Energy Transfer/Endothermic And Exothermic Reactions
1/28
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
29 Terms
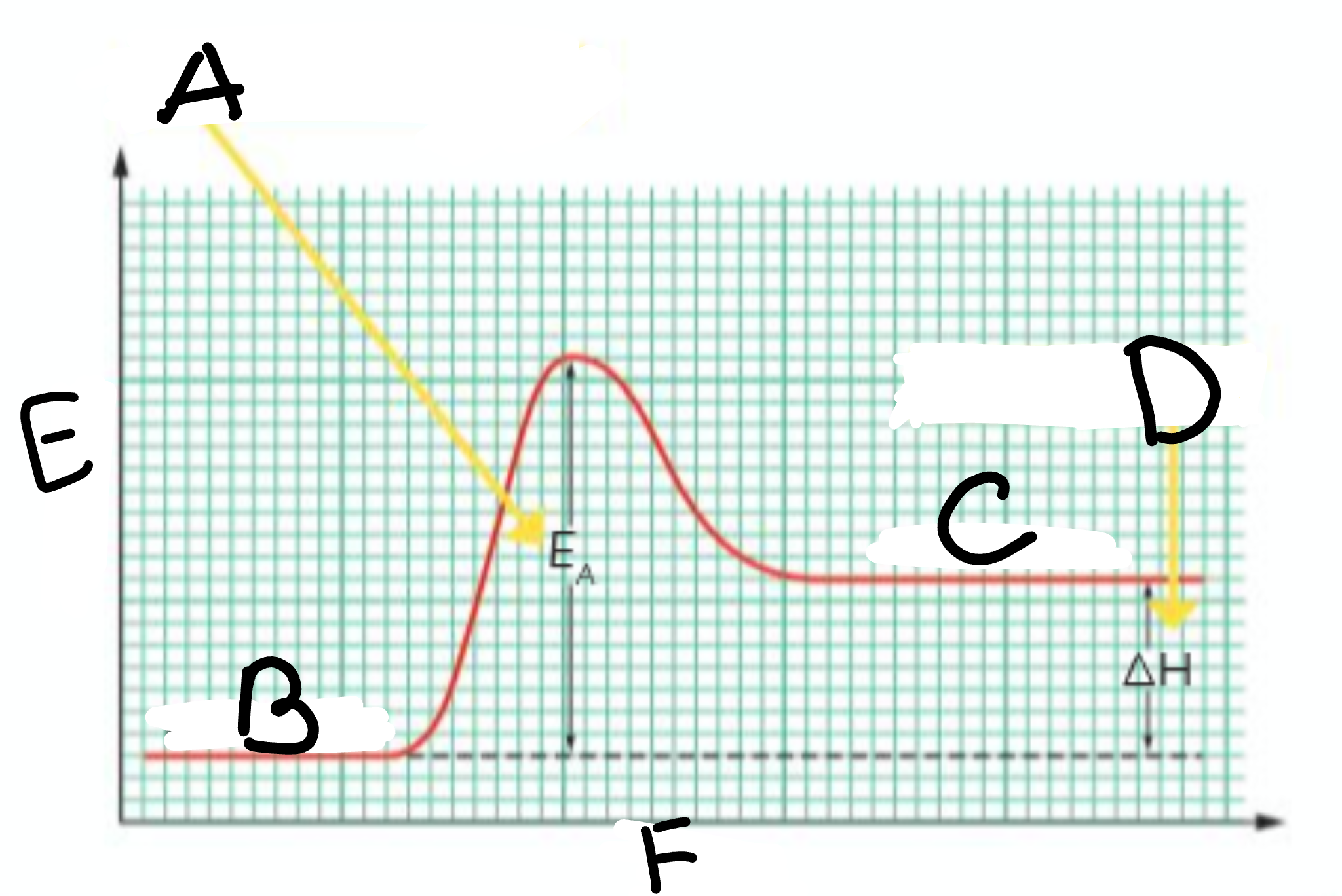
A?
