Rest of micro lectures
1/156
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
157 Terms
fomites
Inanimate objects that may carry microbial contamination
Fomites may be treated
with more aggressive control methods, or for longer time, to achieve lower levels of contamination
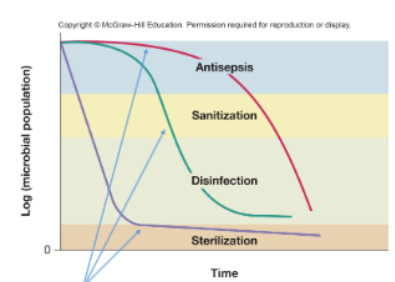
Disinfection
Reduces microbial load of an inanimate item through application of heat or antimicrobial chemical (chlorine, bleach, phenols, glutaraldehyde)
Used on objects and surfaces
Sanitization
Reduces microbial load of inanimate item safe public health levels through application of heat or antimicrobial chemicals (detergents containing phosphates, industrial strength cleaners containg quaternary ammonium compounds)
Used on objects and surfaces
Sterilization
Completely eliminated all vegetative cells, endospores, and viruses from inanimate item (pressurized steam, autoclave, chemicals, radiation)
Used on objects and surfaces
Antisepsis
Reduces microbial load on skin or tissue through application of an antimicrobial chemical (Boric acid, isopropyl alcohol, hydrogen peroxide, iodine)
Apply to living tissue and samples
Degerming
Reduces microbial load on skin or tissue through gentle to firm scrubbing and the use of mild chemicals (soap, alcohol swab)
Apply to living tissue and samples
Aseptic Technique
involves set of protocols that maintain sterility or asepsis, thus preventing contamination of the patient with microbes
Failure of aseptic technique
may put a clinical patient at risk for sepsis, a systematic inflammatory response to a systemic infection
Kill microbes
suffix -cide or -cidal
bactericides kill bacteria
viricides kill viruses
fungicides kill fungi
complete sterility
inhibit growth
suffix - stat or -static
bacteriostatic for bacteria
fungistatic for fungi
in infection, may allow immune system opportunity to clear an infection
Control microbial growth: Physical
Heat, radiation
Control microbial growth: Chemical
Gas, liquid
Control microbial growth: Mechanical removal
Filtration
Control microbial growth: Biological
Virus, toxin
Individual methods are typically tailored to work on
either objects or
living organisms
Treatments
dont kill microorganisms instantly
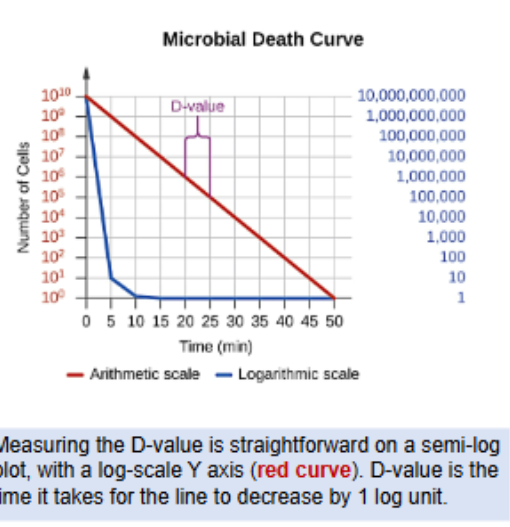
Killing is a probabilistic process
Microbial populations usually die exponentially
Eg, fixed percentage dies per unit time
Can plot on microbial death curve
straight line on semi-log plot
Efficacy of killing agent is measured by D-Value
Decimal reduction time
time to kill 90% = 1 Log10 Unit
Heat: Physical method
Function of temp. and time
Various microorganisms respond differently to high temps. Endospore formers such as Clostridium botulinum are more heat tolerant
Boiling does not kill all microbes
Incineration at very high temp. destroy all microorganisms
Heat: Autoclaves
rely on moist heat sterilization and raise temp. above the boiling point of water; considered most effective method of sterilization
Internal indicators are used to ensure sterilization
heat-sensitive autoclave tape
biological indicator spore test - endospores of the thermophile Geobacillus stearothermophilus to determine whether endospores were killed
Pasteurization
uses heat but does not render the food sterile; it reduces the number of spoilage-causing microbes while maintaining food quality.
LTH
low temp holding 65C 30 min
HTST
High temp short time
72oC - 15 seconds; lowers bacterial numbers while preserving the quality of the milk
UHT
Ultra high temp
138oC >2 seconds; UHT pasteurized milk can be stored for a long time in sealed containers without being refrigerated
Low temp inhibits microbial metabolism
slowing growth
Bacterial cultures and medical specimens
requiring long-term storage or transport are often frozen at ultra-low temperatures (dry ice -70°C or liquid nitrogen tanks -196°C)
Refrigeration
inhibits metabolism
Freezing
stops metabolism, may kill microbes
Pressure High
Denatures proteins and can cause cell lysis
Pressure: Food
kills microbes while maintaining food quality and extending shelf life. High pressure between 100 and 800 MPa (sea level is about 0.1 MPa) kills vegetative cells by denaturing proteins
Pressure: Clinical
hyperbaric oxygen therapy is used to treat infections; patient breathes pure oxygen at ~1 - 3 atmospheres (atm). Inhibits the growth of oxygen-sensitive or anaerobic bacteria
Removing water
slows or halts bacterial growth, without killing microbes.
