Proteins
1/29
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
30 Terms
Proteins are _______ made up of _______ _____ joined by ________ _____
polymers, amino acids, peptide bonds
Main functions of proteins (6)
Enzymes 🌀
Structural proteins 🔨
Contractile proteins 💪
Hormones 🌸
Transport proteins 🚚
Defence proteins 🛡️
enzyme example
amylase
structural proteins example
keratin, found in skin, hair, nails
contractile proteins example
actin and myosin in muscles
hormones example
insulin and growth hormones
transport proteins example
hemoglobin, which transports oxygen
defence proteins example
antibodies that protect the body against disease
all proteins are made of the ______ set of ___ amino acids, connected in different ___________
SAME, 20, combinations
Components of a a general amino acid
Amino group
Carboxyl group
Side chain (R)
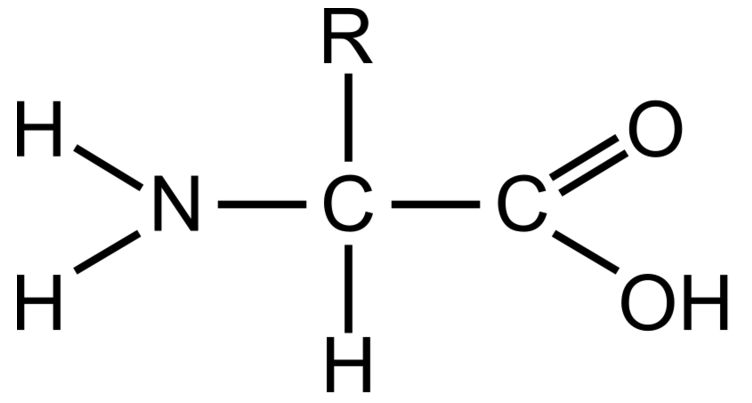
Amino acids’ ____________ and thus ___________ are determined by:
properties, classification, those of their side chain (R)
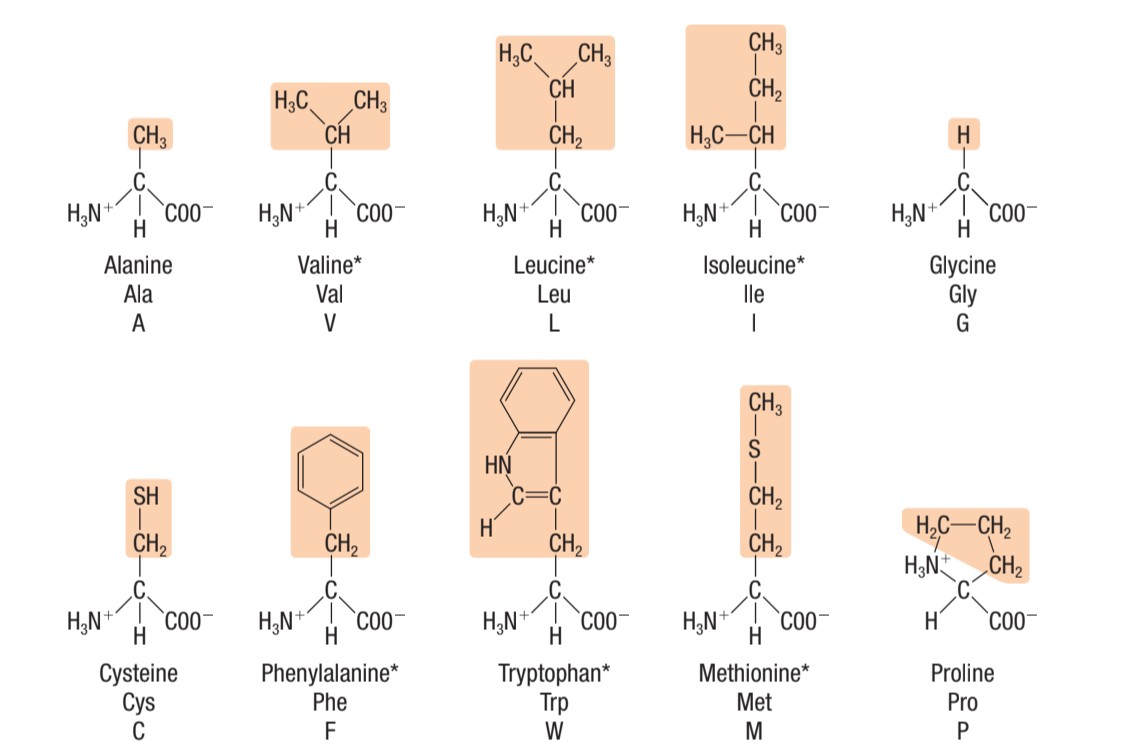
Identify this type of amino acid
non-polar amino acids
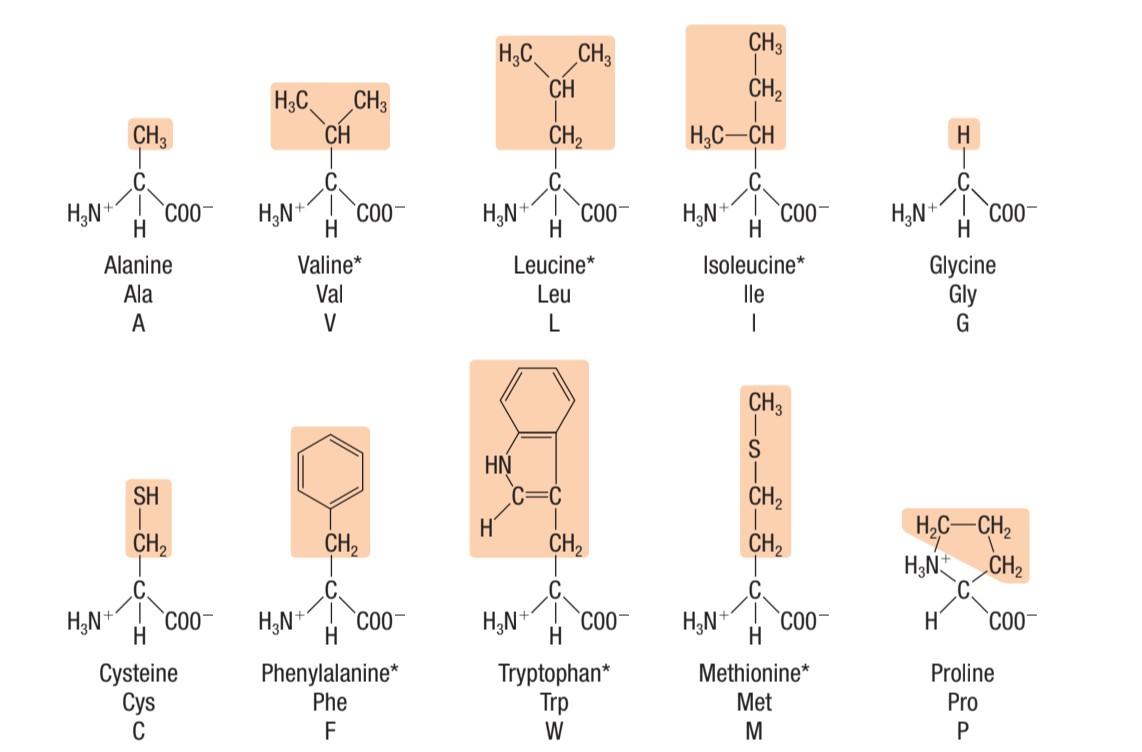
Identify this type of amino acid
polar amino acids (uncharged)
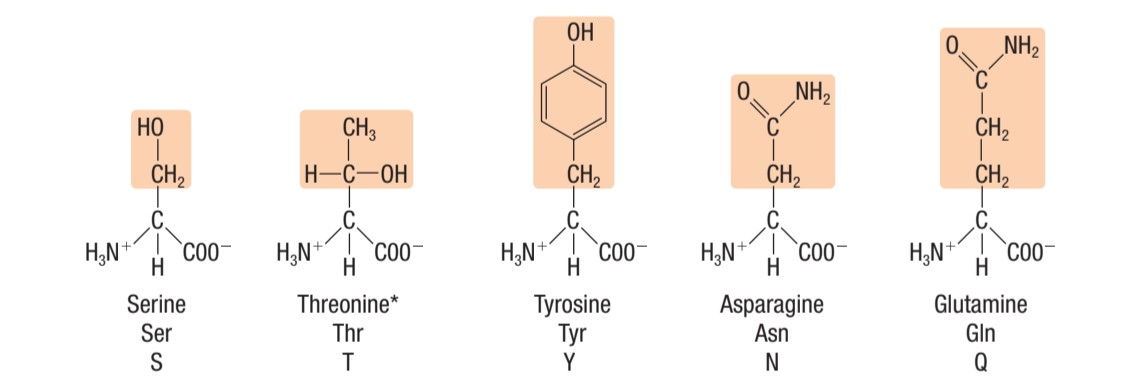
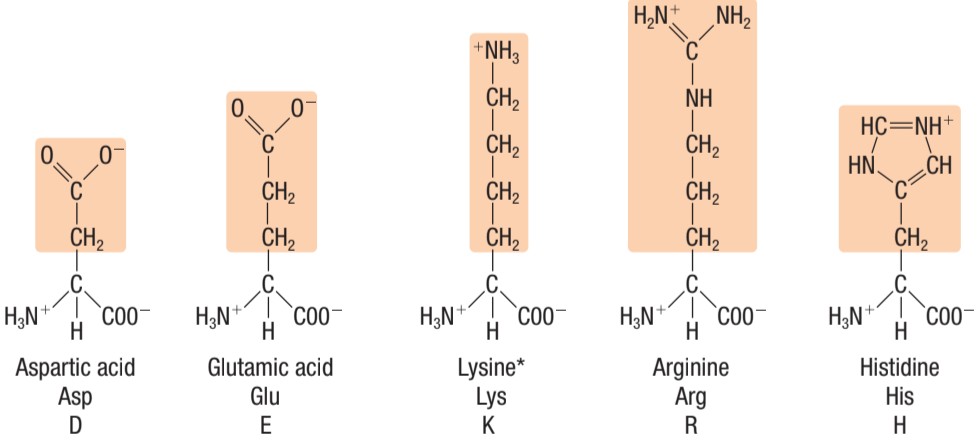