Activation Energy
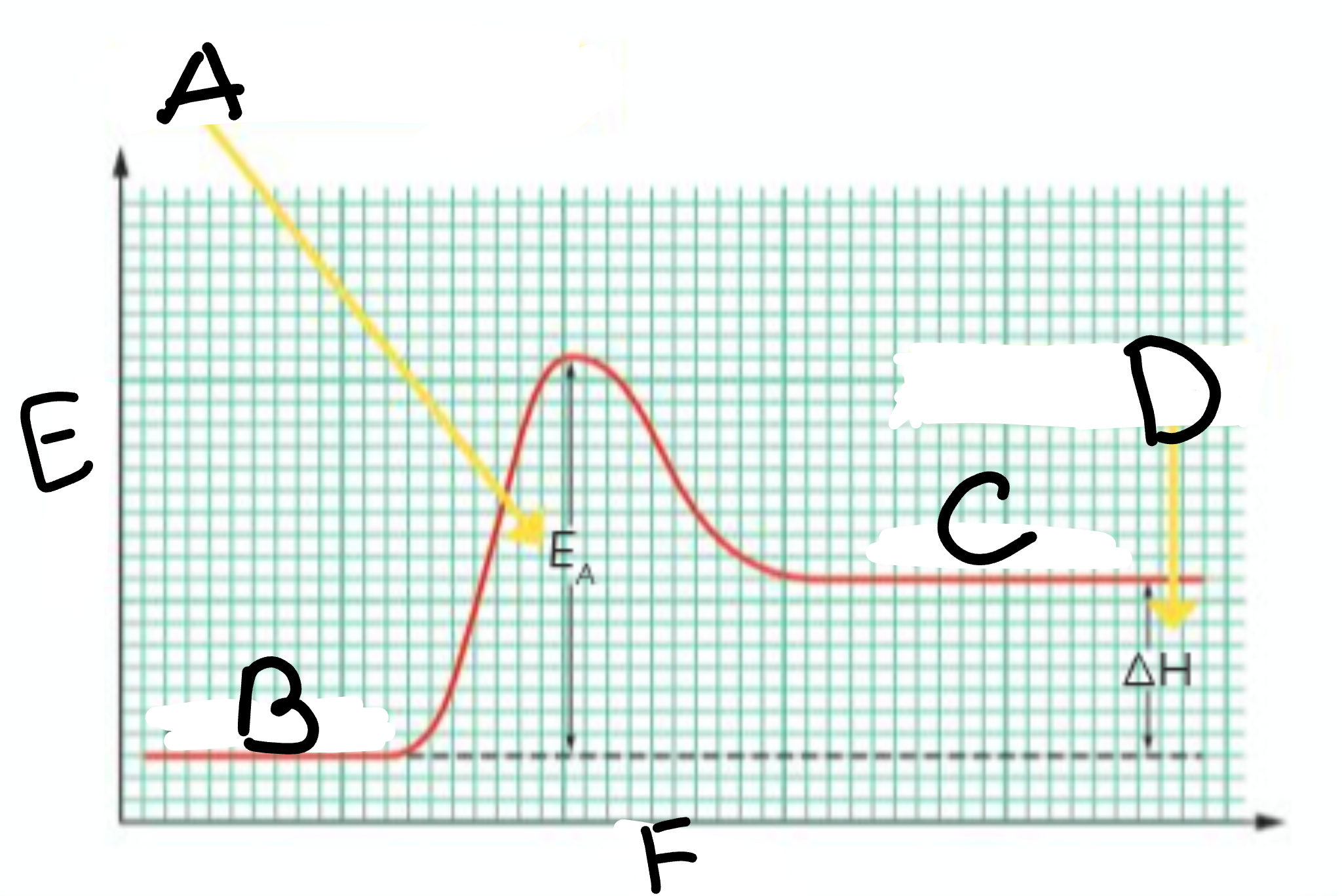
B?