Freeze-drying, or lyophilization
involves freezing and applying vacuum so that water is lost by sublimation. This combines both exposure to cold temperatures and desiccation
Water content/ water activity
can be lowered by adding solutes such as salts or sugars. At very high concentrations of salts or sugars, the amount of available water in microbial cells is reduced dramatically due to osmosis
Ionizing radiation
X-rays, gamma rays, and high-energy electron beams
• Penetrates cells, directly damages biological molecules. Causes DNA
mutations, leading to cell death.
• X-rays and gamma rays penetrate paper and plastic to sterilize packaged
materials.
Nonionizing radiation
Ultraviolet (UV) light
• Less energetic, less penetrating. Used for surface disinfection.
• Causes thymine dimers to form between adjacent thymines in DNA.
Filtration
• Physical separation of microbes from air or liquid.
• Uses filters with pores of specific sizes
• Ideal when liquids contain heat-sensitive components
High efficiency particulate air (HEPA)
filters have pores ~0.3 μm – filter out bacteria, endospores, and many viruses
• Efficiency of HEPA filters 99.97% for particles of 0.3 μm diameter or more
Membrane filters
Porous membranes with defined pore sizes
Microbes removed by physical screening
Cellular microbes ≥ 0.2 μm
Viruses ≥ 0.1 μm
Face masks
• Both surgical masks and KN95 respirators reduce the
outward particle emission rates by 90% and 74%
• For coughing, which produced the highest rates of particle
emission for of all expiratory activities tested, wearing
homemade masks considerably reduced the fraction of
large particles (> 0.8 μm)
chemical agents
chemically modify biological molecules, and can cause damage to proteins or DNA
preservatives act
by lowering the pH
inhibit metabolic pathways
important in microbes
Disk-Diffusion Method
or chemical agents
• Filter disks containing chemical placed on an agar plate inoculated with bacterium
• Compound diffuses and causes a zones of inhibition of microbial growth.
• Size of zone correlates with potency of compound
In-Use Test
determine whether an actively used solution of disinfectant in a clinical setting is microbially contaminated
Koch’s postulates
were the foundation of modern medical microbiology
• causative microbe must be present in diseased organisms, but not healthy organisms
• must be able to isolate the causative organism in pure culture
• must be able to infect a healthy organism with the isolated culture
• must be able to re-isolate microbe from experimentally infected organism
Pathogens
cause infectious disease in host organisms.
The host is a source of nutrients.
primary pathogens
almost always cause disease
• some Salmonella enterica strains; enterohemorrhagic E. coli
opportunistic
many common pathogens
• normally exist outside of the host (e.g., commensals)
• cause infection under the right circumstances
• age, weakened immune system, injury
• Staphylococcus epidermidis
some pathogens are obligate
• cannot exist outside host in the natural environment
• (perhaps can be cultured in the lab)
• Chlamydia, Rickettsia, Mycobacteria
reservoir
a natural population outside of the host
• drinking water supply, soil, etc.
• an animal population – e.g., zoonoses like rabies and influenza
• in organisms that spread the infection – e.g., arthropod vectors like mosquitos carrying malaria
WHERE PATHOGENS OFTEN EXIST
extracellular
• pathogens exist on or in host fluids and tissues, but do not enter host cells
• may move through circulatory system or migrate through the matrix between host cells
• can directly encounter elements of the immune system
•examples:
• E. coli
• Staphylococcus aureus
• Helicobacter pylori
• Borrela burgdorferi
intracellular
• microbes enter and multiply within host cells
• allows them to evade many elements of host immune system
examples:
• Listeria monocytogenes
• Mycobacterium tuberculosis
• Salmonella enterica
• Legionella pneumophila
infections have
a characteristic pattern of progression
Incubation period
pathogen entry, before symptoms
Prodromal stage
first onset if symptoms
Period of illness
disease is most severe, symptoms apparent
Period of decline
body fights off infection
Convalescence
symptoms resolve
Symptoms and disease trajectory
may be diagnostic
infectious dose 50 (ID50)
number of pathogens that will infect 50% of hosts in a specified time
Infectivity
• varies with pathogen, strain, etc.