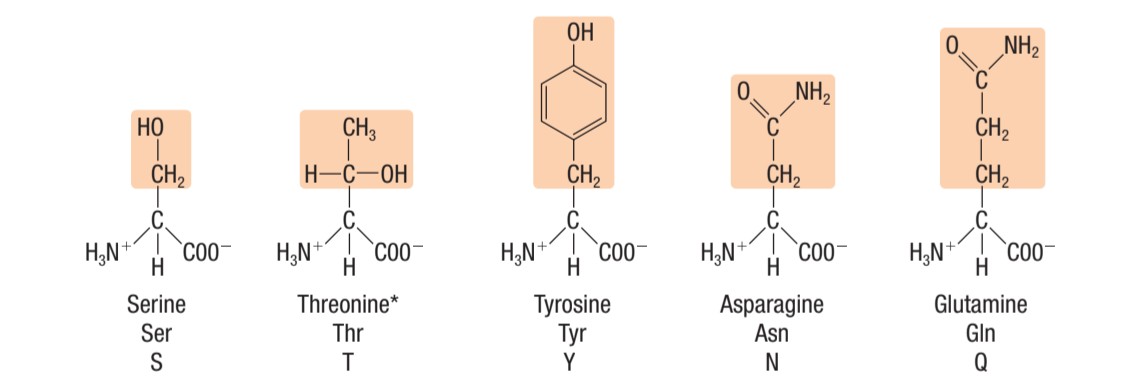
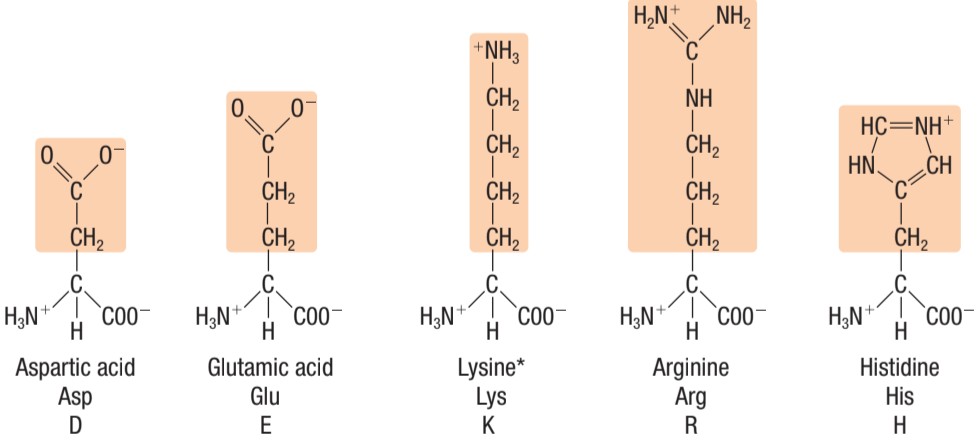
Identify this type of amino acid
LEFT: negatively charged (acidic) amino acids
RIGHT: positively charged (basic) amino acids
4 structure levels of proteins
Primary
Secondary
Tertiary
Quaternary
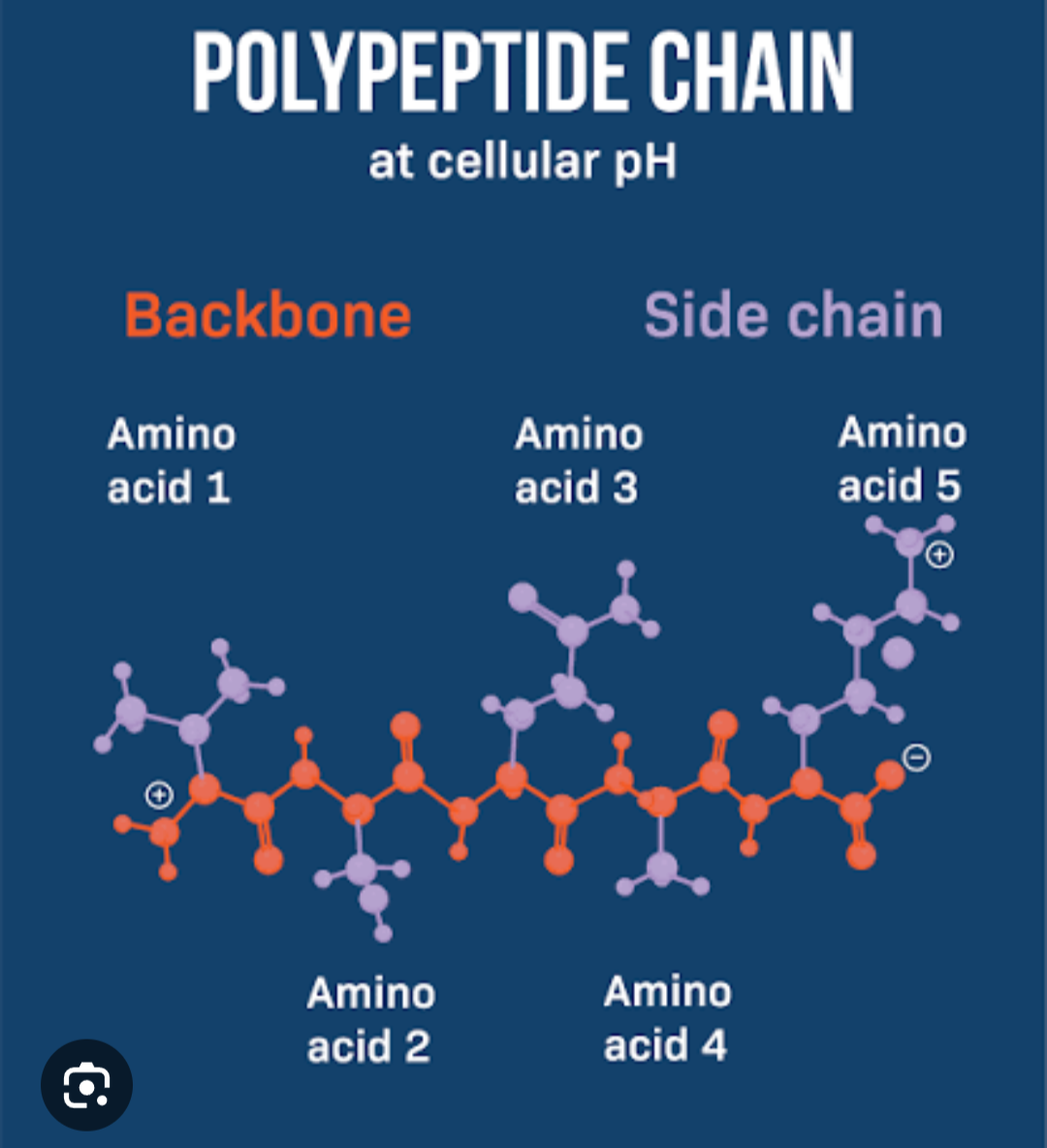
Primary structure
linear sequence of amino acids joined with peptide linkages
forms a polypeptide chain
