Main Group Systems for Activation and Catalysis
1/100
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
101 Terms
What is the TVEC of this complex?
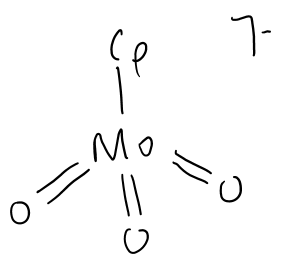
d0 + 3(4) + 6 = 18e
What is the TVEC of this comeplex?
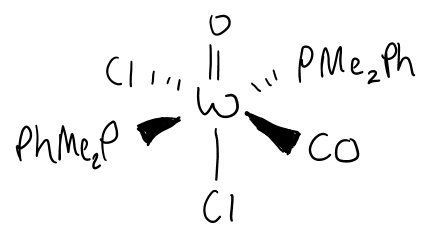
d2 + 5(2) + 4 = 16e in this form.
O can form a triple bond to make it an 18e complex.
Draw the structure formed by group 6 metals in the presence of CO at high pressure.
Draw the structure formed by group 7 metals in the presence of CO at high pressure.
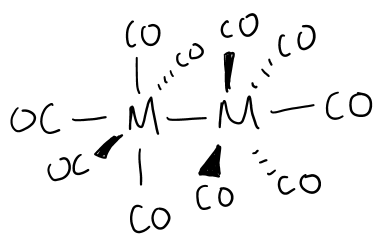
Staggered conformation to reduce steric clash.
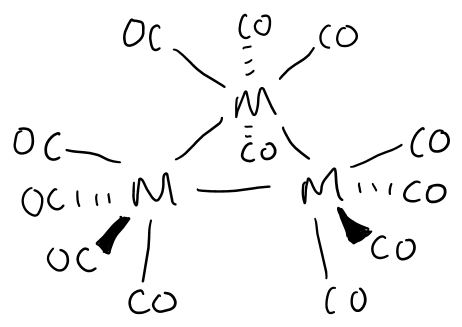
Draw the structure formed by group 8 metals in the presence of CO at high pressure.
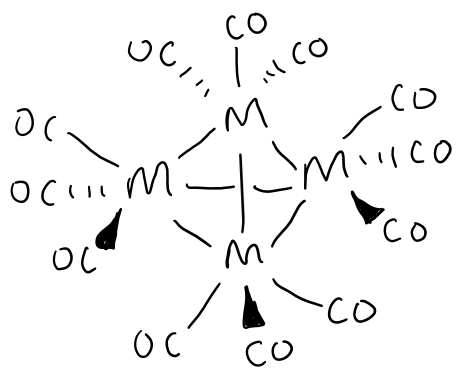
Draw the structure formed by group 9 metals in the presence of CO at high pressure.
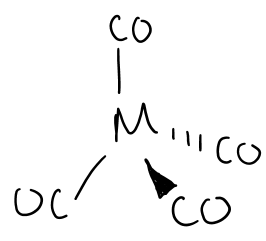
Draw the structure formed by group 10 metals in the presence of CO at high pressure.
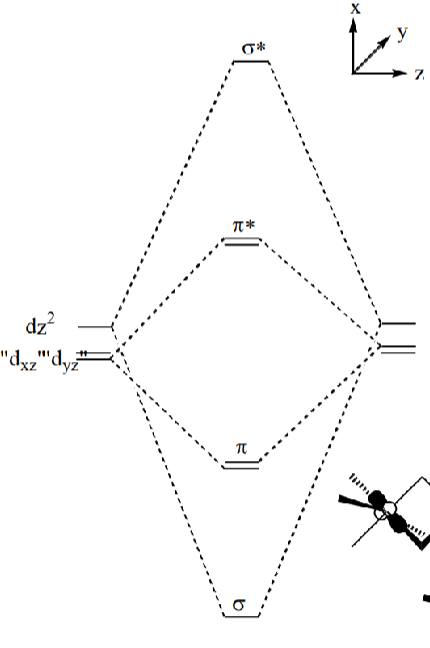
Draw an MO diagram for the bonding between two metals.
Which orbitals have the worst/best overlap?
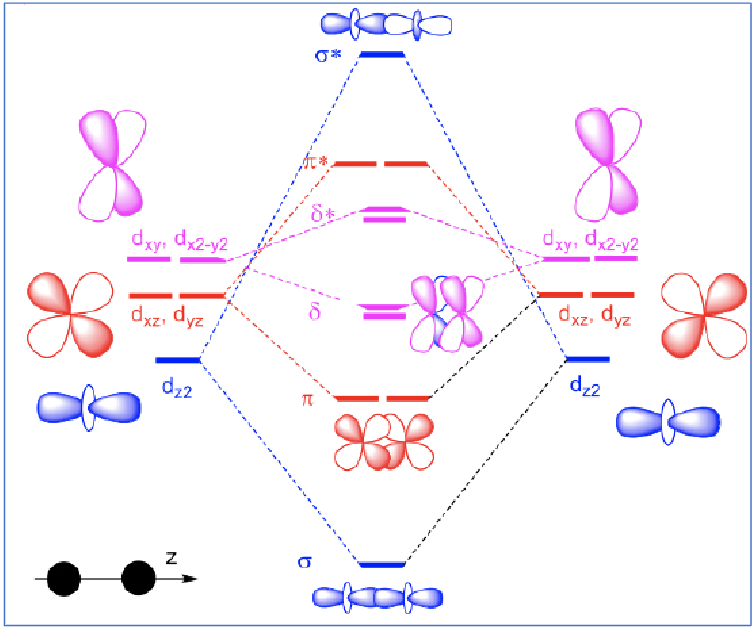
The dz2 orbitals overlap the best. If this is the only occupied MO, then full rotation around the M-M bond is possible to reduce steric clash of ligands:

Explain why a d7 complex has one less ligand than a d6.
The extra electron occupies the dz2 orbital. This prevents one of the z direction ligands from binding, hence giving a square pyramidal geometry.
Draw the MO diagram for M-M bonding of a d7 metal and give the bond order.
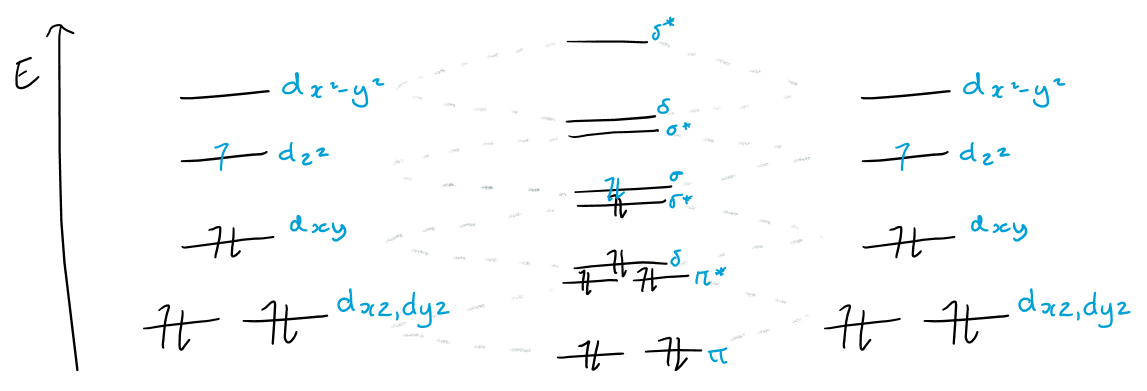
Bond order = 1/2(8 - 6) = 1
Hence metals form a single bond with dz2 orbital, allowing full rotation to produce the lower energy staggered conformation.
How does M-M bond strength change down the d7 group? Why?
The M-M bond gets stronger down the group because the σ - σ* energy gap becomes larger, hence the bonding MO is the more preferred form
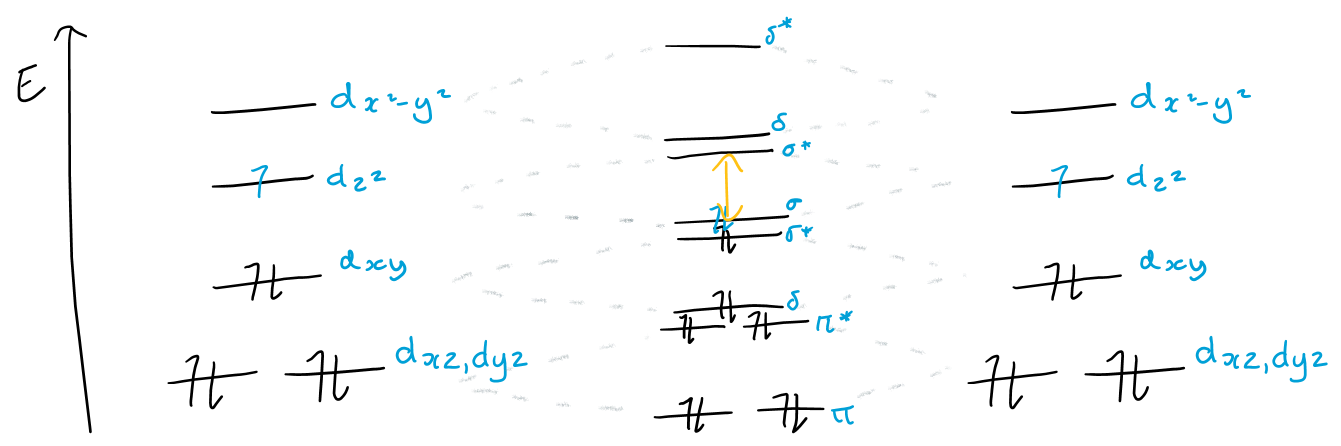
The σ - σ* energy gap gets larger down the group due to improved overlap of the larger d orbitals:
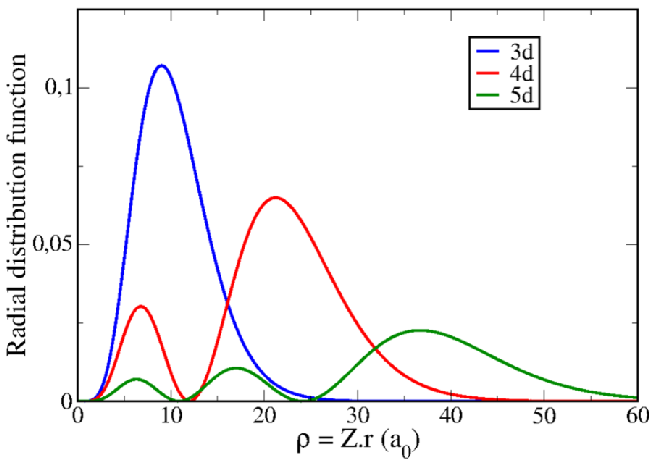
Explain M-M quadruple bonds with an MO diagram.
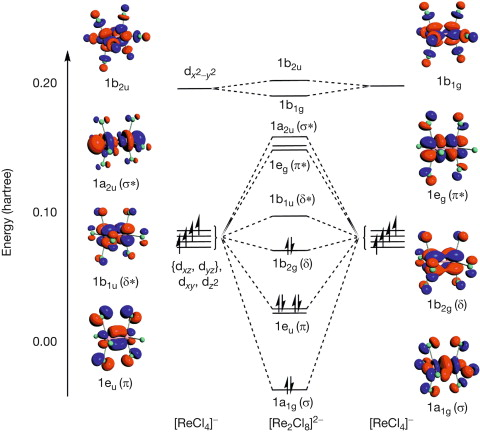
Theres 8 electrons in bonding orbitals, so bond order of 4.
Use a MO diagram to explain how a bond order of 6 can be achieved.
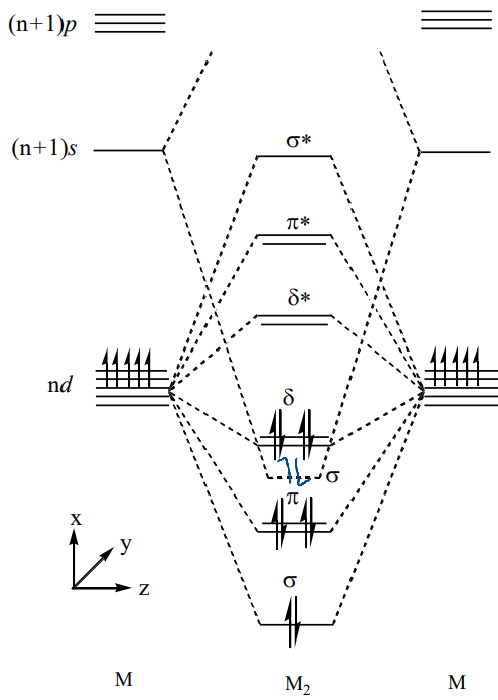
The (n+1)s orbital overlap can be so stabilised that it reaches the bonding manifold and therefore increases the bond order.
Give the oxidation state and therefore d electron count of the metals in this complex
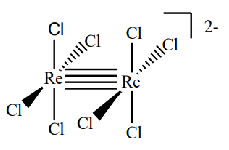
Re(III) so d4
Give the oxidation state and therefore d electron count of the metals in this complex
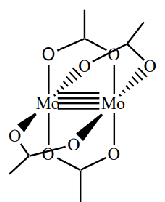
Mo(II) so d4
Give the oxidation state and therefore d electron count of the metals in this complex
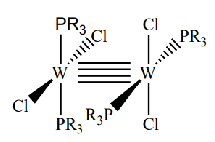
W(II) so d4
Explain why d4 metals form eclipsed dimers using this MO diagram
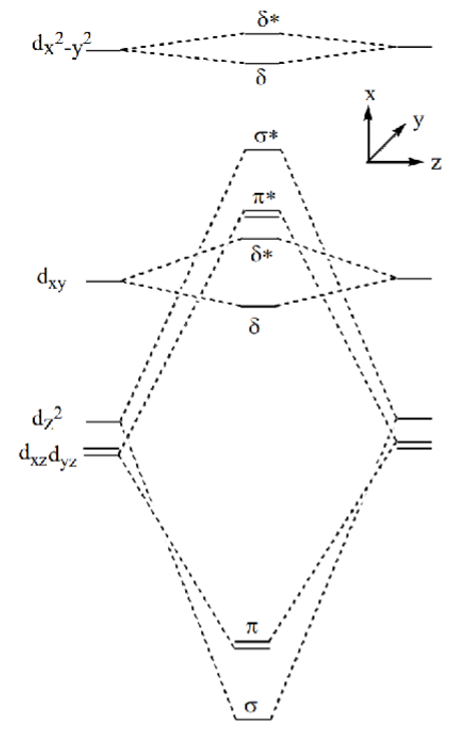
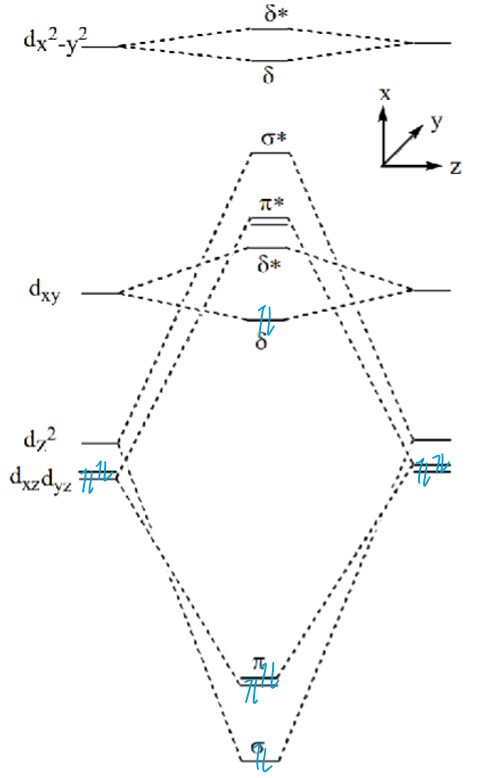
dxy MO is occupied.
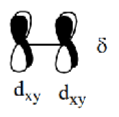
Four overlaps causing fixed orientation of metals and therefore ligands.
What is the difference between these two complexes: Re2Cl82- (group 7) and Os2Cl82- (group 8)
In the Re (d4) complex, the δ dxy MO is occupied, causing fixed eclipsed comformation of the dimer. Bond order is 8 so M-M quadruple bond.