Reactants
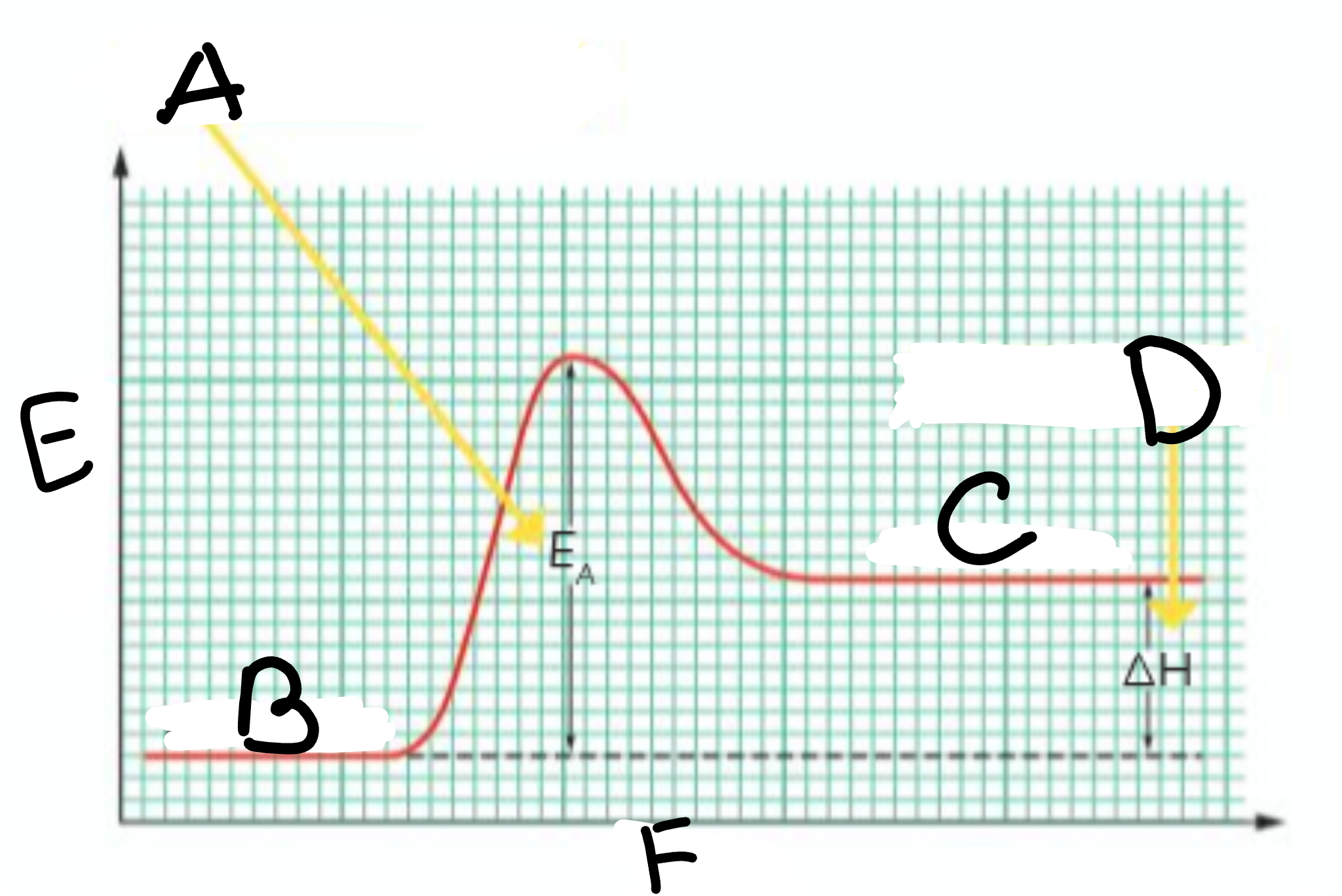
C?