• this is why antisepsis and disinfection are sufficient to reduce incidence of many infections
The dose of pathogens needed to bring on disease varies greatly – can be as few as 1
virulence
the intensity of pathogenicity – degree of harm to host
• may correlate with pathogen lifestyle
• opportunistic pathogens more likely to kill host
• obligate pathogens less likely to severely (or rapidly) harm host
virulence factors
facilitate infection, tissue invasion, or harm
• encoded by genes in the chromosome or on plasmids
• may determine whether opportunistic pathogens can cause infection
• can be acquired by horizontal gene transfer
Genes encoding virulence factors are often found in the
chromosome clustered within
pathogenicity islands
pathogenicity islands
correlate with pathogenicity
• absent in related but non-pathogenic strains or species
• may be transferred by HGT
• sequencing of these regions can determine if strain is virulent
pathogenicity islands encode common phenotypes
• toxin secretion
• pilus, or other features for attachment to host
• iron uptake
• biofilm formation
adherence
once a pathogen has gained entry to host, it must adhere somewhere
• typically recognizes specific host molecules
• mediated by adhesins – typically pili or surface proteins
• in pathogenic E. coli, adhesins target bacteria to sites of infection
• diarrhea: fimbriae bind sugars on intestinal epithelium
• hemolytic uremia: pili bind sugars on kidney cells
• urinary tract infection: pili bind sugars on urethral epithelium
invasiveness
ability to spread to adjacent tissues
secreted exoenzymes may aid spreading:
• break down extracellular matrix
• collagenase breaks down collagen
• hyaluronidase breaks down hyaluronic acid
• degrade carbohydrate-protein matrix between cells
• can also disrupt host cell surface
entry to blood or lymph: bacteremia
presence of bacteria in the blood
entry to blood or lymph: septicemia
pathogens or their toxins in the blood
Exotoxins
factors secreted by bacteria to cause damage to the host
• may induce tissue damage, aiding invasion
• may cause host cell lysis, releasing nutrients
channel-forming toxins
• self-assemble into pores in host-cell plasma membrane
• cause host cell lysis
AB toxins
• A and B components form a complex
• B component attaches to host cell, triggers endocytosis of AB complex
• A component is released, causes toxicity
(many ways to cause cell death)
anthrax
Anthrax toxin
A and B toxin components produced and secreted separately by Bacillus anthrax
Toxin has 3 constituent proteins
• (B) PA – protective antigen
• (A) EF – edema factor
• (A) LF – lethal factor
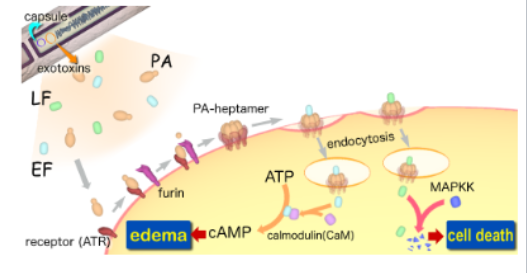
PA
assembles into a pore on the surface of the host cell
Either “A” toxin docks onto the PA pore
d is carried into the cell
lipopolysacchraride
endotoxin
LPS decorating the outer membrane of Gram-negative bacteria can be toxic
• LPS is not secreted, but either shed or released from dying bacteria
lipid A
Toxic
• it potently triggers a blood clotting factor, causing blood clotting
• septic shock
• also triggers fever, other symptoms
endotoxin must be
in the body to trigger these effects
• digestive tract is full of Gram-negative bacteria that do not trigger septic shock
intravenous drugs
must be free of even trace amounts of lipid A
Endotoxin: limulus amebocyte lysate assay
• Horseshoe crab blood cells contain a clotting factor (coagulogen) that responds to endotoxin
• In the presence of endotoxin, coagulogen clots into a gel
• Turns out this is an incredibly sensitive assay
coagulation
endotoxin contamination
no coagulation:
no contamination
Many extracellular pathogens can form biofilms
• in lungs of cystic fibrosis patients
• on implants and catheters
• in chronic wounds
Genes for biofilm formation are associated with
virulence
Pathogen biofilms:
• are associated with chronic infection
• increases virulence
• reduce sensitivity to antibiotics
• form a structural barrier from host immune cells
Biofilms may aid
attachment and invasion
Biofilms may also protect bacteria from immune cells
Immune system clears
most microbes
Many successful pathogens evade immune system
infect immune system cells
diminishing function
• Legionella, Mycobacteria
directly infect adjacent cells
evade antimicrobial peptides and immune cells
• Listeria, Rickettsia
capsules
prevent phagocytosis
• Streptococcus
minimize expression of antigens
Borrelia, Neisseria gonorrhoeae
Listeria monocytogenes
• Gram positive, intracellular pathogen
• virulent food-born pathogen
• meningitis, sepsis, stillbirth
Listeria monocytogenes binds to cell surface
and stimulates endocytosis, even in non-lymphocytes
• evades extracellular immune system components
• secretes enzymes that allow it to destroy vacuolar membrane, escape into cytosol
hijacks the actin cytoskeleton
• stimulates actin polymerization
• propels bacterium around in host cell
termed actin gliding
• actin gliding can project Listeria into adjacent cell
Legionella pneumophila
• Gram negative, intracellular pathogen
• accidental human pathogen – normally infects amoeba and protists
• humans are a dead-end host
• Legionella responds to macrophages as if they were amoeba
Legionella pneumophila endocytosed by host cell
• does not escape vacuolar compartment
• creates a special type 4 secretion system
• secretes hundreds of effector proteins that modulate host cell
• disrupts normal vacuolar trafficking to lysosome
• recruits proteins and lipids to disguise surface of its vacuole
• creates a safe niche for itself inside host cell, where it multiplies