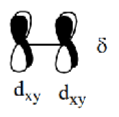
In the Os (d5) complex, the δ* dxy MO is also occupied, which cancels out the δ MO and allows rotation. Bond order is now 6 so M-M triple bond so staggered and eclipsed can both form.
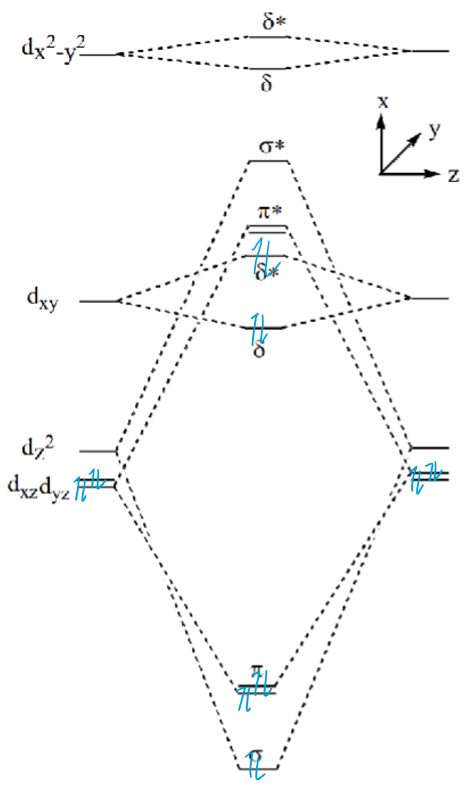
Draw molecular orbital diagrams to explain why d3 (M≡M) complexes can be eclipsed or staggered.
d3 complexes fill the bottom 3 MOs.
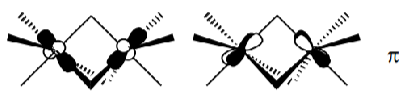
The dxy and dyz orbitals hybridise with dx2-y2 and dxy respectively, causing a tilt. They then overlap as above (with some δ character), giving the eclipsed conformation as the most stable.
Aligning these orbitals in a staggered conformation gives weaker π orbital overlap
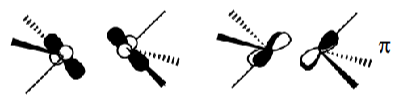
However, large steric bulk of ligands can cause the staggered conformation to be preferred anyway.
Explain the trend shown in this table
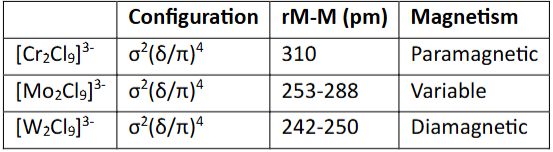
Cr → Mo → W is going down group 6 (d3)
This means that the metals are getting larger (3d → 4d → 5d).
Therefore, there is greater orbital overlap between the metals and thus the M≡M bond is shorter.
This allows the unpaired electron of each metal to pair together, hence making the dimer more diamagnetic.
Mo is variable because the M-M distance depends on the cation in the crystal
Give the expected and observed geometry of a quintuple bond.

Explain the purpose of the ligand in this complex:
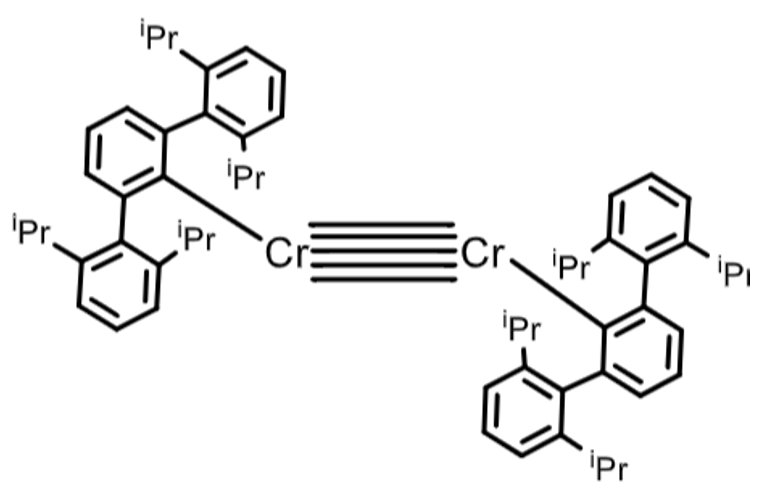
Very bulky terphenyl ligands protect the M centres, which allows the quintuple bond to form
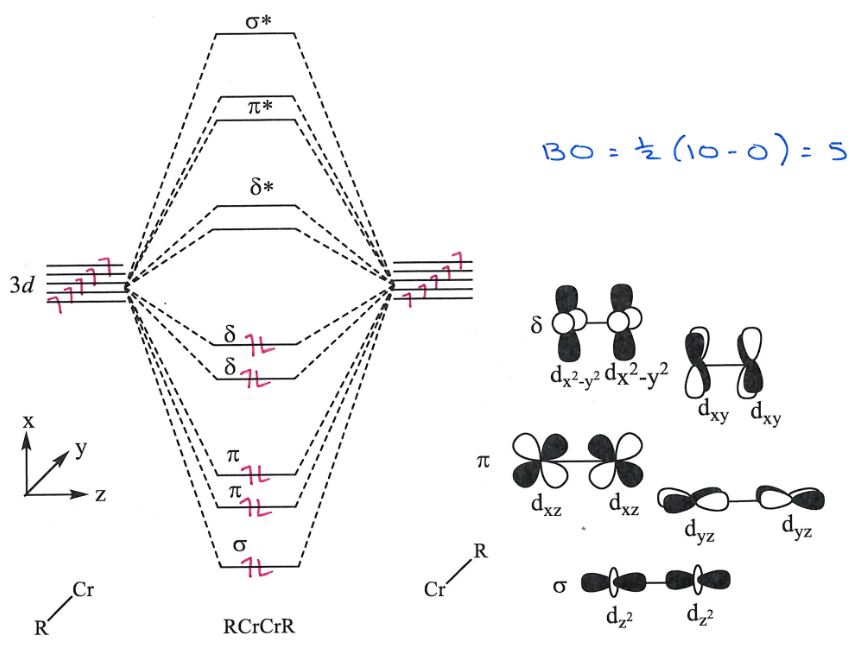
Draw a molecular orbtial diagram showing the bonding in this complex:

Draw the orbitals associated with each bonding orbital.
How do these complexes vary because of the differing ligands?

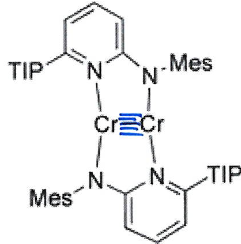
The second complex has a shorter Cr-Cr bond (175 pm) than the first (184 pm) because the ligand is bridging and rigid.
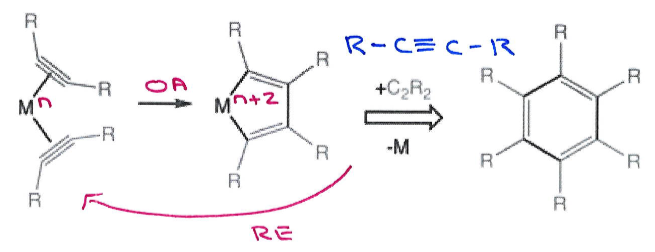
Give the general mechanism for metal-catalysed cyclotrimerisation.
Give the major and minor products of cyclotrimerisation catalysed by this complex:
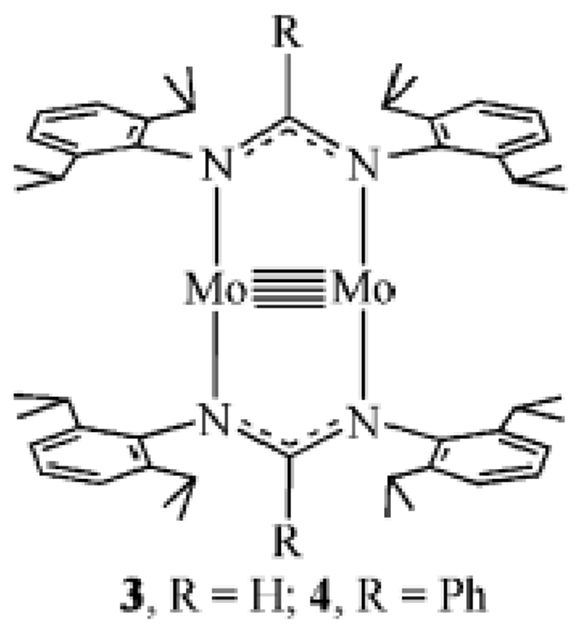
Major:
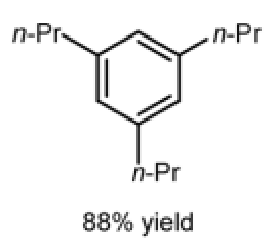
Minor:
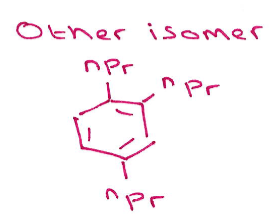
Give the structures of the 2 possible intermediate states formed during cyclotrimerisation using this catalyst:

Major intermediate:
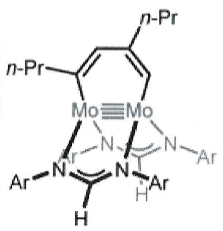
(which gives:)

Minor intermediate:
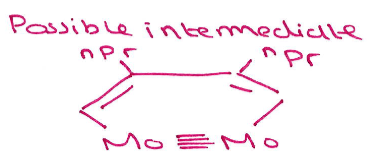
(which gives:)