Products
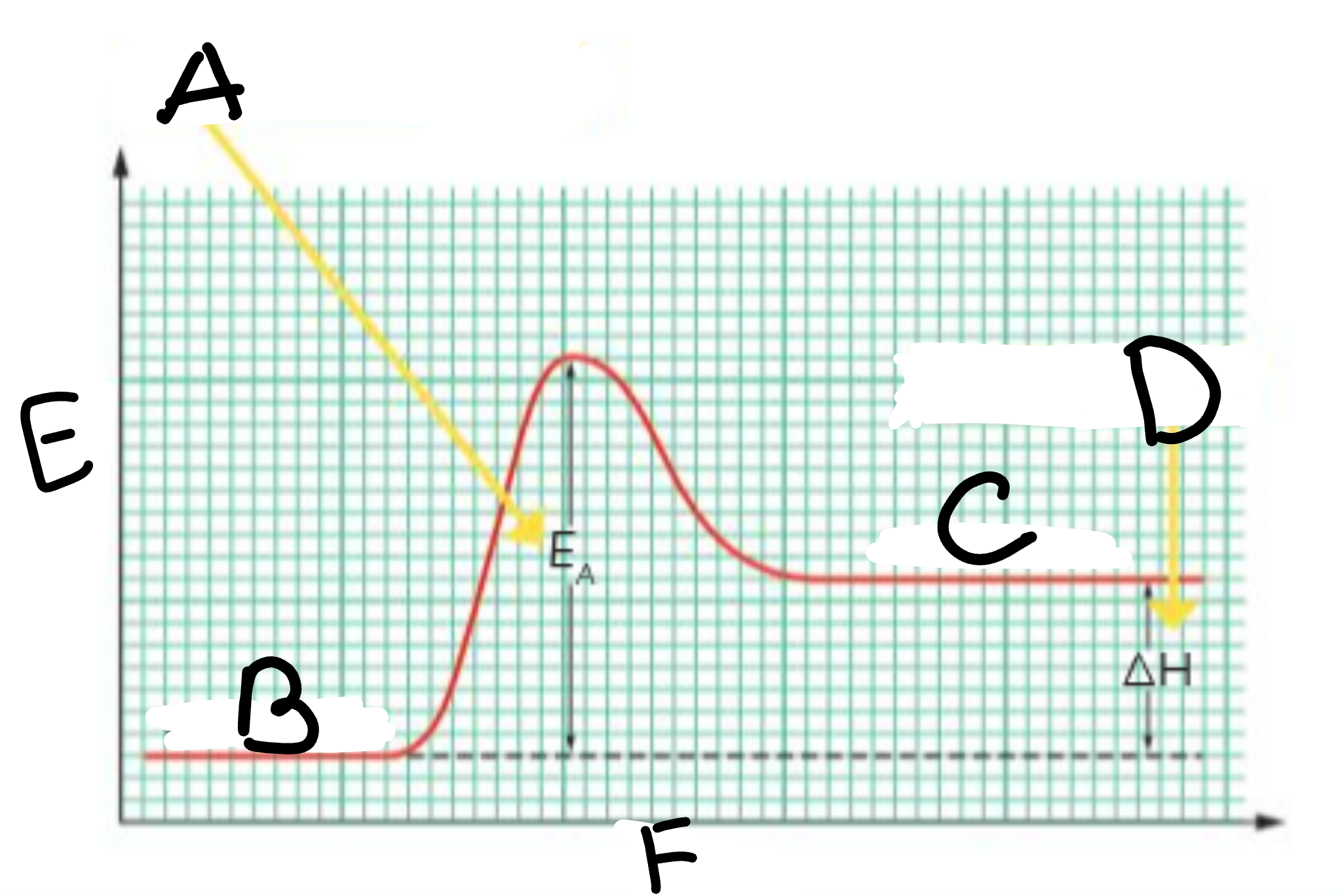
D?
Heat taken in
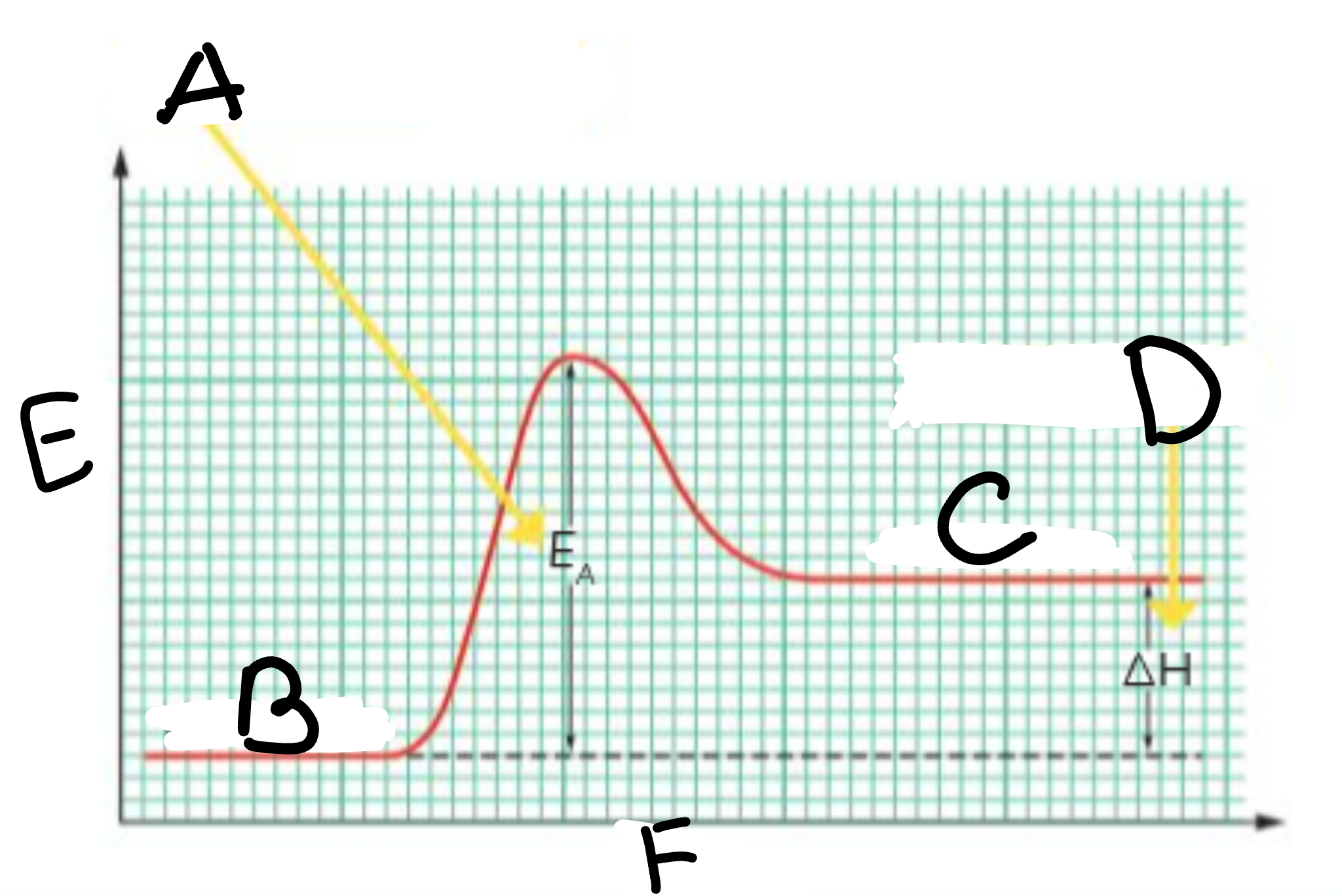
E?
Energy
F?
Course of Reaction
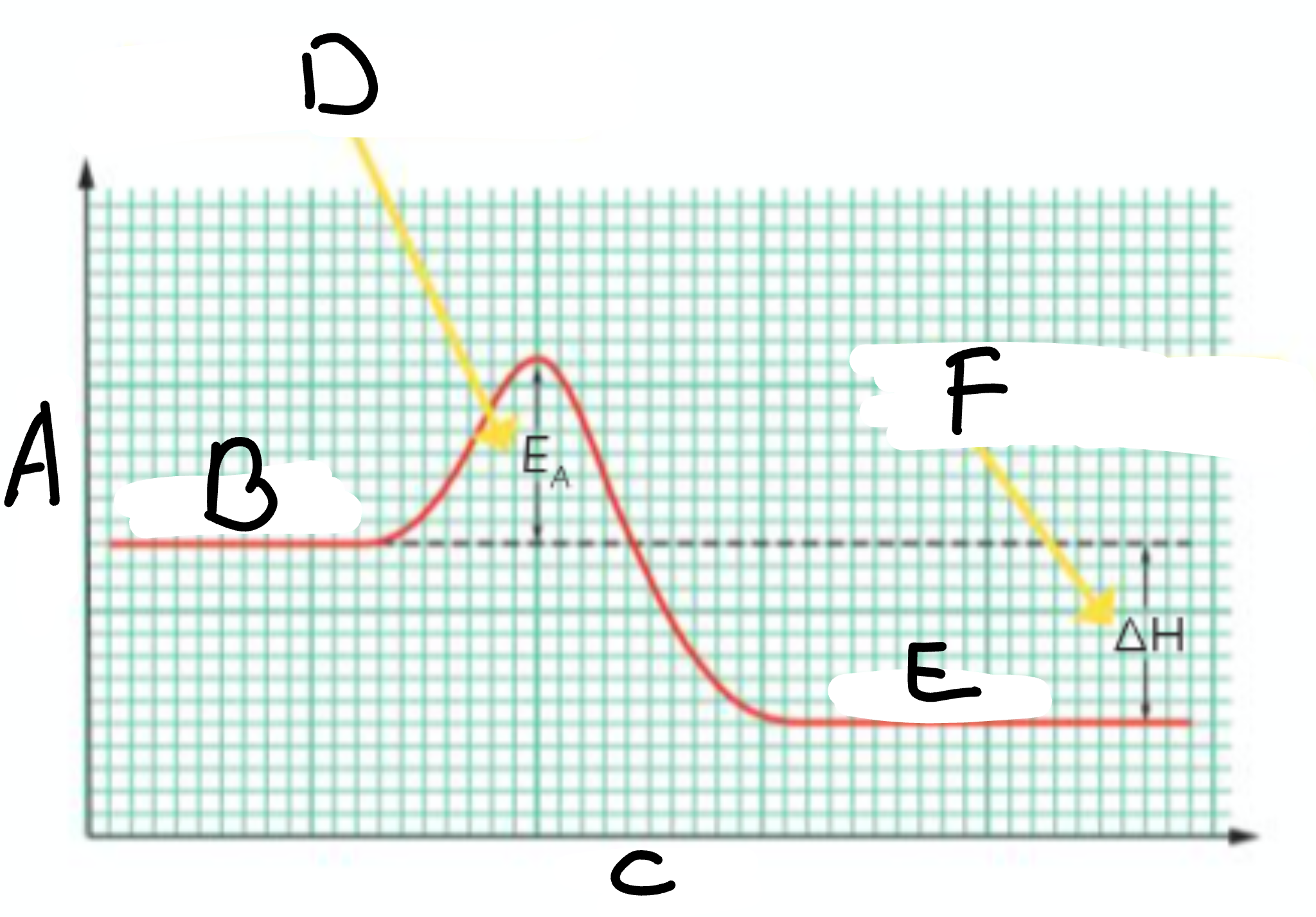
Is this reaction exothermic or endothermic, and why
Exothermic, because the products have less energy than the reactants which means that heat energy was given out into the surroundings
Is this reaction exothermic or endothermic, and why?
Endothermic, as the products have more energy than the reactants, which means that heat was taken in during the reaction
Differences between exothermic and endothermic reactions:
Exothermic give out heat while endothermic give out heat
Exothermic have less energy in their products than in their reactants while endothermic have more energy in their products than their reactants
The temperature goes from 17-19
exothermic reaction
the temperature goes from 19-17
endothermic reaction
activation energy is…
the minimum energy that colliding particles must have for a chemical reaction to occur.
what is activation energy
activation energy is the minimum energy that two colliding particles must have for a reaction to occur.
what is a catalyst
