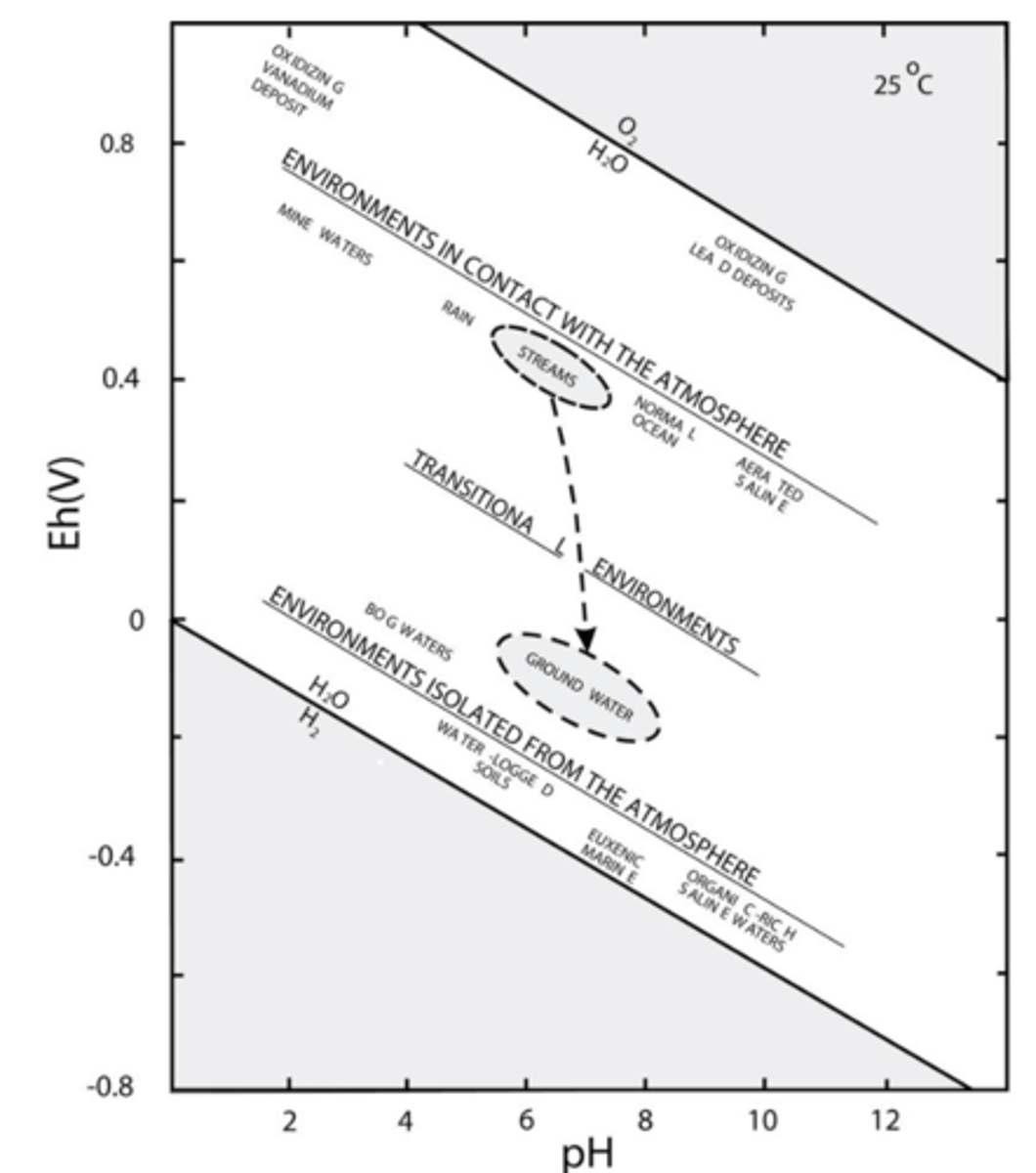
What is oxygen fugacity?