What is significant about this complex and how is it formed?
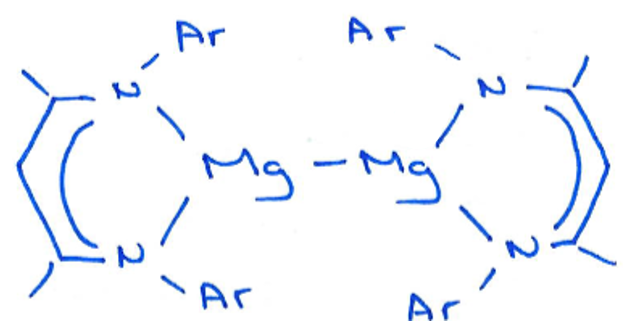
This was the first group 2 complex to contain the alkali metal in a +1 oxidation state.
It is produced by reduction of a +2 Mg complex by excess potassium:
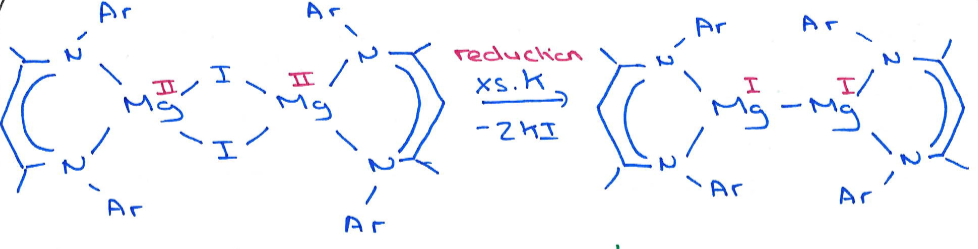
What is the inert pair effect?
The tendency for the outermost s electrons in larger elements to be inert from bonding, thus making lower oxidation states than the group oxidation state more stable.
The larger the element, the more significant the inert pair effect.
This is because, down the group, the preferred electronic ground state changes from triple to singlet, which pairs the electrons and causes them to have a greater ionisation energy.
In groups 13-15, it looks like this:
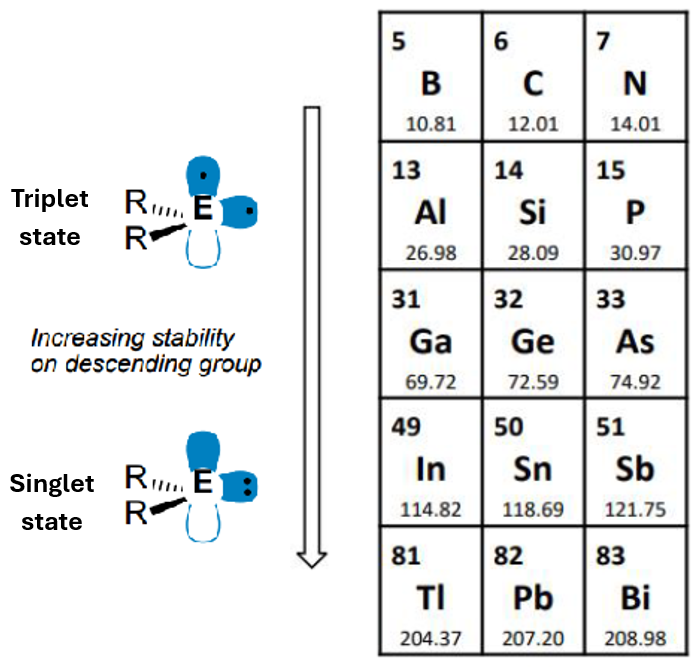
In the singlet state, the p block element is ambiphilic - it has a lone pair of electrons (Lewis base, nucleophilic) and a vacant p orbital (Lewis acid, electrophilic)
Describe how the inert pair effect impacts the stability of different p-block oxidation states.
The tendency for the outermost s electrons in larger elements to be inert from bonding, thus making lower oxidation states than the group oxidation state more stable.
For example, elements in group 3 become more stable in the +1 oxidation state as you go down the group (rather than +3)
The forward reaction becomes more significant:

Stability of these (+1) salts increases down the group:

Describe the impact of the inert pair effect on group 13-15 (p block) elements.
As you go down the groups, the singlet state becomes more stable than the triplet state.
This makes oxidation states lower than the group oxidation state more stable as the s electrons are harder to ionise.
It also makes that element ambiphilic (when in singlet state) - lone pair of electrons (Lewis base, nucleophilic) and a vacant p orbital (Lewis acid, electrophilic)

What is the double bond rule?
That mutiple bonds could not form between main group elements of principle quantum number greater than 2, ie. highlighted elements:
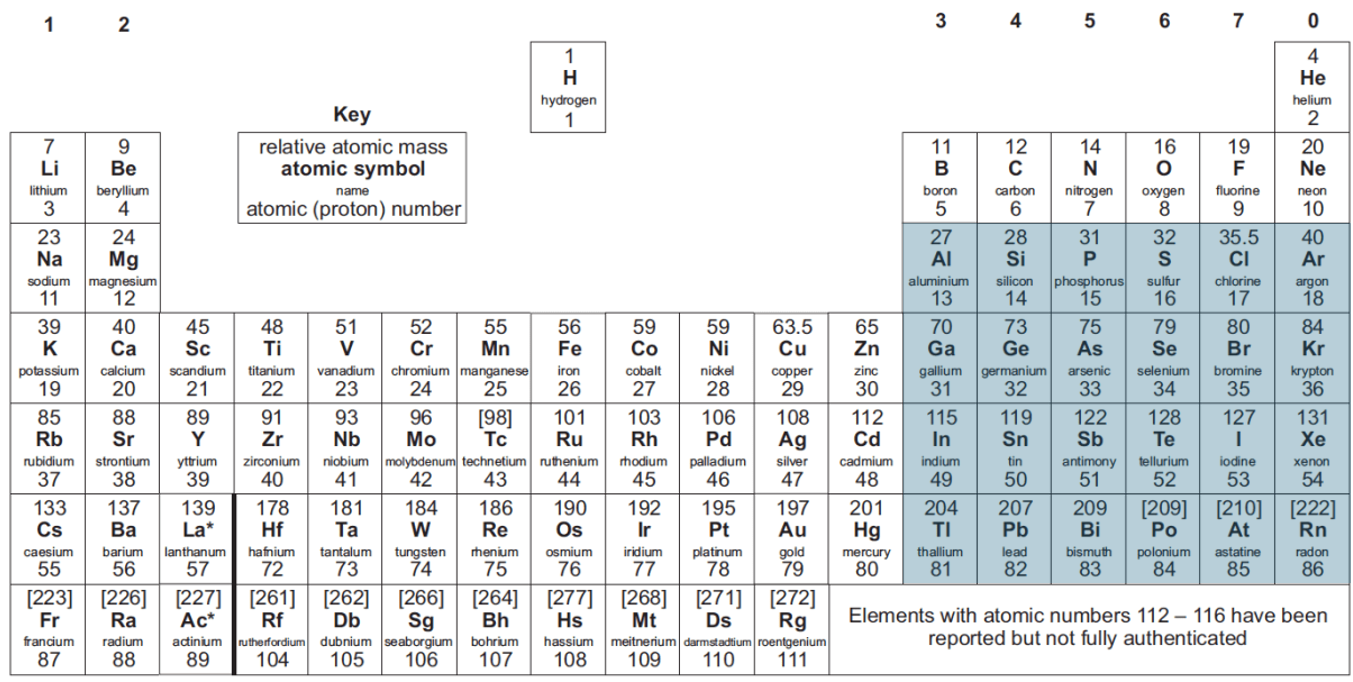
This is because, for these elements, having two σ bonds is stronger than having 1 σ and 1 π, and thus σ bonded polymers readily form.
This rule has been disproven now, provided that there is enough stabilisation.
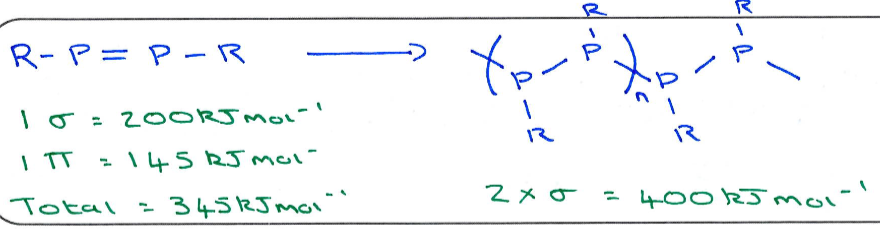
What do these bond energies demonstrate?
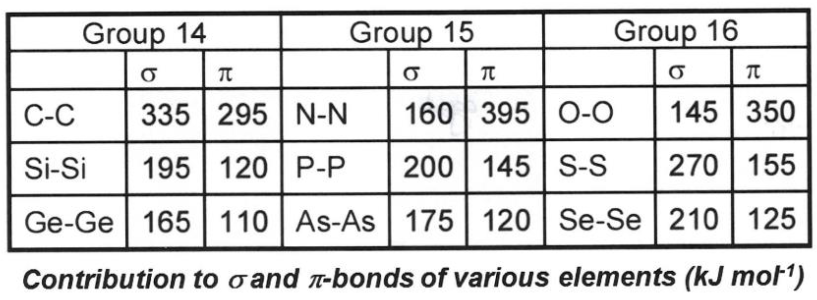
Shows that generally (for row 3 and beyond), pi bonds are weaker than sigma bonds.
This means that main group multiple bond complexes are difficult to form as polymerising to give two sigma bonds is favourable:

This is the Double Bond Rule. It has been broken in recent years but this is the basis for why stabilisation is required to make main group double bonded systems.
What do these bond energies demonstrate about N and O specifically?

N and O have larger π bond energies than σ so prefer to be in multiply bonded systems, unlike other main group elements (Double Bond Rule).
N2: 160 + 2(395) = 950 kJmol-1 - very strongly bonded, this explains why N2 is inert.
What are the products of this reaction? Why is this reaction significant?
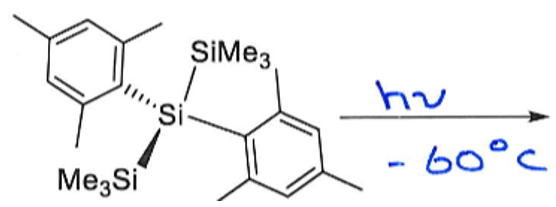
Give the oxidation state of Si in both reactant and products.
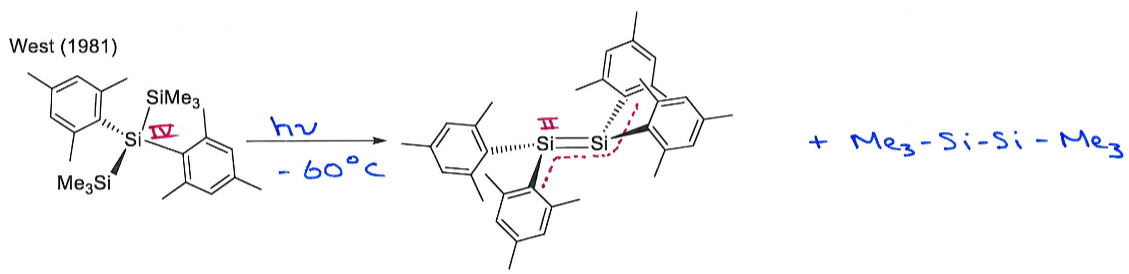
This reaction breaks the Double Bond Rule by having a main group element from row 3 multiply bonded. It is only possible because the bulky ligands provide kinetic stabilisation by shielding the double bond, preventing intermolecular collision.
What are the products of this reaction? Why is this reaction significant?
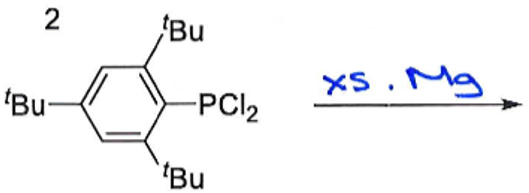
Give the oxidation state of P in both reactant and products.
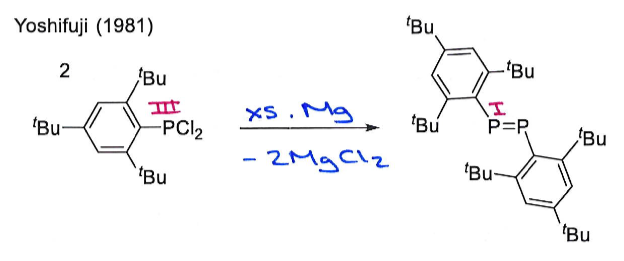
This reaction breaks the Double Bond Rule by having a main group element from row 3 multiply bonded. It is only possible because the bulky ligands provide kinetic stabilisation by shielding the double bond, preventing intermolecular collision.
What are the products of this reaction? Why is this reaction significant?
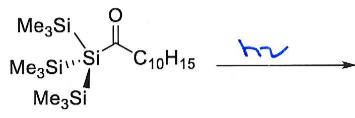
Give the oxidation state of Si in both reactant and products.
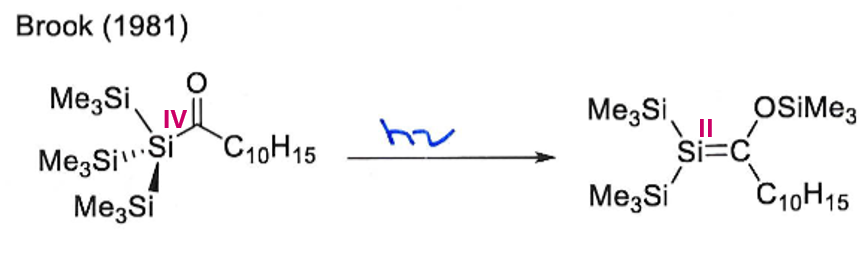
This reaction breaks the Double Bond Rule by having a main group element from row 3 multiply bonded. It is only possible because the bulky ligands provide kinetic stabilisation by shielding the double bond, preventing intermolecular collision.
What is the difference between main group doubly bonded systems and alkenes?
Alkenes are planar.
Main group doulble bonds are not.
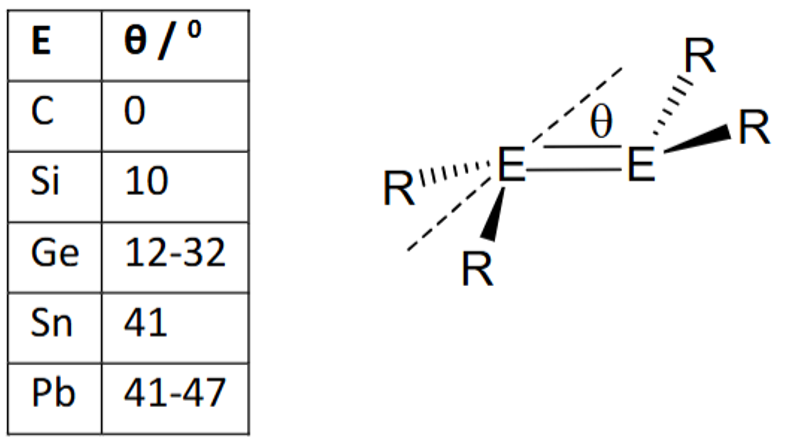
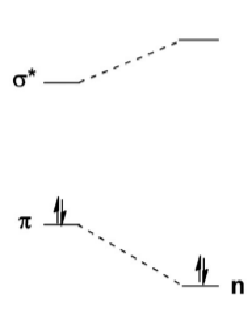
Use molecular orbitals to explain this trend in main group double bond geometries.