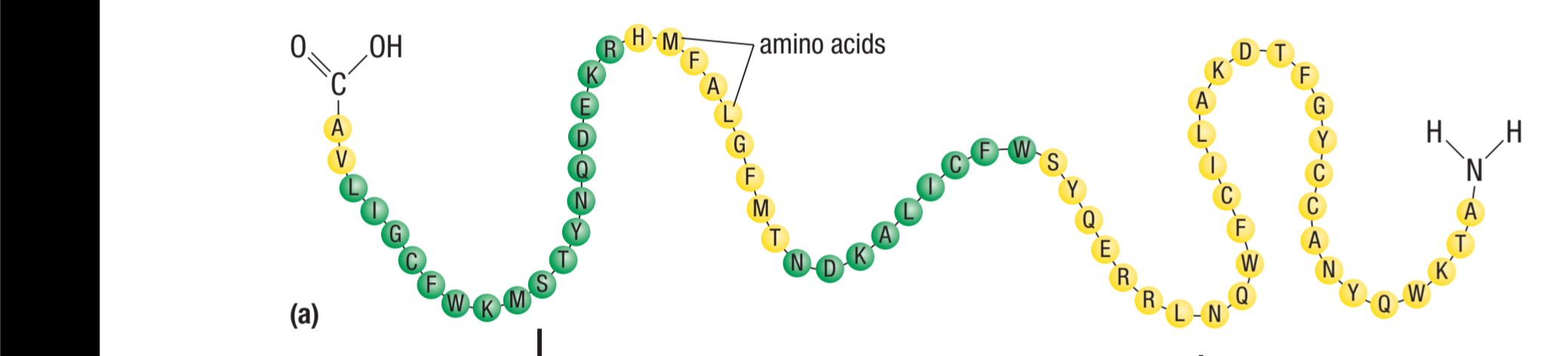
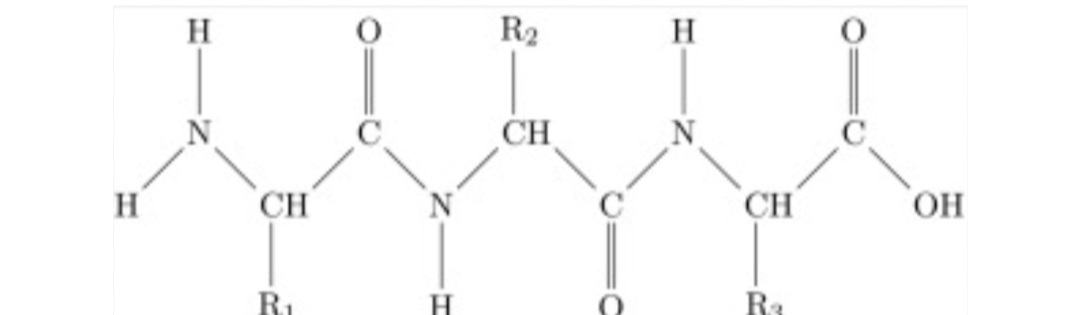
Circle the peptide linkage(s)
Includes the C=O and N-H
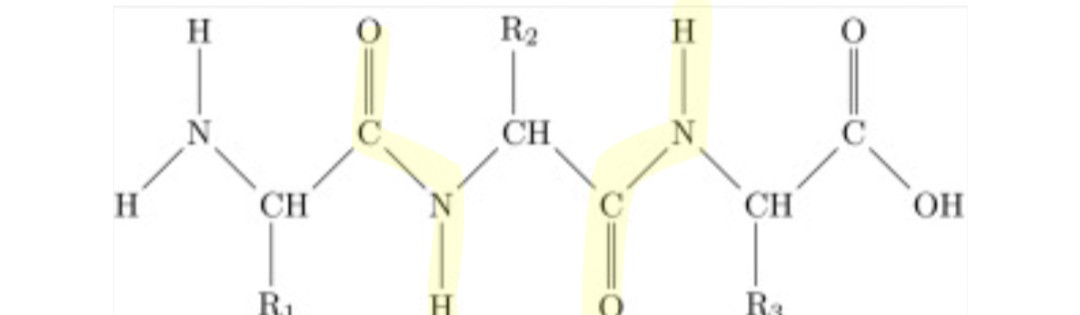
Peptide linkages are a kind of ___________ rxn
Condensation
Secondary structure
Result of H-bonding btn atoms in the amino acid backbone
Two forms: alpha helix and beta pleated sheets
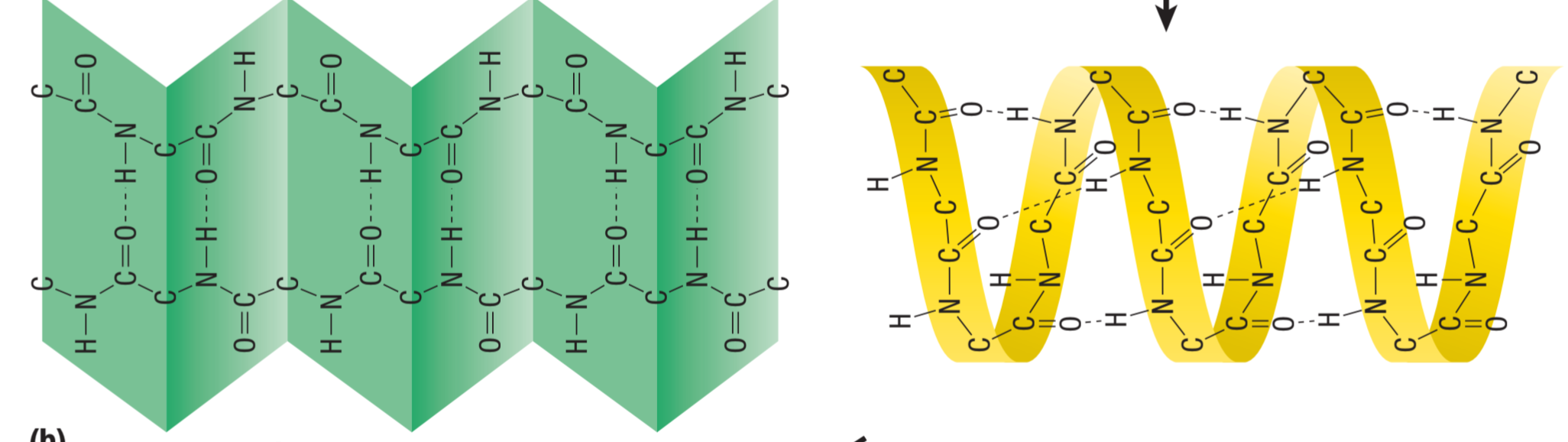
The amino acid backbone