Allows us to test oxygen potential of an environment.
fO2 = log[O2]
Why is Eh-pH of sulfur species important?
useful for understanding acid-rock drainage and transport/deposition of metals in hydrothermal fluids
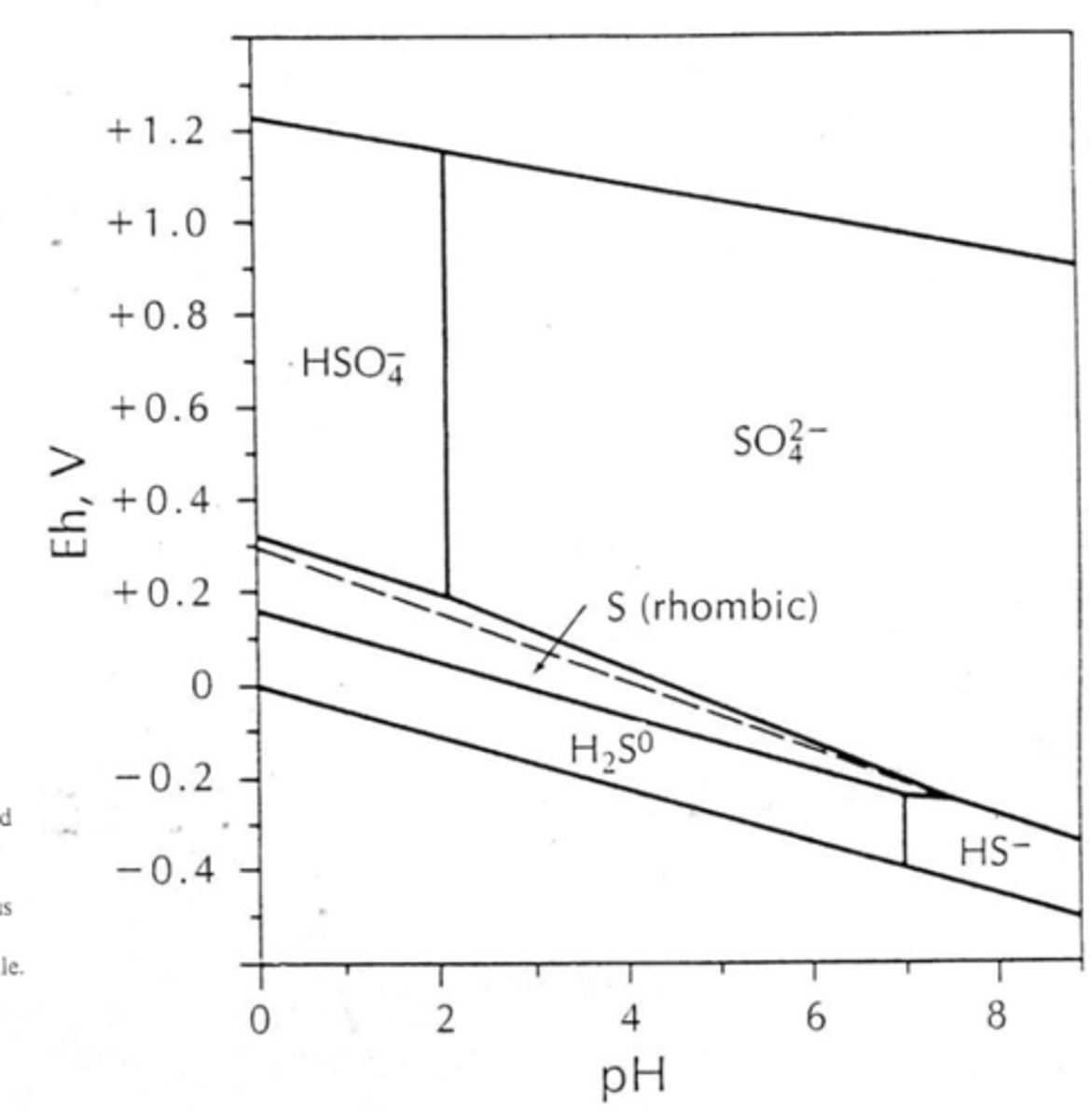
On the Eh-pH plot, what are dotted lines and what are solid lines?
Dotted lines are redox boundaries
Solid lines are mineral stability fields
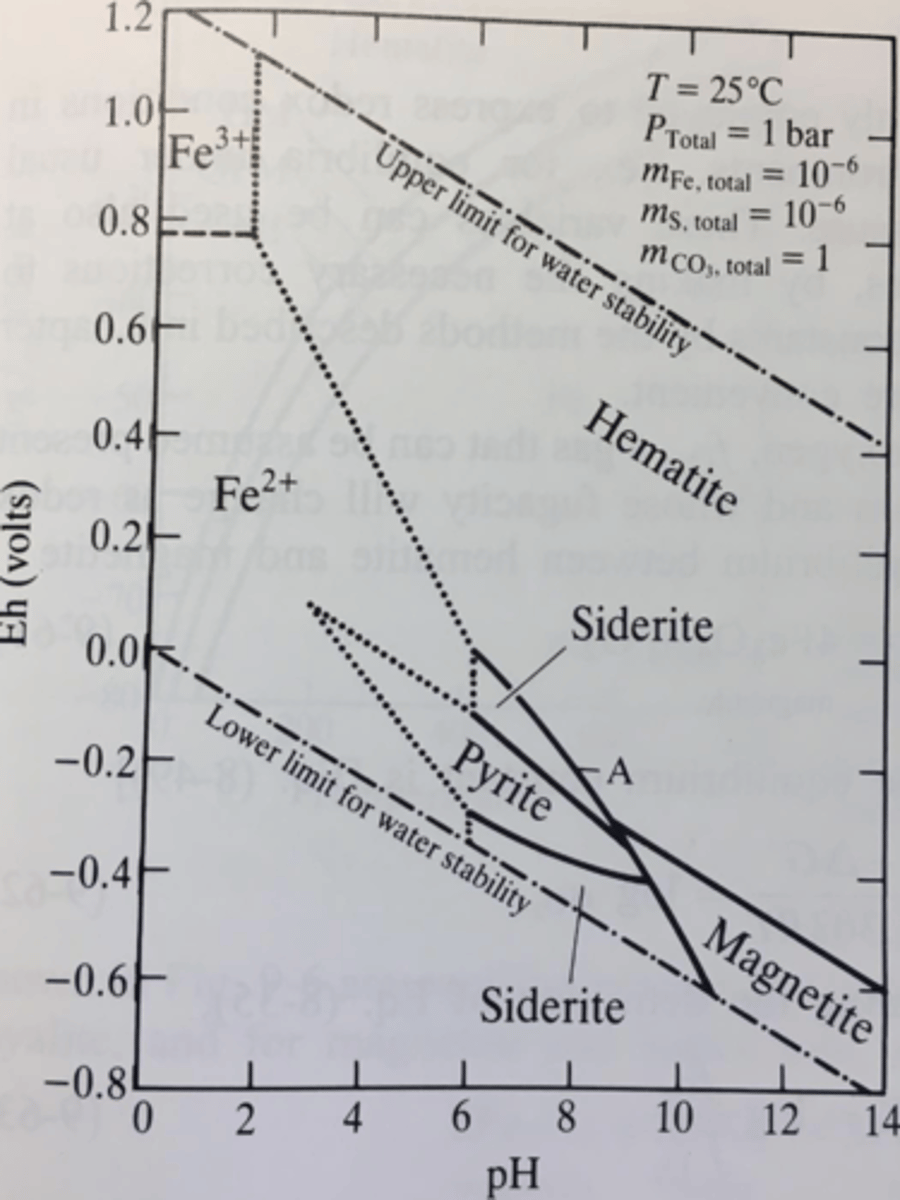
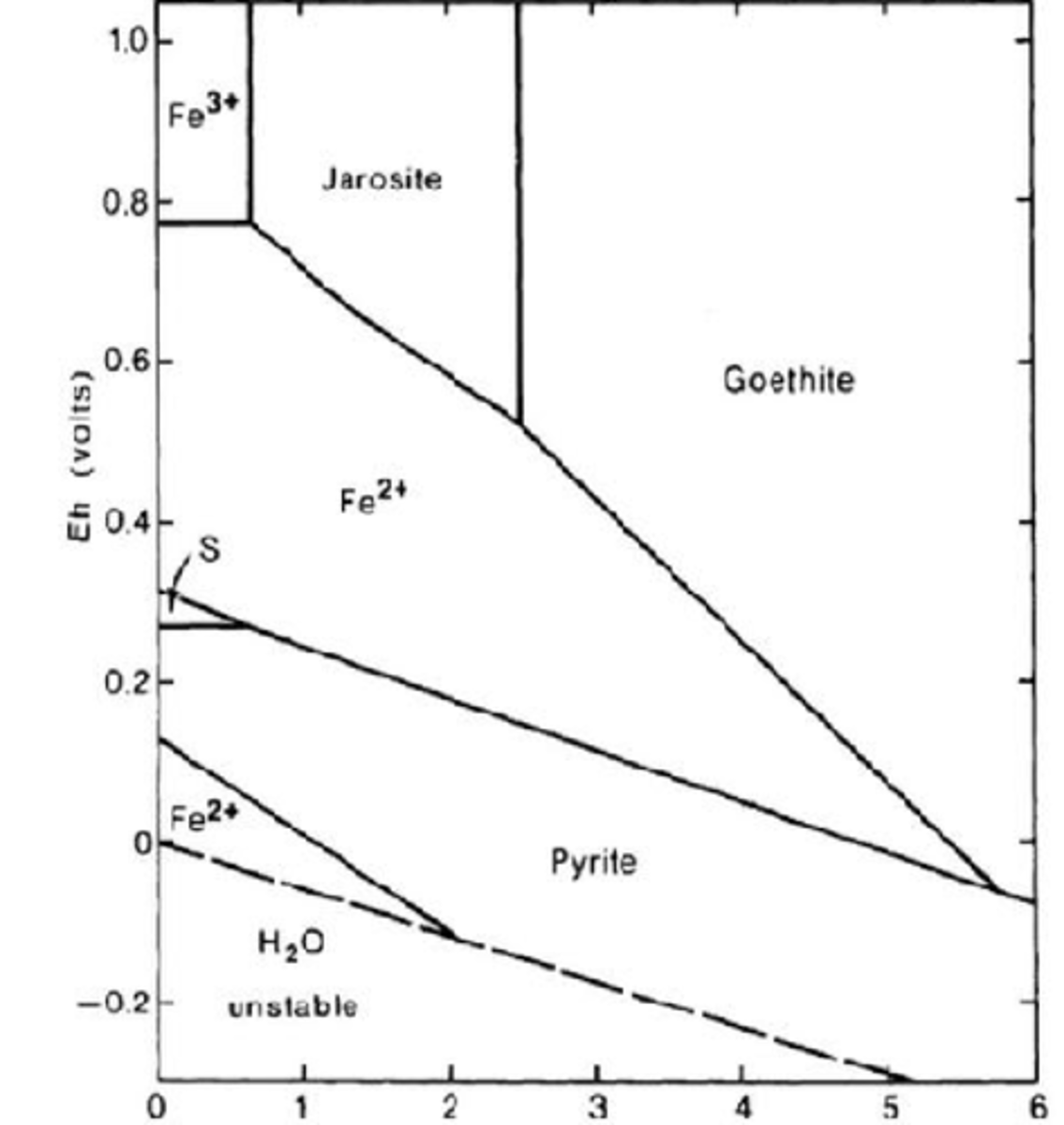
Is Fe3+ stable at ordinary surface conditions? Why? What geological conditions is it found at high concentrations?
No, because it requires highly acidic and highly oxidized environment which is not common on surface. It occurs in acidic mine tailings.
Is higher fugacity more or less oxidizing?
More
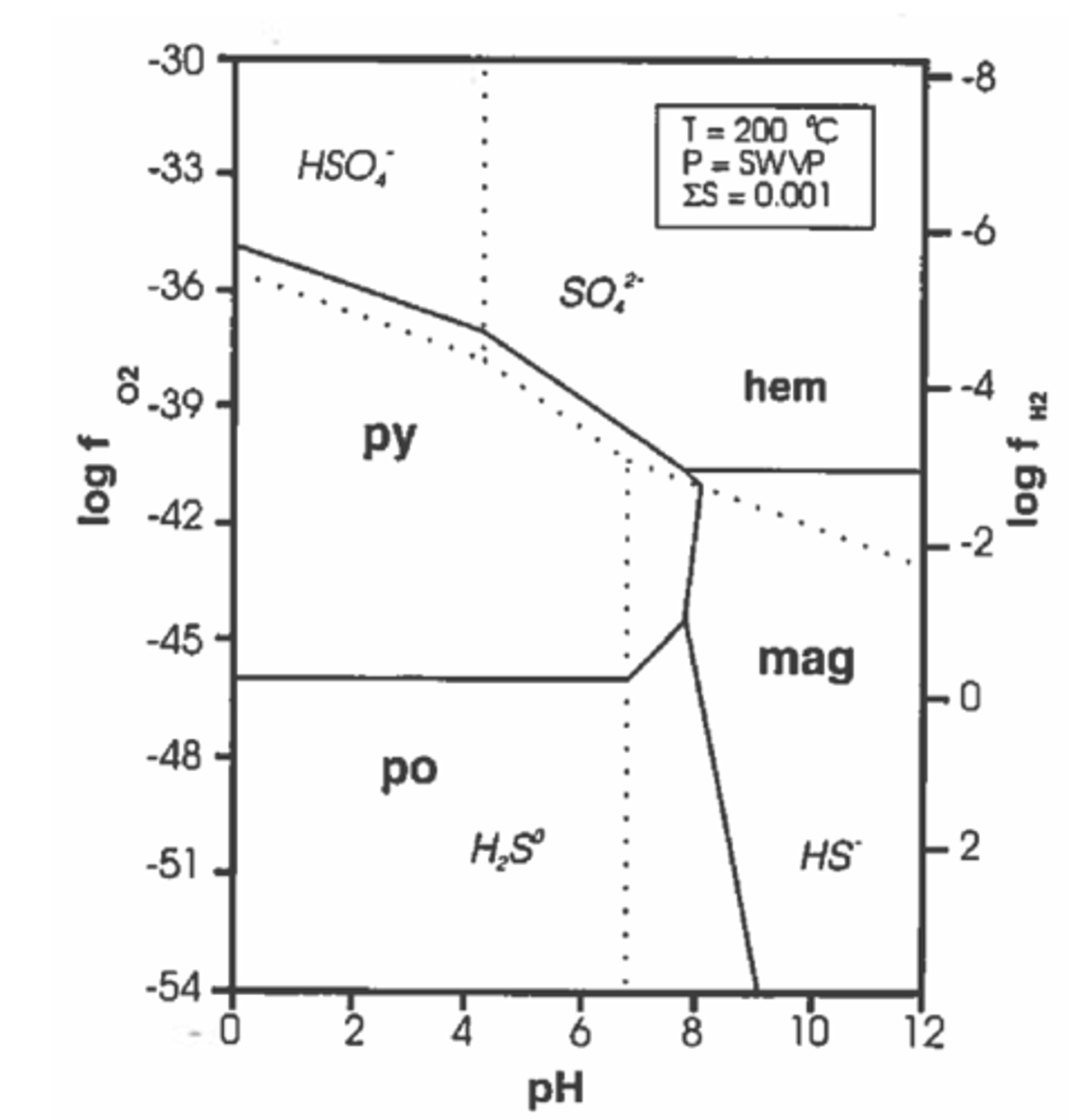
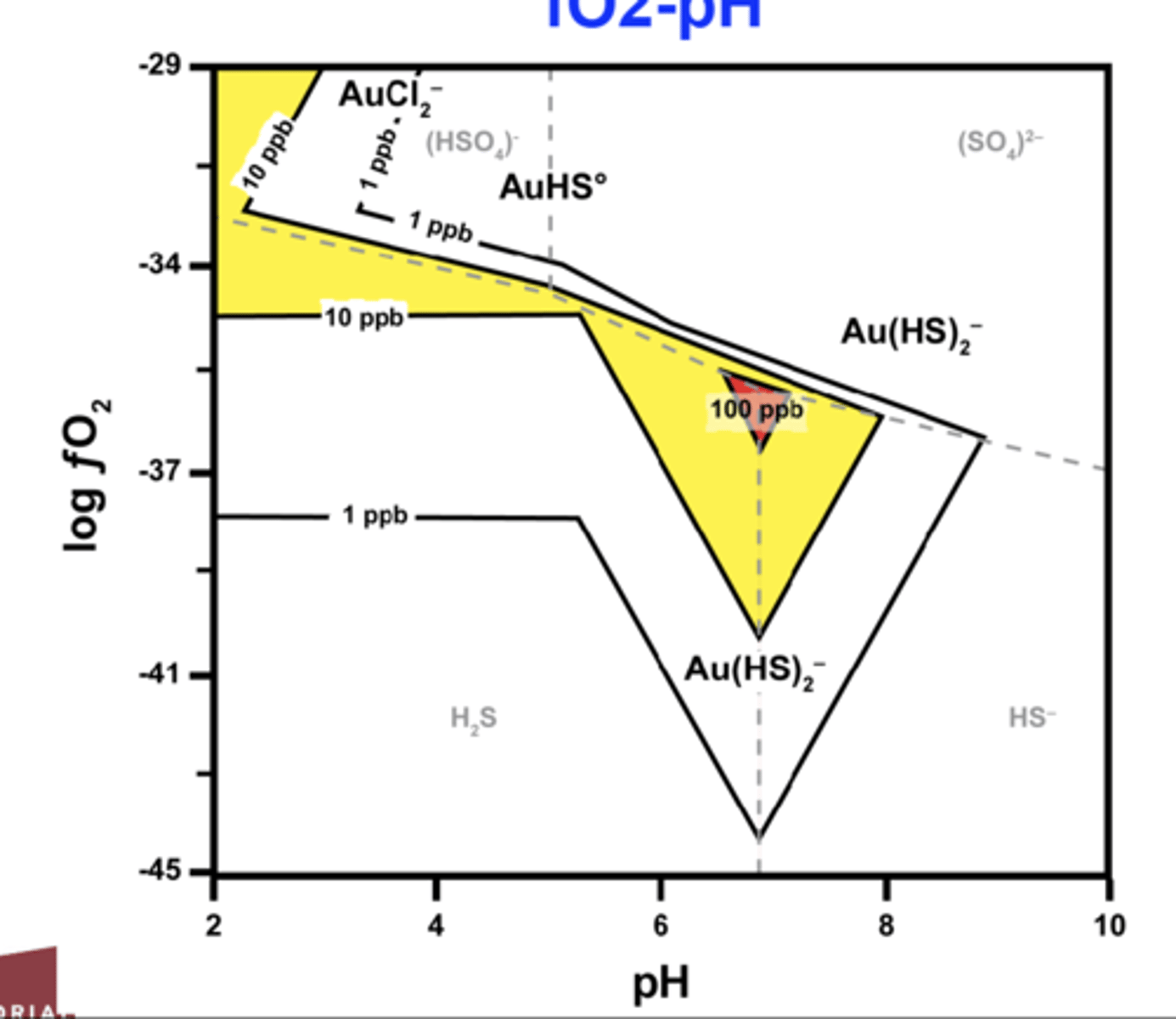
What characteristics does gold-bearing fluid have?
Its dilute, CO2 bearing, and near-neutral pH
What does the formula for pyrite weathering tell us about why acid rock drainage is a problem?
2FeS2(s) + 7O2(g) + 2H2O(l) = 2Fe2+(aq) + 4SO42−(aq) + 4H+(aq)
The 4H+ makes the surroundings very acidic
What can we do to prevent aid rock drainage?
Adding lime, Ca(OH2), neutralizes the acids and creates inert solids that can be removed.
Fe2+(aq) + SO42−(aq) + 4H+(aq) + Ca(OH)2(s) = Fe(OH)2(s) + CaSO4(s) + 2H2O(l)