In C, the valence p orbitals are sp2 hybridised because of the small singlet-triplet energy gap (C is in triplet state):

Further down the group, the singlet-triplet energy gap becomes larger and elements begin to favour the singlet state much more.
This leads to more singlet-like bonding, as below:

Using this diagram, explain why the triplet state is preferred for carbon.
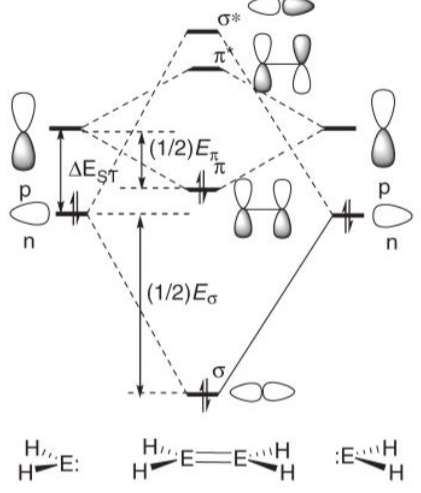
In C, the n-p energy gap is small so one electron occupies each orbital, giving the triplet state.

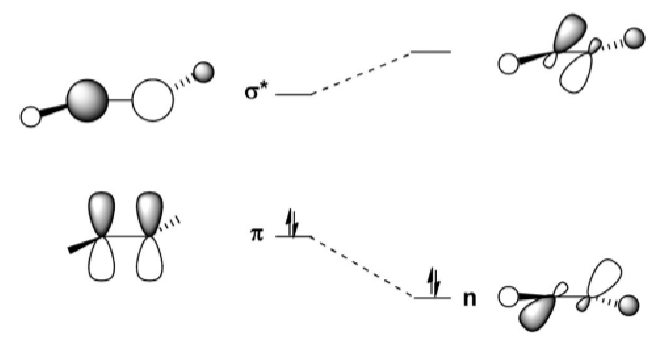
Using this diagram, explain how the geometries of main group double bonded systems changes down the group.

As you go down the group, the inert pair effect causes the singlet state to be more stable than the triplet state. This increases the ΔEST energy gap and also causes the σ* MO to decrease in energy, closer to the π MO.
This increases the σ*-π mixing, which creates 2 new Ms called n+ and n*. The n+ orbital is essentially the π orbital, but is now more stable because of the mixing with the σ*, which stabilises the complex in the trans-bent geometry.
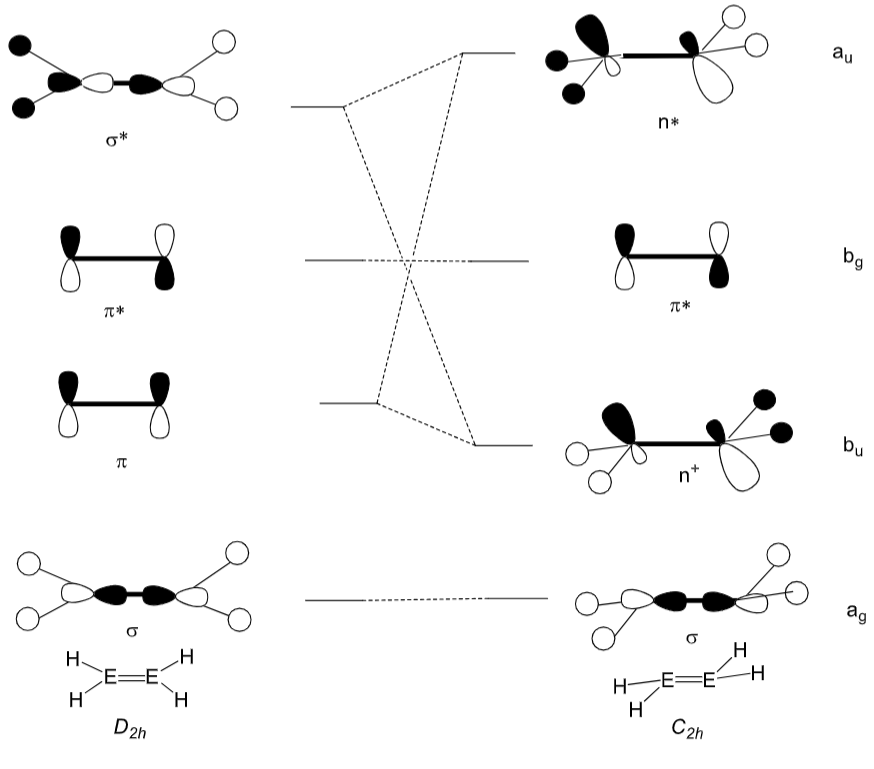
Draw the MOs for an alkene and main group double bond system shown in this MO diagram:

Most important to know is π, σ* and n+.
What are the general steps for preparation of a main group alkyne analogues (triple bonded)?
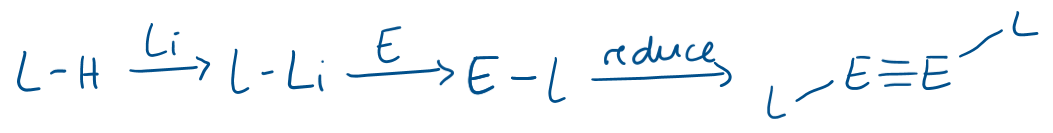
Where L is a very bulky ligand and E is the main group element (Ge, Sn, Pb)
The L-Li → E-L step is often salt metathesis (L-Li + ECl2 → E-L + LiCl).
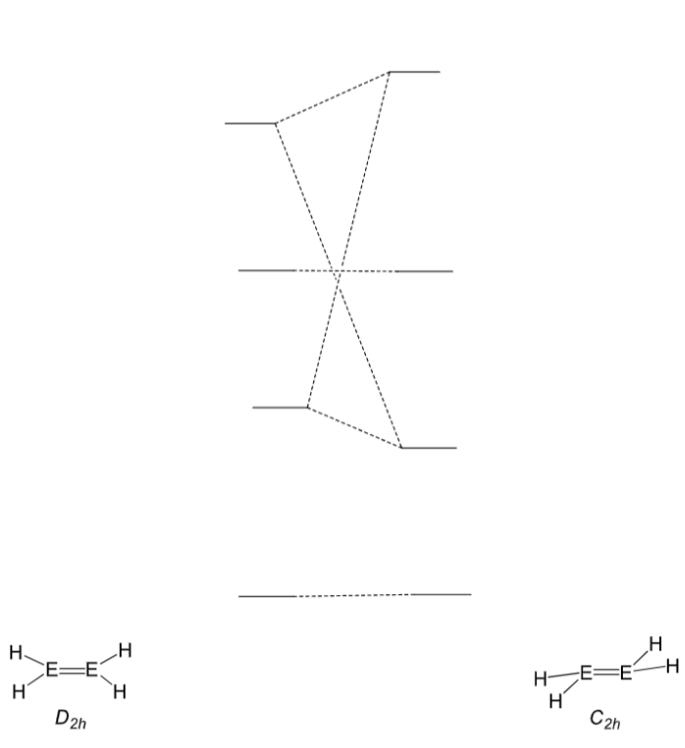
Draw the MOs for an alkene and main group triple bond system shown in this MO diagram:
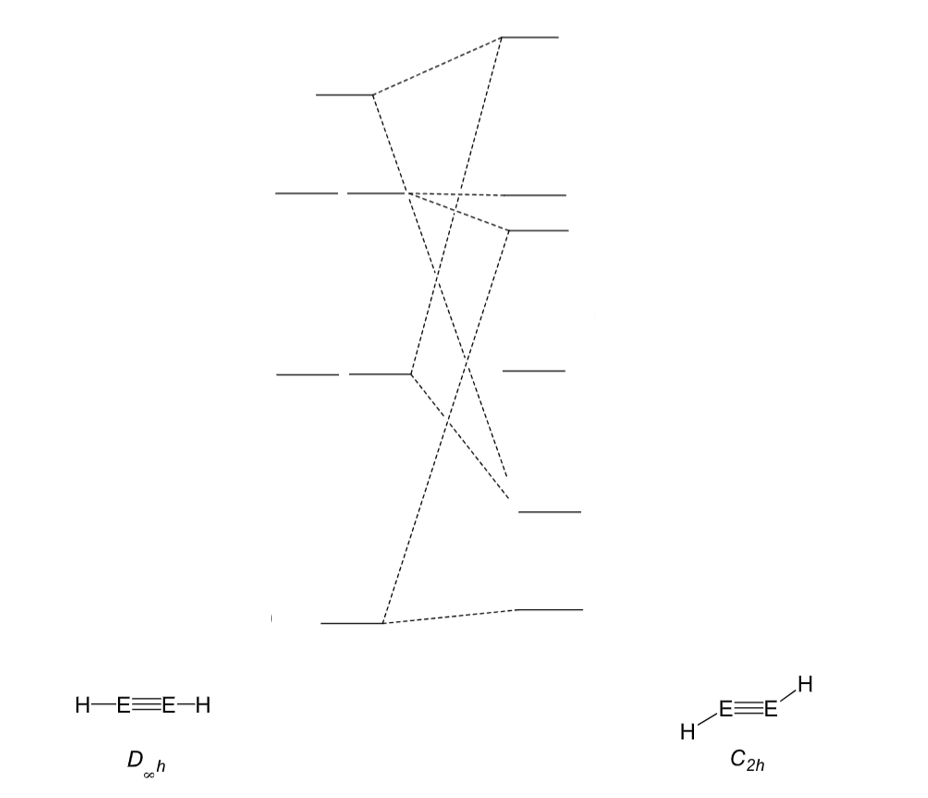
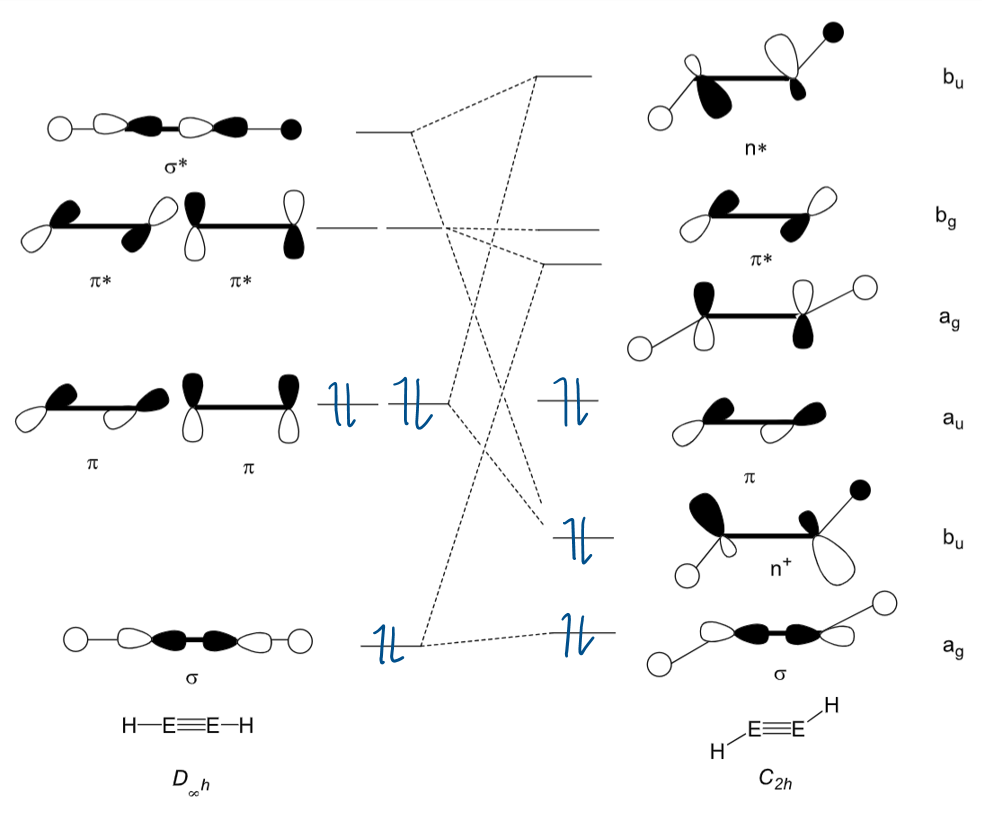
Key ones are n+ and n*
Give an example of a reducing agent used in this course.
KC8
Potassium graphite.
OR
Na napthalide
What reagent is required for this reaction?
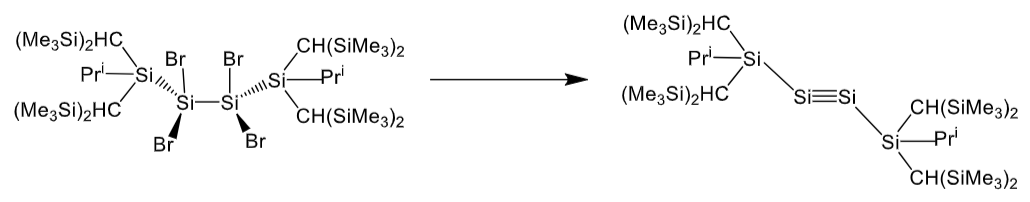
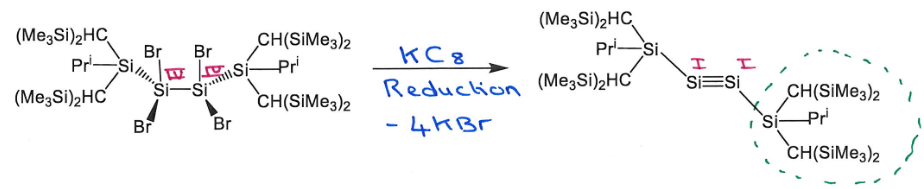
Reduction of SiIV to SiI so reducing agent - KC8 (potassium graphite)
What is the difference between main group triply bonded systems and alkynes?
In alkynes, the C is in the triplet state, so produces a linear triple bond:
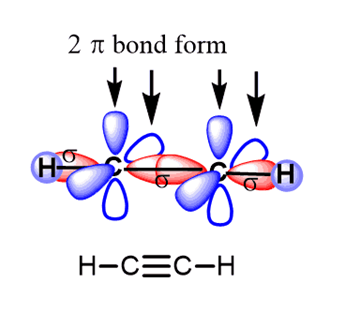
In (other) main group systems, the main group element is in the singlet state. The triply bonded system is therefore not linear:
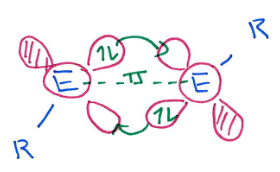
(The circles around the Es are p orbitals in and out if the plane)
How do main group multiple bond systems mimick transition metals?
Main group mutiple bonds have similarly energetically accessible frontier MOs to TMs which enable them to activate small molecules such as hydrogen.
However, they only readily undergo oxidative addition, and reductive elimination is more challenging. So traditional redox processes aren’t possible, but alternative processes are possible.
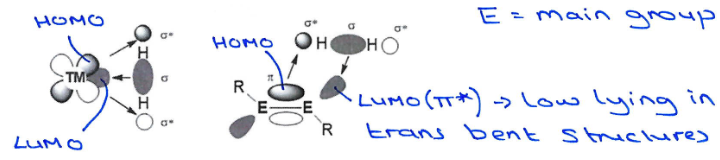
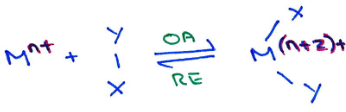
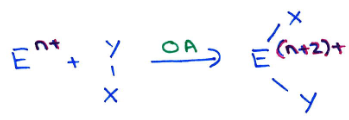
Why don’t main group multiply bonded systems fully mimick transition metals?
While main group systems can activate small molecules by oxidative addition, they become stable at their group oxidation state (E(n+2)+) and then don’t reductively eliminate.