Electronegative Oxygen or Nitrogen with an electropositive Hydrogen further down the polypeptide
⇨ when hydrogen bonding occurs between these atoms, the polypeptide chain coils or folds (secondary structure)
Tertiary structure
complex, 3D structure due to bonding interactions between the R groups
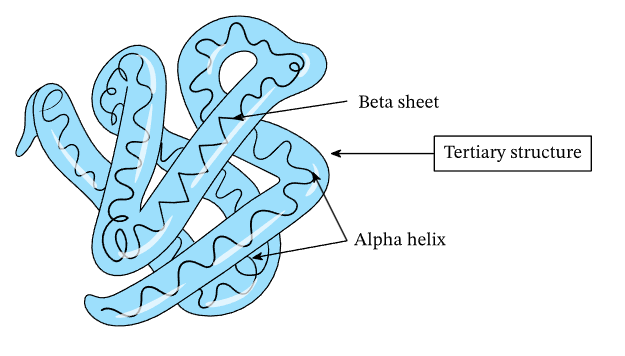
Interactions between R groups that contribute to tertiary structure include: (5)
Disulfide bridges 🌉 (between distant cysteines)
Hydrophilic associations 💧💓
Hydrophobic associations💧❌
Ionic bonds 🧲
Hydrogen bonds
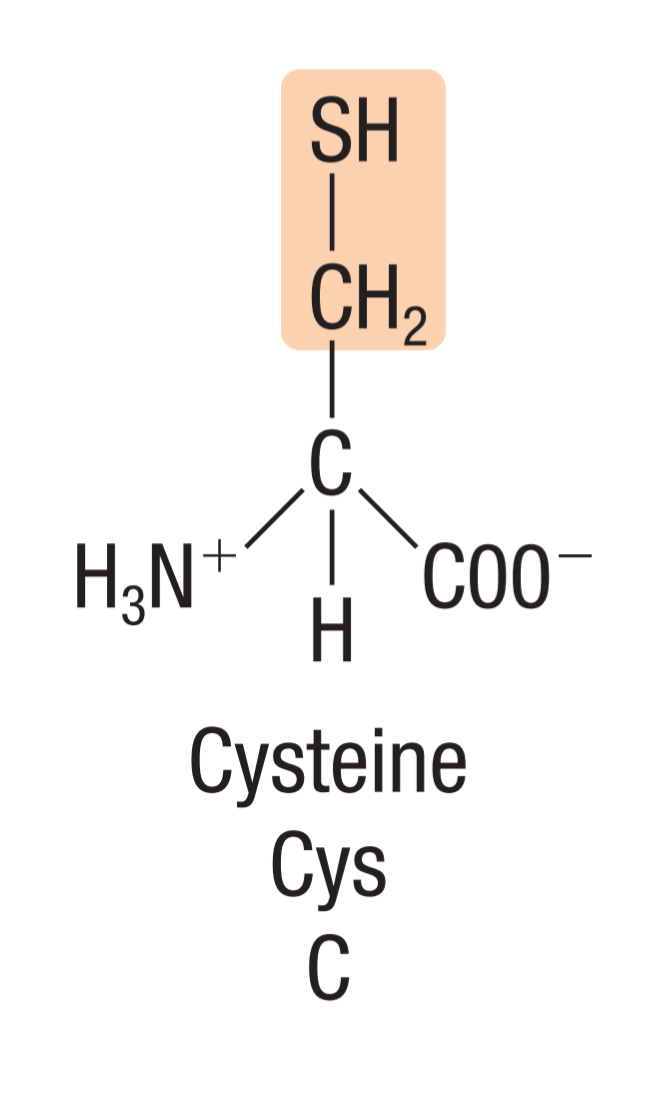
Cystein
a non-polar amino acid with sulfhydryl group (-SH) as its side chain, allowing them to form disulfide bridges/linkages
⇨the two groups react to form a strong S-S covalent bond
Tertiary Structure is critical to the _________ of proteins, especially __________!!