a catalyst is a substance that alters the rate of a chemical reaction without being used in the reaction itself
what is the function of the catalyst
a catalyst works by providing an alternative route for the reaction to occur. This alternative route has a lower activation energy, which means that more molecules have enough energy for effective collisions to occur.
The collision theory is…
a scientific theory that states that for a chemical reaction to occur, the reacting particles must collide with each other. It also states that a collision will only result in the formation of a product if a certain minimum energy is reached in the collision. This type of collision is called an effective collision
why does stirring affect the rate of reaction?
Stirring keeps the reactant particles in motion, which increases the chance of collision.
why does the concentration of the reactants affect the rate of reaction?
When you increase the concentration of the reactants there are more particles, which increases the number of collisions taking place.
why does particle size affect the rate of reaction?
Increasing the surface area of the reactants (either by grinding or powdering them) exposes more particles to the other reactants, which leads to more collisions
why does temperature affect the rate of reaction?
When you add heat the reactant particles gain energy and move faster, which leads to more collisions.
how does increasing the surface area of the reactants increase the rate of reaction
Increasing the surface area of the reactants (by grinding or powdering them) exposes more particles to the other reactant, which leads to more collisions.
Neutralising an acid and a base
exothermic
respiration in living cells
exothermic
concentrated sulfuric acid and water
exothermic
sherbert dissolved in water
endothermic
instant cold packs
endothermic
photosynthesis
endothermic
preparation of carbon dioxide
set up apparatus as shown, add HCl to the buchner flask + stopper flask with para film, observe gas displacing water, remove gas jar when full, test gas for carbon dioxide
preparation of oxygen
set up apparatus as shown, pour hydrogen peroxide through onto the manganese dioxide + stopper with para film, observe the gas displacing the water, remove gas jar when full, test for oxygen.