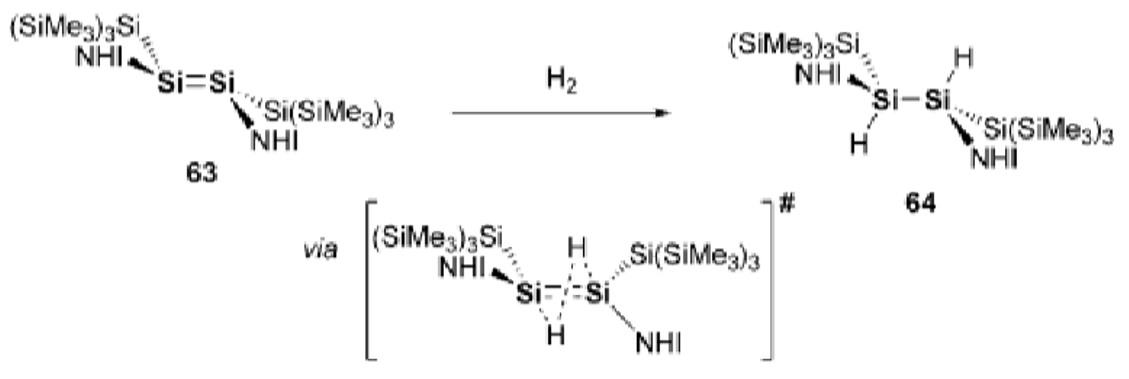
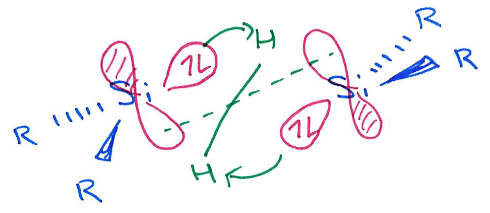
Explain the difference in H2 activation across an alkene vs main group double bond (eg. Si=Si).
In alkenes, H2 adds syn, in main group systems, the H2 adds anti.
This is due to the orientation that H2 approaches in in the transition state.
Give the reagents for eahc of these steps.
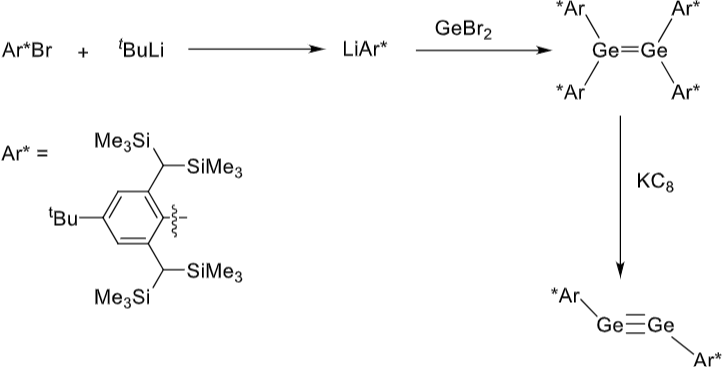
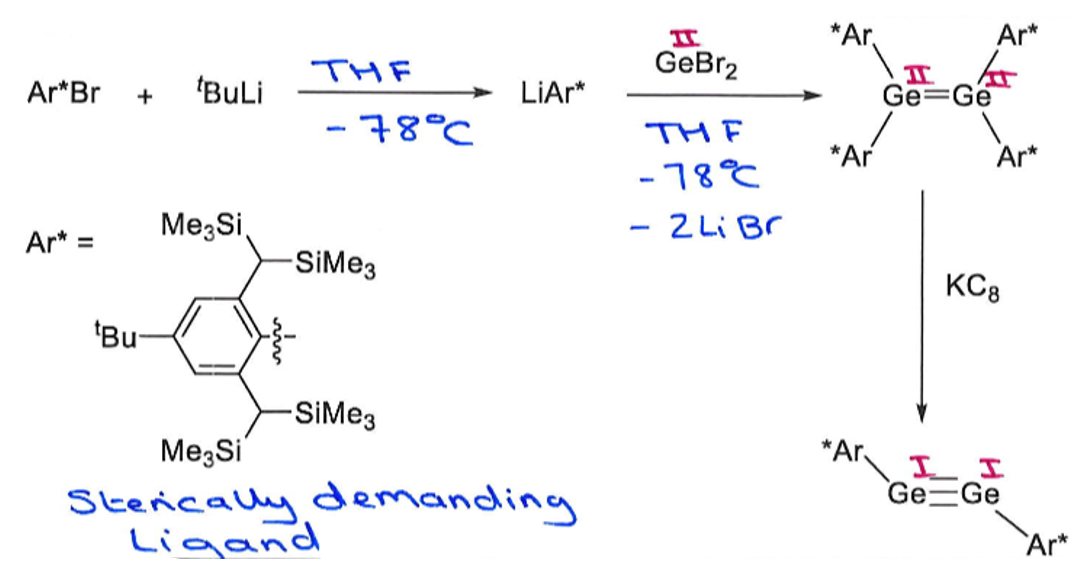
How does main group catalysed cyclotrimerisation differ from that with transition metals?
Key difference: Ge catalysed method gives a different major product to TM catalysed.
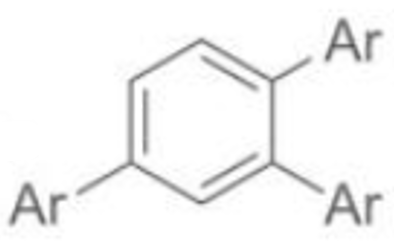
Gives 1,2,4 product as major product, whereas TM catalysed process gave 1,3,5 as major product.
Main group catalytic cycle involves a reversible step which is at equilibrium at 60oC.
Also involves the breaking of the Ge-Ge bond, which doesn’t happen in
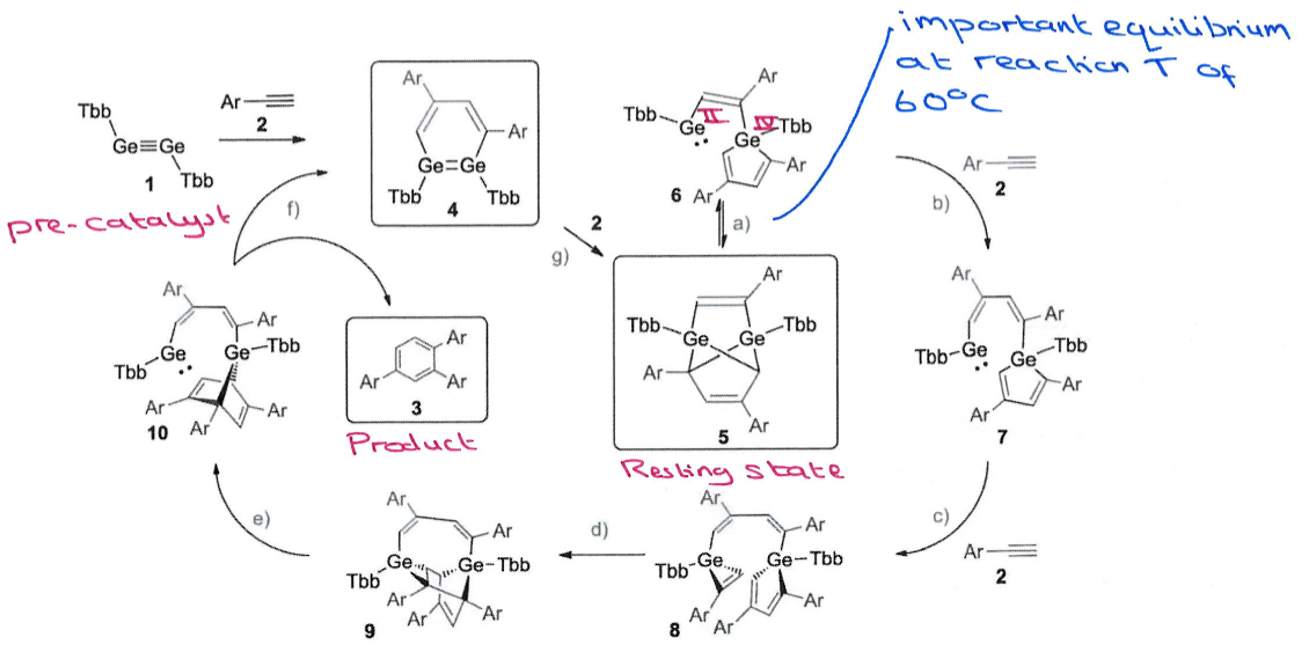
Ge complex 6 has a low valent Ge(II) centre which can activate another substrate molecule to push the reaction forwards.
Why do heavier group 14 mutiply bonded systems generally not perform well as catalysts?
They have weak E-E bonds and tend to dissociate in solution.
Draw the general structure of a phosphine and NHC ligand.
Explain the differences between them as ligands.
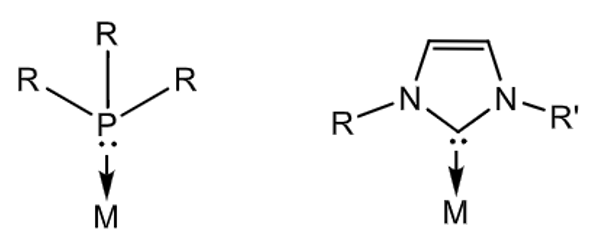
Substituents in phosphines are pointed away from the metal centre, towards the metal in NHCs
In phosphines, changes to sterics and electronic properties of the ligand occur simulaneously when changing R. In NHCs, you can tune sterics and electronics independently
NHCs are stronger σ-donors
π-backbonding from the metal to phosphine, little to none in NHCs
Explain the trend depicted in this graph
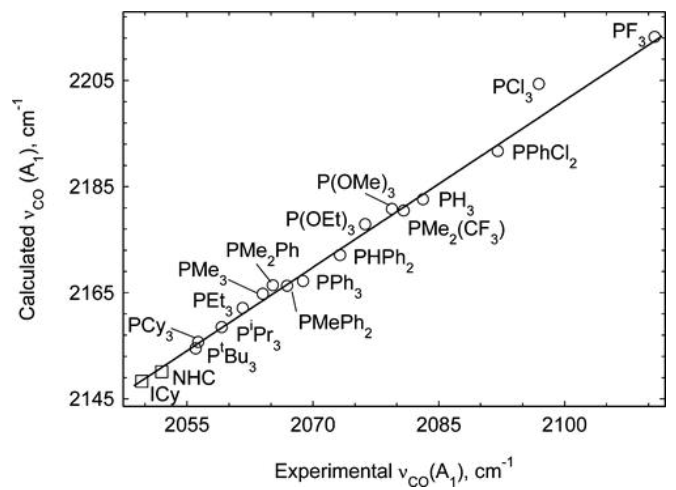
This graph shows the frequency of vibration of a CO ligand on a TM centre with various other ligands bound to the same centre.