FUNCTION, enzymes
Quaternary structure
Occurs when multiple tertiary structures combine to make a functional protein
Many forces hold these subunits together
Hemoglobin is composed of ___ _____________, making it at the _____________ level of structure
4 polypeptides, quaternary
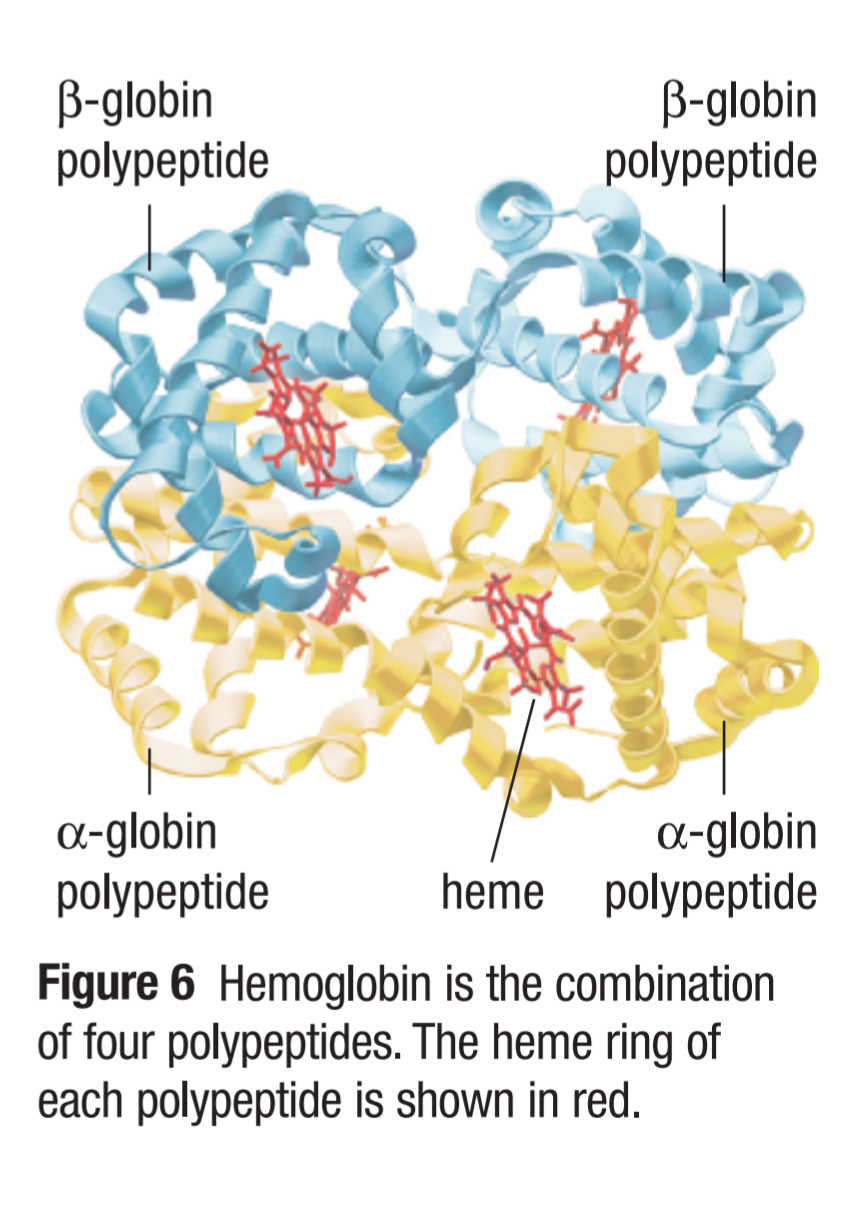
Forces that hold subunits together in a quaternary structure include: (4)
Disulfide linkages
Negatively-charged groups fitting against positively-chatged groups
Hydrogen bonds
Hydrophobic regions of two subunits associating, thus excluding water
Extreme conditions, like _____________ or ___, can unfold a protein, causing ________________
temperature, pH, denaturation
Denaturation
the loss of both the structure and function of a protein, often causing the formation a precipitate (protein is no longer soluble)
The same bonds and forces that fold single polypeptide chains into tertiary structures are also responsible for:
Holding multiple peptide chains together in quaternary structure