BIOL120: Chapter 3 (Protein Structure and Function)
1/20
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
21 Terms
amino acids
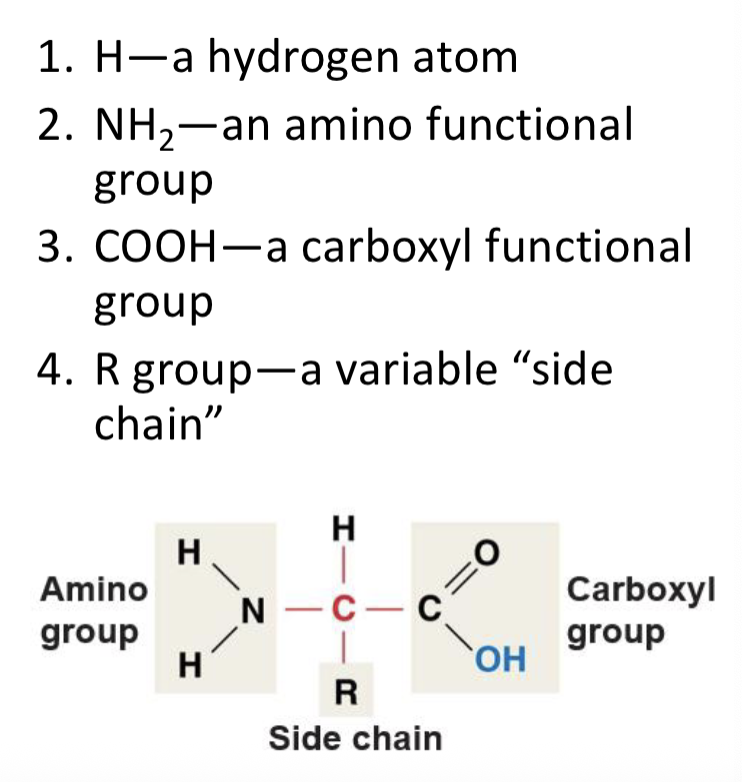
functional groups
amino groups
carboxyl groups
carbonyl groups
hydroxyl groups
phosphate groups
sulfhydryl groups
ionization of amino acids
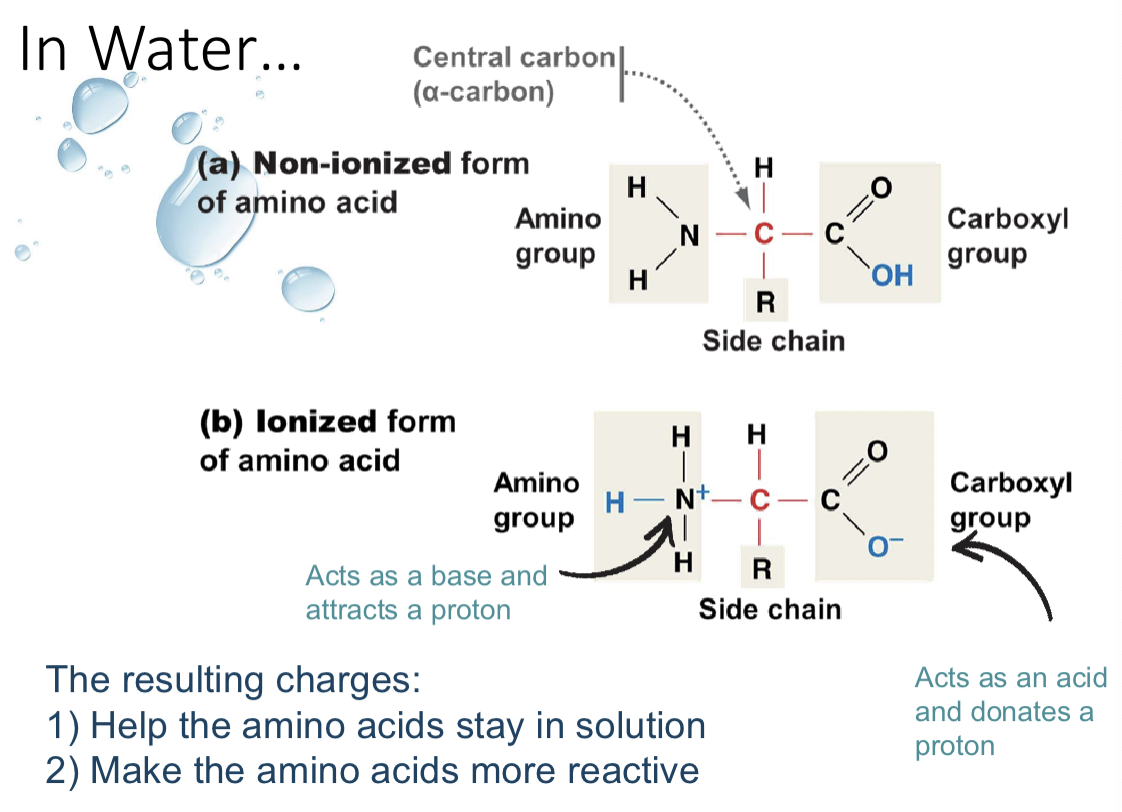
r-group side chains
three types:
charged - acidic (-) and basic (+)
uncharged polar
nonpolar
structural formula for an amino acid

polymerization
the process of linking monomers togethers
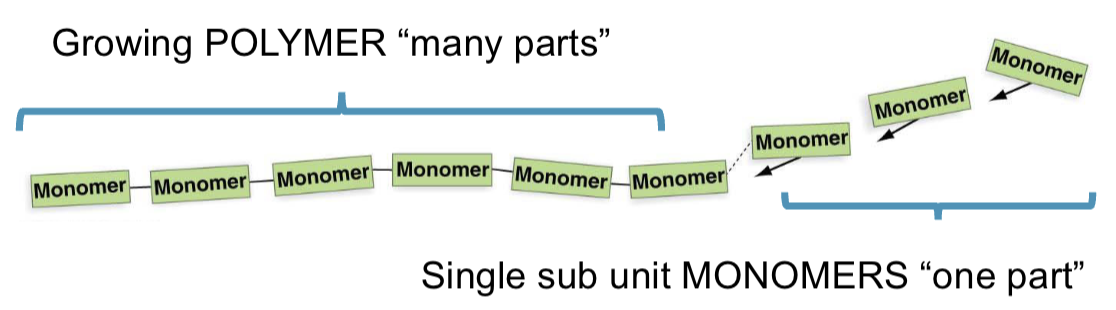
condensation reaction (aka. dehydration synthesis)
monomer in, water out
decreases entropy
requires energy
hydrolysis
water in, monomer out
increases entropy
release energy
peptide bond
amino acids polymerize when a bond forms between a carboxyl group of one amino acid and an amino group of another, resulting in a C-N bond
backbone formation from peptide bonds
r-group orientation: side chains extend out and can interact with each other or water
directionality:
free amino end: n-terminus
free carboxyl end: c-terminus
written with n-terminus on left
flexibility: single bonds on either side of the peptide bond can rotate
proteins
made of polypeptides (>50 linked amino acids), which are made of oligopeptides (<50 linked amino acids), which are made of amino acids (monomer subunits)
protein basic structures
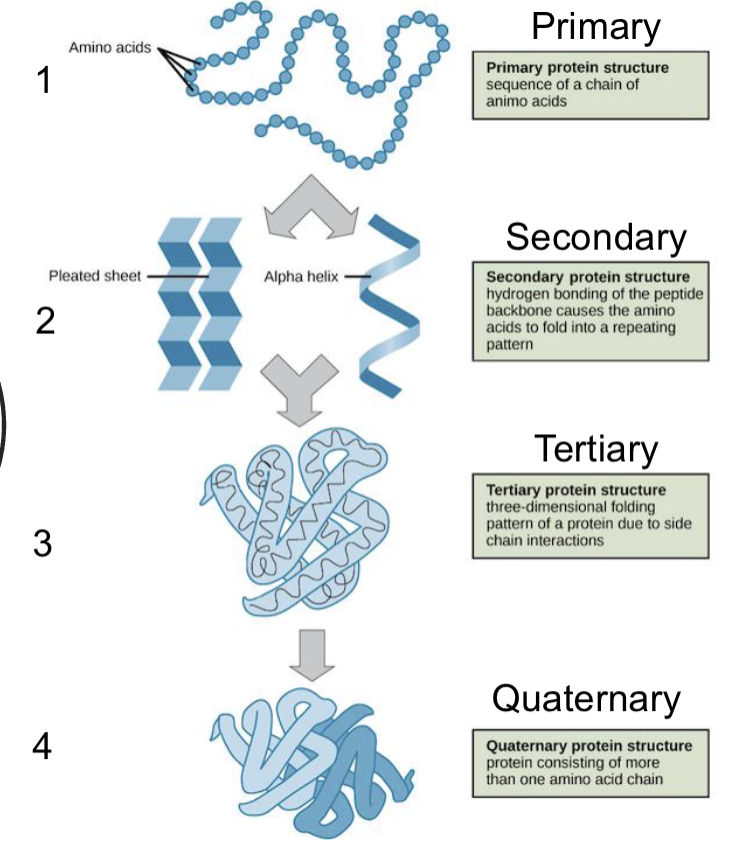
secondary structure
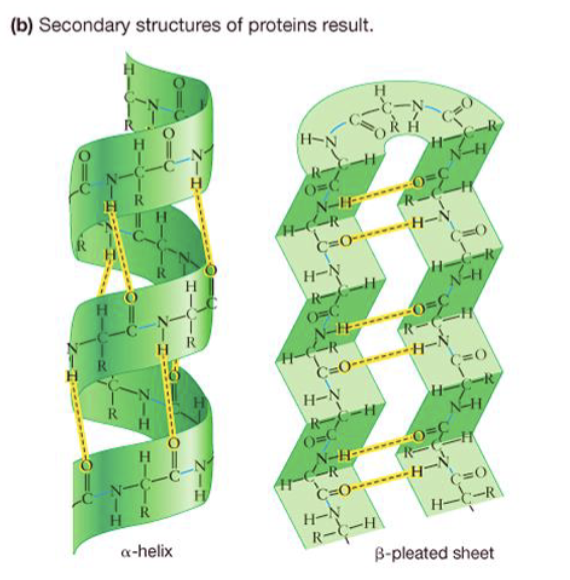
hydrogen bonds (r-group interaction)
form between polar side chains and opposite partial charges
hydrophobic interactions (r-group interaction)
water forces hydrophobic side chains
van der waals interactions (r-group interaction)
weak electrical interactions between hydrophobic side chains
covalent disulfide bonds (r-group interaction)
forms bridges between two sulfhydryl groups
ionic bonds (r-group interaction)
form between groupos with full and opposing charges
normal folding
folding is often spontaneous because of the H bonds and van der waals interactions
the folded molecule is more energeticaly stable
a denatured (unfolded) protein is unable to function
molecular chaperones
help proteins fold correctly
prions
improperly folded forms of normal proteins
induce normal protein molecules to change their shape to the altered form