Stronger σ donation leads to higher electron density on the metal centre.
This means more donation of electron density from the metal into the CO π* antibonding orbital, weakening the CO bond.
Weaker CO bond = vibrates at lower frequency.
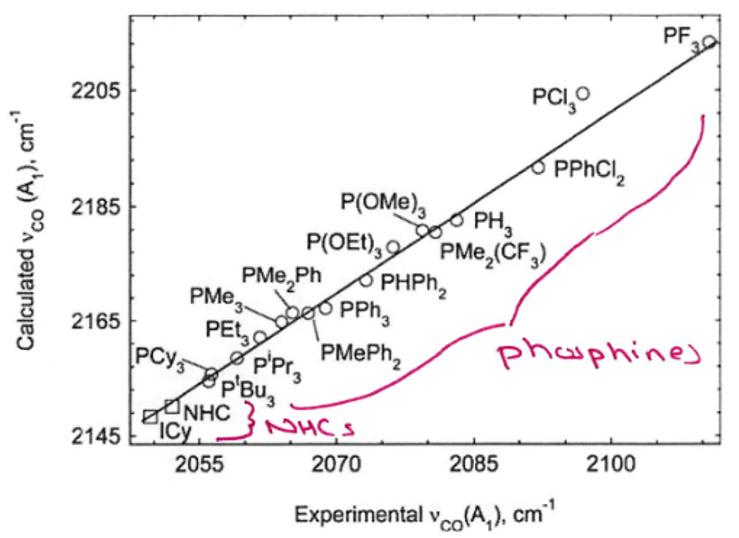
Therefore, this graph shows that NHCs are stronger σ donors than any phosphines.
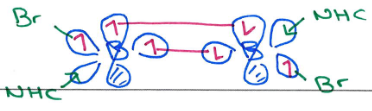
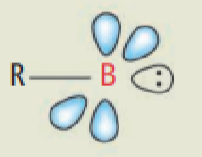
What state does B generally exist in?
What is the consequence of this?
B is a light element (row 2) so favours the triplet state.
B acts like C so B=B systems are planar like alkenes, and B≡B systems are linear like alkynes.
Draw the MOs on B that allow this species to exist
B is row 2 so exists in triplet state.
Gives the reagents required for this reaction
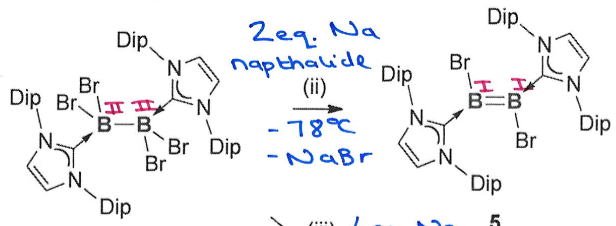
Requires 2 equivalents of reducing agent, eg. Na napthalide
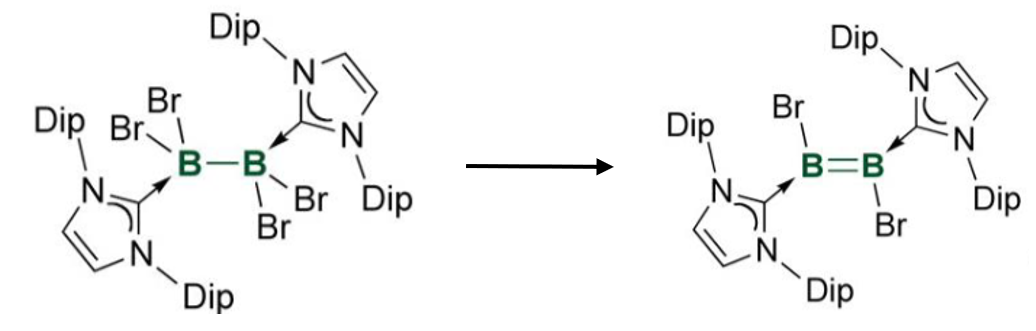
Gives the reagents required for this reaction
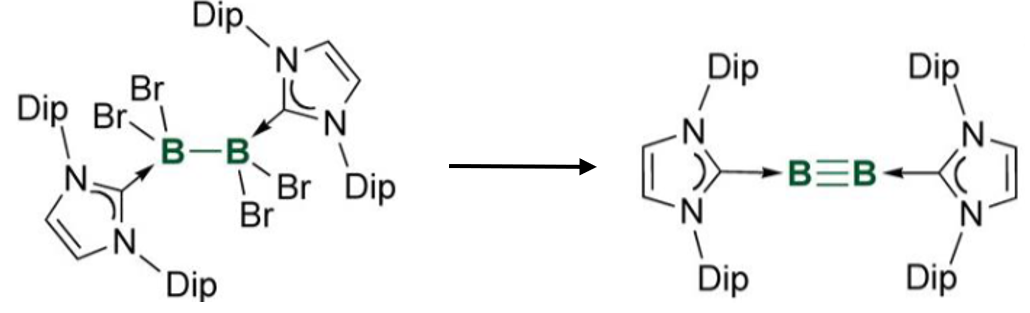
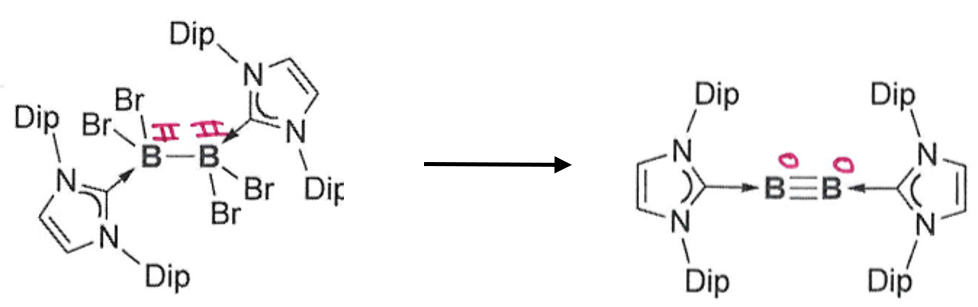
Requires 4 equivalents of reducing agent, eg. Na napthalide
What is comproportionation?
Give an example.
A reaction where two reactants containing the same element in different oxidation states react to form a single product with an intermediate oxidation state.
Boron Example:
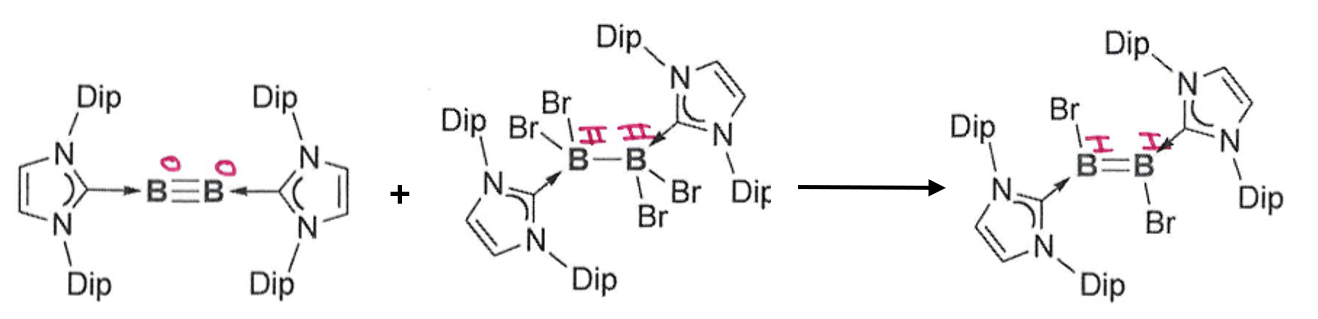
What is the key feature of these boron multiple bond systems?
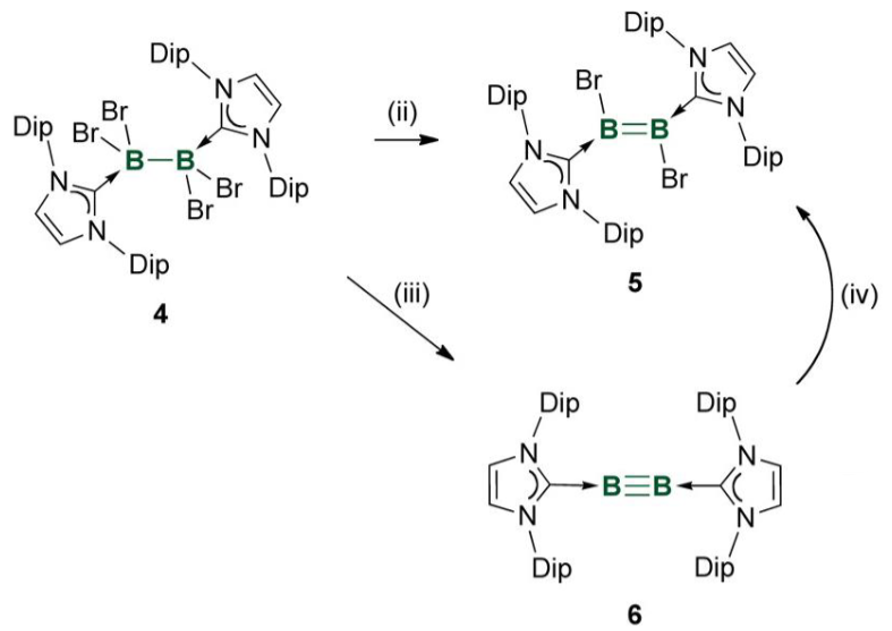
The NHC has very bulky substituents (Dip). This prevents collisions with the multiple bonds and subsequent intermolecular reactions.
What is the role of Na napthalide in chemical reactions?
Na napthalide is a reducing agent.
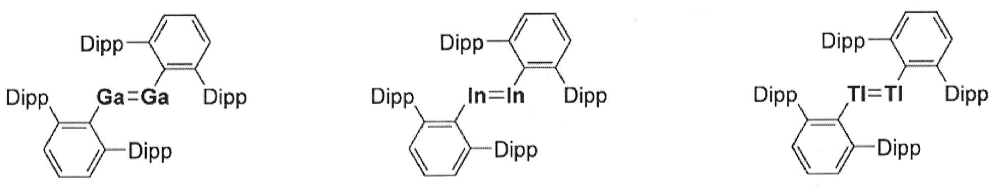
Explain how double bonding differs between B and heavier elements.
B: light element so favours triplet state, gives alkene-like geometry (planar)

Heavier elements: favour singlet state, NHC stabilisation no longer required, gives trans bent geometry of (X-type?) sterically bulky ligands
Examples:
Draw the MOs for lighter (eg. B) group 13 mutipy bonded systems and heavier ones.
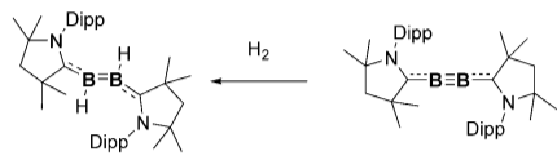
How can this molecule be altered to facilitate activation of H2?
Exchanging the carbene ligand for one that’s more σ donating and π accepting.
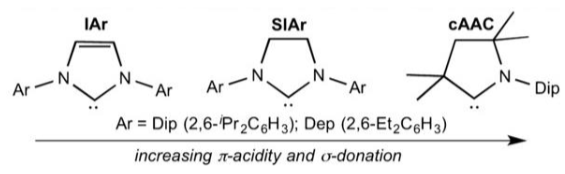
Explain what makes cAAC a strong σ donor.

The N lone pairs donate into the carbene carbon’s p orbital.
Removing one of the nitrogens reduces electron density in this orbital, allowing for more backbonding from the metal/main group centre.
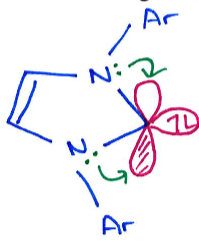
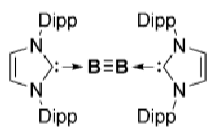
How does this boron complex activate H2 and CO?
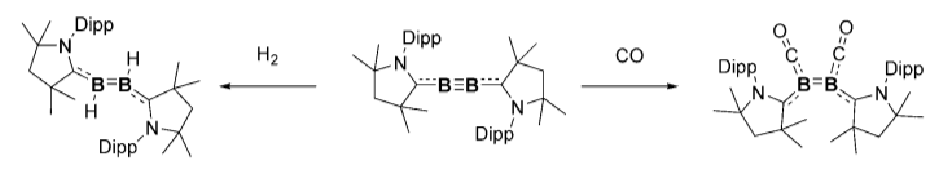
What is the difference between between these two boron complexes?
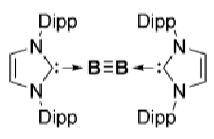
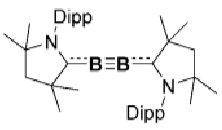
The first one doesn’t activate H2 because the B≡B bond is too strong.
In the second, there is more backbonding from the B to the carbene due to the loss of the nitrogen lone pair, weakening the B≡B bond and allowing it to activate H2.
Give the missing reagent for this reaction and explain its function.
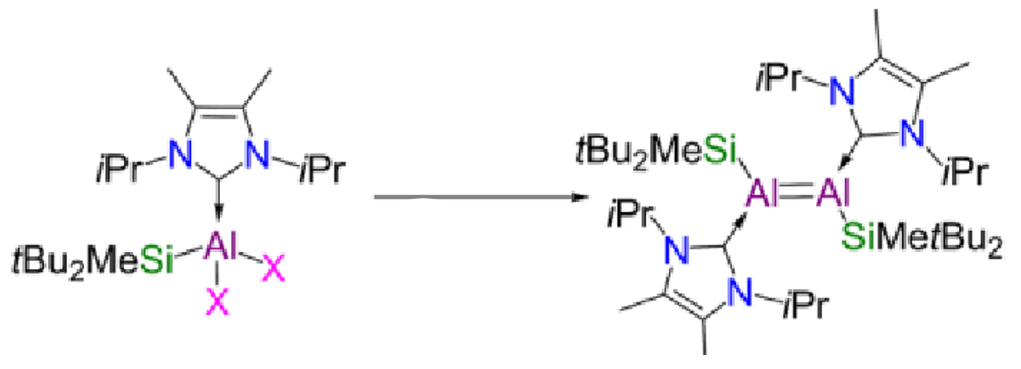
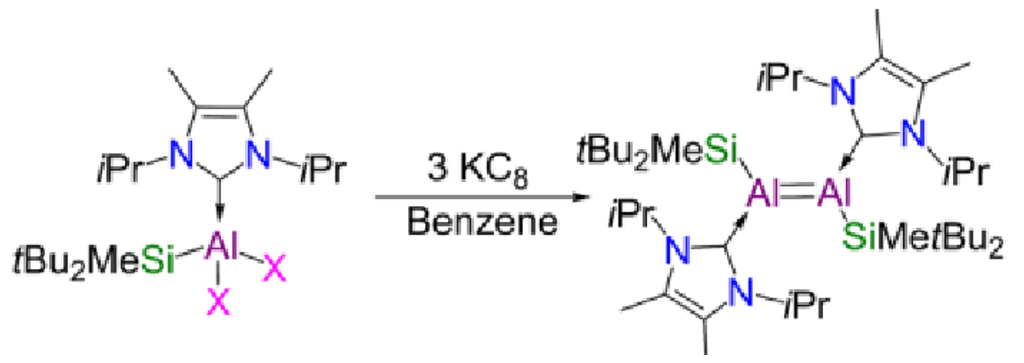
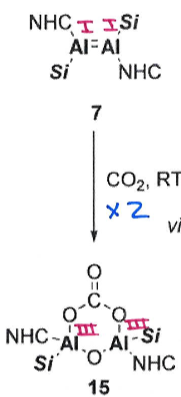
KC8 - potassium graphite
This is a reducing agent which aids the reduction from Al(III) to Al(I).
Give the important features of this catalytic cycle.
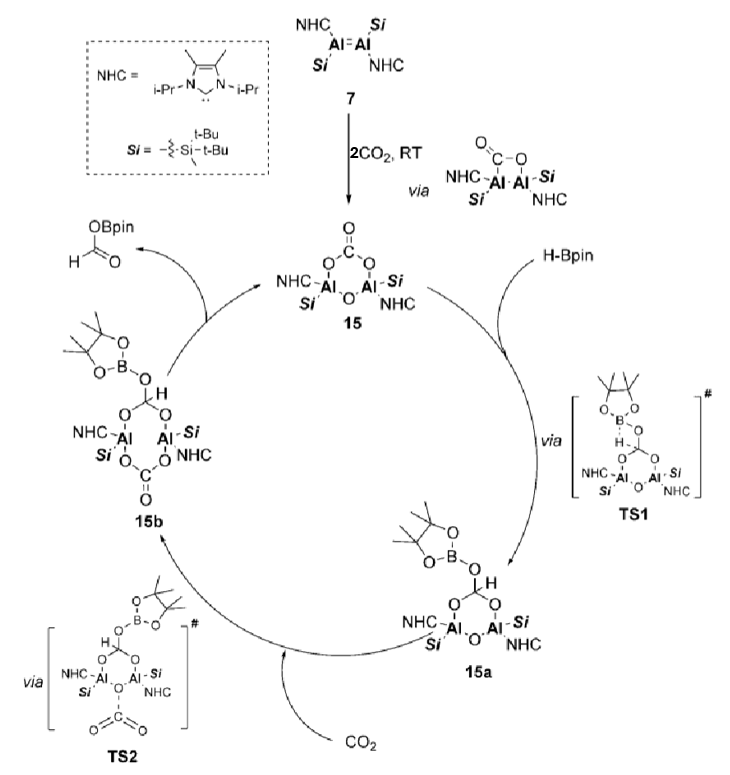
The cycle doesn’t involve an Al-Al bond, but the complex remains intact due to bridging units
This cycle fixates CO2 and then can be used to turn it into a carbonate
Al in the catalytic cycle is Al(III) in every step so no redox activity required, apart from initial fixing of CO2 from precatalyst:
Why is the valorisation of small molecules often difficult?
Small molecules (eg. H2O, CO2, N2, O2, NH3) are often highly thermodynamically stable.
Activation of small molecules requires a catalyst, typically transition metal based, but these are often toxic.
What does low-valent mean?
Low-valent means that the main group element is either in a low oxidation state and/or has a low coordination number.
This generally makes them reactive as they want to get to a higher oxidation state/coordination number as these are generally more stable.
Fill in the electrons and explain their positioning.
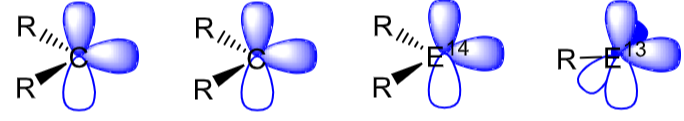
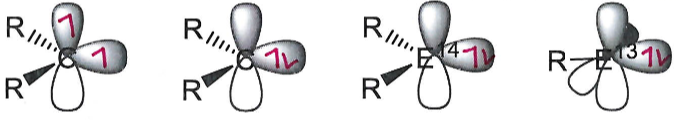
A carbene can be stable in both the singlet or triplet state. This is determined by the non bonding (lone pair) and p orbital energy gap (ΔE). Size of ΔE is dependent on R.

In carbenes where R = N, the N lone pair donates into the p orbital and stabilises the singlet state.
Heavier group 14 elements than carbon are only stable in the singlet state.
Low valent group 13 elements are only stable in the singlet state.
Singlet state main group centres are ambiphilic because of the lone pair (Lewis base) and empty p orbital (Lewis acid).
Draw the valence orbitals and explain their preferred state in each of these main group centres:
Carbene
Heavier group 14 element than C
Low valent group 13 element
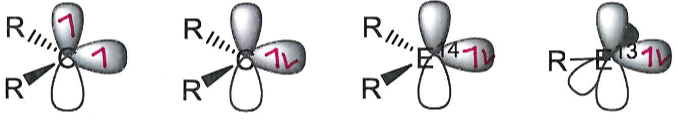
A carbene can be stable in both the singlet or triplet state. This is determined by the non bonding (lone pair) and p orbital energy gap (ΔE). Size of ΔE is dependent on R.

In carbenes where R = N, the N lone pair donates into the p orbital and stabilises the singlet state.
Heavier group 14 elements than carbon are only stable in the singlet state.
Low valent group 13 elements are only stable in the singlet state.
Describe the activity of a singlet state.
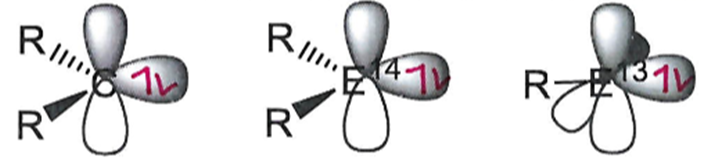
Singlet states are ambiphilic because of the lone pair (Lewis base) and empty p orbital (Lewis acid).
Name and describe these orbitals in this group 13 main group centre.
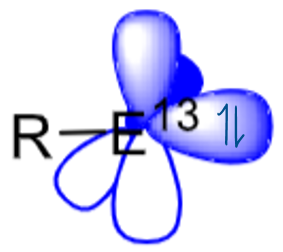

The presence of two unoccupied p orbitals makes group 13 centres even more electron deficient than carbenes/other group 14 centres.
This means that Lewis base stabilisation of these empty orbitals is required to isolate a group 13 complex.
Use molecular orbitals to explain why this boron complex is stable.
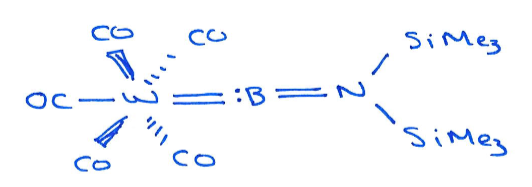
B is group 13, so is in a singlet state. It has one lone pair which it uses to coordinate to the tungsten.
The tungsten then back donates into the empty p orbitals (W0 so lots of available electron density to donate).
The N lone pair also donates into the empty p orbitals.
This provides enough Lewis base stabilisation to give a stable complex.
Use molecular orbitals to explain the stability of this complex.
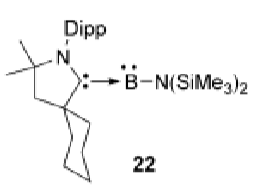
Boron is a group 13 element, so requires Lewis base stabilisation to give a stable compound (ie. donation of electrons into empty p orbitals)

However, Boron is electropositive and doesn’t like to be too electron rich.
Therefore, while the carbene lone pair donates into the empty p orbitals, the carbene’s empty p orbital also accepts electron density from the boron lone pair.
This is possible with cAAC carbenes because the is less electron density in the carbene p orbital because of the loss of nitrogen lone pair on one side.
The N lone pair from the other ligand also stabilises by donating into the empty B p orbitals.
(The result is a linear C-B-C system.)
Give the product of this reaction:
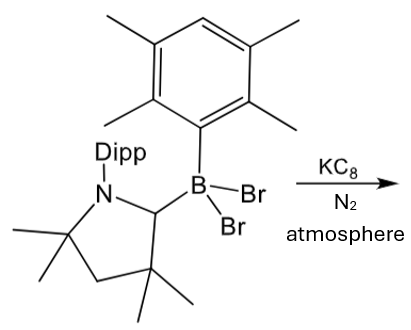
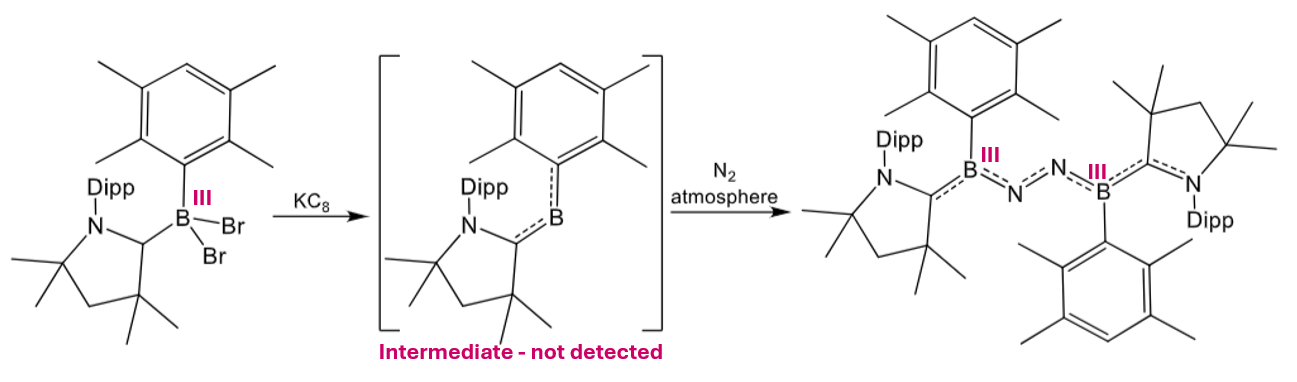
1st step is reduction by potassium graphite.
2nd step is fixation (and reduction) of N2.
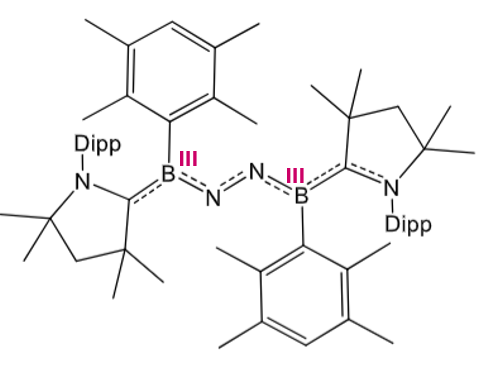
Use molecular orbitals to explain the fixation of N2 in this boron complex.
Boron valence orbitals (2s and 2×2p) hybridise to give three sp2 hybridised lobes in a trigonal planar geometry.
Remaining p orbital contains the lone pair.
Therefore, B orbitals look like this:

Therefore, 3 ligands give trigonal planar geometry around B and there is some pi character to all of the bonds across the middle.
(Remaining valence electron sits in sp2 hybrid orbital and pairs with negatively charged aromatic ligand)
Draw the molecular orbitals involved in TM and main group activation of N2 and explain the similarities and differences.
TM:
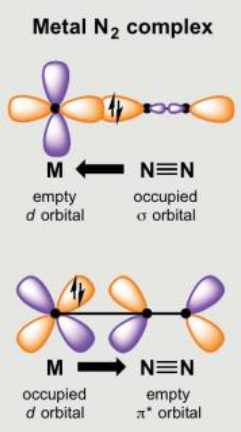
Main group (here its B, group 13):

Similar concepts of σ donation and π* backdonation, but different TM/main group orbitals involved. B gives unique ligand geometry to TMs due to sp orbital hybridisation (red = sp2 hybrids)
Explain the reasoning for the differences in N2 fixation by these two boron complexes.
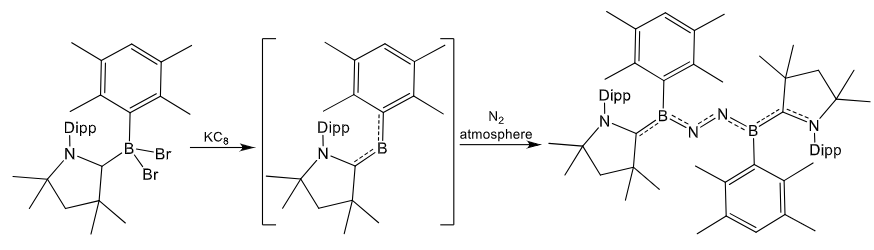
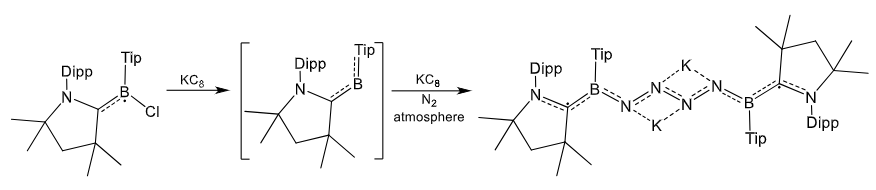
In the second example, the Tip ligand is too bulky for the B2N2 bridge to form.
Therefore, a planar K2N4 unit forms instead.
What happens to this species the presence of a reducing agent and excess cAAC?
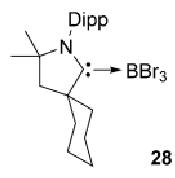
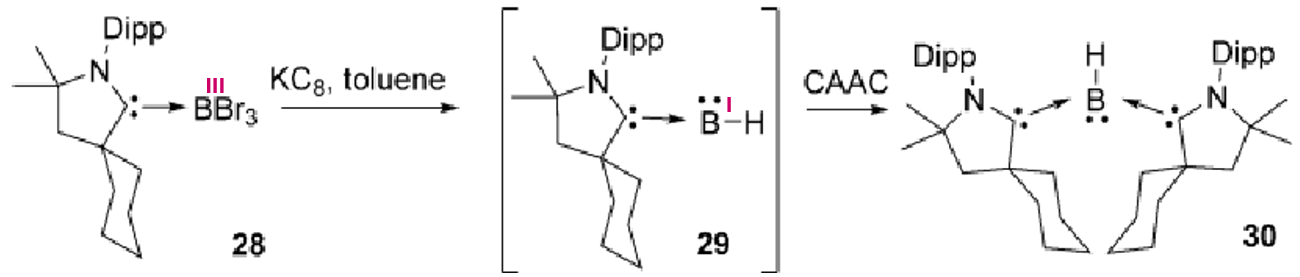
The intermediate complex would just dimerise in solution but excess cAAC allows formation of 30
Draw the molecular orbitals in a linear borylene species.
Draw the molecular orbitals on the boron in this species:
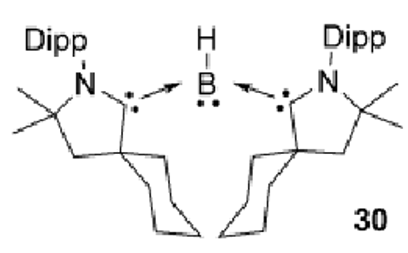
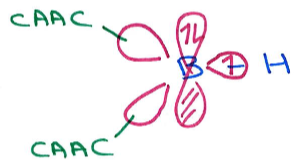
sp2 hybridised orbital gives trigonal planar ligand geometry.
Remaining p orbital contains the lone pair, making this boron a Lewis base (electron pair donor)
Explain this reaction:
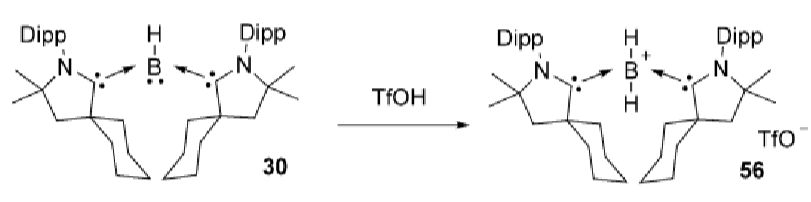
Boron in this species is a Lewis base (lone pair in p orbital):

This reaction proves this Lewis basicity by protonating the boron with triflic acid.
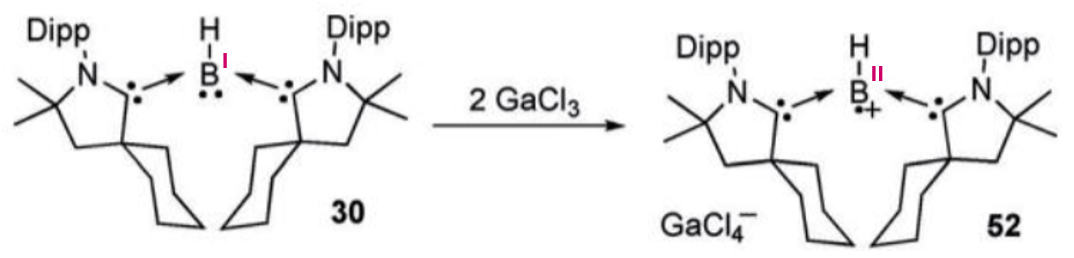
Give the product of this reaction:
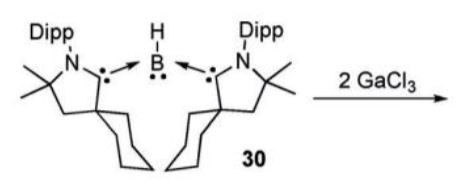
GaCl3 oxidises the boron to give a radical anion.
What is a borane?
What happens when you reduce a borane?
Boranes are a boron bonded to a hydride (B-H).
It produces a radical anion.

What is the function of GaCl3 in this course?
GaCl3 is a mild oxidising agent.
In this course it was demonstrated to oxidise boranes to give radical anions.

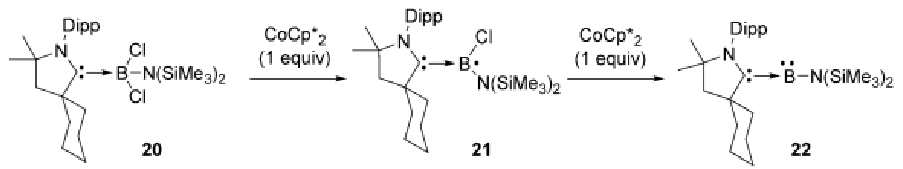
What is the function of CoCp2* in this course?
CoCp2* is a one electron reducing agent.
In this course it was shown to reduce a boron species to a boryl radical and then to a borylene (Lewis base boron)
Draw the molecular orbitals of the Al in this species:

Al (group 3) produces the same sp2 hybrid orbitals as boron.
However, unlike boron, the electrons are all in the hybrid orbitals, not the p orbital.
The unpaired electron pairs with the anionic NAcNAc ligand.
The paired electrons being in the hybrid orbital makes this Al act like a singlet carbene.
The Al is ambiphilic because of the empty p orbital (Lewis acid) and lone pair (Lewis base).
What is the result of Al being ambiphilic?
Its can form structures with itself:
These decompose at higher temperatures.
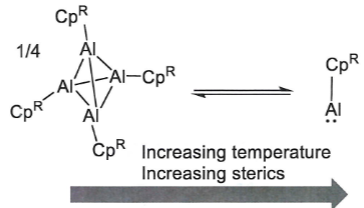
(CpR is just a Cp ligand with an R group)
What happens when these two Al complexes are added together?
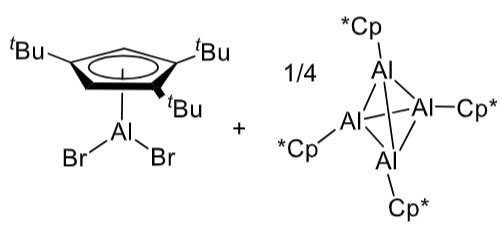
.
The tetrahderal complex can be thought of as a source of AlCp*.
The two Als swap ligands to reduce steric clash, then coordinate to each other. The Al(I) acts as a Lewis base (electron pair donor) and Al(III) as a Lewis acid.
Addition of cAAC ligand then replaces the Al(I) as the Lewis base giving:
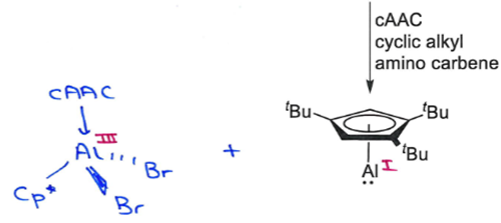
Al(I) in starting material, Al(III) in product - Al is oxidised.
O2 is reduced from O2 to 2O2-
Give the product of the reaction of this Al species with elemental sulfur:

Elemental sulfur:
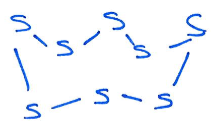
Product:
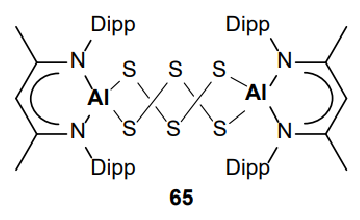
Al(I) in starting material, Al(III) in product - Al is oxidised.
Elemental sulfur (S8) is partially reduced to [S6]4-.
Give the product of the reaction of this Al species with tetraphosporus:

Tetraphosphorus - aka white phosphorus:
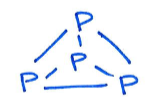
Product:
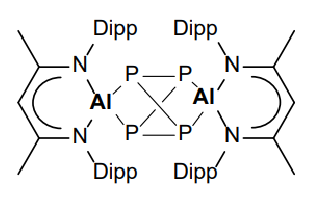
Al(I) in starting material, Al(III) in product - Al is oxidised.
Tetraphosphorus (P4) is partially reduced to [P4]4-.
Describe the difference in activity of B/Al species with heavier group 13 elements in singlet states.
Down the group, the singlet-triplet energy gap gets larger so the lone pair becomes more stable.
This means that heavier element species act more as nucleophiles through the lone pair than B/Al which tend to act more like electrophiles.
What is the product of this reaction?
Ga4I4 + LiAr →
Ga4I4 is a source of GaI.
These species undergo salt metathesis to give GaAr.
This can dimerise under certain conditions into
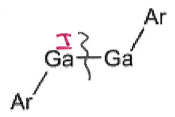
GaAr can then react in a disproportionation reaction with GaI to give GaIIArI + Ga0
2(GaIIArI) can then produce this product under certain conditions:
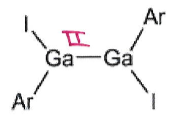
Note that she only expects you to be able to give the first salt metathesis product in an exam, second part is too complex.
What is the product of this reaction?
Ga4I4 + K(C5Me4R) →
Ga4I4 is a source of GaI.
Salt metathesis:
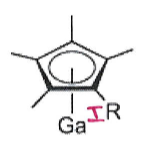
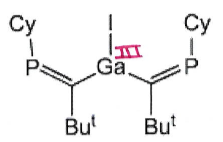
What is the product of this reaction?
Ga4I4 + CyP=C(tBu)MgCl →
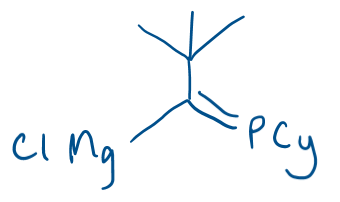
Ga4I4 is a source of GaI.
Reagent is a Grignard.
Product:
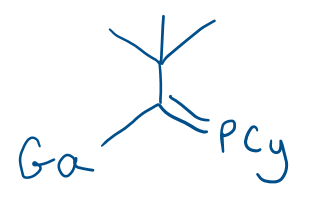
(Also gives MgClI)
Note: while not expected to be able to predict, this product can then go on to give product 28: